Abstract
Soil salinity is one of the ecological problems limiting plant growth and development, especially in semi-arid and arid regions where water availability is very limited. Stevia rebaudiana Bertoni (Asteraceae) is known for its sweetness and non-caloric products (steviol glycosides). It can be considered as a plant for the future sugar industry and human health nutrition. The present work aims to study the modulation of salt stress (NaCl: 80 mM) effects on Stevia rebaudiana Bertoni plants by an exogenous application of salicylic acid (0 mM, 0.1 mM and 0.5 mM). The results obtained show that saline stress influences the growth and development of the plant through several metabolism aspects. The exogenous application of salicylic acid to plants subjected to saline stress has improved plant biomass, chlorophyll content, chlorophyll fluorescence, stomatal conductance, antioxidant enzyme activity, MDA content and electrolyte leakage. All these results show a potential role of salicylic acid in strengthening tolerance mechanisms and mitigating physiological and biochemical damage caused by saline stress on Stevia rebaudiana Bertoni plant.
Zusammenfassung
Die Versalzung des Bodens ist eines der ökologischen Probleme, die das Wachstum und die Entwicklung von Pflanzen einschränken, besonders in semi-ariden und ariden Regionen, in denen die Wasserverfügbarkeit sehr begrenzt ist. Stevia rebaudiana Bertoni (Asteraceae) ist bekannt für ihre Süße und nichtkalorischen Produkte (Steviolglykoside). Sie kann als Pflanze für die künftige Zuckerindustrie und die gesunde Ernährung betrachtet werden. Die vorliegende Arbeit zielt darauf ab, die Modulation der Effekte von Salzstress (NaCl: 80 mM) auf Stevia rebaudiana Bertoni-Pflanzen durch eine exogene Applikation von Salicylsäure (0 mM, 0,1 mM und 0,5 mM) zu untersuchen. Die erzielten Ergebnisse zeigen, dass Salzstress das Wachstum und die Entwicklung der Pflanze durch verschiedene Stoffwechselaspekte beeinflusst. Die exogene Anwendung von Salicylsäure auf Pflanzen, die Salzstress ausgesetzt waren, hat die Pflanzenbiomasse, den Chlorophyllgehalt, die Chlorophyllfluoreszenz, die stomatäre Leitfähigkeit, die antioxidative Enzymaktivität, den MDA-Gehalt und den Elektrolytverlust verbessert. All diese Ergebnisse zeigen eine potenzielle Rolle von Salicylsäure bei der Stärkung von Toleranzmechanismen und der Abschwächung von physiologischen und biochemischen Schäden, die durch Salzstress an der Stevia rebaudiana Bertoni-Pflanze verursacht werden.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nowadays, the problem of salinity is considered one of the most important abiotic factors hindering plant growth and productivity, especially in arid and semi-arid regions (Zhu 2001; Rasool et al. 2013; Soliman and El Shaieny 2014). The excess of NaCl in soil or in irrigation water leads to massive production of reactive oxygen species (ROS) in plants (Arif et al. 2020), which generally causes photosynthetic malfunction and other metabolic disorders (Kalhor et al. 2018; Kapoor and Hasanuzzaman 2020). Salt stress affects all physiological processes of the plant; its effects vary according to the type of salinity, salt concentration, species, variety, plant organ and stage of development (Radhouane 2008; Hniličková et al. 2019).
The tolerance to salt stress is therefore a widely sought-after characteristic in plants of agronomic and medicinal interest to expand their cultivation in saline and water deficit regions. Some molecules such as salicylic acid (SA) have recently attracted the attention of the scientific community for their positive effects on the response of plants to abiotic stress. Salicylic acid is generally synthesized by the plant it self and seems to be involved in establishing resistance mechanisms to several environmental stresses (Mutlu et al. 2016; Shakirova et al. 2016; Sharma et al. 2018; Batista et al. 2019). The exogenous application of SA to plants under different stresses has been studied by several researchers and its role in activating germination, growth and development under saline stress has been reported in beans (Ahmad et al. 2018), wheat (Fardus et al. 2018) and tomato (Jahan et al. 2019). Morever, the use of salicylic acid improves photosynthetic activity, chlorophyll content antioxidant enzymes and mineral nutrition uptake (N, P, K+, Ca2+ and Mg2+) (Anaya et al. 2017; Karlidag et al. 2009). According to Nazar et al. (2011), this phytohormone plays a key role in mitigating damage due to saline stress by regulating certain metabolic and physiological pathways in plants.
Stevia rebaudiana Bertoni is a plant that is attracting a lot of interest nowadays because of its high sweetening power and non-caloricity (Samuel et al. 2018). It is considered one of the most important plants of economic interest in the world. The leaves of the plant contain diterpene-glycosides (steviosides and rebaudiosides), which are widely used as a natural non-caloric sweetener especially for diabetics (Shivanna et al. 2013). According to several researchers, this plant is very water demanding and is very sensitive to the salinisation of irrigation water (Zeng et al. 2013; Cantabella et al. 2017; Gerami et al. 2020). Its cultivation in regions with semi-arid and arid climates, which are characterized by saline water, requires control of the plant’s responses to saline stress. Few studies reported the effect of salicylic acid on stevia growth and development. The aim of this work is to study the influence of a pre-treatment with salicylic acid on the morphological, physiological and biochemical behaviour of Stevia rebaudiana Bertoni plants subjected to saline stress.
Materials and Methods
Plant Growth Analysis
The plant material used in this study consists of young seedlings (2 months after sowing) of Stevia rebaudiana Bertoni (Fig. 1).
The seeds of S. rebaudiana (var. Bertoni) were disinfected for 5 min using NaClO and then washed five times with sterile distilled water. Healthy-looking seeds germinated in honeycombed plates contained peat previously sterilized. After 2 months of germination (Fig. 1). Seedlings of uniform size (six-leaf stage) were selected and transplanted into a 2 kg plastic pots (one plant per pot) filled with sand/peat mixture (5/1). The plants were kept well irrigated (administered twice a week, field capacity of the substrate: 150 ml) for 3 weeks prior to application of the treatments. Once a week, the water is replaced by the nutrient solution (Hoagland and Arnon 1950).
