Abstract
Wood-decaying fungi are essential for the functioning of forest ecosystems. They provide habitat for many other organisms and enable the regeneration of forests throughout the world. Since wood decomposition is a decisive process in nutrient recycling, soil formation and the carbon budget of forest ecosystems, it is receiving increasing attention from forest ecologists, pathologists and managers. Research has focussed on the factors driving the species-richness of wood-decomposing organisms and is moving on to analyse the effects of this species-richness on ecosystem functioning. Coarse woody debris (CWD) and its associated wood-decaying organisms have been drastically reduced in abundance and diversity by forestry and so these features often have potential as conservation indicators. Protective measures at a landscape level are needed for threatened wood-inhabiting fungi. These include restricting salvage operations in windthrow stands, actively encouraging the accumulation of deadwood in forests, and facilitating decay in standing trees by inoculating them with fungi. Here, we aim to collect and summarize recently produced work on deadwood ecology, pointing out research gaps and perspectives.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Deadwoodology, the ecology of deadwood (Grove 2002), is a thriving research field, with wood-decaying fungi playing a major role in it. Wood-decaying fungi are excellent ecosystem engineers, because they directly modulate the availability of resources other than themselves for several other functional groups (Harley 1971; Jones et al. 1994; Krajick 2001; Moore et al. 2004). The fundamental ecological significance of deadwood decomposition in forests has been highlighted in several reviews and conclusions for silviculture have been drawn repeatedly (Table 1). Essentially, modern forestry needs to retain appropriate levels of deadwood in managed forests, ideally in all its forms and density levels, in order to cover the full spectrum of habitat conditions (Samuelsson et al. 1994; MacNally et al. 2001; Berg et al. 2002; Vasiliauskas et al. 2004; Christensen et al. 2005) for the sake of dependent organisms and in order to achieve sustainability of timber production.
Much information on the process of wood-decay in forest ecosystems and on the organisms associated with it is currently scattered in individual papers. Here, focusing mainly on wood-decaying fungi (rather than saproxylic arthropods, molluscs, birds and other vertebrates dependent on deadwood) and on boreal and temperate forests (where most published research on this topic has been carried out), we aim to bring together recently published knowledge and to identify gaps in research dealing with this important part of biodiversity. The review is organized as follows: in the ‘For a functional ecology of decay section’, we highlight the importance of deadwood for a range of organisms, and for the regeneration of forests (Deadwood as Noah’s ark section), we discuss the issue of whether deadwood size or other related factors drive the species-richness of wood-decaying fungi (What is driving the species-richness of wood-decaying fungi? section) and point out that the question of whether the species (and functional) diversity of wood-decaying organisms affects decomposition rates has been seldom addressed, but is relevant in the context of global climate change and carbon sequestration (Species-richness of wood-decaying fungi and ecosystem functioning section). In the ‘Conservation issues in deadwoodology section’, we show that conservation biologists are increasingly focussing on wood-decaying fungi as general indicators of forest ecosystem threat (How threatened are wood-decaying fungi? section). We then draw attention to studies applying the single large or several small? (SLOSS) debate to the conservation of wood-decaying fungi (And how well are we protecting them? section), addressing the consequences of regional habitat fragmentation for the metapopulation persistence of wood-decaying fungi (Landscape perspectives for wood-decaying fungi section) and studying this aspect of biodiversity in tropical forests (Biogeography of CWD and of wood-decaying fungi section). We then attempt a comparison of the amount of deadwood in extratropical versus tropical forests. In the ‘Deadwood management section’, we discuss opportunities provided by various kinds of disturbance (tree pathogens, windthrow and fire) in order to increase the amount of deadwood in managed forests (Creative ideas for a permanent flow of deadwood needed section), we review studies assessing whether management guidelines aiming to improve the presence of deadwood in forests are having success (Are deadwood guidelines having any noticeable effect? section), and identify active morticulture as a new method to encourage wood-decaying organisms in forests where their presence has been unduly suppressed (Active morticulture section). The review is concluded by a recapitulation of some outstanding research questions.
For a functional ecology of decay
Deadwood as Noah’s ark
Standing and fallen decaying trees are species-rich environments, and deadwood has been recognized as providing resources for a variety of living organisms (Table 2). Wood-decay is fundamental to the formation of ecological niches (Odling-Smee et al. 2003), the wide variety of which reflects the potential decay pathways and ecological successions, together with the variety of the starting material. This includes standing deadwood (snags), fallen logs, living trees with heartrot and dying branches. In many cases, species and (meta)populations dependent on deadwood require a particular amount of coarse woody debris (CWD) at a certain stage of decay, thus demonstrating that deadwood niches should be defined quantitatively as well as qualitatively. In Europe an example is the flagship beetle species of tree hollows Osmoderma eremita (Ranius 2002). Niche opportunities for wood-decaying fungi are also shaped by interspecific interactions between different species (Coates and Rayner 1985a, b, c; Boddy 2000) as well as by intraspecific interactions (including nuclear reassortment between conspecific heterokaryons, see Johanneson and Stenlid 2004). Another layer of niche-definition comes from growing evidence that fungal species are often complexes of previously undifferentiated taxonomic units which specialize in different ecological conditions [e.g. Armillaria mellea s.l (e.g. Guillaumin et al. 1993), Heterobasidion annosum s.l. (e.g. Niemelä and Korhonen 1998), Laetiporus spp. (Rogers et al. 1999), Phellinus spp. s.l. (Fischer and Binder 2004) and Phialocephala fortinii s.l. (Grünig et al. 2004)].
In many regions of the world, a list of taxa dependent on decaying wood is equivalent to a coarse inventory of threatened species (see How threatened are wood-decaying fungi? section). The amount of evidence testifying to the ecological utility of CWD accumulated in terrestrial ecosystems is staggering, and an equally fundamental structural and functional role is played in aquatic freshwater (Gurnell et al. 1995; Krajick 2001; Gurnell et al. 2002) and saline (e.g. Robertson 1991; Gilbert and Sousa 2002; Parrent et al. 2004) ecosystems. Indeed, Noah’s ark was made of nothing else but floating deadwood.
Moreover, the presence of dead and decaying wood is a functional requirement not only for animal wildlife, but also for trees. In many cases, the regeneration of a forest only happens when fallen logs decay in situ. Decaying logs create differences in substrate; in turn, different micro-sites allow species with different niche requirements to coexist, thus influencing future stand composition. There are reports of a protective effect of CWD against pathogens of seeds (in Picea engelmannii and Abies lasiocarpa, Zhong and van der Kamp 1999) and of seedlings; in Tsuga canadensis (O’Hanlon-Manners and Kotanen 2004), and in Picea jezoensis (Sakamoto and Miyamoto 2005). The enhanced survival of seedlings of at least some tree species on decaying deadwood in a forest is very widely reported (see also Harmon et al. 1986). Reports come from forests in Japan, Taiwan, Tasmania, Hawaii, Argentina, Chile, pastures in Costa Rica, rainforests in Dominica, forests across USA and Canada, in Sweden, Finland, Poland and the European Alps (Table 3). The mechanisms through which the presence of deadwood facilitates seedling survival include moisture retention, mineral recycling, provision of mycorrhizal fungi, biological control of soil-borne pathogens and, in the case of branchy fallen material, protection against browsing.
What is driving the species-richness of wood-decaying fungi?
Even though wood-inhabiting fungi are important for a whole web of other organisms, they are themselves part of the biodiversity of a forest ecosystem. Strictly speaking, measures aimed to conserve wood-decaying fungi can thus be already justified merely for the sake of these organisms. The question arises as to which factors have an influence on their species-richness.
The species-richness of wood-decaying fungi tends to increase with the amount of substrate (e.g. Sippola and Renvall 1999; Allen et al. 2000; Humphrey et al. 2000; Edman et al. 2004b; Penttilä et al. 2004; Berglund and Jonsson 2005; Heilmann-Clausen and Christensen 2005; Schmit 2005; Sippola et al. 2005; Similä et al. 2006; Ódor et al. 2006). On the basis of the species-area relationship, a 90% reduction in deadwood substrate could be followed by the extinction of 50% of wood-inhabiting species (Siitonen 2001). A positive relationship also exists between CWD size and the number of fruiting fungal species (e.g. Bader et al. 1995; Renvall 1995; Lindblad 1997; Lindhe et al. 2004). But Heilmann-Clausen and Christensen (2004) show in near-natural beech stands in Denmark that, although the number of wood-inhabiting fungal species increases with CWD size (this is expected due to the species-area curve: a sample of a larger volume is likely to contain more species), the number of species per unit of volume decreases.
The occurrence of relatively few fungal species in large CWD units can be explained by the combined effects of a number of factors. The smaller the CWD size, the larger the surface per volume, which implies more space for fungal sporocarps. In order to sample the same CWD volume, the smaller its size, the greater the number of items, and thus the higher the individual cases of fungal infection (e.g. Takahashi and Kagaya 2005). The issue is similar to the collector curve: a sample of more individuals is likely to contain more species. Another reason is that a large item of CWD may allow more room for large fungal individuals to develop, rather than necessarily providing more space for a wider range of species. Conversely, there are some basidiomycetes that may require wood of a certain size before fruiting, although there is evidence from Bavaria that many species of the Corticiaceae and Polyporaceae produce fruit bodies even on rather thin woody substrates (Hahn and Blaschke 2005) and from Switzerland that fine and very fine woody debris, even in intensively managed forests, often provides important refuges for many wood-inhabiting fungi (Küffer and Senn-Irlet 2005a).
Relationships between CWD size and the number of fungal species might to some extent be dependent on the age of the part of the tree concerned, as size is partly related to age (but see Heilmann-Clausen and Christensen 2005). Age involves factors such as the presence of heartwood and the initiation of heartrot while the wood was still standing. Furthermore, the size of the units can influence moisture content as well as temperature, and these factors may affect which species are able to colonize the deadwood (Vasiliauskas et al. 2005). An additional point is that larger logs take longer to decay and thus allow more time for colonization of different species of fungi (Ódor et al. 2006). Hence, from an applied point of view it is important to keep in mind that a wide range of CWD, representing differing sizes and stages of decay, is needed in order to support species of varying requirements (Niemelä et al. 1995; Høiland and Bendiksen 1996; Kruys and Jonsson 1999; Nordén et al. 2004a; Stokland and Kauserud 2004; Selonen et al. 2005; Junninen et al. 2006; Tikkanen et al. 2006). Analogously, both high and low CWD turnover regimes are needed to conserve populations with, respectively, small and large body size, given that size is inversely related to the frequency of disturbances in the ecosystem; e.g. as shown for lichens (Hestmark 1997). A problem here is that some fungal species may be present endophytically and cause decay only after tree death (Boddy 1994; Baum et al. 2003). Some of these fungi occur as mycelia in various tree species, but form fruit bodies only on particular hosts, e.g. Xylariaceae (Whalley 1996).
Species-richness of wood-decaying fungi and ecosystem functioning
Within an ecological context, the term ‘functioning’ has many connotations (Jax 2005) and we focus here on the functional relationships between wood decomposition and species-richness of wood-inhabiting fungi only. It is mainly through fungal activity that organic matter is mineralized in forest ecosystems (Rayner and Boddy 1988). The particular fungal species which colonize functional and dysfunctional wood of veteran trees are also a frequent determinant of the age attained by these trees (Lonsdale 2004). Deadwoodologists have thus investigated the species-richness of wood-decaying fungi at different stages of decomposition (e.g. Lumley et al. 2001; papers reviewed in Heilmann-Clausen and Christensen 2003), but the effect of species-richness on decomposition rates has seldom been addressed (Boddy et al. 1989; Boddy 2000; Heilmann-Clausen and Boddy 2005; DeLong et al. 2005). An exception is a 13-year study of the decomposition of logs of Cyrilla racemiflora in forests of Puerto Rico, which provides evidence that high rates of wood decay are correlated with a high diversity of species and of functional groups of wood-inhabiting organisms (Torres and González 2005). Any influences of a more complex wood-colonizing community on decomposition rates are likely to be concurrent with the effect of seasonal variations in temperature and moisture availability (Progar et al. 2000; see also Toljander et al. 2006).
Moving from species-richness to functional diversity of the decaying community (Heilmann-Clausen 2001; Marcot 2002; Urcelay and Robledo 2004), the question arises of whether this diversity is a factor in determining the different rates of decomposition of CWD of different decay classes and tree species (Mackensen et al. 2003; Yatskov et al. 2003). Answering this question is important because the action of deadwood decomposers and consequent durability of CWD are a source of uncertainty in the role of terrestrial ecosystems as global carbon sinks (Krankina and Harmon 1995; Yin 1999; Chambers et al. 2000; Liski et al. 2003; Mackensen et al. 2003; Yatskov et al. 2003; Hood et al. 2004; Laiho and Prescott 2004; Edman et al. 2006; Liu et al. 2006). On the associated and equally relevant function of wood-decaying fungi as recyclers of nutrients, we refer to the review of Laiho and Prescott (2004) for northern coniferous forests. Similar syntheses are needed for other biomes.
Conservation issues in deadwoodology
How threatened are wood-decaying fungi?
In view of the several ecological functions of wood-decaying fungi, conservation biologists are increasingly focussing on these organisms and on the degree of threat to them (e.g. Molina et al. 2001; Parmasto 2001; Buchanan and May 2003). Thus, for instance, the proportion of fungal species dependent on deadwood is estimated to be 20–25% in Finland (Siitonen 2001). Likewise, 40% of the threatened species in Finland are assumed to be dependent on old-growth forest (Penttilä et al. 2004). In Sweden, 25% of all threatened species are thought to occur mostly in old living trees and fallen logs (Berg et al. 1994), 20% of the 670 native Aphyllophorales are endangered (Edman and Jonsson 2001) and nearly 40% of the 2120 red-listed organisms related to forest and woodland habitats are saproxylic (one fourth of which are fungi; Lindhe et al. 2004). In Bavaria, 25% of wood-decaying fungi species are assessed as threatened (Albrecht 1992). These proportions mean every time something slightly different, but remain invariably substantial.
One thing is clear: both deadwood (e.g. Angelstam et al. 2003; Sippola et al. 2004) and wood-decaying fungi (e.g. Bader et al. 1995; Norstedt et al. 2001) are first-rate indicators for conservation value of forest patches. The question of whether the former or the latter is a better indicator should not lead to a dead-end. We need to preserve both decaying wood and wood-decaying organisms and happily the presence of the two is often positively correlated. Nonetheless, there are hints from boreal Fennoscandia that the use as indicators of corticoids and polypores can provide different management guidelines at different scales (Berglund et al. 2005). This is not surprising, given their different preferences for woody substrate. Moreover, there are differences between rare and common wood-decaying species in their value as indicator species for key woodland habitats in Finland (Sippola et al. 2005). Wood-disk traps colonized by homokaryotic mycelia provide here the way to detect low rates of spore deposition, both in the study of rare species and of long-distance dispersal (Edman and Gustafsson 2003; see also Landscape perspectives for wood-decaying fungi section).
