Abstract
The behavior of five apricots grafted across Blackthorn inter-stem (BI) on Myrobalan stock and on Myrobalan rootstock (MR) was evaluated through vegetative growth, tree mortality, productivity, fruit quality and leaf nutrient composition from the 1st to the 6th leaf. The trial was performed in the Prislonica village (Cacak, Serbia), on a dry, shallow, sandy-loam and acidic soil. Results showed that MR improved tree vigor, yield per tree, cumulative yield, fruit weight, flesh rate, acidity, antioxidant power, leaf N, K, Ca and Mn contents and better balanced nutritional values for macronutrients. BI increased tree mortality, yield efficiency, yield per hectare, total phenolic and total flavonoid contents, and leaf P and Fe amounts. Wide variability among cultivars for evaluated traits was observed on both BI and MR. The more vigorous trees had ‘Biljana’ as compared with others on both BI and MR, whereas the most productive cultivar was ‘Vera’ on BI and ‘Biljana’ on MR. The highest and similar fruit weight had ‘Roxana’ and ‘Harcot’, whereas the highest stone weight and flesh rate was found in ‘Vera’ and ‘Harcot’, respectively, all in both BI and MR. Fruits harvested from ‘Aleksandar’ trees were rich in soluble solids content (SSC), titratable acidity (TA) and total flavonoid content (TFC), ‘Harcot’ in total phenolic content (TPC), and ‘Roxana’ in acidity and total antioxidant capacity (TAC), also on both BI and MR. The better balanced nutritional values were promoted by BI. Regarding cultivars, the best balanced nutritional values for macronutrients were exhibited by ‘Vera’ on BI, and by ‘Harcot’ on MR. For micronutrients, the wider imbalance among nutrients was found in ‘Roxana’ on BI, and in ‘Vera’ on MR.
Zusammenfassung
Das Verhalten von fünf Aprikosensorten, die auf Prunus myrobalana mit Zwischenveredlung Schlehe (BI) und direkt auf Prunus myrobalana (MR) veredelt worden waren, hinsichtlich vegetativem Wachstum, Baumsterblichkeit, Ertragsleistung, Fruchtqualität und Ernährungszustand wurde vom ersten bis zum sechsten Laub überprüft. Der Versuch wurde in Prislonica (Cacak, Serbien) auf einem trockenen, flachgründigen, sandig-lehmigen und sauren Boden durchgeführt. Die Ergebnisse zeigten, dass die Verwendung der Unterlage Prunus myrobalana die Wuchsstärke des Baumes, den Baumertrag, den kumulativen Ertrag, das Fruchtgewicht, den Fruchtfleischanteil, die Säure, das antioxidative Potential und den Blattgehalt der Hauptnährstoffe N, K, Ca und Mn verbessert und zu einem besser ausgewogenen Verhältnis der Makronährstoffe beiträgt. BI erhöhte die Baumsterblichkeit, den spezifischen Ertrag, den Ertrag pro Hektar, die Gesamt-Phenol- und Flavonoid-Gehalte und den Blattgehalt bezüglich P und Fe. Zwischen den Sorten wurde bei den erfassten Merkmalen sowohl bei BI als auch bei MR eine große Schwankungsbreite festgestellt. Verglichen mit den anderen Sorten brachte ‘Biljana’ sowohl auf BI als auch auf MR die wüchsigsten Bäume hervor, wohingegen ‘Vera’ den höchsten Ertrag auf BI und ‘Biljana’ den höchsten Ertrag auf MR erbrachte.
Unabhängig von der Unterlage zeigten sich folgende Ergebnisse: Das höchste Fruchtgewicht hatten, mit geringem Unterschied, die Sorten ‘Roxana’ und ‘Harcot’, während ‘Vera’ den größten Stein-Anteil und ‘Harcot’ den höchsten Fruchtfleischanteil aufwies Früchte, die von der Sorte ‘Aleksandar’ geerntet wurden, waren reich an löslicher Trockensubstanz (SSC), titrierbarer Säure (TA) und absolutem Flavonoid-Gehalt (TFC). Die Sorte ‘Harcot’ hatte einen hohen Gesamt-Phenol-Gehalt und ‘Roxana’ war reich an Säure und hatte ein hohes antioxidatives Potential.
Von BI wurde im Allgemeinen ein besser ausgewogenes Nährstoffverhältnis gefördert. Wenn man die Sorten betrachtet, war das am besten ausgewogene Verhältnis der Makronährstoffe bei ‘Vera’ auf BI und bei ‘Harcot’ auf MR zu finden. Bei ‘Roxana’ auf BI und bei ‘Vera’ auf MR gab es die größten Schwankungen bei den Mikronährstoffen.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
As known, grafts are often used in agriculture to reduce the time to flowering and shorten the breeding program, improve quality and yield of seeds and fruits, improve hardiness in response to environmental conditions and pathogens resistance and change cultivars (Darikova et al. 2011). This is an ancient cloning method that has been used widely for thousands of years. So, most of fruit species and its cultivars in fruit industry, including apricot, are grafted on different rootstocks. Among others, the main factors which determined ideal rootstock are its compatibility with scion cultivar, resistance and/or tolerance to pests and diseases, adaptability to wide range of soil types and climatic conditions (Webster 1995), and also should improve one or more of the following characteristics: cold tolerance, harvest date, internal and external fruit quality, yield, and post-harvest fruit quality (Castle 1995).
Several studies worldwide demonstrated impact of rootstock on apricot tree growth (Southwick and Weis 1998; Basile et al. 2003), yield attributes (Southwick and Yeager 1999; Egea et al. 2004; Sosna and Licznar–Małańczuk 2012), fruit weight and size (Son and Küden 2003; Hernández et al. 2010), fruit chemical composition (Scalzo et al. 2005), physiological parameters (Shackel et al. 1997), leaf nutrient status (Rosati et al. 1997; Velemis et al. 1999), tree survival (Licznar–Małańczuk and Sosna 2005; Sitarek and Bartosiewicz 2011), etc.