The plants were treated with two levels of salicylic acid concentration (0.1 and 0.5 mM) with or without NaCl (80 mM) for 8 weeks. The SA was initially dissolved in ethanol and the concentrations of 0.1 and 0.5 mM were compensated with distilled water with 0.01% Tween-20. The SA solutions were applied exogenously with irrigation water (only once time before the saline treatment). The salt treatment began after 48 h of salicylic acid application and progressed to reach the desired concentration (to avoid osmotic shock).
The applied treatments were
-
0 mM NaCl/0 mM AS (T)
-
80 mM NaCl/0 mM AS (N)
-
0 mM NaCl/0.1 mM AS (S1)
-
0 mM NaCl/0.5 mM AS (S2)
-
80 mM NaCl/0.1 mM AS (C1)
-
80 mM NaCl/0.5 mM AS (C2)
The pots corresponding to the different treatments were arranged in a random pattern with twelve replicates per treatment. The experiment was carried out from March to May in a greenhouse (photoperiod of 16 h/8 h (light/dark), temperature: 25.5 °C and a light: 410 μm−2 s−1) at the Faculty of Sciences Semlalia, Marrakech, Morocco.
Growth Parameters
The shoot height of the plants and their fresh and dry biomass were determined after 2 months of treatment. The weight of the dry matter was measured after 48 h of drying the plant material in the oven at 70 °C with four replicates per treatment.
The leaf area was determined after processing the leaf images with the computer software “Mesurim_Pro” with four replicates per treatment.
Chlorophyll Fluorescence, Stomatal Conductance and Relative Water Content of Leaves
The photochemical efficiency of photosystem II was evaluated by the Fv/Fm ratio, where Fv corresponds to variable fluorescence and Fm to maximum fluorescence. This ratio corresponds to the maximum quantum efficiency of the PSII photochemistry. Measurements were made on mature and healthy leaves using a portable fluorometer (Chlorophyll Fluorometer, OS30P+, OPTI-SCIENCES). A 20 min dark adaptation is applied before each measurement with four replicates per treatment.
The stomatal conductance is measured weekly using a portable porometer (Leaf Porometer LP1989, DecagonDevice, Inc., Washington, USA). Measurements are made between 12:00 and 13:00 on two leaves per plant of the same stage with four replicates for each treatment.
Relative water content is the ratio of the water content of the sample at harvest to the maximum water content when the cells are fully turgid (with four replicates per treatment). It is calculated according to the method described by Turner and Begg (1981).
Electrolyte Leakage
The effect of salinity on membrane permeability has been assessed by the percentage of electrolyte leakage (Syeed et al. 2011). Leaf samples (100 mg) were rinsed several times with distilled water and placed in tubes containing 10 ml of distilled water. The electrical conductivities (C1) of the solutions were then determined using a conductivity meter after incubation for 24 h at 25 °C and stirring at 100 rpm. A second conductivity (C2) of the samples was determined after they were autoclaved at 120 °C for 20 min, followed by stirring for 30 min at 25 °C. The percentage of electrolyte loss was expressed as the ratio of C1 and C2 (with four replicates per treatment) (Electrolyte leakage (%) = (C1/C2) * 100).
Assessment of Membrane Lipid Peroxidation (MDA)
Lipid peroxidation is estimated by determining the quantities of malondialdehyde according to the method of Yin et al. (2009). 100 mg of fresh leaf material is crushed and then homogenised in 0.1% Trichloroacetic acid (TCA). The homogenate is centrifuged at 10,000 g for 15 min. 500 µl of the supernatant is mixed with 1.5 ml of Thiobarbituric acid (TBA) prepared in 20% TCA and incubated at 90 °C for 20 min in a hot-water bath. After stopping the reaction by ice, the samples are centrifuged at 10,000 g for 10 min. The optical density (OD) is read at 532 nm by using a spectrophotometer. The MDA content is determined by using the extinction coefficient 155/Mm/cm.
Hydrogen Peroxide Content
The H2O2 content of plant leaves was determined according to the method of Velikova et al. (2000). An aliquot of 100 mg of fresh material was crushed in 2 ml of TCA (20%) and then centrifuged at 15,000 g for 15 min at 4 °C. The supernatants were then recovered to determine the H2O2 content of these extracts. 0.5 ml of phosphate phosphate buffer (10 mM, pH 7) and 1 ml of iodic potassium (1 M) were added to 0.5 ml of the extract. The OD was then determined at 390 nm after 1 h incubation in the dark. The H2O2 content was expressed as nmol H2O2 per g of fresh weight with reference to a standard range established under the same conditions with known H2O2 concentrations.
Leaf Photosynthetic Pigments
Chlorophyll (a) and (b) concentration of the leaves was measured according to the classical method of Arnon (1949). 100 mg of fresh leaves were crushed with 5 ml of 80% acetone. After centrifugation (10 min at 5000 g) and OD reading at 663 and 645 nm, the quantification of the chlorophyll content (µg/g FM) was determined by using the following relations:
Antioxidant Enzyme Assays
100 mg of fresh cold leaves are milled in 1.5 ml of phosphate buffer (0.1 M pH 7) containing 5% polyvinylpolypyrolidone (pvp) and 1% EDTA. The homogenate is centrifuged at 1200 g for 15 min to recover the supernatant which is stored at −20 °C. The supernatant was used for the determination of soluble protein content and the enzymatic analysis of polyphenoloxidase (PPO), peroxidase (POX) and superoxide dismutase (SOD).
Protein determination is carried out through the Bradford method (1976). 2 ml of the protein extracts are taken from tubes and added to 2 ml of Bradford’s reagent. Optical density (OD) is measured at 595 nm. Polyphenoloxidase (PPO) activity is measured according to the method of Oktay et al. (1995). The reaction medium consists of 600 μl catechol (0.1 M), 100 μl enzyme extract and 3 ml phosphate buffer (0.1 M, pH 7) while the enzyme activity of peroxidase (POX) is determined spectrophotometrically using guaiacol and hydrogen peroxide (H2O2) as enzyme substrates. The reaction mixture contains 100 μl of the enzyme extract, 300 μL of guaiacol (20 mM) and 2 ml of phosphate buffer (0.1 M, pH 6). The reaction is triggered by the addition of 200 μl hydrogen peroxide (0.3% H2O2) and then the kinetics of tetraguaiacol formation is monitored by spectrophotometry at 470 nm (Hori et al. 1997). The activity of superoxide dismutase (SOD) is determined by Beauchamp and Fridovich (1971) by measuring the capacity of this enzyme to inhibit the photochemical reduction of nitro blue tetrazolium (NBT). The absorbance of the reaction product is measured at 560 nm.