Unfortunately, one problem is also that nature reserve selection exercises based on the presence of different kinds of wood-decaying organisms can give different results (e.g. Virolainen et al. 2000; Nordén and Appelqvist 2001; Saetersdal et al. 2005; Juutinen et al. 2006). The way out is again a choice of indicator species from several taxonomic groups, whenever possible not merely involving a single functional group, but reflecting the requirements of different organisms (Jonsson and Jonsell 1999; Berglund and Jonsson 2001; Roberge and Angelstam 2004; Similä et al. 2006). This use of the precautionary principle makes sense also in the light of the still rarely scrutinized relationship between the number of wood-decaying species and tree species-richness in different forest types (but see Hattori 2005; Kulhánková et al. 2006). No significant correlation is found in Finland, probably because of the low number of tree species in boreal forests (Sippola et al. 2004). However, a positive correlation is reported from a nemoral forest in Denmark (Heilmann-Clausen et al. 2005). Moreover, a meta-analysis at a global scale shows a positive relationship between the overall species-richness of macrofungi and that of trees (Schmit et al. 2005; see also Biogeography of CWD and of wood-decaying fungi section). This implies that the protection of the latter can often help to safeguard the former (e.g. Andersson and Östlund 2004). But more research is needed to address whether this is true more specifically for wood-decaying fungi and other ecological groups (e.g. mycorrhizal fungi, litter-decaying organisms and successors to saproxylic organisms or fungi associated with charred deadwood). This is currently unclear because analyses of the community structure or rate of decomposition by fungi in CWD have been predominantly based on single tree species.
And how well are we protecting them?
For a given amount of money invested in conservation, the effectiveness of protective measures can be very different depending on the number, size and location of protected areas. Hence, the SLOSS debate is being kindled in the conservation of wood-decaying fungi. Götmark and Thorell (2003) find a negative density-area relationship between the number of large trees, snags and logs per unit area and the reserve size in southern Sweden. This result would imply that smaller protection areas could be more efficient, since they seem to contain a higher density of deadwood. Similarly, Berglund and Jonsson (2003) find that rare species are disproportionately well-represented in small patches of high habitat quality in Sweden, but on the other hand they also find, from species-accumulation curves, that large old-growth forest patches are more species-rich in polypore species than combinations of small patches of an equal area. The importance of small habitat patches is confirmed by Rolstad et al. (2004), who find in southeastern Norway that Norway spruce (Picea abies) CWD in concentrated patches of <1 ha supports wood-decaying fungi just as well as CWD spread out on a larger area. This is not confirmed by Gjerde et al. (2004), who show that in the 5% most species-rich 1-ha woodland patches across Norway, only 20–25% of red-listed species (not only including polypores, but also macrolichens, bryophytes and vascular plants) are represented. In order to protect the other 75–80% of endangered species, mature forests need thus to be preserved in more extensive patches. Also the findings by Penttilä et al. (2004) in Finland suggest that the conservation of threatened polypore species may be better served by enlarging the size of patches of suitable habitat (old-growth forests) than by attempting to increase the overall quality of managed forests.
More generally, how are we to assess the effectiveness of protected areas for the conservation of fungal wood-decaying species? Kruys et al. (1999) state that, given that <1% of the forest land in the non-mountainous regions of Sweden is protected, in view of inevitable local extinctions over time, protected areas can at best function as temporary refuges and sources of dispersal for threatened wood-inhabiting species. They thus argue that the whole landscape should be managed taking into account deadwood retention objectives (see also Ranius and Kindvall 2004). Currently, however, where sanitation measures are the rule, the average deadwood volumes in managed forests are so low that many species dependent on deadwood are confined to protected areas (Rouvinen and Kouki 2002, see also Ranius and Fahrig 2006). Complementarity analyses (e.g. Virolainen et al. 2000; Juutinen and Mönkkönen 2004; Mayer et al. 2006) are needed so as to select a network of protected forests containing enough naturally occurring deadwood that would be needed to meet conservation targets within a given country or supranational region. If such an approach is ignored by politicians and foresters, the science will become meaningless. Even in Europe, the last temperate forest which is believed to retain some primeval areas, Bialowieza, Poland, is rapidly vanishing (Wesolowski 2005).
Landscape perspectives for wood-decaying fungi
There is an increasing recognition that the conservation biology of saproxylic organisms has to be addressed from the perspective of the landscape and not only of the forest stand (e.g. Gu et al. 2002; Berglund et al. 2005; Jonsson et al. 2005; Vanderpoorten et al. 2005; Ekbom et al. 2006; Junninen et al. 2006). This approach can benefit from regional gap analyses, such as Angelstam et al. (2003) present for a suite of birds, a beetle and for lichens dependent on a humid micro-climate. They show that patches of a certain size and connectivity are needed for the persistence of the target species; the inference is that the same is needed for wood-decaying fungi. Additionally, the quality of the landscape matrix should be taken into account, so as to improve the chances of success when implementing conservation measures for fungi dependent on decaying wood. In Scotland, Humphrey et al. (2000) show that the number of macrofungal species of conservation importance (including mainly mycorrhizal species and only a few wood-decaying species) in conifer plantations is higher if plantations are close to patches of natural forest. However, they add that the dispersal ecology of these species is poorly understood and that in many cases propagules may have been transported in association with the root system of planting stock. Along with Sverdrup-Thygeson and Lindemayer (2003), these authors argue that the matrix in which suitable habitat patches are embedded has to be understood not only spatially but also chronologically; i.e. in terms of the continuity of the presence of deadwood. In Sweden, where spatial and temporal patterns of human activity can be related to the present-day local and regional presence distribution of old-growth forest, this activity has evidently influenced the abundance of viable spores of two endangered wood-decaying organisms, Fomitopsis rosea and Phlebia centrifuga (Edman et al. 2004a, b). Similarly, the importance of chronological continuity for the survival of species of saproxylic invertebrates has been demonstrated in the UK (Alexander 1998).
The above data suggest that the negative effects of lack of deadwood are operating not only through loss of habitat but also through fragmentation (Kouki et al. 2001; Berglund and Jonsson 2003, 2005; Pharo et al. 2004). This is confirmed by modelling, experimental and empirical work. Strange et al. (2004) apply a variety of site-selection strategies to a Danish database of occurrences of wood-decaying species, subject to a budget constraint. They show that the inclusion of spatial connectivity in models would markedly affect the selection of reserves in an intended network. Edman et al. (2004c) experimentally placed freshly cut logs in Swedish plots containing varying amounts of CWD and thus differing in their pool of wood-decaying species. Despite the potentially confounding transmission of species from stands where logs were cut, colonization by fungal species is found to be higher in sites rich in deadwood. Siitonen et al. (2005) find less old-growth indicator fungi at the edge of forests in comparison with interiors within a fragmented landscape in eastern Finland, but this edge effect is not simple, since it varies with the age of the forest because of interacting factors.
All these studies imply that, although fungal species can maintain viable populations by dispersing within old-growth stands (e.g. Komonen 2005; see also Kauserud et al. 2005), their dispersal can be disrupted by forest fragmentation. Therefore, conservation measures are inadequate if they aim merely to increase the amount of CWD in managed forests without connecting forest patches (see also Penttilä et al. 2006). Although the relationships between forest history, habitat fragmentation and dispersal of wood-decay fungi are still rather poorly understood, especially for some species (e.g. F. rosea, Högberg and Stenlid 1999; Kauserud and Schumacher 2003a), it is becoming increasingly clear that fragmentation can be a problem, particularly for wood-decaying species which show limited gene flow between regions (e.g. Datronia caperata, Parrent et al. 2004). On the contrary, the detrimental effects of fragmentation may be more easily overcome by species of wood-inhabiting basidiomycetes whose spores remain viable during long-distance dispersal (e.g. Trichaptum abietinum, Kauserud and Schumacher 2003b; or Fomitopsis pinicola, Högberg et al. 1999), although their spore load at long distances is inevitably much smaller than that of locally established species (Rishbeth 1959; de Jong et al. 1990; Nordén 1997; Nordén and Larsson 2000; Hallenberg and Küffer 2001).
Biogeography of CWD and of wood-decaying fungi
Traditionally, research on wood-decaying fungi has concentrated mainly on temperate and boreal ecosystems in developed countries (Harmon et al. 1986). Yet, the same questions of conservation and functional ecology are being increasingly investigated in temperate and boreal regions of developing countries (e.g. Mukhin and Kotiranta 2001; Greslebin and Rajchenberg 2003; Dai et al. 2004; Afyon et al. 2005) and in warm mixed and tropical forests (e.g. Lodge and Cantrell 1995; Rodriguez et al. 1995; Ryvarden 1998; Goes-Neto et al. 2000; Lindblad 2001; Ortega and Navarro 2004; Groposo and Loguercio-Leite 2005; Torres and González 2005). Is the level of threat of wood-decaying fungi in the tropics similar to that in boreal regions? The answer might be determined by the greater rarity of fungal tropical species (Gilbert et al. 2002) and their lower host specialization (Lindblad 2000; Gilbert and Sousa 2002), both possibly influenced by higher tree species diversity (Ferrer and Gilbert 2003). However, the assessment of rarity of wood-decaying tropical species may be biased by a relative lack of studies.
Not only the status of wood-decaying fungi but also that of deadwood has gone largely unreported from tropical regions (Delaney et al. 1998; Grove 2001). But studies are beginning to fill this gap. Are levels of standing and fallen CWD lower in temperate and boreal old-growth forests than in tropical rainforests? This would be expected on the basis of high productivity in the tropics, given that there is a positive relationship between productivity and deadwood volumes within temperate and boreal forests (Harris 1999; Spetich et al. 1999; Feller 2003). On the other hand, the rate of decomposition is also higher at the higher mean temperatures of tropical forests and is often accelerated by the activities of termites. From a study of available data, we have not been able to find evidence that CWD is more abundant in the tropics. The volume of CWD (average 118 m3 ha−1, SD 40 m3 ha−1) reported from nine recent rainforest surveys we were able to find [multiple plots in Borneo and plots in Ecuador (Gale 2000), tropical Australia (Grove 2001), Costa Rica (Clark et al. 2002) and Brazil (Keller et al. 2004; Rice et al. 2004)] does not differ significantly (ANOVA: F 1,17 = 1.4; P = 0.25), from that of nine selected old-growth or mature forests at higher latitudes [average 94 m3 ha−1, SD 43 m3 ha−1; plots in Canada, Alberta (Lee et al. 1997) and Newfoundland (Sturtevant et al. 1997), USA, New York (McGee et al. 1999), Sweden (Jonsson 2000), multiple plots in Canada, Ontario (Pedlar et al. 2002) and in Finland (Rouvinen et al. 2002)]. Similarly high values are reported also from temperate rainforests of Chile (Carmona et al. 2002), from unmanaged forests of southern New South Wales (MacNally et al. 2002b) and from Fagus forests in different temperate countries (Piovesan et al. 2005).
A higher species-richness of wood-decaying organisms at tropical latitudes might be extrapolated from the findings (1) of positive site productivity–diversity relationships for polypores in six forests over a latitudinal extent of 1,350 km in Norway (Gjerde et al. 2005), (2) of a higher species-richness of wood-inhabiting aphyllophoroid basidiomycetes in fallen woody material at lower altitudes in Swiss forests (Küffer and Senn-Irlet 2005b) and (3) of a positive species–energy availability (expressed in terms both of log volume and density) relationship for macrofungal wood-inhabiting species in Puerto Rico (Schmit 2005). Assuming such a pattern, a mechanism enabling more fungal species to coexist in tropical deadwood could be a finer partitioning of resources. This would also assume a comparable level of CWD volumes in tropical forests and extra-tropical ones, which may be expected given that not only deadwood production but also decay rates increase with increasing forest productivity (Stephenson and van Mantgem 2005; Storaunet et al. 2005). This is also confirmed by analyses of Pregitzer and Euskirchen (2004), and of Woldendorp and Keenan (2005). However, any lack of statistical significance in the differences in deadwood volumes between biogeographical regions may be the consequence of confounding factors (e.g. survey methodology, plot area, topography and recent disturbance).
Deadwood management
Creative ideas for a permanent flow of deadwood needed
At the risk of circularity, one argument for the importance of gap perturbations and stand-replacing events due to supposed forest ‘pests’ and tree pathogens is that their by-product is a continuous supply of deadwood in forests (Harmon et al. 1986; McGee 2000; Rouvinen et al. 2002; Müller-Using and Bartsch 2003; Storaunet and Rolstad 2004; McPherson et al. 2005). This unrelenting deadwood flow is part of the ecological memory of forests (Peterson 2002) and should be emulated by forest managers. The next step for applied ecosystem management is indeed to allow an acceptable amount of disease in a particular forested landscape, gauging it on the baseline provided by an understanding of the conditions that predated modern forestry (e.g. Edmonds et al. 2000; Manion 2003; Crow and Perera 2004). The view that tree decay and diseases are necessarily deleterious is rather anthropocentric (Cooke and Rayner 1984). The incidence of damage has, however, sometimes been increased by human activity, which has been a factor in the chronological changes in the incidence and severity of forest disease. For instance, in many managed forests, the occurrence of H. annosum s.l. has long been exacerbated by an unnatural abundance of cut stumps, which act as a selective substrate for this and similar root rot fungi (Redfern and Stenlid 1998). Stumps probably also favour Armillaria spp. (Prospero et al. 2003). The potential use of non-pathogenic saprotrophic wood-decaying fungi as antagonistic factors against H. annosum s.l. is reviewed by Holdenrieder and Greig (1998) and Korhonen and Holdenrieder (2005).
Windthrow gaps, if left to their natural development, are nowadays potential deadwood islands in a CWD-deprived matrix (Bouget and Duelli 2004). They provide a great opportunity to increase deadwood abundance in managed forests faster than can be achieved applying biodiversity-oriented silvicultural practices (Ranius and Kindvall 2004). In central European forests, this opportunity was arguably missed with the overzealous clearing of stands overthrown by Hurricane Lothar at the end of 1999. In Switzerland, as of November 2004, 80% of the ca. 14 million m3 of wood-windthrow had been cleared (BUWAL 2004). It has been shown that for Hurricane Hugo in the United States the economic surplus generated by salvage averaged $6.25 million for each additional percentage in the volume of salvaged timber up to the observed salvage rate (16%) (Prestemon and Holmes 2004). But when salvage rates are much higher, it may be not only ecologically but also economically sensible to retain windthrow trees in the forests (Baur et al. 2003).
Are deadwood guidelines having any noticeable effect?