However, the controversies regarding rootstocks for the apricot speak of the very complex nature of this problem and the need to study it with a view to establishing the most appropriate rootstocks for each or group of apricot cultivar(s) in different agro-ecological conditions (Son and Küden 2003).
The inter-stem is a part of a tree trunk that it is grafted between the rootstock and the scion. It has been used with different fruit species to modulate tree size, tree survival, alleviate incompatibility, fruit production and quality, and aging of the tree (Vachůn 1983; Gil-Izquierdo et al. 2004). However, the trees grafted with inter-stem are more expensive, and grafting delays the production phase.
As known, rootstocks derived from plums are more tolerant to compact soils and waterlogging than other species of Prunus L., a fundamental reason for their use as rootstocks for apricots. In the most apricot growing-area worldwide (Poland, Russia, Romania, Hungary, France, Czech Republic, Switzerland, Bulgaria, Serbia, etc.), the most popular rootstock for apricots are seedlings of Myrobalan (P. cerasifera Ehrh.), which is too vigorous for high soil fertility and, thus, not adapted for high-density planting (Monney et al. 2010; Sitarek and Bartosiewicz 2011). Also, grafting incompatibility sometimes can be observed between Myrobalan and apricots (Licznar–Małańczuk and Sosna 2005).
In past few decades, in new Serbian apricot orchards, Blackthorn (P. spinosa L.) as inter-stem between Myrobalan stocks and apricots is used to decrease the thickness of the trunk at the grafting point in order to suppress tree growth and increase number of trees per unit area (Djuric and Keserovic 1999). However, influence of Blackthorn inter-stem on apricots behavior has been poorly studied yet.
For these reasons, the main objective of this study was focused on the influence of Blackthorn inter-stem and Myrobalan rootstocks on vegetative growth, tree mortality, productivity, fruit quality parameters and leaf nutrient composition in five apricots grown under Serbian conditions on dry, shallow and acidic soil from the 1st to the 6th leaf.
Material and Methods
Trial Design and Weather Conditions
This study was conducted in private orchard in Prislonica village (43º53′ N, 20°21′ E, 340 m a.s.l.), Cacak region in western Serbia from 2008 to 2013. The trial was established in the spring 2008 with five apricot cultivars, three new Serbian (‘Aleksandar’, ‘Biljana’, ‘Vera’), one Canadian (‘Harcot’), and one Afgan (‘Roxana’). These cultivars were comprised in two sub-trials at the same orchard. In the first (BI), cultivars were grafted across Blackthorn inter-stem (P. spinosa L.) on Myrobalan stock (P. cerasifera Ehrh.), and planted at 5 × 2 m (1000 trees ha−1); in the second (MR), cultivars were grafted directly on Myrobalan seedlings rootstock and planted at 5.5 × 3 m (606 trees ha−1). In both cases, trunk height was between 60 and 70 cm. The six representative trees of each rootstock-cultivar or rootstock-inter-stem-cultivar combination in four replicates (n = 24) were selected for sampling and data collection.
Orchard was established at early spring of 2008; training system was open vase with three primary boughs. Row orientation is east-west. Plantation was managed following the standard procedures, except irrigation. Orchard was fertilised with 500 kg NPK ha−1 (15:15:15) complex mineral fertiliser in autumn and with 300 kg calcium ammonium nitrate (CAN) ha−1 [N mineral fertiliser containing 27 % of N total (NTOT)] in early spring every year, respectively, starting in 2010 (3rd leaf). Cattle manure in amount of 50 t ha−1 was added to soil in late autumn 2007, i.e. before planting. Tree growth is controlled by early summer pruning between 15 June and 15 July. Pruning and harvesting are performed without the use of ladders because they are small trees.
Soil analyses were done prior to the experiment. The orchard has a sandy-loam soil texture with 1.62 % organic matter and low soil pH (4.86) in 0–30 cm soil depth. Contents of NTOT, available P2O5 and K2O, CaO and MgO are 0.16 %, 178 µg g−1, 220 µg g−1, 0.39 % and 6.2 µg g−1 on dry matter basis, respectively. Also, soil contains 78 µg g−1 Fe, 7.8 µg g−1 Mn, 1.6 µg g−1 Cu, 0.52 µg g−1 Zn and 2.3 µg g−1 B. Hence, soil is rich source in P2O5, K2O and Fe, whereas other macro- and micronutrients are in a medium to low range (Ankerman and Large 1977).
The long-term (1965–2010) average annual temperature was 11.3 °C, with an average air temperature during growing cycle of 17.0 °C, and total annual precipitation was 690.2 mm. In the period April–October from 2009 to 2013, mean monthly air temperatures were considerably higher than long-term averages, while rainfall had lower values in general, especially in July and August.
Tree Growth and Yield Characteristics
Trunk circumference was measured during the end of vegetative cycle from 2008 to 2013 at 20 cm above the ground level using caliper gauge Sttarret 727 (Athol, MA, USA), and the trunk cross-sectional area (TCSA, cm2) was calculated. Yield per tree (kg), cumulative yield per tree (kg) and yield efficiency (total cumulative yield per final TCSA, kg cm−2) of each sub-trial were computed from the harvest data from 2009 to 2013. Yield per hectare (t) was calculated. Yield was measured with ACS System Electronic Scale (Zhejiang, China). The percentage of dead trees was determined every year at the end of growing cycle.
Fruit Quality Analysis
The analyzed fruits were sampled in 2012 and 2013 at commercial maturity stage (fully-colored). Fruits from each cultivar on both BI and MR were randomly harvested from six trees in four replicates (n = 24), and 50 representative apricots were processed for all the physical and chemical analysis. Apricots were harvested from different parts of the trees to avoid fruit position effect (Taylor et al. 1993).