The catalase assay is carried out according to the method of Montavon et al. (2007), 100 mg of fresh leaf tissue is homogenised in 50 mmol phosphate buffer (pH 7) containing 150 mmol NaCl and 0.5 mmol EDTA. One unit of CAT activity corresponds to the amount of enzyme required to reduce 1 μmol hydrogen peroxide (H2O2) per minute.
Statistical Analysis
Statistical analysis of the data is carried out by analysis of variance (ANOVA) to determine differences between treatments. The LSD test is used to compare the averages of the different treatments to the 5% threshold using SPSS for Windows software.
Results
Growth Parameters
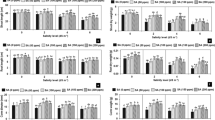
Measurements of growth parameters of Stevia rebaudiana Bertoni under the different concentrations of salicylic acid and NaCl are shown in Table 1, Fresh weight of the aerial and root parts as well as the stem length of Stevia rebaudiana Bertoni were significantly reduced by 41, 32 and 34% respectively in conditions of stress. Furthermore, the exogenous application of salicylic acid improved significantly the biomass of plants in the presence or absence of saline stress (80 mM). It should also be noted that the 0.5 mM concentration of salicylic acid gave good results compared to the 0.1 mM concentration of salicylic acid under saline stress conditions.
Leaf Area
The effect of NaCl results in a significant reduction (32%) of the leaf area in plants of Stevia rebaudiana Bertoni (Fig. 2). Also, the addition of SA to non-stressed plants caused a significant increase in leaf area, especially the 0.5 mM concentration. In addition, the combination of NaCl (80 mM) with salicylic acid (0.1 and 0.5 mM) induces a significant improvement (41 and 63% respectively) of the leaf area compared to the stressed plants not treated with SA.
Influence of salicylic acid (0, 0.1 and 0.5 mM) and/or NaCl (0 and 80 mM) on leaf area of Stevia rebaudiana Bertoni after 8 weeks of treatment. The values represent the mean of 4 replicates ± standard deviation. Bars of each parameter labeled by different lettersare significantly different at 5% significance level by using the LSD test
Chlorophyll Fluorescence
To estimate the effect of salicylic acid on the photosynthetic activity of Stevia rebaudiana Bertoni under saline stress (80 mM), the photochemical efficiency of photosystem II (Fv/Fm) was evaluated (Fig. 3). It can be seen that this parameter underwent a significant regression under the influence of salt stress. This reduction is significatively smaller in plants treated with salicylic acid in the presence of NaCl.
Influence of salicylic acid (0, 0.1 and 0.5 mM) and/or NaCl (0 and 80 mM) on chlorophyll fluorescence of Stevia rebaudiana Bertoni after 8 weeks of treatment. The values represent the mean of 4 replicates ± standard deviation. Bars of each parameter labeled by different lettersare significantly different at 5% significance level using the LSD test
Stomatal Conductance
The study of stomatal conductance (Fig. 4) was significantly reduced under the effect of salt stress (72% compared to control). However, under treatment with salicylic acid (0.5 mM), non-stressed plants recorded an 18% increase compared to control plants. Yet, the combination of SA with NaCl reduced the harmful effects of saline stress on the plants gas exchanges, it was also noted that the highest values were obtained with the 0.5 mM concentration of SA.
Influence of salicylic acid (0, 0.1 and 0.5 mM) and/or NaCl (0 and 80 mM) on stomatal conductance of Stevia rebaudiana Bertoni after 8 weeks of treatment. The values represent the mean of 4 replicates ± standard deviation. Bars of each parameter labeled by different lettersare significantly different at 5% significance level using the LSD test
Relative Water Content (RWC)
According to the analysis of Fig. 5, the relative water content of the leaves of Stevia rebaudiana Bertoni is very high (79%) in controlled plants, while the values recorded in stressed plants with 80 mM NaCl are very low (29%). Moreover, addition of SA caused a significant increase (<0.05) of relative water content, especially with the 0.5 mM concentration. These results show that salicylic acid positively influenced the water uptake by stressed plants.
Influence of salicylic acid (0, 0.1 and 0.5 mM) and/or NaCl (0 and 80 mM) on relative water content of Stevia rebaudiana Bertoni after 8 weeks of treatment. The values represent the mean of 4 replicates ± standard deviation. Bars of each parameter labeled by different lettersare significantly different at 5% significance level using the LSD test
Electrolyte Leakage
Statistical analysis of the results in Fig. 6 shows that salt stress caused a significant increase in electrolyte leakage in all stressed plants with or without salicylic acid. Plants treated with salicylic acid and NaCl showed a low loss of electrolytes compared to plants treated with NaCl alone.
Influence of salicylic acid (0, 0.1 and 0.5 mM) and/or NaCl (0 and 80 mM) on the electrolyte leakage of Stevia rebaudiana Bertoni after 8 weeks of treatment. The values represent the mean of 4 replicates ± standard deviation. Bars of each parameter labeled by different lettersare significantly different at 5% significance level using the LSD test
Membrane Lipid Peroxidation
Under the salt stress conditions, the accumulation of MDA (Fig. 7) is very high (35 nmol/g fw) compared to control plants (15 nmol/g FM). The application of salicylic acid at 0.1 and 0.5 mM to the stressed plants decreased significantly (p < 0.05) the MDA level.
Influence of salicylic acid (0, 0.1 and 0.5 mM) and/or NaCl (0 and 80 mM) on MDA content of Stevia rebaudiana Bertoni after 8 weeks of treatment. The values represent the mean of 4 replicates ± standard deviation. Bars of each parameter labeled by different lettersare significantly different at 5% significance level using the LSD test
Hydrogen Peroxide Content
The results relative to the effect of NaCl on the H2O2 content of the plants of Stevia rebaudiana Bertoni are presented in Fig. 8, the salinity of the growing medium caused an increase in the quantity of H2O2 in the leaves, and this increase is less intense when salicylic acid is applied under salt stress. The interaction between salicylic acid and NaCl has a significant effect on the H2O2 content.