With the possible exception of tropical forests (Keller et al. 2004), managed forests generally show CWD levels significantly lower than those of unmanaged forests (e.g. Guby and Dobbertin 1996; Green and Peterken 1997; Fridman and Walheim 2000; Jenkins et al. 2004; Gibb et al. 2005; Köster et al. 2005; Marage and Lemperiere 2005; Storaunet et al. 2005; Ekbom et al. 2006), except that the differences may be statistically non-significant when logging is selective (Rouvinen et al. 2002). This implies that, in many cases, the naturalness of a forest can be assessed from the amount of deadwood present (Rouvinen et al. 2005). Issues surrounding deadwood are therefore becoming relevant also for forest certification, with best practice guidelines often encouraging the retention of a higher volume of deadwood in managed forests.
But are these guidelines having real consequences; i.e. have levels of CWD been increasing over the last years in managed forests, as is reported in protected forests: e.g. an old-growth oak forest in IN, USA (Spetich and Parker 1998), and sub-alpine pine forests in the European Alps (Risch et al. 2003)? In selected Appalachian water catchments, CWD is more abundant in areas harvested 18 years earlier but managed under best practice (including retention of 15 m unharvested zones along streams) than in unharvested control plots. Also, the various decay classes of CWD are more evenly represented in plots harvested by best management practices than in conventionally harvested plots, in which CWD consists mostly of slash left behind after harvest (McClure et al. 2004). Similarly, the application of best management practices is reported to have led to an increase in snag density in northern Arizona mixed-conifer forests between 1997 and 2002 (Ganey and Vojta 2005). The latter study does not, however, show the same effect in ponderosa forests and there have even been suggestions that intensive forest management can unintentionally produce more snags thanks to an increased live tree density following clear felling (Ferguson and Archibald 2002). Due to a relative lack of empirical data on the subject, simulations have been used to predict the amount of CWD produced from long-term application of deadwood-friendly management practices (Ranius et al. 2003). Modelling work in Sweden suggests that adopting certification requirements will double the amount of CWD in forests, but that this will take more than one century (Ranius and Kindvall 2004).
Sweden is one of the countries at the forefront in this field. In that country, the Forest Stewardship Council requires that in certified forests representative fresh windfalls are retained (FSC 2005). The unconditional ban on cutting in the few remaining European old-growth stands (advocated by Bobiec et al. 2000) could thus be extended to a large-scale case-by-case presumption against clearing deadwood from stands following windthrow (see Creative ideas for a permanent flow of deadwood needed section). But such a ban may be superfluous in marginal regions, since extreme weather may act on the supply side not only positively (with increased output due to salvage operations) but also negatively (with decreased output due to more difficult access to forests, and also with lower quality of timber) (DeWalle et al. 2003). What is clear is that we need CWD management practices that achieve a sustainable protection of deadwood biodiversity, while minimizing economic drawbacks to the forestry sector (Schiegg 1998; Heilmann-Clausen and Christensen 2004; Ranius et al. 2005; Jonsson et al. 2006). In the long-term, CWD reduction in managed forests may end up in reducing site productivity and tree height (Debeljak 2006).
Because of conflicting interests, this is indeed a politically difficult topic. For instance, in fire-prone forest ecosystems there is the potential for a conflict between biodiversity enhancement as well as forest regeneration facilitation through deadwood retention and fire risk reduction through fuel management (e.g. Ucitel et al. 2003; Bury 2004; Sanchez-Flores and Yool 2004; Donato et al. 2006; Passovoy and Fulé 2006). In regions afflicted by storms, the numerous damaged trees in a windthrow area, although desirable for all the reasons discussed in this review, can become a source of infestation by bark beetles (Wichmann and Ravn 2001; Eriksson et al. 2005), with perceived risk rates of further infestation often significantly higher than actual ones (e.g. Eriksson et al. 2006; Flint 2006). The presence or absence of particular bark beetles can have major implications for a policy of retaining deadwood. For example, Ips typographus is not native to the UK, where there is thus much less obsession with sanitation than in many continental countries. This could change, however, if I. typographus were to become established in the UK (Evans 1997).
On the particular issue of bark beetles, the need is greater than ever, not only for further research, but also for more public relations work by forest managers informing people that moderate populations of bark beetle can in certain cases contribute to overall ecosystem functioning (see Bouget and Duelli 2004; Fayt et al. 2005). For instance, they can help to diversify forests which have become spruce monocultures. This can be considered desirable where these are outside their natural range and do not have a slope protection value (Wermelinger 2004). In the latter context, broken trees, snags and logs are beneficial inasmuch as they confer protection against avalanches and rock fall (e.g. Frey and Thee 2002; Kupferschmid et al. 2003; Schönenberger et al. 2005; Ammann 2006). The problem is that on steep slopes avalanches and gravity itself tend to make deadwood rarer than elsewhere. Hence, here rapid decay is not welcome, and models estimating time from tree death to the present time (Storaunet 2004) need to incorporate decay and mortality factors (e.g. Janisch and Harmon 2002; Rademacher and Winter 2003; Garber et al. 2005; Mäkinen et al. 2006) so as to build tools predicting how long a perceptible protective effect will last (e.g. Kupferschmid and Bugmann 2005; Ammann 2006; see also Russell et al. 2006).
Active morticulture
Steep protection forests aside, the creative catabolic transformation of CWD into biodiversity, energy, CO2 and nutrients (Swift 1977) should be facilitated by forest management in equally creative ways. Of course, the provision of CWD at an adequate spatiotemporal scale needs to be balanced against timber production (and fire risk in dry climate; Are deadwood guidelines having any noticeable effect? section). However, given that clean forestry has been a long-standing operating paradigm, there is scope for some exaggeration on the other side now. If morticulture as a new paradigm means connecting dead and live trees (Harmon 2001), then, along with the traditional planting of a tree for every new-born child, a dead tree per hectare of logged forest should be retained as well (Harris 2001). Similarly, salvage logging in burnt forests should retain large-diameter snags in clumps so as to preserve habitat for cavity-excavating species (Chambers and Mast 2005).
Understanding the interconnections between saproxylic organisms (e.g. Torgersen and Bull 1995; Bull and Wales 2001), even though praiseworthy, is not enough and needs to be complemented with the active creation of dead trees by cutting tops and girdling (e.g. Aulen 1991; Hallett et al. 2001; Cavalli and Mason 2003; Penttilä et al. 2004; Lilja et al. 2005; Ranius et al. 2005) or by inoculating standing living trees with wood-decaying fungi, where this has been shown usefully to hasten the development of decay-based habitat (e.g. Baker et al. 1996; Lewis 1998; Carey et al. 1999; Jack et al. 2003). There is also evidence that mechanical harvesters can effectively spread fungal inoculum across logs (Uzunović et al. 2004). It may, however, require time to obtain results and to convince people that these actions are not vandalism. In the Coast Range of Oregon, Filip et al. (2004) show that inoculation of Douglas-fir trees with Phellinus pini and Fomitopsis cajanderi creates internal decay within 5 years, but that a longer time is needed for the decay column to grow large enough to be used by cavity-nesting birds. In the Gila National forest, New Mexico, Parks et al. (1999) find that 20% of 102 ponderosa pines killed so as to reduce the spread of dwarf mistletoe (Arceuthobium spp.) contained woodpecker cavities 7–9 years after the treatment, which consisted either of basal burning or girdling.
Spreading wood-decaying fungi from tree to tree may be nothing unnatural, since woodpeckers themselves appear to contribute to the process (Farris et al. 2004). However, this kind of commendable hands-on research is hampered by the need to obtain results in a short period. In the dry sclerophyll forests of southeast Queensland, the average tree age for cavities to begin to appear is estimated at 186–230 years in five of six tree species [the sixth species needs longer; Wormington et al. (2003)]. It should thus be mandatory for long-term grants to be allocated in order to allow researchers to monitor the development of snags created with different methods and of decay in trees inoculated with different fungi until significant differences become evident (Brandeis et al. 2002).
Research gaps
The main conclusions of this review are the following:
-
(1).
Research on deadwood-dependent fungi shows that these organisms are a vital link in the functioning of forest ecosystems.
-
(2).
Forest management practices which threaten these fungi need to be modified in order to achieve sustainability of forest ecosystems.
-
(3).
A whole series of management options is available to increase the presence of deadwood and its dependent organisms in forests, balancing this requirement with the risk of fires and upsurges in bark beetle populations.
Notwithstanding the studies discussed in this and in other reviews (Table 1), wood-decaying fungi are still a neglected part of biodiversity relative to other taxa. Figure 1 shows that the proportion of studies of species-richness dealing with fungi in general (studies of wood-rotting fungi are in turn only a fraction of this proportion) is only a tiny slice of the whole cake currently analyzing biodiversity.
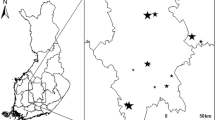
Proportion of papers dealing with species-richness of different taxa in a random sample of 100 papers per year retrieved with the keyword ‘species-richness’ in the UK version of Web of Science© (1991–2004; n = 1,400). Note: microbes do not include micro-fungi, and invertebrates other than insects do not include Protozoa (which were grouped together with microbes)
Given their functional importance in ecosystems (Schwarze et al. 2000; For a functional ecology of decay section), lignicolous fungi have been the object of many empirical studies. In general, however, much of the research on organisms dependent on decaying wood (Deadwood as Noah’s ark section), although substantial, has been descriptive and could have been more compelling if experimentally proving that (1) augmenting levels of deadwood is followed by an increased species abundance (MacNally et al. 2002a; Pretty and Dobson 2004; Patrick et al. 2006) or (2) that a depletion of CWD is followed by a reduction in abundance, as considered by McCay and Komoroski (2004). The latter authors point out that weak treatment effects may be due to CWD levels already being lower than normal in control plots. Research is needed to unravel the influence of various factors (deadwood size, diversity, age and decay class) not only on the species-richness of wood-decaying fungi (What is driving the species-richness of wood-decaying fungi? section), but also on their species-composition (e.g. Rubino and McCarthy 2003; Nordén et al. 2004b; Heilmann-Clausen and Christensen 2005; Küffer and Senn-Irlet 2005a, b). It would be interesting to establish (1) whether pioneer tree species tend, possibly for reasons of wood chemistry, to be colonized by a different functional group of wood-decaying species and (2) whether CWD from such trees is an exception to positive relationship between unit size and fungal species-richness (Küffer and Senn-Irlet 2000; Nordén and Paltto 2001; see also Hibbett and Donoghue 2001). As for ecosystem functioning (Species-richness of wood-decaying fungi and ecosystem functioning section), there is scope for addressing the interspecific interactions between the vast diversity of saprotrophs and the very few fungal pathogens inhabiting wood (which are potentially controlled by other saprotrophic organisms) and the therefore low risk of diseases posed by CWD accumulation (Hamdan et al. 2005; Woods et al. 2006). Similarly, we need data on potential influences on tree health by the presence of CWD in forests. What about deadwood dearth as a limitating factor for tree establishment at the treeline in different regions of the world? We would like to see more investigations of the role of the functional diversity of lignicolous fungi in the context of climate change and carbon sequestration (Treseder and Allen 2000), and there is much potential for forest canopy cranes to be used also for deadwood and mycological studies (Ishii and Kadotani 2006; Unterseher and Tal 2006).
As for outstanding conservation issues in deadwoodology (Conservation issues in deadwoodology section), in order to make a proper measurement of global trends in the status of biodiversity threat (How threatened are wood-decaying fungi? section), wood-decaying organisms should be included in red list indices. It would be interesting to know whether the level of threat to wood-decaying organisms in different countries is significantly affected by some other factor (e.g. forest cover or deadwood removal by humans). Are the statistics for deadwood availability and for the status of threatened species, as established in some countries, similar in different biogeographical regions? Is the conservation budget of countries a significant factor here? Similarly, we need to know more about the importance of edge effects in the SLOSS debate (And how well are we protecting them? section). For example, is the value of small remnants of old-growth habitats diminished by fragmentation (Aune et al. 2005)? There are contradicting results from studies evaluating the effect of habitat fragmentation on spore dispersal and viability of metapopulations of wood-decaying fungi. The issue would thus benefit from further investigation.
At the landscape level (Landscape perspectives for wood-decaying fungi section), there is the need for further integration of human impacts on wood-decaying fungi: e.g. environmental pollution (Stavishenko et al. 2002) or genetic impacts on the resident mycobiota by large-scale application of single genotypes (Vainio et al. 2001). Another neglected area of research is the availability of CWD in urban areas. At the biogeographical level (Biogeography of CWD and of wood-decaying fungi section), there is a whole series of outstanding research questions involving, e.g. latitudinal gradients in the species-richness of wood-decaying fungi, the influence of energy availability on large-scale patterns in mycodiversity and mainland versus island comparisons in the diversity and abundance of wood-decaying organisms (Huhndorf et al. 2004).
Regarding deadwood management (Deadwood management section), there is not only the necessity of a permanent flow of deadwood in forests (Creative ideas for a permanent flow of deadwood needed section), but of a constant monitoring of this process (Are deadwood guidelines having any noticeable effect? section). Hurricanes, ‘pest’ outbreaks, and forest fires can provide an increase of deadwood as well as many research opportunities to follow in situ the succession of wood-decaying organisms on freshly colonized substrates. We need more comparative research on how different cultures perceive the presence and abundance of deadwood in managed and natural forests (e.g. Kohsaka and Handoh 2006), as well as public relations work in relation to the ecological role of bark beetles as deadwood providers. Active morticulture is just one of the many fascinating research and management avenues in current deadwoodology.