Fruit and stone weights (g) were measured using a digital scale FCB 6 K (Kern & Sohn GmbH, Belingen, Germany). Pulp yield or flesh rate (%) was calculated as [(fruit weight − stone weight)/fruit weight] × 100. Flesh firmness (kg cm−2) was determined with a penetrometer Bertuzzi FT-327 (Facchini, Alfonsine, Italy) fitted with an 8 mm cylindrical plunger at three equatorial positions on each fruit at 120°.
After assessment of flesh firmness, fruit juice was extracted by homogenising fruit flesh in a blender. Soluble solids content (SSC, °Brix) of the juice were measured in each fruit with a hand refractometer Milwaukee MR 200 (ATC, Rocky Mount, USA). Titratable acidity (TA, % of malic acid) was determined using an automatic titration apparatus Titrino 719 S (Metrohm, Herisau, Switzerland) with 0.1 mol L−1 NaOH up to pH 8.1. The SSC/TA ratio or maturity index was determined.
The content of bioactive compounds (TPC, TFC and TAC) analyzed using UV–visible spectrophotometer MA9523-SPEKOL 211 (Iskra, Horjul, Slovenia). The TPC was determined with Folin–Ciocalteu’s phenol reagent according to the method of Singleton and Rossi (1965) with some modifications, using gallic acid as a standard. Fruit TPC was expressed as mg gallic acid equivalent (GAE) g−1 dry weight (dw). The TFC was determined colorimetrically as described previously (Brighente et al. 2007). Fruit TFC was determined from a rutin standard curve and expressed as mg rutin equivalent (RUE) g−1 dw. The TAC was evaluated by the phosphor-molybdenum method (Prieto et al. 1999). Ascorbic acid (AA) was used as standard and the TAC is expressed as milligrams of AA per gram of dw (mg AA g−1 dw). Content of bioactive compounds are expressed as means ± SE of triplicate analyses per each sub-trial treatment for 2012 and 2013.
Leaf Nutrient Analysis
Leaf samples (approximately 100 leaves per each treatment) for macro- and micronutrients content analyses were collected from the middle parts of non-bearing shoots from current season of the evaluated cultivar trees on each BI and MR treatment at 120 DAFB in 2012 and 2013. Each nutrient was determined according to routine laboratory procedures using adequate equipments (Milosevic and Milosevic 2011a, b).
The DOP index (deviation from optimum percentage) was assessed for the diagnosis of the nutritional status of trees (Montañés et al. 1993). The DOP index was calculated from leaf chemical analysis by applying the following general formula:
where: C n = foliar content of the tested nutrient, and C o = critical optimum nutrient content for apricot; both values given on a dry matter basis. The C o has been taken from optimum values for apricot proposed by Leece and van den Ende (1975). Besides, it provides the general nutritional status of nutrients through the ƩDOP index, obtained by adding the values of DOP indices irrespective of sign. The lower the ƩDOP, the greater is the intensity of balance among nutrients.
Statistical Analysis
Data were subjected to analysis of variance (ANOVA) and means were separated by LSD significant difference test (P ≤ 0.05), when F test indicated significant variable effects using the Microsoft Excel software package (Microsoft Corporation, Roselle, IL, USA).
Results and Discussion
Tree Growth and Productivity
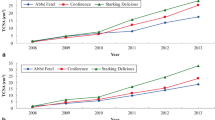
Tree growth, as measured by TCSA, significantly differed between BI and MR, starting from 3rd to 6th leaf (Fig. 1). On both BI and MR, the most intensive tree growth rate was exhibited by ‘Biljana’. In the case of BI, ‘Roxana’ had much lower intensity of tree growth than ‘Biljana’, followed by ‘Vera’, ‘Harcot’ and ‘Aleksandar’ (Fig. 1a). On MR, after ‘Biljana’, intensive tree growth was found in ‘Aleksandar’, followed by ‘Vera’, ‘Roxana’ and ‘Harcot’ (Fig. 1b).
In the first 2 years of trial, impact of rootstock and inter-stem on TCSA was similar and there were no differences between BI and MR and among cultivars at the same rootstock or inter-stem. However, in the next years, especially in 2013, differences were significant (Table 1). BI reduced tree vigor by about 19 % than MR. This result showed that apricots grafted across BI on Myrobalan stock grows more slowly when compared with apricots directly grafted on Myrobalan rootstock. Hence, apricots on BI can be planted at shorter distances apart, resulting in reduced tree growth, although Guerriero et al. (1986) mentioned that the closest spacing did not always reduce tree vigor.
Final TCSA values showed that invigorating MR was related to the expected apricot tree vigor (Son and Küden 2003; Sitarek and Bartosiewicz 2011), whereas BI suppressed tree growth (Djuric and Keserovic 1999). Some authors reported that, among others, wider planting distance generally improved apricot tree growth (Guerriero and Scalabrelli 1989; Kumar et al. 2013). In this case, tree growth was higher and hence gave more stem girth due to light competition among trees which was smaller when compared with closer spacing apparently due to an adequate supply of photosynthates and metabolites (Restrepo-Díaz et al. 2010). Several reports have documented that in grafted trees, the control of plant size is mainly exerted across the rootstock, although the mechanism by which the rootstock regulates scion vigor has not been identified (Basile et al. 2003). Therewith, rootstocks generally affect tree water relations because trees grafted on invigorating rootstocks have consistently higher stem water potentials than trees grafted on dwarfing rootstocks (Rosati et al. 1997). In addition, Blackthorn being a less vigorous, i.e. dwarf inter-stem with a high capacity to the control tree growth (Djuric and Keserovic 1999), probably through reducing of water potential and other physiological procesess (Shackel et al. 1997). This may also be due to some form of graft-incompatibility between BI and some scion cultivars which occurred in our trial, and manifested with a high graft union swelling (Fig. 2a). Graft union in MR manifested with a slight swelling (Fig. 2b). Therefore, it is necessary to carry out selection of Blackthorn in order to find types with good compatibility with apricot cultivars (Palk 1984).