Influence of salicylic acid (0, 0.1 and 0.5 mM) and/or NaCl (0 and 80 mM) on H2O2 content of Stevia rebaudiana Bertoni after 8 weeks of treatment. The values represent the mean of 4 replicates ± standard deviation. Bars of each parameter labeled by different lettersare significantly different at 5% significance level using the LSD test
Leaf Photosynthetic Pigments
Analysis of the chlorophylls (a) and (b) contents (Fig. 9) shows that under salt stress, Stevia rebaudiana Bertoni plants experienced a remarkable and statistically significant drop compared to control plants of about 51% for chlorophyll (a) and 56% for chlorophyll (b). On the other hand, stressed plants treated with SA showed significantly higher levels of chlorophyll (a) and (b) compared to plants treated with saline stress alone.
Influence of salicylic acid (0, 0.1 and 0.5 mM) and/or NaCl (0 and 80 mM) on chlorophyll (a) and (b) contents of Stevia rebaudiana Bertoni after 8 weeks of treatment. The values represent the mean of 4 replicates ± standard deviation. Bars of each parameter labeled by different lettersare significantly different at 5% significance level using the LSD test
Protein Content
The exploitation of the results of Fig. 10 allows to underline that salicylic acid has no significant effect on the protein content of Stevia rebaudiana Bertoni, it was also detected that salinity causes a significant increase (p < 0.05) of the foliar protein content, while the addition of salicylic acid at 0.1 and 0.5 mM to the plants under stress conditions (80 mM) induced a significant decrease (p < 0.05) of the protein content (equal to 41 and 36% respectively).
Influence of salicylic acid (0, 0.1 and 0.5 mM) and/or NaCl (0 and 80 mM) on protein content of Stevia rebaudiana Bertoni after 8 weeks of treatment. The values represent the mean of 4 replicates ± standard deviation. Bars of each parameter labeled by different lettersare significantly different at 5% significance level using the LSD test
Antioxidant Enzymes Activity
Polyphenoloxidases (PPO)
The activity of polyphenoloxidase (Fig. 11) was significantly influenced by NaCl (80 mM) and/or salicylic acid, a significant increase was observed in plants exposed to salt stress. Indeed, the application of salicylic acid at 0.1 and 0.5 mM on Stevia rebaudiana Bertoni plants under stress conditions increased the activity of this enzyme.
Influence of salicylic acid (0, 0.1 and 0.5 mM) and/or NaCl (0 and 80 mM) on PPO of Stevia rebaudiana Bertoni after 8 weeks of treatment. The values represent the mean of 4 replicates ± standard deviation. Bars of each parameter labeled by different lettersare significantly different at 5% significance level using the LSD test
Peroxidases (POX)
Fig. 12 illustrates the effect of saline treatment and/or salicylic acid on the plants of Stevia rebaudiana Bertoni, it was found that there is an increase (34.37%) in the activity of these enzymes in the presence of saline stress compared to the non-stressed control. However, in the presence of salicylic acid, the activity of peroxidase was significantly increased in the presence or absence of saline stress.
Influence of salicylic acid (0, 0.1 and 0.5 mM) and/or NaCl (0 and 80 mM) on POX of Stevia rebaudiana Bertoni after 8 weeks of treatment. The values represent the mean of 4 replicates ± standard deviation. Bars of each parameter labeled by different lettersare significantly different at 5% significance level using the LSD test
Superoxide Dismuatase (SOD)
The data presented in the figure below (Fig. 13) show that salinity significantly increased SOD activity by approximately 32% compared to control, treatment with salicylic acid alone had no effect on the control plants, on the other hand, under saline stress, salicylic acid at 0.1 and 0.5 mM significantly increased SOD with percentages of 16 and 22% respectively compared to the stressed plants.
Influence of salicylic acid (0, 0.1 and 0.5 mM) and/or NaCl (0 and 80 mM) on SOD of Stevia rebaudiana Bertoni after 8 weeks of treatment. The values represent the mean of 4 replicates ± standard deviation. Bars of each parameter labeled by different lettersare significantly different at 5% significance level using the LSD test
Catalases (CAT)
The catalase activity in the leaves is significantly affected by salinity and salicylic acid (Fig. 14), an increase in this enzyme is observed in stressed plants compared to the control plants, however, the combination of salicylic acid with NaCl can lead to a higher elevation compared to the other treatments.
Influence of salicylic acid (0, 0.1 and 0.5 mM) and/or NaCl (0 and 80 mM) on CAT of Stevia rebaudiana Bertoni after 8 weeks of treatment. The values represent the mean of 4 replicates ± standard deviation. Bars of each parameter labeled by different lettersare significantly different at 5% significance level using the LSD test
Discussion
Salicylic acid is an endogenous growth hormone, which can improve the plant’s response to stress conditions (biotic and abiotic) by increasing the resistance of the plant to System Acquired Resistance (SAR) (Khan et al. 2015). Several researchers have demonstrated the interest of using this molecule to improve plant tolerance against environmental constraints such as salt stress (Simaei et al. 2012; Miao et al. 2020; Souana et al. 2020).
The results obtained show that salt affects negatively the weight of the fresh and dry matter and the stem length as well as the leaf area of the plant. These data are similar to those observed by Zhu (2001) who reported that the reduction in the growth of the aerial vegetative apparatus is an adaptive capacity necessary for the survival of plants exposed to abiotic stress. Indeed, this effect on the development allows the plant to accumulate energy and nutrient reserves to combat stress. Reduction in the number of leaves and leaf area of plants could be explained by the early senescence of leaves with a decrease in the mitotic activity of epidermal cells. This poor development is generally due to the adaptive mechanism of plants that aims to limit plant leaf transpiration during adverse conditions (Ghanem et al. 2008; Santos et al. 2016). According to Ordóñez et al. (2018), stevia can tolerate water with electrical conductivity up to 2.0 mS cm−1, and that at higher salinity (≥80 mM), the biomass yield was significantly reduced (Mahajan et al. 2020).
The exogenous application of salicylic acid improved the biomass of Stevia rebaudiana Bertoni seedlings in the presence or absence of NaCl (80 mM), which is consistent with the results in maize (Gunes et al. 2007), strawberry (Karlidag et al. 2009), tomato (Mimouni et al. 2016) and rice (Jini and Joseph 2017), which demonstrated that pre-treatment with salicylic acid under conditions of salt stress has positive effects on plant growth parameters. This effect might be related to the effect of salicylic acid in inhibition of Cl− and Na+ accumulation and increased the K+ content under salt stress (Jini and Joseph 2017).