References
Afyon A, Konuk M, Yagiz D, Helfer S (2005) A study of wood decaying macrofungi of the western Black Sea Region, Turkey. Mycotaxon 93:319–322
Albrecht L (1991) Die Bedeutung des toten Holzes im Wald. Forstwissenschaftliches Centralblatt 110:106–113
Albrecht L (1992) The importance of natural forest reserves for species protection in woodlands. Forstwissenschaftliches Centralblatt 111:214–224
Alexander KNA (1998) The links between forest history and biodiversity: the invertebrate fauna of ancient pasture woodland in the British Isles and its conservation. In: Kirby K, Watkins C (eds) The ecological history of European forests. CAB International, Wallingford, pp 73–80
Alkaslassy E (2005) Abundance of plethodontid salamanders in relation to coarse woody debris in a low elevation mixed forest of the western cascades. Northwest Sci 79:156–163
Allen RB, Buchanan PK, Clinton PW, Cone AJ (2000) Composition and diversity of fungi on decaying logs in a New Zealand temperate beech (Nothofagus) forest. Can J For Res 30:1025–1033
Ammann M (2006) Schutzwirkung abgestorbener Bäume gegen Naturgefahren. Dissertation ETH Zürich, CH, 189 pp http://e-collection.ethbib.ethz.ch/cgi-bin/show.pl?type=diss&nr=16638
Ammer U (1991) Konsequenzen aus den Ergebnissen der Totholzforschung für die forstliche Praxis. Forstwissenschaftliches Centralblatt 110:149–157
Andersson LI, Hytteborn H (1991) Bryophytes and decaying wood—a comparison between managed and natural forest. Holarctic Ecol 14:121–130
Andersson R, Östlund L (2004) Spatial patterns, density changes and implications on biodiversity for old trees in the boreal landscape of northern Sweden. Biol Conserv 118:443–453. doi:10.1016/j.biocon.2003.09.020
Angelstam PK, Butler R, Lazdinis M, Mikusinski G, Roberge JM (2003) Habitat thresholds for focal species at multiple scales and forest biodiversity conservation—dead wood as an example. Ann Zool Fenn 40:473–482
Aulen G (1991) Increasing insect abundance by killing deciduous trees—a method of improving the food situation for endangered woodpeckers. Holarctic Ecol 14:68–80
Aune K, Jonsson BG, Moen J (2005) Isolation and edge effects among woodland key habitats in Sweden: is forest policy promoting fragmentation? Biol Conserv 124:89–95. doi:10.1016/j.biocon.2005.01.015
Bader P, Jansson S, Jonsson BG (1995) Wood-inhabiting fungi and substratum decline in selectively logged boreal spruce forests. Biol Conserv 72:355–362. doi:10.1016/0006-3207(94)00029-P
Baker FA, Daniels SE, Parks CA (1996) Inoculating trees with wood decay fungi with rifle and shotgun. West J Appl For 11:13–15
Baum S, Sieber TN, Schwarze FWMR, Fink S (2003) Latent infections of Fomes fomentarius in the xylem of European beech (Fagus sylvatica). Mycol Prog 2:141–148. doi:10.1007/s11557-006-0052-5
Baur P, Bernath K, Holthausen N, Roschewitz A (2003) LOTHAR Ökonomische Auswirkungen des Sturms Lothar im Schweizer Wald, Teil I. Einkommens- und Vermögenswirkungen für die Waldwirtschaft und gesamtwirtschaftliche Beurteilung des Sturms. Umwelt-Materialien Nr. 157. Bundesamt für Umwelt, Wald und Landschaft, Bern
Berg Å, Ehnström B, Gustasson L, Hallingbäck T, Jonsell M, Weslien J (1994) Threatened plant, animal, and fungus species in Swedish forests: distribution and habitat associations. Conserv Biol 8:718–731. doi:10.1046/j.1523-1739.1994.08030718.x
Berg Å, Gardenfors U, Hallingback T, Noren M (2002) Habitat preferences of red-listed fungi and bryophytes in woodland key habitats in southern Sweden—analyses of data from a national survey. Biodivers Conserv 11:1479–1503. doi:10.1023/A:1016271823892
Berglund H, Jonsson BG (2001) Predictability of plant and fungal species-richness of old-growth boreal forest islands. J Veg Sci 12:857–866
Berglund H, Jonsson BG (2003) Nested plant and fungal communities; the importance of area and habitat quality in maximizing species capture in boreal old-growth forests. Biol Conserv 112:319–328. doi:10.1016/S0006-3207(02)00329-4
Berglund H, Jonsson BG (2005) Verifying an extinction debt among lichens and fungi in Northern Swedish boreal forests. Conserv Biol 19:338–348. doi:10.1111/j.1523-1739.2005.00550.x
Berglund H, Edman M, Ericson L (2005) Temporal variation of wood-fungi diversity in boreal old-growth forests: implications for monitoring. Ecol Appl 15:970–982
Bobiec A, van der Burgt H, Meijer K, Zuyderduyn C, Haga J, Vlaanderen B (2000) Rich deciduous forests in Bialowieza as a dynamic mosaic of developmental phases: premises for nature conservation and restoration management. For Ecol Manage 130:159–175. doi:10.1016/S0378-1127(99)00181-4
Boddy L (1994) Latent decay fungi—the hidden foe? Arboric J 18:113–135
Boddy L (2000) Interspecific combative interactions between wood-decaying basidiomycetes. FEMS Microbiol Ecol 31:185–194. doi:10.1111/j.1574-6941.2000.tb00683.x
Boddy L, Owens EM, Chapela IH (1989) Small-scale variation in decay-rate within logs one year after felling—effect of fungal community structure and moisture-content. FEMS Microbiol Ecol 62:173–184. doi:10.1111/j.1574-6968.1989.tb03691.x
Bouget C, Duelli P (2004) The effects of windthrow on forest insect communities: a literature review. Biol Conserv 118:281–299. doi:10.1016/j.biocon.2003.09.009
Bowman JC, Sleep D, Forbes GJ, Edwards M (2000) The association of small mammals with coarse woody debris at log and stand scales. For Ecol Manage 129:119–124. doi:10.1016/S0378-1127(99)00152-8
Brandeis TJ, Newton M, Filip GM, Cole EC (2002) Cavity-nester habitat development in artificially made Douglas-fir snags. J Wildl Manage 66:625–633
Brang P, Moran J, Puttonen P, Vyse A (2003) Regeneration of Picea engelmannii and Abies lasiocarpa in high-elevation forests of south-central British Columbia depends on nurse logs. For Chron 79:273–279
Bratton JH (2003) Habitat management to conserve fungi: a literature review. Countryside Council for Wales, Natural Science Report, Nr 03/10/1
Buchanan PK, May TW (2003) Conservation of New Zealand and Australian fungi. NZ J Bot 41:407–421
Bull EL, Wales BC (2001) Effects of disturbance on birds of conservation concern in eastern Oregon and Washington. Northwest Sci 75:166–173
Bury RB (2004) Wildfire, fuel reduction, and herpetofaunas across diverse landscape mosaics in Northwestern forests. Conserv Biol 18:968–975. doi:10.1111/j.1523-1739.2004.00522.x
Butler J, Alexander K, Green T (2002) Decaying wood: an overview of its status and ecology in the United Kingdom and continental Europe. US Forest Service, PSW-GTR-181, pp 11–19
Butler R, Angelstam P, Ekelund P, Schlaepfer R (2004) Dead wood threshold values for the three-toed woodpecker presence in boreal and sub-Alpine forest. Biol Conserv 119:305–318. doi:10.1016/j.biocon.2003.11.014
Butts SR, McComb WC (2000) Associations of forest-floor vertebrates with coarse woody debris in managed forests of western Oregon. J Wildl Manage 64:95–104
BUWAL (2004) Lothar. Rechenschaftsbericht. Materielle und finanzielle Bilanz 2000–2003. Bundesamt für Umwelt, Wald und Landschaft, Bern
Carey AB, Kershner J, Biswell B, de Toledo LD (1999) Ecological scale and forest development: squirrels, dietary fungi, and vascular plants in managed and unmanaged forests. Wildl Monogr 142:5–71
Carmona MR, Armesto JJ, Aravena JC, Perez CA (2002) Coarse woody debris biomass in successional and primary temperate forests in Chiloe Island, Chile. For Ecol Manage 164:265–275. doi:10.1016/S0378-1127(01)00602-8
Cavalli R, Mason F (eds) (2003) Tecniche di Ripristino del Legno Morto per la Conservazione delle Faune Saproxiliche. Gianluigi Arcari Editore, Mantova
Chambers CL, Mast JN (2005) Ponderosa pine snag dynamics and cavity excavation following wildfire in northern Arizona. For Ecol Manage 216:227–240. doi:10.1016/j.foreco.2005.05.033
Chambers JQ, Higuchi N, Schimel JP, Ferreira LV, Melack JM (2000) Decomposition and carbon cycling of dead trees in tropical forests of the central Amazon. Oecologia 122:380–388. doi:10.1007/s004420050044
Christensen M, Hahn K, Mountford EP, Ódor P, Standovár T, Rozenbergar D, Diaci J, Wijdeven S, Meyer P, Winter S, Vrska T (2005) Dead wood in European beech (Fagus sylvatica) forest reserves. For Ecol Manage 210:267–282. doi:10.1016/j.foreco.2005.02.032
Christie DA, Armesto JJ (2003) Regeneration microsites and tree species coexistence in temperate rain forests of Chiloe Island, Chile. J Ecol 91:776–784. doi:10.1046/j.1365-2745.2003.00813.x
Clark DB, Clark DA, Brown S, Oberbauer SF, Veldkamp E (2002) Stocks and flows of coarse woody debris across a tropical rain forest nutrient and topography gradient. For Ecol Manage 164:237–248. doi:10.1016/S0378-1127(01)00597-7
Coates D, Rayner ADM (1985a) Fungal population and community development in cut beech logs. I. Establishment via the aerial cut surface. New Phytol 101:153–171
Coates D, Rayner ADM (1985b) Fungal population and community development in cut beech logs. II. Establishment via the buried cut surface. New Phytol 101:173–181
Coates D, Rayner ADM (1985c) Fungal population and community development in cut beech logs. III. Spatial dynamics, interactions and strategies. New Phytol 101:183–198
Conner RN, Miller OK, Adkisson CS (1976) Woodpecker dependence on trees infected by fungal heart rots. Wilson Bull 88:575–581
Cooke RC, Rayner ADM (1984) Ecology of Saprotrophic Fungi. Longman, London
Cornett MW, Reich PB, Puettmann KJ (1997) Canopy feedbacks and microtopography regulate conifer seedling distribution in two Minnesota conifer-deciduous forests. Ecoscience 4:353–364
Cornett MW, Puettmann KJ, Frelich LE, Reich PB (2001) Comparing the importance of seedbed and canopy type in the restoration of upland Thuja occidentalis forests of northeastern Minnesota. Rest Ecol 9:386–396. doi:10.1046/j.1526-100X.2001.94008.x
Crow TR, Perera AH (2004) Emulating natural landscape disturbance in forest management—an introduction. Landsc Ecol 19:231–233. doi:10.1023/B:LAND.0000030762.86156.5d
Dai YC, Wei YL, Wang Z (2004) Wood-inhabiting fungi in southern China—2. Polypores from Sichuan Province. Ann Bot Fenn 41:319–329
de Jong MD, Scheepens PC, Zadoks JC (1990) Risk analysis for biological control: a Dutch case study in biocontrol of Prunus serotina by the fungus Chondrostereum purpureum. Plant Dis 74:189–194
Debeljak M (2006) Coarse woody debris in virgin and managed forest. Ecol Indic 6:733–742. doi:10.1016/j.ecolind.2005.08.031
Delaney M, Brown S, Lugo AE, Torres-Lezama A, Quintero NB (1998) The quantity and turnover of dead wood in permanent forest plots in six life zones of Venezuela. Biotropica 30:2–11. doi:10.1111/j.1744-7429.1998.tb00364.x
DeLong SC, Daniels LD, Heemskerk B, Storaunet KO (2005) Temporal development of decaying log habitats in wet spruce–fir stands in east-central British Columbia. Can J For Res 35:2841–2850
DeWalle DR, Buda AR, Fisher A (2003) Extreme weather and forest management in the mid-Atlantic region of the United States. North J Appl For 20:61–70
Diaci J, Pisek R, Boncina A (2005) Regeneration in experimental gaps of subalpine Picea abies forest in the Slovenian Alps. Eur J For Res 124:29–36. doi:10.1007/s10342-005-0057-7
Donato DC, Fontaine JB, Campbell JL, Robinson WD, Kauffman JB, Law BE (2006) Post-wildfire logging hinders regeneration and increases fire risk. Science 311:352–352. doi:10.1126/science.1122855
Dovciak M, Reich PB, Frelich LE (2003) Seed rain, safe sites, competing vegetation, and soil resources spatially structure white pine regeneration and recruitment. Can J For Res 33:1892–1904
Edman M, Jonsson BG (2001) Spatial pattern of downed logs and wood-decaying fungi in an old-growth Picea abies forest. J Veg Sci 12:609–620
Edman M, Gustafsson M (2003) Wood-disk traps provide a robust method for studying spore dispersal of wood-decaying basidiomycetes. Mycologia 95:553–556
Edman M, Gustafsson M, Stenlid J, Ericson L (2004a) Abundance and viability of fungal spores along a forestry gradient—responses to habitat loss and isolation? Oikos 104:35–42. doi:10.1111/j.0030-1299.2004.12454.x
Edman M, Gustafsson M, Stenlid J, Jonsson BG, Ericson L (2004b) Spore deposition of wood-decaying fungi: importance of landscape composition. Ecography 27:103–111. doi:10.1111/j.0906-7590.2004.03671.x
Edman M, Kruys N, Jonsson BG (2004c) Local dispersal sources strongly affect colonization patterns of wood-decaying fungi on spruce logs. Ecol Appl 14:893–901
Edman M, Moeller R, Ericson L (2006) Effects of enhanced tree growth rate on the decay capacities of three saprotrophic wood-fungi. For Ecol Manage 232:12–18. doi:10.1016/j.foreco.2006.05.001
Edmonds RL, Agee JK, Gara RI (2000) Forest health and protection. McGraw-Hill, Boston
Ehnström B (2001) Leaving dead wood for insects in boreal forests—suggestions for the future. Scand J For Res Suppl 3:91–98. doi:10.1080/028275801300090681
Ekbom B, Schroeder LM, Larsson S (2006) Stand specific occurrence of coarse woody debris in a managed boreal forest landscape in central Sweden. For Ecol Manage 221:2–12. doi:10.1016/j.foreco.2005.10.038
Eichrodt R (1970) Über die Bedeutung von Moderholz für die natürliche Verjüngung im subalpinen Fichtenwald. Beiheft Schw Z Forstw 45:1–122
Eriksson M, Pouttu A, Roininen H (2005) The influence of windthrow area and timber characteristics on colonization of wind-felled spruces by Ips typographus (L.). For Ecol Manage 216:105–116. doi:10.1016/j.foreco.2005.05.044
Eriksson M, Lilja S, Roininen H (2006) Dead wood creation and restoration burning: implications for bark beetles and beetle induced tree deaths. For Ecol Manage 231:205–213. doi:10.1016/j.foreco.2006.05.050