Graft union of apricot grafted across Blackthorn inter-stem on Myrobalan stock (a) and directly on Myrobalan rootstock (b). In the first case (a), graft union is indicated by a high swelling between scion and inter-stem, and also between inter-stem and stock. In the second case (b), graft union is indicated by a slight swelling between scion and rootstock
Regarding cultivars, ‘Biljana’ gave more vigorous trees than other four cultivars in both BI and MR. These results are expected and in accordance with previous works on apricot, indicating strong cultivar effect on this trait (Velemis et al. 1999; Kumar et al. 2013). At this connection, the enumerated cultivars in general, except ‘Biljana’, are more suitable for dense planting. However, behavior of some apricots in both BI and MR was not consistent. For example, ‘Aleksandar’ on MR had high vigor, similar to ‘Biljana’, whereas on BI had the lowest.
In the first bearing year (2009) yields per tree were minor (~ 1.5 kg), and there were no statistically significant differences between MR and BI and among cultivars at the same graft combination (Fig. 3). During the next cropping years (2010–2013), the yields considerable increased on both MR and BI, and differences between them were significant (data not shown, except for 2013).
On BI, yields of ‘Vera’ and ‘Biljana’ constantly increased during above period (Fig. 3a). Yields of ‘Roxana’ and ‘Harcot’ had maximum values in 5th leaf, and decreased in 6th leaf. Yield of ‘Aleksandar’ increased from 2nd to 4th leaf, decreased in 5th leaf, and again increased in last year of trial. In the case of MR, yields of all cultivars also increased from 2010 to 2011, and decreased in next year (2012), except for ‘Harcot’, with significant differences among cultivars (data not shown) (Fig. 3b). In 2013, yields of ‘Aleksandar’, ‘Biljana’ and ‘Vera’ increased. Yields of ‘Roxana’ and ‘Harcot’ decreased from 4th to 6th leaf and from 5th to 6th leaf, respectively.
Generally, our data for yields tendency are in accordance with results of Guerriero et al. (1986) who reported that apricots started to produce in the second year of growing and was continually productive also in the third and fourth years. On the other hand, irregularity of yield, occurred in our study, is one of the main handicaps in apricot fruit production (Bassi 2001), and tended to be very high in some years and very low in the following year (Vachůn 2001), which confirmed our results. The reasons for this phenomenon are numerous, and have been shown to be due to different problems concerning climatic adaptation (winter killing of flower buds, blossoms killed by spring frosts, high crop load in a previous season, etc.) and flower bud development (Egea et al. 2004). Data from Table 1 showed that final yield per tree and hectare, cumulative yield and yield efficiency significantly varied between both MR and BI and among cultivars grafted on them. MR induced higher yield per tree and cumulative yield (Sitarek and Bartosiewicz 2011), whereas BI induced higher yield per hectare and yield efficiency (Djuric and Keserovic 1999). On this way, Guerriero and Scalabrelli (1989) noted that increasing tree density resulted in lower yield per tree, but increased yield per ha during the early years of cropping, which partially confirmed our results.
Regarding cultivars, ‘Vera’ on BI had higher final yield per tree, cumulative yield and yield per unit area than others. The better yield efficiency was exhibited by ‘Aleksandar’ as compared to others due to its high productivity and lower TCSA, whereas the lowest yield per tree, cumulative yield and yield per ha was observed in ‘Roxana’. Contrary to our results, some authors reported that ‘Roxana’ grafted across BI on Myrobalan stock gave the highest yield per tree and hectare as compared with other cultivars (Djuric and Keserovic 1999). Growing conditions and cultural practices also could account for these differences.
Behavior of cultivars grafted on MR is different as compared with BI. ‘Vera’ on BI and ‘Biljana’ on MR had the highest final yield per tree and per hectare, whereas ‘Roxana’ and ‘Harcot’ on both BI and MR had the lowest. In addition, ‘Biljana’ is the cultivar with the lowest cumulative yield and yield efficiency, probably due to its high tree vigor. Strong impact of apricot genotype on tree productivity was previously reported (Vachůn 2001).
Tree mortality
During the first 6 years after planting, we observed apricot tree mortality on both inter-stem and rootstock treatments (Fig. 4), being higher on BI (16.67 %) than on MR (10.83 %). This phenomenon was more intensive in last year of trial (2013), indicating some form of delayed graft-incompatibility (Hernández et al. 2010). Licznar–Małańczuk and Sosna (2005) also reported lower trees mortality on Myrobalan seedlings than on Somo seedlings. However, high mortality of apricots on Myrobalan may also occur (Southwick and Weis 1998; Bassi 2001) as a result of incompatibility between scion and rootstock. Contrary, Dimitrova and Marinov (2002) never detected graft incompatibility in apricots grafted on Myrobalan seedling rootstock under Bulgarian environment. On the other hand, too much of a dwarfing effect along with the relative high mortality of trees in our trial can be a sign that there is an incompatibility between apricots and BI (Palk 1984). It seems that usefulness of unproved BI for apricot is limited due to the relatively high tree mortality, i.e. short life span of trees.
Behavior of cultivars regarding tree mortality was different. Unacceptable, three Serbian cultivars (‘Aleksandar’, ‘Biljana’ and ‘Vera’) had lower capacity to control tree survival as compared with Canadian cultivar ‘Harcot’, especially with Afgan cultivar ‘Roxana’ from Irano-Caucasian eco-geographical group. The highest tree mortality on both BI and MR was observed in ‘Biljana’ (high vigor), and the lowest in ‘Roxana’ (less vigor). Due to the different cultivars evaluated, our results are not always comparable to the literature. So, according to Djuric and Keserovic (1999), survival of nine apricot genotypes (including ‘Roxana’) on BI during the first 6 years after planting was more than 90 % of all planted trees in most cultivars, whereas Vachůn (1983) reported that some apricot cultivars as an inter-stem for ‘Velkopavlovická’ cultivar induced their tree mortality, about 5 % per year. In an earlier study, the mortality rate of ‘Harcot’ on Myrobalan at 8th year after planting was 10 % (Licznar–Małańczuk and Sosna 2005), whereas Sitarek and Bartosiewicz (2011) noted tree mortality of ‘Morden 604’ and ‘Miodowa’ apricots at trees age like ours between 66.7 and 87.5 % in average.