In this work, chlorophyll fluorescence, specifically the maximum efficiency of PSII (Fv/Fm), was used to evaluate the impact of salicylic acid and/or salt stress on the photosynthetic activity of Stevia rebaudiana Bertoni. The Fv/Fm ratio was negatively influenced by salt in this experiment. This reduction generally implies damage in the photochemical efficiency of PSII (Mehta et al. 2010). However, treatment with salicylic acid had a positive effect on the Fv/Fm ratio. Methenni et al. (2018) also reported a similar increase in photosynthetic activity antioxidant system under saline condition. Several studies have reported that pre-treatment with salicylic acid restored the functioning of the photosystem II reaction centre by restoring the activity of protein D1 and protein kinase (Zhao et al. 2011; Anaya et al. 2017).
Determination of stomatal conductance is one of the simplest and most non-destructive techniques, indicating the plant’s foliar transpiration rate. In this experiment, stomatal conductance decreased significantly under the stress conditions. These results are consistent with those of Mahlooji et al. (2018) who reported that plant survival under stress conditions requires a reduction in gas exchange (with stomatal closure), which leads to a drop in the rate of CO2 assimilation necessary for photosynthesis (Schulze and Hall 1982). Addition of SA improved stomatal conductance under both saline and non-saline conditions. These results are consistent with those presented by Ma et al. (2017) who reported that under saline stress, salicylic acid improves stomatal density and stomatal conductance, and this is due to the fact that salicylic acid limits ethylene formation by inhibiting the activity of 1‑aminocyclopropane-1-carboxylic acid (ACC) synthase (ACS) (Lotfi et al. 2020). This improvement results in a reduction in the toxic effects of salt stress. A decrease in RWC is considered to be one of the first symptoms of water shortage in plant tissues (Valentovic et al. 2006). The results obtained showed that the relative water content of the leaves decreased significantly in stressed plants. According to Khator et al. (2020), this decrease can be explained by the low turgidity due to the limited availability of water for the cell extension process. These results also agree with those found by Karlidag et al. (2009) who found that salinity negatively affects the relative water content in strawberries. In the presence of salicylic acid and under salt stress conditions, the relative water content was significantly increased in the leaves of Stevia rebaudiana Bertoni, these results are in agreement with those found by Sabzmeydani et al. (2020) on Kentucky bluegrass.
The disruption of photosynthesis is also due to the reduction in the content of chlorophyll pigments (a and b) under conditions of saline stress. In the presence of saline treatment, Stevia rebaudiana reduced the content of its chlorophyll pigments. Zhang et al. (2018) reported that NaCl causes a drop in photosynthetic pigment content in Castor bean. This reduction is attenuated by the exogenous application of salicylic acid that may have a a protective effect on chloroplasts and the thylakoid membrane against the oxidative effects of salt stress as demonstrated by Bose et al. (2017).
Membrane permeability and MDA content are considered among the methods for assessing oxidative effects on plants (Astaneh et al. 2019). Under salt stress, a high accumulation of Na+ and Cl− ions in plant cells causes a disturbance in the membrane permeability of the cells (Kaya et al. 2018). Application of salicylic acid (0.1 or 0.5 mM) on Stevia rebaudiana Bertoni plants, significantly reduced the loss of electrolytes and the accumulation of MDA, a marker of membrane lipid peroxidation, under stressful conditions. This is consistent with the results of Kaya et al. (2020) in peppers and El-Esawi et al. (2017) in rosemary under salt stress. On the other hand, the H2O2 content was decreased by the addition of salicylic acid, which is consistent with the results found by Alsahli et al. (2019), who showed that salicylic acid contributes to the neutralization of H2O2 caused by the stress, which explains the potential role of salicylic acid in protecting the cell membrane against the adverse effects of salt stress.
According to the results found, Stevia rebaudiana Bertoni increased the activity of the antioxidant enzymes specifically peroxidase (POX), polyphenoloxidase (PPO), superoxide dismutase (SOD) and catalase (CAT) under saline stress. The increased activity of these enzymes plays a very important role in the elimination of ROS under saline conditions (Ahanger and Agarwal 2017). Induction of these enzymes in plants has also been reported in several species in response to saline stress (Vighi et al. 2017; Ghaderi et al. 2018; Srivastava and Shahi 2018; Tahjib-Ul-Arif et al. 2018). Our results also show that the treatment of plants subjected to salt stress with salicylic acid clearly helped the plants to further increase the activity of the enzymes studied compared to plants treated only with salt. Recently, a study conducted on radish (Bukhat et al. 2020), reported that the application of salicylic acid induced an increase in antioxidant enzymes with the aim of strengthening the antioxidant system against reactive oxygen species (ROS) such as superoxide (O2▪), hydrogen peroxide (H2O2), hydroxyl radical (OH−), and singlet oxygen (1 O2), confirming the results found in this study. Thus, salinity tolerance in stevia rebaudiana was associated with the induction of an antioxidant enzyme system by the application of salicylic acid, which improved the plant performance as well as protected against various damages caused by salt stress.
Conclusion
The exposure of Stevia rebaudiana Bertoni to salt stress influenced all the morphological, physiological and biochemical parameters studied through the reduction of the biomass, the disturbance of the photosynthetic apparatus and the deterioration of the plant’s cell membrane. An exogenous pre-treatment with salicylic acid improves the plant’s tolerance to saline stress. This positive effect of salicylic acid can be explained by the enhanced growth and improved pigment content, relative water content and photosynthesis. In addition, salicylic acid, especially the 0.5 mM SA concentration, plays a very important role in reducing the oxidative effects of stress through the activation of a range of antioxidant enzymes. We can therefore consider salicylic acid to be one of the techniques that can be used to improve the tolerance of this plant to saline conditions.