Evans AM, Clinton PW, Allen RB, Frampton CM (2003) The influence of logs on the spatial distribution of litter-dwelling invertebrates and forest floor processes in New Zealand forests. For Ecol Manage 184:251–262. doi:10.1016/S0378-1127(03)00158-0
Evans HF (1997) The present position of forest entomology in Great Britain. Forestry 70:327–336. doi:10.1093/forestry/70.4.327
Farris KL, Huss MJ, Zack S (2004) The role of foraging woodpeckers in the decomposition of ponderosa pine snags. Condor 106:50–59
Fayt P, Machmer MM, Steeger C (2005) Regulation of spruce bark beetles by woodpeckers—a literature review. For Ecol Manage 206:1–14. doi:10.1016/j.foreco.2004.10.054
Feller MC (2003) Coarse woody debris in the old-growth forests of British Columbia. Environ Rev 11:S135–S157
Ferguson SH, Archibald DJ (2002) The 3/4 power law in forest management: how to grow dead trees. For Ecol Manage 169:283–292. doi:10.1016/S0378-1127(01)00766-6
Ferrer A, Gilbert GS (2003) Effect of tree host species on fungal community composition in a tropical rain forest in Panama. Divers Distrib 9:455–468. doi:10.1046/j.1472-4642.2003.00039.x
Filip GM, Parks CG, Baker FA, Daniels SE (2004) Artificial inoculation of decay fungi into Douglas-Fir with rifle or shotgun to produce wildlife trees in western Oregon. West J Appl For 19:211–215
Fischer M, Binder M (2004) Species recognition, geographic distribution and host-pathogen relationships: a case study in a group of lignicolous basidiomycetes, Phellinus s.l. Mycologia 96:799–811
Flint CG (2006) Community perspectives on spruce beetle impacts on the Kenai Peninsula, Alaska. For Ecol Manage 227:207–218. doi:10.1016/j.foreco.2006.02.036
Franklin JF, Shugart HH, Harmon ME (1987) Tree death as an ecological progress. Bioscience 37:550–556
Frey W, Thee P (2002) Avalanche protection of windthrow areas: a ten year comparison of cleared and uncleared starting zones. For Snow Landsc Res 77:89–107
Fridman J, Walheim M (2000) Amount, structure, and dynamics of dead wood on managed forestland in Sweden. For Ecol Manage 131:23–36. doi:10.1016/S0378-1127(99)00208-X
FSC (2005) Swedish FSC standard for forest certification. Endorsed by the board of directors of FSC Sweden, 050901. Accessed online 1st Nov 2005 at http://www.fsc-sweden.org/Portals/0/ Documents/ Swedish%20FSC%20standard_050907.pdf
Gale N (2000) The aftermath of tree death: coarse woody debris and the topography in four tropical rain forests. Can J For Res 30:1489–1493
Ganey JL, Vojta SC (2005) Changes in snag populations in Northern Arizona mixed-conifer and ponderosa pine forests, 1997–2002. For Sci 51:396–405
Garber SM, Brown JP, Wilson DS, Maguire DA, Heath LS (2005) Snag longevity under alternative silvicultural regimes in mixed-species forests of central Maine. Can J For Res 35:787–796
Gibb H, Ball JP, Johansson T, Atlegrim O, Hjalten J, Danell K (2005) Effects of management on coarse woody debris volume and composition in boreal forests in northern Sweden. Scand J For Res 20:213–222. doi:10.1080/02827580510008392
Gilbert GS, Sousa WP (2002) Host specialization among wood-decay polypore fungi in a Caribbean mangrove forest. Biotropica 34:396–404. doi:10.1111/j.1744-7429.2002.tb00553.x
Gilbert GS, Ferrer A, Carranza J (2002) Polypore fungal diversity and host density in a moist tropical forest. Biodivers Conserv 11:947–957. doi:10.1023/A:1015896204113
Gjerde I, Satersdal M, Rolstad J, Blom HH, Storaunet KO (2004) Fine-scale diversity and rarity hotspots in northern forests. Conserv Biol 18:1032–1042. doi:10.1111/j.1523-1739.2004.00526.x
Gjerde I, Sætersdal M, Rolstad J, Storaunet KO, Blom HH, Gundersen V, Heegaard E (2005) Productivity-diversity relationships for plants, bryophytes, lichens, and polypore fungi in six northern forest landscapes. Ecography 28:705–720. doi:10.1111/j.2005.0906-7590.04249.x
Goes-Neto A, Loguercio-Leite C, Guerrero RT (2000) Taxonomy and qualitative ecological aspects of poroid hymenochaetales in a Brazilian seasonal tropical forest. Mycotaxon 76:197–211
Götmark F, Thorell M (2003) Size of nature reserves: densities of large trees and dead wood indicate high value of small conservation forests in southern Sweden. Biodivers Conserv 12:1271–1285. doi:10.1023/A:1023000224642
Gray AN, Spies TA (1997) Microsite controls on tree seedling establishment in conifer forest canopy gaps. Ecology 78:2458–2473
Green P, Peterken GF (1997) Variation in the amount of dead wood in the woodlands of the Lower Wye Valley, UK, in relation to the intensity of management. For Ecol Manage 98:229–238. doi:10.1016/S0378-1127(97)00106-0
Greslebin AG, Rajchenberg M (2003) Diversity of Corticiaceae sens. lat. in Patagonia, Southern Argentina. NZ J Bot 41:437–446
Groposo C, Loguercio-Leite C (2005) Contribution to the lignocellulolytic fungi (Basidiomycetes) of the Atlantic rain forest in southern Brazil. Mycotaxon 92:103–106
Grove SJ (2001) Extent and composition of dead wood in Australian lowland tropical rainforest with different management histories. For Ecol Manage 154:35–53. doi:10.1016/S0378-1127(00)00618-6
Grove SJ (2002) Saproxylic insect ecology and the sustainable management of forests. Ann Rev Ecol Syst 33:1–23. doi:10.1146/annurev.ecolsys.33.010802.150507
Grünig CR, McDonald BA, Sieber TN, Rogers SO, Holdenrieder O (2004) Evidence for subdivision of the root-endophyte Phialocephala fortinii into cryptic species and recombination within species. Fungal Genet Biol 41:676–687. doi:10.1016/j.fgb.2004.03.004
Gu WD, Heikkila R, Hanski I (2002) Estimating the consequences of habitat fragmentation on extinction risk in dynamic landscapes. Landsc Ecol 17:699–710. doi:10.1023/A:1022993317717
Guby NAB, Dobbertin M (1996) Quantitative estimates of coarse wooded debris and standing trees in selected Swiss forests. Glob Ecol Biogeogr Lett 5:327–341
Guillaumin J-J, Mohammed C, Anselmi N, Courtecuisse R, Gregory SC, Holdenrieder O, Intini M, Lung B, Marxmüller H, Morrison D, Rishbeth J, Termorshuizen AJ, Tirro B, Van Dam B (1993) Geographical distribution and ecology of the Armillaria species in Western Europe. Eur J For Pathol 23:321–341
Gurnell AM, Gregory KJ, Petts GE (1995) The role of coarse woody debris in forest aquatic habitats—implications for management. Aquat Conserv—Mar Freshw Ecosyst 5:143–166
Gurnell AM, Piegay H, Swanson FJ, Gregory SV (2002) Large wood and fluvial processes. Freshw Biol 47:601–619. doi:10.1046/j.1365-2427.2002.00916.x
Hahn VC, Blaschke M (2005) Ökologische Studie von Rindenpilzen und Porlingen an Totholz in einem Naturwaldreservat und forstlich bewirtschafteten Vergleichsflächen. Allg Forst Jagdz 176:161–169
Hallenberg N, Küffer N (2001) Long-distance spore dispersal in wood-inhabiting Basidiomycetes. Nord J Bot 21:431–436
Hallett JG, Lopez T, O’Connell MA, Borysewicz MA (2001) Decay dynamics and avian use of artificially created snags. Northwest Sci 75:378–386
Hamdan A, Sieber TN, Holdenrieder O (2005) Decay fungi in Norway spruce snags. In: Manka M, Lakomy P (eds) Root and butt rots of forest trees. Proceedings of the 11th int conf root and butt rots. Poznan and Bialowieza, Poland, IUFRO, pp 126–130, 16–22 Aug 2004
Harley JL (1971) Fungi in ecosystems. J Ecol 59:653–668
Harmon ME (2001) Moving towards a new paradigm for woody detritus management. Ecol Bull 49:269–278
Harmon ME, Franklin JF (1989) Tree seedlings on logs in Picea-Tsuga forests of Oregon and Washington. Ecology 70:48–59
Harmon ME, Franklin JF, Swanson FJ, Sollins P, Gregory SV, Lattin JD, Anderson NH, Cline SP, Aumen NG, Sedell JR, Lienkaemper GW, Cromack K, Cummins KW (1986) Ecology of coarse woody debris in temperate ecosystems. Adv Ecol Res 15:133–302
Harris RB (1999) Abundance and characteristics of snags in western Montana forests. USDA, Forest Service. RMRS-GTR 31:1–19
Harris RB (2001) Observations on the use of stubs by wild birds: a 10-year update. J Ecosyst Manage 1:19–23
Hattori T (2005) Diversity of wood-inhabiting polypores in temperate forests with different vegetation types in Japan. Fungal Divers 18:73–88
Hautala H, Jalonen J, Laaka-Lindberg S, Vanha-Majamaa I (2004) Impacts of retention felling on coarse woody debris (CWD) in mature boreal spruce forests in Finland. Biodivers Conserv 13:1541–1554. doi:10.1023/B:BIOC.0000021327.43783.a9
Heilmann-Clausen J (2001) A gradient analysis of communities of macrofungi and slime moulds on decaying beech logs. Mycol Res 105:575–596. doi:10.1017/S0953756201003665
Heilmann-Clausen J, Boddy L (2005) Inhibition and stimulation effects in communities of wood decay fungi: exudates from colonized wood influence growth by other species. Microbiol Ecol 49:399–406. doi:10.1007/s00248-004-0240-2
Heilmann-Clausen J, Christensen M (2003) Fungal diversity on decaying beech logs—implications for sustainable forestry. Biodivers Conserv 12:953–973. doi:10.1023/A:1022825809503
Heilmann-Clausen J, Christensen M (2004) Does size matter? On the importance of various dead wood fractions for fungal diversity in Danish beech forests. For Ecol Manage 201:105–117. doi:10.1016/j.foreco.2004.07.010
Heilmann-Clausen J, Christensen M (2005) Wood-inhabiting macrofungi in Danish beech-forests—conflicting diversity patterns and their implications in a conservation perspective. Biol Conserv 122:633–642. doi:10.1016/j.biocon.2004.10.001
Heilmann-Clausen J, Aude E, Christensen M (2005) Cryptogam communities on decaying deciduous wood—does tree species diversity matter? Biodivers Conserv 14:2061–2078. doi:10.1007/s10531-004-4284-x
Heinemann K, Kitzberger T, Veblen TT (2000) Influences of gap microheterogeneity on the regeneration of Nothofagus pumilio in a xeric old-growth forest of northwestern Patagonia, Argentina. Can J For Res 30:25–31
Heinemann K, Kitzberger T (2006) Effects of position, understorey vegetation and coarse woody debris on tree regeneration in two environmentally contrasting forests of north-western Patagonia: a manipulative approach. J Biogeogr 33:1357–1367. doi:10.1111/j.1365-2699.2006.01511.x
Hestmark G (1997) Gap-dynamics, recruitment and individual growth in populations of Lasallia pustulata. Mycol Res 101:1273–1280. doi:10.1017/S0953756297003997
Hibbett DS, Donoghue MJ (2001) Analysis of character correlations among wood decay mechanisms, mating systems, and substrate ranges in Homobasidiomycetes. Syst Biol 50:215–242. doi:10.1080/10635150121079
Hirayama K, Sakimoto M (2005) Seedling demography and establishment of Cryptomeria japonica in a cool-temperate, old-growth, conifer hardwood forest in the snowy region of Japan. J For Res 10:67–71. doi:10.1007/s10310-004-0104-0
Hiura T, Sano J, Konno Y (1996) Age structure and response to fine scale disturbances of Abies sachalinensis, Picea jezoensis, Picea glehnii, and Betula ermanii growing under the influence of a dwarf bamboo understory in northern Japan. Can J For Res 26:289–297
Hodge SJ, Peterken GF (1998) Deadwood in British forests: priorities and a strategy. Forestry 71:99–112. doi:10.1093/forestry/71.2.99
Högberg N, Holdenrieder O, Stenlid J (1999) Population structure of the wood decay fungus Fomitopsis pinicola. Heredity 83:354–360
Högberg N, Stenlid J (1999) Population genetics of Fomitopsis rosea—a wood-decay fungus of the old-growth European taiga. Mol Ecol 8:703–710. doi:10.1046/j.1365-294X.1999.00561.x
Høiland K, Bendiksen E (1996) Biodiversity of wood-inhabiting fungi in a boreal coniferous forest in Sor-Trondelag County, Central Norway. Nord J Bot 16:643–659
Holdenrieder O, Greig BJW (1998) Biological methods of control. In: Woodward S, Stenlid J, Karjalainen K, Huttermann A (eds) Heterobasidion annosum: biology, ecology, impact and control. CAB International, Wallingford, pp 235–258
Hood IA, Beets PN, Kimberley MO, Gardner JF, Oliver GR, Pearce S (2004) Colonisation of podocarp coarse woody debris by decomposer basidiomycete fungi in an indigenous forest in the central North Island of New Zealand. For Ecol Manage 196:311–325. doi:10.1016/j.foreco.2004.03.024
Hornberg G, Ohlson M, Zackrisson O (1997) Influence of bryophytes and microrelief conditions on Picea abies seed regeneration patterns in boreal old-growth swamp forests. Can J For Res 27:1015–1023
Hou P, Pan C (2001) Coarse woody debris and its function in forest ecosystem. Chin J Appl Ecol 12:309–314
Huhndorf SM, Lodge DJ, Wang C-J, Stokland JN (2004) Macrofungi on woody substrata. In: Mueller GM, Bills GF, Foster MS (eds) Biodiversity of fungi. Inventory and monitoring methods. Elsevier, Amsterdam, pp 159–163
Humphrey JW (2005) Benefits to biodiversity from developing old-growth conditions in British upland spruce plantations: a review and recommendations. Forestry 78:33–53. doi:10.1093/forestry/cpi004
Humphrey JW, Newton AC, Peace AJ, Holden E (2000) The importance of conifer plantations in northern Britain as a habitat for native fungi. Biol Conserv 96:241–252. doi:10.1016/S0006-3207(00)00077-X
Humphrey JW, Davey S, Peace AJ, Ferris R, Harding K (2002) Lichens and bryophyte communities of planted and semi-natural forests in Britain: the influence of site type, stand structure and deadwood. Biol Conserv 107:165–180. doi:10.1016/S0006-3207(02)00057-5
Hunziker U, Brang P (2005) Microsite patterns of conifer seedling establishment and growth in a mixed stand in the southern Alps. For Ecol Manage 210:67–79. doi:10.1016/j.foreco.2005.02.019