Fruit Physical Characteristics
There were significant differences in fruit weight and flesh rate at harvest between MR and BI, being higher in the case of MR, whereas differences in stone weight and flesh firmness were not significant (Table 2). Effect of rootstock (Hernández et al. 2010; Sitarek and Bartosiewicz 2011) or inter-stem (Webster 1995) on fruit physical traits was previously reported. Similarly, Kumar et al. (2013) reported that wider spacing which coincides with MR in our study, induced better above traits when compared with closer spacing, which partially confirmed our results.
Significant differences among cultivars on BI or MR for all evaluated fruit physical attributes were observed (Table 2), and behavior of cultivars is consistent in both MR and BI treatments. Hence, ‘Roxana’ and ‘Harcot’ are the cultivars with the highest and similar fruit weight, whereas the lowest was found in ‘Vera’ in general. Contrary, this cultivar had the highest stone weight and, logically, the lowest flesh rate. Stone weight of ‘Aleksandar’ and ‘Harcot’ is the lowest and statistically similar. The best flesh rate was observed in ‘Harcot’, whereas the poorest flesh firmness had ‘Aleksandar’. Discrepancy of cultivars behavior was observed for flesh firmness because ‘Biljana’ on BI and ‘Roxana’ on MR had the best values. Moreover, cultivars with higher flesh firmness had a better resistance of fruits to post-harvest damage.
Generally, fruit weight was affected by crop load, and there was a correspondence between low yield and large fruit weight (Egea et al. 2004), which confirmed our results. Previous works on apricot also reported a high variability among cultivars regarding fruit weight, flesh rate and flesh firmness (Vachůn 2003; Leccese et al. 2010), indicating strong genotype depending traits. Otherwise, larger apricot fruits are commonly preferred by consumers (Hegedüs et al. 2010). In this study, all cultivars showed flesh firmness values between 3.0 and 0.5 kg cm−2, suitable for consumers and the apricot industry (Cemagref 1981).
Fruit Chemical Characteristics
There were significant differences between BI and MR concerning the percentage TA, whereas their impact on SSC and maturity index was not significant (Table 3). According to Monastra and Salvador (1995), Myrobalan GF-31 gave good results from the point of view of fruit quality and yield in South Italy. The impacts induced by rootstocks (Sosna and Licznar–Małańczuk 2012) and/or inter-stems (Djuric and Keserovic 1999) on controlling apricot tree growth and productivity are well known. However, influence on internal fruit quality is not always clear. So, Hernández et al. (2010) reported that rootstocks had no influence on TA and SSC/TA ratio, but influenced SSC, whereas Sitarek and Bartosiewicz (2011) noted that SSC was not depending on the rootstocks. Controversies regarding rootstock or inter-stem influence on SSC and acidity found by different authors indicated that this segment of apricot trees was not main factor which determined level of these phytochemicals and their ratio (Castle 1995; Webster 1995). Nevertheless, Monastra and Salvador (1995) concluded that Myrobalan GF-31 gave good results from the point of view of fruit quality in South Italy.
Similarly to fruit physical traits, the SSC, TA and maturity index significantly varied among cultivars and their behavior is generally consistent on both BI and MR (Table 3). Fruit of ‘Aleksandar’ is particularly rich in SSC (consisting mostly of sugars) and had the highest maturity index value, whereas fruit of ‘Roxana’ contains higher acidity as compared to others. ‘Biljana’ and ‘Vera’ grafted on MR had similar SSC to ‘Aleksandar’. The lowest SSC had ‘Harcot’, whereas the lowest acidity had ‘Aleksandar’ on BI and ‘Vera’ on MR. From this point, fruits of ‘Roxana’ and ‘Harcot’ are good for diabetic patients as it contains low sugar content (Dauchet and Dallongeville 2008). However, Kumar et al. (2013) found that ‘Harcot’ had higher SSC/TA ratio when compared with some Indian apricots. These discrepancies for same cultivar can be explained with fact that, beside genotype, SSC and SSC/TA ratio depends on the fruit maturity stage, position in canopy, cultural practices, cultivation system, season (year) and also weather conditions (Taylor et al. 1993; Leccese et al. 2010). Additionally, malic acid has been suggested the major organic acids component of apricot flesh fruit (Gurrieri et al. 2001), whereas values of SSC > 12°Brix are representative of an excellent gustative quality in some fresh apricots, plums, nectarines and peaches (Crisosto et al. 2006), which is the case in our study.
As known, fruits of apricot are high source of certain phytochemicals that have beneficial effects on human metabolism. Among these compounds, TPC, TFC and TAC are the most important (Leccese et al. 2010; Hegedüs et al. 2010). In the present study, BI significantly promoted TPC and TFC, whereas MR improved antioxidant power (Table 4). Several authors reported that, among others, bioactive compounds depend on the rootstock (Scalzo et al. 2005) or inter-stem (Gil-Izquierdo et al. 2004). In general, our data can be connected with results of Font i Forcada et al. (2014) who reported that most vigorous and high-yielding rootstocks have been shown to induce lower fruit quality, including bioactive compounds, probably due to the higher strength of vegetative growth versus fruit quality. However, in a study of Scalzo et al. (2005), invigorating Myrobalan 29 C rootstock induced higher TPC and TAC than other less vigorous rootstocks. Discrepancies among our results and data of different authors can be explained with fact that influence of same rootstock on scion in different environmental conditions have not always been similar (Hernández et al. 2010) or positive (Darikova et al. 2011).