References
Ahanger MA, Agarwal RM (2017) Salinity stress induced alterations in antioxidant metabolism and nitrogen assimilation in wheat (Triticum aestivum L) as influenced by potassium supplementation. Plant Physiol Biochem 115:449–460
Ahmad P, Alyemeni MN, Ahanger MA, Egamberdieva D, Wijaya L, Alam P (2018) Salicylic acid (SA) induced alterations in growth, biochemical attributes and antioxidant enzyme activity in faba bean (Vicia faba L.) seedlings under NaCl toxicity. Russ J Plant Physiol 65(1):104–114. https://doi.org/10.1134/S1021443718010132
Alsahli A, Mohamed AK, Alaraidh I, Al-Ghamdi A, Al-Watban A, El-Zaidy M, Alzahrani SM (2019) Salicylic acid alleviates salinity stress through the modulation of biochemical attributes and some key antioxidants in wheat seedlings. Pak J Bot 51(5):1551–1559. https://doi.org/10.30848/PJB2019-5(12)
Anaya F, Fghire R, Wahbi S, Loutfi K (2017) Antioxidant enzymes and physiological traits of Vicia faba L. as affected by salicylic acid under salt stress. J Mater Environ Sci 8(7):2549–2563
Arif Y, Singh P, Siddiqui H, Bajguz A, Hayat S (2020) Salinity induced physiological and biochemical changes in plants: an omic approach towards salt stress tolerance. Plant Physiol Biochem 156:64–77. https://doi.org/10.1016/j.plaphy.2020.08.042
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24(1):1
Astaneh RK, Bolandnazar S, Nahandi FZ, Oustan S (2019) Effects of selenium on enzymatic changes and productivity of garlic under salinity stress. S Afr J Bot 121:447–455. https://doi.org/10.1016/j.sajb.2018.10.037
Batista VCV, Pereira IMC, de Oliveira Paula-Marinho S, Canuto KM, Pereira RDCA, Rodrigues THS et al (2019) Salicylic acid modulates primary and volatile metabolites to alleviate salt stress-induced photosynthesis impairment on medicinal plant Egletes viscosa. Environ Exp Bot 167:103870. https://doi.org/10.1016/j.envexpbot.2019.103870
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44(1):90370–90378. https://doi.org/10.1016/0003-2697
Bose J, Munns R, Shabala S, Gilliham M, Pogson B, Tyerman SD (2017) Chloroplast function and ion regulation in plants growing on saline soils: lessons from halophytes. J Exp Bot 68(12):3129–3143. https://doi.org/10.1093/jxb/erx142
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Bukhat S, Manzoor H, Zafar ZU, Azeem F, Rasul S (2020) Salicylic acid induced photosynthetic adaptability of raphanus sativus to salt stress is associated with antioxidant capacity. J Plant Growth Regul 39(2):809–822. https://doi.org/10.1007/s00344-019-10024-z
Cantabella D, Piqueras A, Acosta-Motos JR, Bernal-Vicente A, Hernández JA, Díaz-Vivancos P (2017) Salt-tolerance mechanisms induced in stevia rebaudiana Bertoni: effects on mineral nutrition, antioxidative metabolism and steviol glycoside content. Plant Physiol Biochem 115:484–496. https://doi.org/10.1016/j.plaphy.2017.04.023
El-Esawi MA, Elansary HO, El-Shanhorey NA, Abdel-Hamid AM, Ali HM, Elshikh MS (2017) Salicylic acid-regulated antioxidant mechanisms and gene expression enhance rosemary performance under saline conditions. Front Physiol 8:716. https://doi.org/10.3389/fphys.2017.00716
Fardus J, Matin MA, Hasanuzzaman M, Hossain MA, Hasanuzzaman M (2018) Salicylic acid-induced improvement in germination and growth parameters of wheat under salinity stress. J Anim Plant Sci 28:197–207
Gerami M, Majidian P, Ghorbanpour A, Alipour Z (2020) Stevia rebaudiana Bertoni responses to salt stress and chitosan elicitor. Physiol Mol Biol Plants. https://doi.org/10.1007/s12298-020-00788-0
Ghaderi N, reza Hatami M, Mozafari A, Siosehmardeh A (2018) Change in antioxidant enzymes activity and some morpho-physiological characteristics of strawberry under long-term salt stress. Physiol Mol Biol Plants 24(5):833–843. https://doi.org/10.1007/s12298-018-0535-2
Ghanem ME, Albacete A, Martínez-Andújar C, Acosta M, Romero-Aranda R, Dodd IC, Pérez-Alfocea F (2008) Hormonal changes during salinity-induced leaf senescence in tomato (Solanum lycopersicum L.). J Exp Bot 59(11):3039–3050. https://doi.org/10.1093/jxb/ern153
Gunes A, Inal A, Alpaslan M, Eraslan F, Bagci EG, Cicek N (2007) Salicylic acid induced changes on some physiological parameters symptomatic for oxidative stress and mineral nutrition in maize (Zea mays L.) grown under salinity. J Plant Physiol 164(6):728–736. https://doi.org/10.1016/j.jplph.2005.12.009
Hniličková H, Hnilička F, Orsák M, Hejnák V (2019) Effect of salt stress on growth, electrolyte leakage, Na+ and K+ content in selected plant species. Plant Soil Environ 65(2):90–96. https://doi.org/10.17221/620/2018-PSE
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Circular. California agricultural experiment station, 347, 2nd edn.