Ishii H, Kadotani T (2006) Biomass and dynamics of attached dead branches in the canopy of 450-year-old Douglas-fir trees. Can J For Res 36:378–389
Jabin M, Mohr D, Kappes H, Topp W (2004) Influence of deadwood on density of soil macro-arthropods in a managed oak-beech forest. For Ecol Manage 194:61–69. doi:10.1016/j.foreco.2004.01.053
Jack SB, Parks CG, Stober JM, Engstrom RT (2003) Inoculating red heart fungus (Phellinus pini) to create nesting habitat for the red-cockaded woodpecker. In: Proceedings of the Red Cockaded Woodpecker Symp, pp 1–18
Jackson JA, Jackson BJS (2004) Ecological relationships between fungi and woodpecker cavity sites. Condor 106:37–49
Janisch JE, Harmon ME (2002) Successional changes in live and dead wood carbon stores: implications for net ecosystem productivity. Tree Phys 22:77–89
Jax K (2005) Function and “functioning” in ecology: what does it mean? Oikos 111:641–648. doi:10.1111/j.1600-0706.2005.13851.x
Jenkins MA, Webster CR, Parker GR, Spetich MA (2004) Coarse woody debris in managed Central Hardwood Forests of Indiana, USA. For Sci 50:781–792
Johanneson H, Stenlid J (2004) Nuclear reassortment between vegetative mycelia in natural populations of the basidiomycete Heterobasidion annosum. Fungal Genet Biol 41:563–570. doi:10.1016/j.fgb.2004.01.002
Jones CG, Lawton JH, Shachak M (1994) Organisms as ecosystem engineers. Oikos 69:373–386
Jonsell M, Nordlander G (2002) Insects in polypore fungi as indicator species: a comparison between forest sites differing in amounts and continuity of dead wood. For Ecol Manage 157:101–118. doi:10.1016/S0378-1127(00)00662-9
Jonsson BG (2000) Availability of course wood debris in a boreal old growth Picea abies forest. J Veg Sci 11:51–56
Jonsson BG, Jonsell M (1999) Exploring potential biodiversity indicators in boreal forests. Biodivers Conserv 8:1417–1433. doi:10.1023/A:1008900309571
Jonsson BG, Kruys N, Ranius T (2005) Ecology of species living on dead wood—lessons for dead wood management. Silva Fenn 39:289–309
Jonsson M, Ranius T, Ekvall H, Bostedt G, Dahlberg A, Ehnstrom B, Nordén B, Stokland JN (2006) Cost-effectiveness of silvicultural measures to increase substrate availability for red-listed wood-living organisms in Norway spruce forests. Biol Conserv 127:443–462. doi:10.1016/j.biocon.2005.09.004
Junninen K, Similä M, Kouki J, Kotiranta H (2006) Assemblages of wood-inhabiting fungi along the gradients of succession and naturalness in boreal pine-dominated forests in Fennoscandia. Ecography 29:75–83. doi:10.1111/j.2005.0906-7590.04358.x
Juutinen A, Mönkkönen M (2004) Testing alternative indicators for biodiversity conservation in old-growth boreal forests: ecology and economics. Ecol Econ 50:35–48. doi:10.1016/j.ecolecon.2004.02.006
Juutinen A, Mönkkönen M, Sippola A-L (2006) Cost-efficiency of decaying wood as a surrogate for overall species-richness in boreal forests. Conserv Biol 20:74–84. doi:10.1111/j.1523-1739.2006.00306.x
Kappes H (2005) Influence of coarse woody debris on the gastropod community of a managed calcareous beech forest in western Europe. J Molluscan Stud 71:85–91. doi:10.1093/mollus/eyi011
Kappes H, Topp W (2004) Emergence of Coleoptera from deadwood in a managed broadleaved forest in central Europe. Biodivers Conserv 13:1905–1924. doi:10.1023/B:BIOC.0000035873.56001.7d
Kauserud H, Schumacher T (2003a) Genetic structure of Fennoscandian populations of the threatened wood-decay fungus Fomitopsis rosea (Basidiomycota). Mycol Res 107:155–163. doi:10.1017/S0953756203007214
Kauserud H, Schumacher T (2003b) Regional and local population structure of the pioneer wood-decay fungus Trichaptum abietinum. Mycologia 95:416–425
Kauserud H, Lie M, Stensrud O, Ohlson M (2005) Molecular characterization of airborne fungal spores in boreal forests of contrasting human disturbance. Mycologia 97:1215–1224
Keller M, Palace M, Asner GP, Pereira R, Silva JNM (2004) Coarse woody debris in undisturbed and logged forests in the eastern Brazilian Amazon. Glob Change Biol 10:784–795. doi:10.1111/j.1529-8817.2003.00770.x
Kennedy PG, Quinn T (2001) Understory plant establishment on old-growth stumps and the forest floor in western Washington. For Ecol Manage 154:193–200. doi:10.1016/S0378-1127(00)00622-8
Kohsaka R, Handoh IC (2006) Perceptions of “close-to-nature forestry” by German and Japanese groups: inquiry using visual materials of “cut” and “dead” wood. J For Res 11:11–19. doi:10.1007/s10310-005-0177-4
Komonen A (2003) Distribution and abundance of insect fungivores in the fruiting bodies of Fomitopsis pinicola. Ann Zool Fenn 40:495–504
Komonen A (2005) Local spatial pattern in the occurrence of two congeneric wood-decaying fungi in an old-growth boreal forest. Scand J For Res 20:393–399. doi:10.1080/02827580500281983
Korhonen K, Holdenrieder O (2005) Neue Erkenntnisse über den Wurzelschwamm (Heterobasidion annosum s.l.)—eine Literaturübersicht. Forst und Holz 60:206–210
Köster K, Jogiste K, Tukia H, Niklasson M, Mols T (2005) Variation and ecological characteristics of coarse woody debris in Lahemaa and Karula National Parks, Estonia. Scand J For Res 20(Suppl 6):102–111. doi:10.1080/14004080510042137
Kouki J, Lofman S, Martikainen P, Rouvinen S, Uotila A (2001) Forest fragmentation in Fennoscandia: linking habitat requirements of wood-associated threatened species to landscape and habitat changes. Scand J For Res 16:27–37. doi:10.1080/028275801300090564
Krajick K (2001) Defending deadwood. Science 293:1579–1581. doi:10.1126/science.293.5535.1579
Krankina ON, Harmon ME (1995) Dynamics of the dead wood carbon pool in northwestern Russian boreal forests. Water Air Soil Poll 82:227–238. doi:10.1007/BF01182836
Kruys N, Jonsson BG (1999) Fine woody debris is important for species-richness on logs in managed boreal spruce forests of northern Sweden. Can J For Res 29:1295–1299
Kruys N, Fries C, Jonsson BG, Lämås T, Ståhl G (1999) Wood-inhabiting cryptogams on dead Norway spruce (Picea abies) trees in managed Swedish boreal forests. Can J For Res 29:178–186
Kruys N, Jonsson BG, Stahl G (2002) A stage-based matrix model for decay-class dynamics of woody debris. Ecol Appl 12:773–781
Küffer N, Senn-Irlet B (2000) Diversity and ecology of corticioid basidiomycetes in green alder stands in Switzerland. Nova Hedwigia 71:131–143
Küffer N, Senn-Irlet B (2005a) Influence of forest management on the species-richness and composition of wood-inhabiting basidiomycetes in Swiss forests. Biodivers Conserv 14:2419–2435. doi:10.1007/s10531-004-0151-z
Küffer N, Senn-Irlet B (2005b) Diversity and ecology of wood-inhabiting aphyllophoroid basidiomycetes on fallen woody debris in various forest types in Switzerland. Mycol Prog 4:77–86. doi:10.1007/s11557-006-0110-z
Kulhánková A, Béguiristain T, Moukoumi J, Berthelin J, Ranger J (2006) Spatial and temporal diversity of wood decomposer communities in different forest stands, determined by ITS rDNA targeted TGGE. Ann For Sci 63:547–556. doi:10.1051/forest:2006037
Kupferschmid AD, Bugmann H (2005) Predicting decay and ground vegetation development in Picea abies snag stands. Plant Ecol 179:247–268. doi:10.1007/s11258-005-0903-1
Kupferschmid AD, Brang P, Schönenberger W, Bugmann H (2003) Decay of Picea abies snag stands on steep mountain slopes. For Chron 79:247–252
Kuuluvainen T, Juntunen P (1998) Seedling establishment in relation to microhabitat variation in a windthrow gap in a boreal Pinus sylvestris forest. J Veg Sci 9:551–562
Kuuluvainen T, Kalmari R (2003) Regeneration microsites of Picea abies seedlings in a windthrow area of a boreal old-growth forest in southern Finland. Ann Bot Fenn 40:401–413
Lack AJ (1991) Dead logs as a substrate for rain forest trees in Dominica. J Trop Ecol 7:401–405
Laiho R, Prescott CE (2004) Decay and nutrient dynamics of coarse woody debris in northern coniferous forests:a synthesis. Can J For Res 34:763–777
Lee P, Sturgess K (2001) The effects of logs, stumps, and root throws on understory communities within 28-year-old aspen-dominated boreal forests. Can J Bot 79:905–916
Lee PC, Crites S, Nietfeld M, VanNguyen H, Stelfox JB (1997) Characteristics and origins of deadwood material in aspen-dominated boreal forests. Ecol Appl 7:691–701
Lee SD (2004) Population dynamics and demography of deermice (Peromyscus maniculatus) in heterogeneous habitat: role of coarse woody debris. Pol J Ecol 52:55–62
Lewis C (1998) Creating snags and wildlife trees in commercial forest landscapes. West J Appl For 13(3):97–101
Liao CC, Chou CH, Wu JT (2003) Regeneration patterns of yellow cypress on down logs in mixed coniferous-broadleaf forest of Yuanyang Lake Nature Preserve, Taiwan. Bot Bull Acad Sinica 44:229–238
Lilja S, De Chantal M, Kuuluvainen T, Vanha-Majamaa I, Puttonen P (2005) Restoring natural characteristics in managed Norway spruce [Picea abies (L.) Karst.] stands with partial cutting, dead wood creation and fire: immediate treatment effects. Scand J For Res 20(Suppl 6):68–78. doi:10.1080/14004080510040977
Lindblad I (1997) Wood-inhabiting fungi on fallen logs of Norway spruce: relations to forest management and substrate quality. Nord J Bot 18:243–255
Lindblad I (2000) Host specificity of some wood-inhabiting fungi in a tropical forest. Mycologia 92:399–405
Lindblad I (2001) Diversity of poroid and some corticoid wood-inhabiting fungi along the rainfall gradient in tropical forests, Costa Rica. J Trop Ecol 17:353–369. doi:10.1017/S0266467401001249
Lindhe A, Asenblad N, Toresson HG (2004) Cut logs and high stumps of spruce, birch, aspen and oak—nine years of saproxylic fungi succession. Biol Conserv 119:443–454. doi:10.1016/j.biocon.2004.01.005
Liski J, Korotkov AV, Prins CFL, Karjalainen T, Victor DG, Kauppi PE (2003) Increased carbon sink in temperate and boreal forests. Clim Change 61:89–99. doi:10.1023/A:1026365005696
Liu WH, Bryant DM, Hutyra LR, Saleska SR, Hammond-Pyle E, Curran D, Wofsy SC (2006) Woody debris contribution to the carbon budget of selectively logged and maturing mid-latitude forests. Oecologia 148:108–117. doi:10.1007/s00442-006-0356-9
Lodge DJ, Cantrell S (1995) Fungal communities in wet tropical forests—variation in time and space. Can J Bot 73:S1391–S1398
Lonsdale D (2004) Aging processes in trees and their relationships with decay fungi. In: Nicolotti G, Gonthier P (eds) The trees of history. Protection and exploitation of veteran trees. Proc Int Cong, Torino, Italy, pp 23–30, 1–2 Apr 2004
Lumley TC, Gignac LD, Currah RS (2001) Microfungus communities of white spruce and trembling aspen logs at different stages of decay in disturbed and undisturbed sites in the boreal mixedwood region of Alberta. Can J Bot 79:76–92
Lusk CH (1995) Seed size, establishment sites and species coexistence in a Chilean rainforest. J Veg Sci 6:249–256
Mackensen J, Bauhus J, Webber E (2003) Decomposition rates of coarse woody debris—a review with particular emphasis on Australian tree species. Aust J Bot 51:27–37. doi:10.1071/BT02014
MacNally R, Parkinson A, Horrocks G, Conole L, Tzaros C (2001) Relationships between terrestrial vertebrate diversity, abundance and availability of coarse woody debris on south-eastern Australian floodplains. Biol Conserv 99:191–205. doi:10.1016/S0006-3207(00)00180-4
MacNally R, Horrocks G, Pettifer L (2002a) Experimental evidence for potential beneficial effects of fallen timber in forests. Ecol Appl 12:1588–1594
MacNally R, Parkinson A, Horrocks G, Young M (2002b) Current loads of coarse woody debris on southeastern Australian floodplains: evaluation of change and implications for restoration. Restor Ecol 10:627–635. doi:10.1046/j.1526-100X.2002.01043.x
Mäkinen H, Hynynen J, Siitonen J, Sievanen R (2006) Predicting the decomposition of Scots pine, Norway spruce, and birch stems in Finland. Ecol Appl 16:1865–1879
Manion PD (2003) Evolution of concepts in forest pathology. Phytopathology 93:1052–1055
Marage D, Lemperiere G (2005) The management of snags: a comparison in managed and unmanaged ancient forests of the Southern French Alps. Ann For Sci 62:135–142. doi:10.1051/forest:2005005
Marcot BG (2002) An ecological functional basis for managing wood decay elements for wildlife. USDA Forest Service, PSW-GTR-181
Maser C, Trappe JM, Nussbaum RA (1978) Fungal—small mammal interrelationships with emphasis on Oregon coniferous forests. Ecology 59:799–809
Mayer AL, Kauppi PE, Tikka PM, Angelstam PK (2006) Conservation implications of exporting domestic wood harvest to neighboring countries. Environ Sci Pollut 9:228–236. doi:10.1016/j.envsci.2005.12.002
McCay TS, Komoroski MJ (2004) Demographic responses of shrews to removal of coarse woody debris in a managed pine forest. For Ecol Manage 189:387–395. doi:10.1016/j.foreco.2003.09.005
McClelland BR, McClelland PT (1999) Pileated woodpecker nest and roost trees in Montana: links with old-growth and forest “health”. Wildl Soc Bull 27:846–857
McClure JM, Kolka RK, White A (2004) Effect of forest harvesting best management practices on coarse woody debris distribution in stream and riparian zones in three Appalachian watersheds. Water Air Soil Pollut 4:245–261. doi:10.1023/B:WAFO.0000012815.30596.97
McComb W, Lindenmayer D (1999) Dying, dead, and down trees. In: Hunter ML (ed) Maintaining biodiversity in forest ecosystems. Cambridge University Press, Cambridge, pp 335–372
McGee GG (2000) The contribution of beech bark disease-induced mortality to coarse woody debris loads in northern hardwood stands of Adirondack Park, New York, USA. Can J For Res 30:1453–1462