On both BI and MR, differences among cultivars for TPC, TFC and TAC were significant, and their behavior is consistent (Table 4). The average of the last 2 years of analysis shows that fruits of ‘Harcot’ and ‘Aleksandar’ contained the highest TPC and TFC, respectively, whereas ‘Roxana’ is the cultivar with the highest antioxidant power, all on both BI and MR. In contrast, ‘Roxana’ had the lowest TPC, while ‘Harcot’ and ‘Aleksandar’ had the lowest TFC and TAC, respectively. Also, ‘Harcot’ on BI had similar antioxidant power as compared to ‘Aleksandar’. This is consistent with the reports of previous studies, indicating that bioactive compounds of apricot depends mainly on the scion genotype (Scalzo et al. 2005), and also may be influenced by lesser important factors such as maturity stage, length of the fruit period development, cultivation system, climatic conditions (Hegedüs et al. 2010; Leccese et al. 2010) and, of course, by rootstock (Scalzo et al. 2005) or inter-stem (Gil-Izquierdo et al. 2004). It seems that different chemical traits observed in five apricots suggest that regulation of fruit quality mainly depended on cultivar per se, although different size-controlling rootstock and/or inter-stem with different planting distance also plays an important role in fruit chemical composition (Kumar et al. 2013).
Leaf Chemical Composition
Ten essential macro- and micronutrients were determined in apricot leaves at 120 DAFB; their amounts are presented in Tables 5 and 6. According to the results obtained, their levels significantly varied between BI and MR. However, differences among cultivars on BI or MR in the most cases, especially for micronutrients amount, were not significant.
Regarding leaf macronutrients, leaf N, K and Ca contents were higher on MR, whereas P content was higher on BI (Table 5). Difference between BI and MR for leaf Mg was not significant. It could be said that root system of Myrobalan has a better uptake capacity for the most of macronutrients (Southwick and Weis 1998). Contrary, Blackthorn as a dwarf inter-stem reduces the ability of the Myrobalan stock to adopt these elements, probably due to membrane selectivity of its graft union, so it can be the primary reason for having lower nutrients content (Marschner 1995; Velemis et al. 1999). On this line, BI may have reduced N, K, Ca, probably Mg uptake capacity and, as a consequence, may require more careful fertilizer management than MR in soil conditions like ours. The differences among rootstocks nutrient uptake were previously described (Rosati et al. 1997).
Behavior of cultivars on BI and MR was inconsistent (Table 5), showing a very complex accumulation pattern of macronutrients in apricot trees (Leece and van den Ende 1975). On BI, leaf N and K contents in cultivars were statistically similar. On the other hand, the largest variation among cultivars occurred for leaf P, Ca and Mg. Thus, leaf of ‘Biljana’ contained the highest levels of P and Ca, and the lowest Mg amount, whereas other four cultivars contained the highest level of Mg with no significant differences among them. The lowest leaf P and Ca was observed in ‘Vera’ and ‘Aleksandar’, respectively.
On MR, differences among cultivars for leaf Mg were not significant, whereas differences for other macronutrients were found. Hence, leaf of ‘Aleksandar’ contained the highest levels of N and K, whereas ‘Vera’ and ‘Biljana’ contained the highest leaf P and Ca, respectively. ‘Roxana’ and ‘Vera’ are the cultivars with the lowest and similar leaf N. The lowest leaf P, K and Ca amounts were observed in ‘Harcot’, ‘Biljana’ and ‘Aleksandar’, respectively. Cultivar effect on macronutrients content in apricot leaf can be explained with the genetic effect leading to different nutrient uptake capacity, i.e. better mineral uptake efficiency of scion cultivars (Rosati et al. 1997; Velemis et al. 1999).
The present study showed that some leaf micronutrients level significantly differed between BI and MR (Table 6), although several authors reported that leaf micronutrients amount was less influenced by rootstock than macronutrients (Moreno et al. 1996). BI promoted leaf Fe and Zn levels, whereas MR improved leaf Mn. Differences between BI and MR for leaf Cu and B amounts were not significant. On this line, it can be said that rootstock and/or inter-stem used have different uptake capacity of some micronutrients, as previously reported (Rosati et al. 1997; Milosevic and Milosevic 2011a).
Regarding behavior of cultivars on BI or MR, we found significant differences only for leaf Mn and Cu in the first case and for leaf Fe in the second (Table 6). So, ‘Harcot’, ‘Vera’ and ‘Aleksandar’ on BI expressed similar and higher leaf Mn when compared with ‘Roxana’ and ‘Biljana’. Moreover, ‘Harcot’ on MR had the highest leaf Cu, while ‘Roxana’ and ‘Aleksandar’ had the lowest, and with no significant differences between them. ‘Biljana’ and ‘Vera’ accumulated an intermediate level of Cu and were different from all other cultivars. On both BI and MR treatments, differences among cultivars for leaf Zn and B levels were not found (Velemis et al. 1999). Contrary, in our earlier study (Milošević and Milošević 2011b) we found that seven inter-stems originated from local and introduced plums significantly modulated leaf micronutrients level in ‘Vera’ apricot. These differences may be connected with very complex nature of physiological rootstock–scion or stock–inter-stem–scion relationship, as previously reported (Güleryüz et al. 1996; Rosati et al. 1997).
Deviation from Optimum Percentage (DOP) and ƩDOP Index
To assess the optimum mineral nutrition, the DOP index was determined from apricot leaf mineral elements at 120 DAFB. There was a good relationship between the nutritive state of plant and soil (Montañés et al. 1993).