Hori K, Wada A, Shibuta T (1997) Changes in phenoloxidase activities of the galls on leaves of Ulmus davidana formed by Tetraneura fuslformis (Homoptera: Eriosomatidae). Appl Entomol Zool 32(2):365–371. https://doi.org/10.1303/aez.32.365
Jahan MS, Wang Y, Shu S, Zhong M, Chen Z, Wu J, Guo S (2019) Exogenous salicylic acid increases the heat tolerance in Tomato (Solanum lycopersicum L) by enhancing photosynthesis efficiency and improving antioxidant defense system through scavenging of reactive oxygen species. Sci Hortic 247:421–429. https://doi.org/10.1016/j.scienta.2018.12.047
Jini D, Joseph B (2017) Physiological mechanism of salicylic acid for alleviation of salt stress in rice. Rice Sci 24(2):97–108. https://doi.org/10.1016/j.rsci.2016.07.007
Kalhor MS, Aliniaeifard S, Seif M, Asayesh EJ, Bernard F, Hassani B, Li T (2018) Enhanced salt tolerance and photosynthetic performance: Implication of ɤ‑amino butyric acid application in salt-exposed lettuce (Lactuca sativa L.) plants. Plant Physiol Biochem 130:157–172. https://doi.org/10.1016/j.plaphy.2018.07.003
Kapoor RT, Hasanuzzaman M (2020) Exogenous kinetin and putrescine synergistically mitigate salt stress in Luffa acutangula by modulating physiology and antioxidant defense. Physiol Mol Biol Plants. https://doi.org/10.1007/s12298-020-00894-z
Karlidag H, Yildirim E, Turan M (2009) Salicylic acid ameliorates the adverse effect of salt stress on strawberry. Sci Agric 66(2):180–187. https://doi.org/10.1590/S0103-90162009000200006
Kaya C, Ashraf M, Alyemeni MN, Ahmad P (2020) The role of endogenous nitric oxide in salicylic acid-induced up-regulation of ascorbate-glutathione cycle involved in salinity tolerance of pepper (Capsicum annuum L.) plants. Plant Physiol Biochem 147:10–20. https://doi.org/10.1016/j.plaphy.2019.11.040
Kaya C, Aydemir S, Akram NA, Ashraf M (2018) Epibrassinolide application regulates some key physio-biochemical attributes as well as oxidative defense system in maize plants grown under saline stress. J Plant Growth Regul 37(4):1244–1257. https://doi.org/10.1007/s00344-018-9830-y
Khan MIR, Fatma M, Per TS, Anjum NA, Khan NA (2015) Salicylic acid-induced abiotic stress tolerance and underlying mechanisms in plants. Front Plant Sci 6:462. https://doi.org/10.3389/fpls.2015.00462
Khator K, Mahawar L, Shekhawat GS (2020) NaCl induced oxidative stress in legume crops of Indian Thar Desert: an insight in the cytoprotective role of HO1, NO and antioxidants. Physiol Mol Biol Plants 26(1):51–62. https://doi.org/10.1007/s12298-019-00728-7
Lotfi R, Ghassemi-Golezani K, Pessarakli M (2020) Salicylic acid regulates photosynthetic electron transfer and stomatal conductance of mung bean (Vigna radiata L.) under salinity stress. Biocatal Agric Biotechnol. https://doi.org/10.1016/j.bcab.2020.101635
Ma X, Zheng J, Zhang X, Hu Q, Qian R (2017) Salicylic acid alleviates the adverse effects of salt stress on dianthus superbus (caryophyllaceae) by activating photosynthesis, protecting morphological structure, and enhancing the antioxidant system. Front Plant Sci 8:600. https://doi.org/10.3389/fpls.2017.00600
Mahajan M, Sharma S, Kumar P, Pal PK (2020) Foliar application of KNO3 modulates the biomass yield, nutrient uptake and accumulation of secondary metabolites of Stevia rebaudiana under saline conditions. Ind Crops Prod 145:112102
Mahlooji M, Sharifi RS, Razmjoo J, Sabzalian MR, Sedghi M (2018) Effect of salt stress on photosynthesis and physiological parameters of three contrasting barley genotypes. Photosynthetica 56(2):549–556. https://doi.org/10.1007/s11099-017-0699-y
Mehta P, Jajoo A, Mathur S, Bharti S (2010) Chlorophyll a fluorescence study revealing effects of high salt stress on photosystem II in wheat leaves. Plant Physiol Biochem 48(1):16–20. https://doi.org/10.1016/j.plaphy.2009.10.006
Methenni K, Abdallah MB, Nouairi I, Smaoui A, Zarrouk M, Youssef NB (2018) Salicylic acid and calcium pretreatments alleviate the toxic effect of salinity in the oueslati olive variety. Sci Hortic 233:349–358
Miao Y, Luo X, Gao X, Wang W, Li B, Hou L (2020) Exogenous salicylic acid alleviates salt stress by improving leaf photosynthesis and root system architecture in cucumber seedlings. Sci Hortic 272:109577
Mimouni H, Wasti S, Manaa A, Gharbi E, Chalh A, Vandoorne B, Ahmed HB (2016) Does salicylic acid (SA) improve tolerance to salt stress in plants? A study of SA effects on tomato plant growth, water dynamics, photosynthesis, and biochemical parameters. OMICS 20(3):180–190. https://doi.org/10.1089/omi.2015.0161
Montavon P, Kukic KR, Bortlik K (2007) A simple method to measure effective catalase activities: optimization, validation, and application in green coffee. Anal Biochem 360(2):207–215. https://doi.org/10.1016/j.ab.2006.10.035
Mutlu S, Atıcı Ö, Nalbantoğlu B, Mete E (2016) Exogenous salicylic acid alleviates cold damage by regulating antioxidative system in two barley (Hordeum vulgare L.) cultivars. Front Life Sci 9(2):99–109. https://doi.org/10.1080/21553769.2015.1115430
Nazar R, Iqbal N, Syeed S, Khan NA (2011) Salicylic acid alleviates decreases in photosynthesis under salt stress by enhancing nitrogen and sulfur assimilation and antioxidant metabolism differentially in two mungbean cultivars. J Plant Physiol 168(8):807–815. https://doi.org/10.1016/j.jplph.2010.11.001
Oktay M, Küfreviolu I, Kocaçalişkan I, Şaklrolu H (1995) Polyphenoloxidase from Amasya apple. J Food Science 60(3):494–496. https://doi.org/10.1111/j.1365-2621.1995.tb09810.x
Ordóñez YBM, Jaramillo GR, Chalé JT (2018) Efecto de la calidad del agua y sustratos en la producción de plántulas de estevia („Stevia rebaudiana” Bertoni). Rev Mex Cienc Agríc 21:4284–4295
Radhouane L (2008) Salinity effect on germination, growth, and grain production of some autochthonous pear millet ecotypes (Pennisetum glaucum (L.) R. Br.). C R Biol 331(4):278–286. https://doi.org/10.1016/j.crvi.2008.01.006