McGee GG (2001) Stand-level effects on the role of decaying logs as vascular plant habitat in Adirondack northern hardwood forests. J Torrey Bot Soc 128:370–380
McGee GG, Leopold DJ, Nyland RD (1999) Structural characteristics of old-growth, maturing, and partially cut northern hardwood forests. Ecol Appl 9:1316–1329
McKenny HJA, Kirkpatrick JB (1999) The role of fallen logs in the regeneration of tree species in Tasmanian mixed forest. Aust J Bot 47:745–753. doi:10.1071/BT98001
McPherson BA, Mori SR, Wood DL, Storer AJ, Svihra P, Kelly NM, Standiford RB (2005) Sudden oak death in California: disease progression in oaks and tanoaks. For Ecol Manage 213:71–89. doi:10.1016/j.foreco.2005.03.048
Meyer P (1999) Dead wood research in forest reserves of Northwest-Germany: methodology and results. Forstwissenschaftliches Centralblatt 118:167–180
Molina R, Pilz D, Smith J, Dunham S, Dreisbach T, O’Dell T, Castellano M (2001) Conservation and management of forest fungi in the Pacific Northwestern United States: an integrated ecosystem approach. In: Moore D, Nauta MM, Evans SE, Rotheroe M (eds) Fungal conservation—issues and solutions. Cambridge University Press, Cambridge, pp 19–63
Moore JC, Berlow EL, Coleman DC, de Ruiter PC, Dong Q, Hastings A, Johnson NC, McCann KS, Melville K, Morin PJ, Nadelhoffer K, Rosemond AD, Post DM, Sabo JL, Scow KM, Vanni MJ, Wall DH (2004) Detritus, trophic dynamics and biodiversity. Ecol Lett 7:584–600. doi:10.1111/j.1461-0248.2004.00606.x
Mori A, Mizumachi E, Osono T, Doi Y (2004) Substrate-associated seedling recruitment and establishment of major conifer species in an old-growth subalpine forest in central Japan. For Ecol Manage 196:287–297. doi:10.1016/j.foreco.2004.03.027
Motta R, Berretti R, Lingua E, Piussi P (2006) Coarse woody debris, forest structure and regeneration in the Valbona Forest Reserve, Paneveggio, Italian Alps. For Ecol Manage 235:155–163. doi:10.1016/j.foreco.2006.08.007
Mukhin VA, Kotiranta H (2001) Wood-decaying fungi of northernmost forests in river Khatanga basin. Mikologiya I Fitopatologiya 35:41–47
Müller-Using S, Bartsch N (2003) Dynamics of woody debris in a beech stand (Fagus sylvatica L.) in Solling. Input, causes and decomposition of woody debris. Allg For Jagdz 174:122–130
Narukawa Y, Yamamoto S (2003) Development of conifer seedlings roots on soil and fallen logs in boreal and subalpine coniferous forests of Japan. For Ecol Manage 175:131–139. doi:10.1016/S0378-1127(02)00125-1
Narukawa Y, Iida S, Tanouchi H, Abe S, Yamamoto SI (2003) State of fallen logs and the occurrence of conifer seedlings and saplings in boreal and subalpine old-growth forests in Japan. Ecol Res 18:267–277. doi:10.1046/j.1440-1703.2003.00553.x
Niemelä T, Korhonen K (1998) Taxonomy of the genus Heterobasidion. In: Woodward S, Stenlid J, Karjalainen R, Hüttermann A (eds) Heterobasidion annosum: biology, ecology, impact and control. CABI, Wallingford, pp 27–33
Niemelä T, Renvall P, Penttilä R (1995) Interactions of fungi at late stages of wood decomposition. Ann Bot Fenn 32:141–152
Noguchi M, Yoshida T (2004) Tree regeneration in partially cut conifer-hardwood mixed forests in northern Japan: roles of establishment substrate and dwarf bamboo. For Ecol Manage 190:335–344. doi:10.1016/j.foreco.2003.10.024
Nordén B (1997) Genetic variation within and among populations of Fomitopsis pinicola (Basidiomycetes). Nord J Bot 17:319–329
Nordén B, Larsson KH (2000) Basidiospore dispersal in the old-growth forest fungus Phlebia centrifuga (Basidiomycetes). Nord J Bot 20:215–219
Nordén B, Appelqvist T (2001) Conceptual problems of ecological continuity and its bioindicators. Biodivers Conserv 10:779–791. doi:10.1023/A:1016675103935
Nordén B, Paltto H (2001) Wood-decay fungi in hazel wood: species-richness correlated to stand age and dead wood features. Biol Conserv 101:1–8. doi:10.1016/S0006-3207(01)00049-0
Nordén B, Gotmark F, Tonnberg M, Ryberg M (2004a) Dead wood in semi-natural temperate broadleaved woodland: contribution of coarse and fine dead wood, attached dead wood and stumps. For Ecol Manage 194:235–248. doi:10.1016/j.foreco.2004.02.043
Nordén B, Ryberg M, Gotmark F, Olausson B (2004b) Relative importance of coarse and fine woody debris for the diversity of wood-inhabiting fungi in temperate broadleaf forests. Biol Conserv 117:1–10. doi:10.1016/S0006-3207(03)00235-0
Norstedt G, Bader P, Ericson L (2001) Polypores as indicators of conservation value in Corsican pine forests. Biol Conserv 99:347–354. doi:10.1016/S0006-3207(00)00220-2
Odling-Smee FJ, Laland KN, Feldman MW (2003) Niche construction—the neglected process in evolution. Princeton University Press, Princeton
Ódor P, Van Hees AFM (2004) Preferences of dead wood inhabiting bryophytes for decay stage, log size and habitat types in Hungarian beech forests. J Bryol 26:79–95. doi:10.1179/037366804225021038
Ódor P, Heilmann-Clausen J, Christensen M, Aude E, van Dort KW, Piltaver A, Siller I, Veerkamp MT, Walleyn R, Standovar T, van Hees AFM, Kosec J, Matocec N, Kraigher H, Grebenc T (2006) Diversity of dead wood inhabiting fungi and bryophytes in semi-natural beech forests in Europe. Biol Conserv 131:58–71. doi:10.1016/j.biocon.2006.02.004
O’Hanlon-Manners DL, Kotanen PM (2004) Logs as refuges from fungal pathogens for seeds of eastern hemlock (Tsuga canadensis). Ecology 85:284–289
Økland B (1996) Unlogged forests: important sites for preserving the diversity of mycetophilids (Diptera: Sciaroidea). Biol Conserv 76:297–310. doi:10.1016/0006-3207(95)00129-8
Ortega A, Navarro FB (2004) A myco-ecological analysis (lignicolous Aphyllophorales sensu lato, Basidiomycota) of the Abies pinsapo, Quercus and Pinus forests of Andalusia (southern Spain). Nova Hedwigia 78:485–499. doi:10.1127/0029-5035/2004/0078-0485
Parent S, Simard MJ, Morin H, Messier C (2003) Establishment and dynamics of the balsam fir seedling bank in old forests of northeastern Quebec. Can J For Res 33:597–603
Parish R, Antos JA (2005) Advanced regeneration and seedling establishment in small cutblocks in high-elevation spruce-fir forest at Sicamous Creek, southern British Columbia. Can J For Res 35:1877–1888
Parks CG, Shaw DC (1996) Death and decay: a vital part of living canopies. Northwest Sci 70:46–53
Parks CG, Conklin DA, Bednar L, Maffei H (1999) Woodpecker use and fall rates of snags created by killing ponderosa pine infected with dwarf mistletoe. USDA FS PNW Research Paper Nr. 515
Parmasto E (2001) Fungi as indicators of primeval and old-growth forests deserving protection. In: Moore D, Nauta MM, Evans SE, Rotheroe M (eds) Fungal conservation—issues and solutions. Cambridge University Press, Cambridge, pp 81–88
Parrent JL, Garbelotto M, Gilbert GS (2004) Population genetic structure of the polypore Datronia caperata in fragmented mangrove forests. Mycol Res 108:403–410. doi:10.1017/S0953756204009773
Parsons S, Lewis KJ, Psyllakis JM (2003) Relationships between roosting habitat of bats and decay of aspen in the sub-boreal forests of British Columbia. For Ecol Manage 177:559–570. doi:10.1016/S0378-1127(02)00448-6
Passovoy MD, Fulé PZ (2006) Snag and woody debris dynamics following severe wildfires in northern Arizona ponderosa pine forests. For Ecol Manage 223:237–246. doi:10.1016/j.foreco.2005.11.016
Patrick DA, Hunter ML, Calhoun AJK (2006) Effects of experimental forestry treatments on a Maine amphibian community. For Ecol Manage 234:323–332. doi:10.1016/j.foreco.2006.07.015
Pedlar JH, Pearce JL, Venier LA, McKenney DW (2002) Coarse woody debris in relation to disturbance and forest type in boreal Canada. For Ecol Manage 158:189–194. doi:10.1016/S0378-1127(00)00711-8
Penttilä R, Siitonen J, Kuusinen M (2004) Polypore diversity in managed and old-growth boreal Picea abies forests in southern Finland. Biol Conserv 117:271–283. doi:10.1016/j.biocon.2003.12.007
Penttilä R, Lindgren M, Miettinen O, Rita H, Hanski I (2006) Consequences of forest fragmentation for polyporous fungi at two spatial scales. Oikos 114:225–240. doi:10.1111/j.2006.0030-1299.14349.x
Peterson CJ, Haines BL (2000) Early successional patterns and potential facilitation of woody plant colonization by rotting logs in premontane Costa Rican pastures. Restor Ecol 8:361–369. doi:10.1046/j.1526-100x.2000.80051.x
Peterson GD (2002) Contagious disturbance, ecological memory, and the emergence of landscape pattern. Ecosystems 5:329–338. doi:10.1007/s10021-001-0077-1
Pharo EJ, Lindenmayer DB, Taws N (2004) The effects of large-scale fragmentation on bryophytes in temperate forests. J Appl Ecol 41:910–921. doi:10.1111/j.0021-8901.2004.00947.x
Piovesan G, Di Filippo A, Alessandrini A, Biondi F, Schirone B (2005) Structure, dynamics and dendroecology of an old-growth Fagus forest in the Apennines. J Veg Sci 16:13–28
Porter AD, St Clair CC, de Vries A (2005) Fine-scale selection by marten during winter in a young deciduous forest. Can J For Res 35:901–909
Pregitzer KS, Euskirchen ES (2004) Carbon cycling and storage in world forests: biome patterns related to forest age. Glob Change Biol 10:2052–2077. doi:10.1111/j.1365-2486.2004.00866.x
Prestemon JP, Holmes TP (2004) Market dynamics and optimal timber salvage after a natural catastrophe. For Sci 50:495–511
Pretty JL, Dobson M (2004) The response of macroinvertebrates to artificially enhanced detritus levels in plantation streams. Hydr Earth Syst Sci 8:550–559
Progar RA, Schowalter TD, Freitag CM, Morrell JJ (2000) Respiration from coarse woody debris as affected by moisture and saprotroph functional diversity in Western Oregon. Oecologia 124:426–431. doi:0.1007/PL00008868
Prospero S, Holdenrieder O, Rigling D (2003) Primary resource capture in two sympatric Armillaria species in managed Norway spruce forests. Mycol Res 107:329–338. doi:10.1017/S0953756203007275
Rademacher C, Winter S (2003) Coarse woody debris in natural beech forests: generic predictions of the simulation model BEFORE-CWD of quantity, spatial distribution and availability. Forstwissenschaftliches Centralblatt 122:337–357
Ranius T (2002) Osmoderma eremita as an indicator of species-richness of beetles in tree hollows. Biodivers Conserv 11:931–941. doi:10.1023/A:1015364020043
Ranius T, Kindvall O (2004) Modelling the amount of coarse woody debris produced by the new biodiversity-oriented silvicultural practices in Sweden. Biol Conserv 119:51–59. doi:10.1016/j.biocon.2003.10.021
Ranius T, Fahrig L (2006) Targets for maintenance of dead wood for biodiversity conservation based on extinction thresholds. Scand J For Res 21:201–208. doi:10.1080/02827580600688269
Ranius T, Kindvall O, Kruys N, Jonsson BG (2003) Modelling dead wood in Norway spruce stands subject to different management regimes. For Ecol Manage 182:13–29. doi:10.1016/S0378-1127(03)00027-6
Ranius T, Ekvall H, Jonsson M, Bostedt G (2005) Cost-efficiency of measures to increase the amount of coarse woody debris in managed Norway spruce forests. For Ecol Manage 206:119–133. doi:10.1016/j.foreco.2004.10.061
Rayner ADM, Boddy L (1988) Fungal decomposition of wood: its biology and ecology. Wiley, New York
Redfern DB, Stenlid J (1998) Spore dispersal and infection. In: Woodward S, Stenlid J, Karjalainen K, Huttermann A (eds) Heterobasidion annosum: biology, ecology, impact and control. CABI, Wallingford, pp 105–141
Renvall P (1995) Community structure and dynamics of wood-rotting basidiomycetes on decomposing conifer trunks in northern Finland. Karstenia 35:1–51
Rice AH, Pyle EH, Saleska SR, Hutyra L, Palace M, Keller M, de Camargo PB, Portilho K, Marques DF, Wofsy SC (2004) Carbon balance and vegetation dynamics in an old-growth Amazonian forest. Ecol Appl 14:S55–S71
Risch AC, Nagel LM, Schutz M, Krusi BO, Kienast F, Bugmann H (2003) Structure and long-term development of subalpine Pinus montana Miller and Pinus cembra L. forests in the Central European Alps. Forstwissenschaftliches Centralblatt 122:219–230
Rishbeth J (1959) Dispersal of Fomes annosus Fr. and Peniophora gigantea (Fr.) Massee. Trans Br Mycol Soc 42:243–260
Roberge JM, Angelstam P (2004) Usefulness of the umbrella species concept as a conservation tool. Conserv Biol 18:76–85. doi:10.1111/j.1523-1739.2004.00450.x
Robertson AI (1991) Plant animal interactions and the structure and function of mangrove forest ecosystems. Aust J Ecol 16:433–443
Rodriguez C, Burdsall HH, Volk TJ (1995) Wood-decay basidiomycetes from the state of Bolivar in Southeastern Venezuela. Mycotaxon 53:377–389
Rogers S, Holdenrieder O, Sieber TN (1999) Intraspecific comparisons of Laetiporus sulphureus isolates from broadleaf and coniferous trees in Europe. Mycol Res 103:1245–1251. doi:10.1017/S0953756299008564
Rolstad J, Saetersdal M, Gjerde I, Storaunet KO (2004) Wood-decaying fungi in boreal forest: are species-richness and abundances influenced by small-scale spatiotemporal distribution of dead wood? Biol Conserv 117:539–555. doi:10.1016/j.biocon.2003.09.008
Rooney TP, Waller DM (1998) Local and regional variation in hemlock seedling establishment in forests of the upper Great Lakes region, USA. For Ecol Manage 111:211–224. doi:10.1016/S0378-1127(98)00333-8
Rothman JM, Van Soest PJ, Pell AN (2006) Decaying wood is a sodium source for mountain gorillas. Biol Lett 2:321–324. doi:10.1098/rsbl.2006.0480
Rouvinen S, Kouki J (2002) Spatiotemporal availability of dead wood in protected old-growth forests: a case study from boreal forests in eastern Finland. Scand J For Res 17:317–329. doi:10.1080/02827580260138071