Data from Table 7 expressed that leaf nutrients level in both experimental trial, except leaf P, were lower then optimum according to reference values proposed by Leece and van den Ende (1975) for apricot trees. According to above authors, the negative DOP indexes (DOP < 0) indicate a general problem with soil availability of these nutrients or with nutrient uptake. The excessive leaf P content observed on both BI and MR and in all cultivars was attributed to an excessive P fertilization and its high content in the soil. On this line, fertilization with P in similar soil conditions is very harmful because its excess can cause antagonism with other nutrients, i.e. block Mg, Fe, Zn and Cu (Johnson and Uriu 1989). From this point, an adequate P management implies monitoring the available soil P level. According to Sánches-Rodrígues et al. (2013), the P fertilization should take into account soil P level and crop P uptake in order to reduce the risk of environmental pollution and production costs.
Deficiency N indicative of N:P, N:K and/or N:Mg imbalances, as previously reported (Leece and van den Ende 1975). Interestingly, in apricots on MR, leaf N amount were slightly higher than optimum in ‘Aleksandar’ and slightly lower in others (DOPN ≈ 0). Moreover, soil in our trial is acidic, sandy-loam, clean texture due to the high stakes of sand fractions inclined to high losses soil N. It seems that N fertilization in this orchard requires a new program because unbalanced N fertilization can lead to soil pH decrease (Milošević and Milošević 2011b). Although soil contains high amount of available K2O, leaf K content is lower than normal (DOPK < 0), except in leaf of ‘Aleksandar’ on MR (DOPK > 0). This is probably due to its unavailability in the soil and/or due to its fixation by clay particles (Johnson and Uriu 1989). Also, high content of available elements in the soil did not always result in high nutrient uptake by plant due to the imbalance of elements (Leece and van den Ende 1975). Also, above authors reported that Myrobalan stock, unlike others such as apricot and peach stock, does not readily accumulate K. High deficiency of Ca in the present study is generally induced by acidic soil, its low content in the soil and antagonism with some element such as Mg, since Ca counteracts the potential harmful effects of Mg and other divalent cations (Shear 1975). Also, immobility of Ca in plant tissues and its very low redistribution to other plant tissues and organs is well known (Marschner 1995). Moreover, Mg deficiency developed on trees in the high P treatments (Taylor and Goubran 1975), which is case in our study.
ƩDOP index for macronutrients significantly varied between BI and MR and among cultivars at the same treatment (Table 7). So, invigorating MR showed better balanced nutritional values then BI, probably due to its better mineral uptake efficiency. This confirms good adaptation of MR without inter-stem to shallow, dry, sandy-loam and acidic soils (Son and Küden 2003).
In the case of cultivars on BI, ‘Vera’ showed the best balanced nutritional values, whereas in ‘Biljana’ we found the wider imbalance in nutritional values for macronutrients. Contrary to behavior on BI, ‘Vera’ on MR showed a wider imbalance in nutritional values, whereas the best balance among macronutrients was observed in ‘Harcot’. Generally, cultivars with good balanced nutritional values showed the best yield efficiency, probably due to a better efficiency in the use of available nutrients and its lower vigor.
Regarding leaf micronutrients, we found very high insufficiency (DOP < 0) in the most cases, except for leaf Mn in ‘Biljana’ and ‘Harcot’ on MR, leaf Cu in ‘Biljana’, ‘Vera’ and ‘Harcot’ on BI, and ‘Roxana’ on MR, respectively (Table 7). In these situations, leaf Mn and Cu amounts are higher than normal (DOP > 0). Deficiency of Cu in apricot orchards are rare (Güleryüz et al. 1996), although some cases of Cu insufficiency has been previously described (Milosevic and Milosevic 2011a).
Leaf Fe level was insufficient although its amount in soil was high (Ankerman and Large 1977), indicating insufficient uptake of this nutrient. Probably, deficiency of leaf Fe and also Zn might be due to the antagonistic effect on P excessive or Zn deficiency in the soil (Tisdale and Nelson 1966). In our trial, there were no visible symptoms of Fe chlorosis. Recently, several authors discarded to use leaf Fe amount as a measure of the Fe status in the some fruit trees; the reason being that leaf Fe level is not generally well correlated with leaf chlorophyll content under field conditions (Benítez et al. 2002). Mn is less mobile in plant tissues (Mengel et al. 2001). The negative DOPMn in the present study may be linked with its low amount in soil and also lack of solubility or losses in sandy-loam and acidic soil, which is in harmony with previous data (Marschner 1995).
Generally, B deficiency was observed mostly, especially in soils like ours and soils with low content of organic matter. Moreover, most B in soils is unavailable to plants (Johnson and Uriu 1989). In our trial, the very high B deficiency was associated with its low content in soil, low availability and/or with unfavorable soil physical-chemical properties (Mengel et al. 2001).
ANOVA indicated that both BI and MR promoted similar ƩDOP index values (Table 7), indicating that these parts of trees was not main factor which determined balance among micronutrients. Contrary, differences among cultivars were significant. On BI, ‘Roxana’ showed a wider imbalance in nutritional values, whereas ‘Aleksandar’ showed a better balance in nutritional values. On MR, the better balance among micronutrients was observed in ‘Biljana’, whereas the highest imbalance in nutritional values was registered in ‘Vera’. As known, the imbalance of nutrients negatively affected tree growth, fruit quality and yield (Tisdale and Nelson 1966; Marschner 1995).