Rasool S, Ahmad A, Siddiqi TO, Ahmad P (2013) Changes in growth, lipid peroxidation and some key antioxidant enzymes in chickpea genotypes under salt stress. Acta Physiol Plant 35(4):1039–1050
Sabzmeydani E, Sedaghathoor S, Hashemabadi D (2020) Progesterone and salicylic acid elevate tolerance of Poa pratensis to salinity stress. Russ J Plant Physiol 67(2):285–293. https://doi.org/10.1134/S1021443720020132
Samuel P, Ayoob KT, Magnuson BA, Wölwer-Rieck U, Jeppesen PB, Rogers PJ, Mathews R (2018) Stevia leaf to stevia sweetener: exploring its science, benefits, and future potential. J Nutr 148(7):1186S–1205S. https://doi.org/10.1093/jn/nxy102
Santos JB, Gheyi HR, de Lima GS, Xavier DA, Cavalcante LF, Centeno CRM (2016) Morfofisiologia e produção do algodoeiro herbáceo irrigado com águas salinas eadubado com nitrogênio. Comun Sci 7(1):86–96. https://doi.org/10.14295/cs.v7i1.1158
Schulze ED, Hall AE (1982) Stomatal responses, water loss and CO 2 assimilation rates of plants in contrasting environments. In: Physiological plant ecology II. Springer, Berlin, Heidelberg, New York, pp 181–230 https://doi.org/10.1007/978-3-642-68150-9_8
Shakirova FM, Allagulova CR, Maslennikova DR, Klyuchnikova EO, Avalbaev AM, Bezrukova MV (2016) Salicylic acid-induced protection against cadmium toxicity in wheat plants. Environ Exp Bot 122:19–28. https://doi.org/10.1016/j.envexpbot.2015.08.002
Sharma M, Gupta SK, Majumder B, Maurya VK, Deeba F, Alam A, Pandey V (2018) Proteomics unravel the regulating role of salicylic acid in soybean under yield limiting drought stress. Plant Physiol Biochem 130:529–541. https://doi.org/10.1016/j.plaphy.2018.08.001
Shivanna N, Naika M, Khanum F, Kaul VK (2013) Antioxidant, anti-diabetic and renal protective properties of Stevia rebaudiana. J Diabetes Complicat 27(2):103–113. https://doi.org/10.1016/j.jdiacomp.2012.10.001
Simaei M, Khafari Nejad RA, Bernard F (2012) Exogenous application of salycilic acidand nitric oxide on the ionic contents and enzymatic activities in NaCl-stressed soybean plants. Am J Plant Sci 3:1495–1503
Soliman WS, El-Shaieny AAH (2014) Effect of saline water on germination and early growth stage of five Apiaceae species. Afr J Agric Res 9:713–719
Souana K, Taïbi K, Abderrahim LA, Amirat M, Achir M, Boussaid M, Mulet JM (2020) Salt-tolerance in Vicia faba L. is mitigated by the capacity of salicylic acid to improve photosynthesis and antioxidant response. Sci Hortic 273:109641
Srivastava M, Shahi S (2018) Effect of salinity on morpho-physiological aspects, antioxidant enzymatic studies and yield attributes in wheat genotypes. Plant Physiol Rep 23(2):385–392. https://doi.org/10.1007/s40502-018-0375-7
Syeed S, Anjum NA, Nazar R, Iqbal N, Masood A, Khan NA (2011) Salicylic acid-mediated changes in photosynthesis, nutrients content and antioxidant metabolism in two mustard (Brassica juncea L.) cultivars differing in salt tolerance. Acta Physiol Plant 33(3):877–886. https://doi.org/10.1007/s11738-010-0614-7
Tahjib-Ul-Arif M, Siddiqui MN, Sohag AAM, Sakil MA, Rahman MM, Polash MAS, Tran LSP (2018) Salicylic acid-mediated enhancement of photosynthesis attributes and antioxidant capacity contributes to yield improvement of maize plants under salt stress. J Plant Growth Regul 37(4):1318–1330. https://doi.org/10.1007/s00344-018-9867-y
Turner NC, Begg JE (1981) Plant-water relations and adaptation to stress. Plant Soil 58(1–3):97–131. https://doi.org/10.1007/BF02180051
Valentovic P, Luxova M, Kolarovic L, Gasparikova O (2006) Effect of osmotic stress on compatible solutes content, membrane stability and water relations in two maize cultivars. Plant Soil Environ 52(4):184
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants: protective role of exogenous polyamines. Plant Sci 151(1):59–66. https://doi.org/10.1016/S0168-9452(99)00197-1
Vighi IL, Benitez LC, Amaral MN, Moraes GP, Auler PA, Rodrigues GS, Braga EJB (2017) Functional characterization of the antioxidant enzymes in rice plants exposed to salinity stress. Biol Plant 61(3):540–550. https://doi.org/10.1007/s10535-017-0727-6
Yin D, Chen S, Chen F, Guan Z, Fang W (2009) Morphological and physiological responses of two chrysanthemum cultivars differing in their tolerance to waterlogging. Environ Exp Bot 67(1):87–93. https://doi.org/10.1016/j.envexpbot.2009.06.006
Zeng J, Chen A, Li D, Yi B, Wu W (2013) Effects of salt stress on the growth, physiological responses, and glycoside contents of Stevia rebaudiana Bertoni. J Agric Food Chem 61(24):5720–5726. https://doi.org/10.1021/jf401237x
Zhang T, Hu Y, Zhang K, Tian C, Guo J (2018) Arbuscular mycorrhizal fungi improve plant growth of Ricinus communis by altering photosynthetic properties and increasing pigments under drought and salt stress. Ind Crops Prod 117:13–19. https://doi.org/10.1016/j.indcrop.2018.02.087
Zhao HJ, Zhao XJ, Ma PF, Wang YX, Hu WW, Li LH, Zhao YD (2011) Effects of salicylic acid on protein kinase activity and chloroplast D1 protein degradation in wheat leaves subjected to heat and high light stress. Acta Ecol Sin 31(5):259–263. https://doi.org/10.1016/j.chnaes.2011.06.006
Zhu JK (2001) Plant salt tolerance. Trends Plant Sci 6(2):66–71. https://doi.org/10.1016/S1360-1385(00)01838-0
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
I. Janah, A. Elhasnaoui, O. Issa Ali, K. Lamnai, S. Aissam and K. Loutfi declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Janah, I., Elhasnaoui, A., Issa Ali, O. et al. Physiochemical Responses of Stevia rebaudiana Bertoni Subjected to Sodium Chloride (NaCl) Salinity and Exogenous Salicylic Acid Application. Gesunde Pflanzen 73, 509–520 (2021). https://doi.org/10.1007/s10343-021-00570-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10343-021-00570-6