Rouvinen S, Kuuluvainen T, Karjalainen L (2002) Coarse woody debris in old Pinus sylvestris dominated forests along a geographic and human impact gradient in boreal Fennoscandia. Can J For Res 32:2184–2200
Rouvinen S, Rautiainen A, Kouki J (2005) A relation between historical forest use and current dead woody material in a boreal protected old-growth forest in Finland. Silva Fenn 39:21–36
Rubino DL, McCarthy BC (2003) Composition and ecology of macrofungal and myxomycete communities on oak woody debris in a mixed-oak forest of Ohio. Can J For Res 33:2151–2163
Russell RE, Saab VA, Dudley JG, Rotella JJ (2006) Snag longevity in relation to wildfire and postfire salvage logging. For Ecol Manage 232:179–187. doi:10.1016/j.foreco.2006.05.068
Ryvarden L (1998) African polypores—a review. Belg J Bot 131:150–155
Saetersdal M, Gjerde I, Blom HH (2005) Indicator species and the problem of spatial inconsistency in nestedness patterns. Biol Conserv 122:305–316. doi:10.1016/j.biocon.2004.07.020
Sakamoto Y, Miyamoto T (2005) Racodium snow blight in Japan. For Pathol 35:1–7. doi:10.1111/j.1439-0329.2004.00383.x
Samuelsson J, Gustafsson L, Ingelog T (1994) Dying and dead trees: a review of their importance for biodiversity. Swedish Threatened Species Unit, Uppsala
Sánchez-Flores E, Yool SR (2004) Site environment characterization of downed woody fuels in the Rincon Mountains, Arizona: regression tree approach. Int J Wildland Fire 13:467–477. doi:10.1071/WF04015
Santiago LS (2000) Use of coarse woody debris by the plant community of a Hawaiian montane cloud forest. Biotropica 32:633–641. doi:10.1111/j.1744-7429.2000.tb00510.x
Schiegg K (1998) Totholz bringt Leben in den Wirtschaftswald. Schweiz Z Forstw 149:784–794
Schmit JP (2005) Species-richness of tropical wood-inhabiting macrofungi provides support for species-energy theory. Mycologia 97:751–761
Schmit JP, Mueller GM, Leacock PR, Mata JL, Wu QX, Huang YG (2005) Assessment of tree species-richness as a surrogate for macrofungal species-richness. Biol Conserv 121:99–110. doi:10.1016/j.biocon.2004.04.013
Schönenberger W, Noack A, Thee P (2005) Effect of timber removal from windthrow slopes on the risk of snow avalanches and rockfall. For Ecol Manage 213:197–208. doi:10.1016/j.foreco.2005.03.062
Schwarze FWMR, Engels J, Mattheck C (2000) Fungal strategies of wood decay in trees. Springer, Berlin
Selonen VAO, Ahlroth P, Kotiaho JS (2005) Anthropogenic disturbance and diversity of species: polypores and polypore-associated beetles in forest, forest edge and clear-cut. Scand J For Res 20(Suppl 6):49–58. doi:10.1080/14004080510041002
Siitonen J (2001) Forest management, coarse woody debris and saproxylic organisms: Fennoscandian boreal forests as an example. Ecol Bull 49:11–41
Siitonen P, Lehtinen A, Siitonen M (2005) Effects of forest edges on the distribution, abundance, and regional persistence of wood-rotting fungi. Conserv Biol 19:250–260. doi:10.1111/j.1523-1739.2005.00232.x
Simard MJ, Bergeron Y, Sirois L (1998) Conifer seedling recruitment in a southeastern Canadian boreal forest: the importance of substrate. J Veg Sci 9:575–582
Simard MJ, Bergeron Y, Sirois L (2003) Substrate and litterfall effects on conifer seedling survivorship in southern boreal stands of Canada. Can J For Res 33:672–681
Similä M, Kouki J, Mönkkönen M, Sippola A-L, Huhta E (2006) Co-variation and indicators of species diversity: can richness of forest-dwelling species be predicted in northern boreal forests? Ecol Indic 6:686–700. doi:10.1016/j.ecolind.2005.08.028
Sippola A-L, Renvall P (1999) Wood-decomposing fungi and seed-tree cutting: a 40-year perspective. For Ecol Manage 115:183–201. doi:10.1016/S0378-1127(98)00398-3
Sippola A-L, Similä M, Mönkkönen M, Jokimäki J (2004) Diversity of polyporous fungi (Polyporaceae) in northern boreal forests: effects of forest site type and logging intensity. Scand J For Res 19:152–163. doi:10.1080/02827580410026294
Sippola A-L, Mönkkönen M, Renvall P (2005) Polypore diversity in the herb-rich woodland key habitats of Koli National Park in eastern Finland. Biol Conserv 126:260–269. doi:10.1016/j.biocon.2005.06.002
Slocum MG (2000) Logs and fern patches as recruitment sites in a tropical pasture. Restor Ecol 8:408–413
Spetich MA, Parker GR (1998) Distribution of biomass in an Indiana old-growth forest from 1926 to 1992. Am Midl Nat 139:90–107
Spetich MA, Shifley SR, Parker GR (1999) Regional distribution and dynamics of coarse woody debris in midwestern old-growth forests. For Sci 45:302–313
Stavishenko IV, Zalesov SV, Luganskii NA, Kryazhevskikh NA, Morozov AE (2002) Communities of wood-attacking fungi in the region of oil and gas production. Russ J Ecol 33:161–169. doi:10.1023/A:1015423422744
Steeger C, Hitchcock CL (1998) Influence of forest structure and diseases on nest-site selection by red-breasted nuthatches. J Wildl Manage 62:1349–1358
Stephenson NL, van Mantgem PJ (2005) Forest turnover rates follow global and regional patterns of productivity. Ecol Lett 8:524–531. doi:10.1111/j.1461-0248.2005.00746.x
Stewart JD, Landhausser SM, Stadt KJ, Lieffers VJ (2001) Predicting natural regeneration of white spruce in boreal mixedwood understories. For Chron 77:1006–1013
Stöckli B (1995) Moderholz für die Naturverjüngung im Bergwald; Anleitung zum Moderanbau. Wald Holz 76:16:8–14
Stokland J, Kauserud H (2004) Phellinus nigrolimitatus—a wood-decomposing fungus highly influenced by forestry. For Ecol Manage 187:333–343. doi:10.1016/j.foreco.2003.07.004
Storaunet KO (2004) Models to predict time since death of Picea abies snags. Scand J For Res 19:250–260. doi:10.1080/02827580410024142
Storaunet KO, Rolstad J (2004) How long do Norway spruce snags stand? Evaluating four estimation methods. Can J For Res 34:376–383
Storaunet KO, Rolstad J, Gjerde I, Gundersen VS (2005) Historical logging, productivity, and structural characteristics of boreal coniferous forests in Norway. Silva Fenn 39:429–442
Strange N, Christensen M, Heilmann-Clausen J (2004) Some policy implications of biodiversity conservation in Danish natural forests. Scand J For Res 19:138–149. doi:10.1080/14004080410034227
Sturtevant BR, Bissonette JA, Long JN, Roberts DW (1997) Coarse woody debris as a function of age, stand structure, and disturbance in boreal Newfoundland. Ecol Appl 7:702–712
Sugita H, Tani M (2001) Difference in microhabitat-related regeneration patterns between two subalpine conifers, Tsuga diversifolia and Abies mariesii, on Mount Hayachine, northern Honshu, Japan. Ecol Res 16:423–433. doi:10.1046/j.1440-1703.2001.00408.x
Sugita H, Nagaike T (2005) Microsites for seedling establishment of subalpine conifers in a forest with moss-type undergrowth on Mt. Fuji, central Honshu, Japan. Ecol Res 20:678–685. doi:10.1007/s11284-005-0086-1
Suter W, Schielly B (1998) Liegendes Totholz: ein wichtiges Strukturmerkmal für die Habitatqualität von Kleinsäugern und kleinen Carnivoren im Wald. Schweiz Z Forstw 149:795–807
Sverdrup-Thygeson A, Lindemayer DB (2003) Ecological continuity and assumed indicator fungi in boreal forest: the importance of the landscape matrix. For Ecol Manage 174:353–363. doi:10.1016/S0378-1127(02)00043-9
Swift MJ (1977) The ecology of wood decomposition. Sci Prog 64:175–199
Szewczyk J, Szwagrzyk J (1996) Tree regeneration on rotten wood and on soil in old-growth stand. Vegetatio 122:37–46
Takahashi K (1997) Regeneration and coexistence of two subalpine conifer species in relation to dwarf bamboo in the understorey. J Veg Sci 8:529–536
Takahashi KH, Kagaya T (2005) Guild structure of wood-rotting fungi based on volume and decay stage of coarse woody debris. Ecol Res 20:215–222. doi:10.1007/s11284-004-0025-6
Takahashi M, Sakai Y, Ootomo R, Shiozaki M (2000) Establishment of tree seedlings and water-soluble nutrients in coarse woody debris in an old-growth Picea-abies forest in Hokkaido, northern Japan. Can J For Res 30:1148–1155
Tikkanen O-P, Martikainen P, Hyvärinen E, Junninen K, Kouki J (2006) Red-listed boreal forest species of Finland: associations with forest structure, tree species, and decaying wood. Ann Zool Fenn 43:373–383
Toljander YK, Lindahl BD, Holmer L, Högberg NOS (2006) Environmental fluctuations facilitate species co-existence and increase decomposition in communities of wood decay fungi. Oecologia 148:625–631. doi:10.1007/s00442-006-0406-3
Torgersen TR, Bull EL (1995) Down logs as habitat for forest-dwelling ants—the primary prey of pileated woodpeckers in northeastern Oregon. Northwest Sci 69:294–303
Torres JA, González G (2005) Wood decomposition of Cyrilla racemiflora (Cyrillaceae) in Puerto Rican dry and wet forests: a 13-year case study. Biotropica 37:452–456. doi:10.1111/j.1744-7429.2005.00059.x
Treseder KK, Allen MF (2000) Black boxes and missing sinks: fungi in global change research. Mycol Res 104:1282–1283. doi:10.1017/S0953756200229778
Ucitel D, Christian DP, Graham JM (2003) Vole use of coarse woody debris and implications for habitat and fuel management. J Wildl Manage 67:65–72
Unterseher M, Tal O (2006) Influence of small scale conditions on the diversity of wood decay fungi in a temperate, mixed deciduous forest canopy. Mycol Res 110:169–178. doi:10.1016/j.mycres.2005.08.002
Urcelay C, Robledo G (2004) Community structure of polypores (Basidiomycota) in Andean alder wood in Argentina: functional groups among wood-decay fungi? Aust Ecol 29:471–476. doi:10.1111/j.1442-9993.2004.01387.x
Uzunović A, O’Callahan D, Kreber B (2004) Mechanical tree harvesters spread fungal inoculum onto freshly felled Canadian and New Zealand pine logs. For Prod J 54:34–40
Vainio EJ, Lipponen K, Hantula J (2001) Persistence of a biocontrol strain of Phlebiopsis gigantea in conifer stumps and its effects on within-species genetic diversity. For Pathol 31:285–295
Vanderpoorten A, Sotiaux A, Engels P (2005) A GIS-based survey for the conservation of bryophytes at the landscape scale. Biol Conserv 121:189–194. doi:10.1016/j.biocon.2004.04.018
Vasiliauskas R, Vasiliauskas A, Stenlid J, Matelis A (2004) Dead trees and protected polypores in unmanaged north-temperate forest stands of Lithuania. For Ecol Manage 193:355–370. doi:10.1016/j.foreco.2004.01.048
Vasiliauskas R, Lygis V, Larsson K-H, Stenlid J (2005) Airborne fungal colonisation of coarse woody debris in North Temperate Picea abies forest: impact of season and local spatial scale. Mycol Res 109:487–496. doi:10.1017/S0953756204002084
Virolainen KM, Ahlroth P, Hyvarinen E, Korkeamaki E, Mattila J, Paivinen J, Rintala T, Suomi T, Suhonen J (2000) Hot spots, indicator taxa, complementarity and optimal networks of taiga. Proc R Soc Lond B 267:1143–1147. doi:10.1098/rspb.2000.1120
Wermelinger B (2004) Ecology and management of the spruce bark beetle Ips typographus—a review of recent research. For Ecol Manage 202:67–82. doi:10.1016/j.foreco.2004.07.018
Wesolowski T (2005) Virtual conservation: how the European Union is turning a blind eye to its vanishing primeval forests. Conserv Biol 19:1349–1358. doi:10.1111/j.1523-1739.2005.00265.x
Whalley AJS (1996) The xylariaceous way of life. Mycol Res 100:897–922
Wichmann L, Ravn HP (2001) The spread of Ips typographus (L.) (Coleoptera, Scolytidae) attacks following heavy windthrow in Denmark, analysed using GIS. For Ecol Manage 148:31–39. doi:10.1016/S0378-1127(00)00477-1
Woldendorp G, Keenan RJ (2005) Coarse woody debris in Australian forest ecosystems: a review. Aust Ecol 30:834–843. doi:10.1111/j.1442-9993.2005.01526.x
Woods CM, Woodward S, Pinard MA, Redfern DB (2006) Colonization of Sitka spruce stumps by decay-causing hymenomycetes in paired inoculations. Mycol Res 110:854–868. doi:10.1016/j.mycres.2006.02.007
Wormington KR, Lamb D, McCallum HI, Moloney DJ (2003) The characteristics of six species of living hollow-bearing trees and their importance for arboreal marsupials in the dry sclerophyll forests of southeast Queensland, Australia. For Ecol Manage 182:75–92. doi:10.1016/S0378-1127(03)00010-0
Yatskov M, Harmon ME, Krankina ON (2003) A chronosequence of wood decomposition in the boreal forests. Can J For Res 33:1211–1226
Yin XW (1999) The decay of forest woody debris: numerical modeling and implications based on some 300 data cases from North America. Oecologia 121:81–98. doi:10.1007/s004420050909
Zhong JW, van der Kamp PJ (1999) Pathology of conifer seed and timing of germination in high-elevation subalpine fir and Engelmann spruce forests of the southern interior of British Columbia. Can J For Res 29:187–193
Zielonka T, Piatek G (2004) The herb and dwarf shrubs colonization of decaying logs in subalpine forest in the Polish Tatra Mountains. Plant Ecol 172:63–72. doi:10.1023/B:VEGE.0000026037.03716.fc
Acknowledgments
Many thanks to S. Bertolo, T. Coch, I. Currado, J. F. Dobremez, A. Fischlin, K. J. Gaston, E. Lange, M. Haywood, M. Jeger, H. Keene, M. Lauerer, L. Paul, R. Smith, J.-P. Sorg, P. H. Warren, P. J. Weisberg, R. Whittaker and R. Winkler for discussing ideas contained in this review, and to two anonymous reviewers for helpful comments on a previous version of the draft.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Rainer Matyssek.
Rights and permissions
About this article
Cite this article
Lonsdale, D., Pautasso, M. & Holdenrieder, O. Wood-decaying fungi in the forest: conservation needs and management options. Eur J Forest Res 127, 1–22 (2008). https://doi.org/10.1007/s10342-007-0182-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-007-0182-6