Conclusions
The results obtained in this study indicated that, in dry, sandy-loam and acidic soil growing conditions, trees grafted on Myrobalan rootstock appears to induce higher tree vigor, higher yield per tree, largest fruits with higher flesh rate, and higher acidity, total antioxidant power and leaf macronutrients content with better balanced nutritional values for macronutrients. Blackthorn inter-stem showed a tendency to induce higher yield efficiency, yield per hectare, smaller fruits, higher levels of TPC and TFC and poorer balanced nutritional values for macronutrients. Due to the higher mortality of trees and smaller fruits on Blackthorn inter-stem, its use in a commercial orchard in growing conditions like ours can be limited. Regarding cultivars, it seems that ‘Roxana’ and ‘Harcot’, somewhat ‘Vera’, possess more positive evaluated traits in general, especially on Myrobalan rootstock, and can be recommended to growers. Hence, values of DOP and ƩDOP for leaf macro- and micronutrients indicated that fertilization with organic and mineral fertilisers in this orchard was inadequate and requires a completely new approach. Liming is another operation required for this soil. We assume that under more controlled fertilization and irrigation system, productivity, tree survival, fruit quality and nutritional status of apricot trees may be better. However, further investigations are necessary before making the final conclusions regarding all evaluated traits on both Myrobalan rootstock and Blackthorn inter-stem.
Abbreviations
- BI:
-
Blackthorn inter-stem
- MR:
-
Myrobalan rootstock
- TCSA:
-
trunk cross sectional area
- SSC:
-
soluble solids content
- TA:
-
titratable acidity
- TPC:
-
total phenolic content
- TFC:
-
total flavonoid content
- TAC:
-
total antioxidant capacity
References
Ankerman D, Large R (1977) Minor elements. In: Ankerman D (ed) Soil and plant analysis. Soil Science Society of America, Inc., Madison, pp 34–45
Basile B, Marshal J, DeJong TM (2003) Daily shoot extension growth of peach trees growing on rootstocks that reduce scion growth is related to daily dynamics of stem water potential. Tree Physiol 23:695–704
Bassi D (2001) Apricot culture: present and future. Acta Hortic 488:35–40
Benítez ML, Pedrajas VM, del Campillo MC, Torrent J (2002) Iron chlorosis in olive in relation to soil properties. Nutr Cycl Agroecosys 62:47–52
Brighente IMC, Dias M, Verdi LG, Pizzolatti MG (2007) Antioxidant activity and total phenolic content of some Brazilian species. Pharm Biol 45:156–161
Castle WS (1995) Rootstock as a fruit quality factor in citrus and deciduous tree crops. New Zeal J Crop Hortic Sci 23:383–394.
Cemagref (1981) La qualité gustative des fruits. Méthodes, pratiques d’analyse. Ministry of Agriculture, France, p 150
Crisosto CH, Crisosto GM, Echevarria G, Puy J (2006) Segregation of peach and nectarine [Prunus persica (L.) Batsch] cultivars according to their organoleptic characteristics. Postharvest Biol Technol 39:10–18
Darikova JA, Savva YV, Eugene A, Vaganov EA, Grachev AM, Kuznetsova GV (2011) Grafts of woody plants and the problem of incompatibility between scion and rootstock (a review). J Siber Fed Univer Biol 4:54–63
Dauchet L, Dallongeville J (2008) Fruit and vegetables and cardiovascular disease: epidemiological evidence from the non-western world. Brit J Nutr 99:219–220
Djuric B, Keserovic Z (1999) Study on the possibilities of use of black thorn (Prunus spinosa L.) as an interstock in apricot. Acta Hortic 488:533–538
Dimitrova M, Marinov P (2002) Myrobalan (P. cerasifera Ehrh.) as a rootstock for apricot. Acta Hortic 577:315–318
Egea J, Ruiz D, Martínez-Gómez P (2004) Influence of rootstock on the productive behaviour of ‘Orange Red’ apricot under Mediterranean conditions. Fruits 59:367–373
Font i Forcada C, Gogorcena Y, Moreno MA (2014) Agronomical parameters, sugar profile and antioxidant compounds of “Catherine” peach cultivar influenced by different plum rootstocks. Int J Mol Sci 15:2237–2254
Gil-Izquierdo A., Riquelme MA, Porras I, Ferreres F (2004) Effect of the rootstock and interstock grafted in lemon tree [Citrus limon (L.) Burm.] on the flavonoid content of lemon juice. J Agr Food Chem 52:324–331
Guerriero R, Scalabrelli G (1989) Six year on apricot high density plantings. Acta Hortic 243:365–372
Guerriero R, Scalabrelli G, Franceschini M (1986) Trial on the maximum limit of apricot planting density. Acta Hortic 192:99–106
Güleryüz M, Bolat İ, Pirlak L, Eşítken A, Ercíşlí S (1996) Investigations on determination of nutritional status of apricot (cv. Hasanbey) grown in Erzincan. Turk J Agric For 20:479–487
Gurrieri F, Audergon JM, Albagnac G, Reich M (2001) Soluble sugars and carboxylic acids in ripe apricot fruit as parameters for distinguishing different cultivars. Euphytica 117:183–189
Hegedüs A, Engel R, Abrankó L, Balogh E, Blázovics A, Hermán R, Halász J, Ercisli S, Pedryc A, Stefanovits-Bányai E (2010) Antioxidant and antiradical capacities in apricot (Prunus armeniaca L.) fruits: variations from genotypes, years, and analytical methods. J Food Sci 75:722–730
Hernández F, Pinochet J, Moreno MA, Martínez JJ, Legua P (2010) Performance of Prunus rootstocks for apricot in Mediterranean conditions. Sci Hortic 124:354–359
Johnson RS, Uriu K (1989) Mineral nutrition. In: Larue J, Johnson RS (eds) Peach, plum and nectarine: growing and handling for fresh market. University of California, Division of Agriculture Resource, Oakland, pp 68–81
Kumar D, Ahmed N, Verma MK, Dar TA (2013) Growth, yield, quality and leaf nutrient status as influenced by planting densities and varieties of apricot. Indian J Hortic 70:195–199
Leccese A, Bureau S, Reich M, Renard MG, Audergon JM, Mennone C, Bartolini S, Viti R (2010) Pomological and nutraceutical properties in apricot fruit: cultivation systems and cold storage fruit management. Plant Foods Hum Nutr 65:112–120
Leece DR, van den Ende B (1975) Diagnostic leaf analysis for stone fruit. 6. Apricot. Aust J Exp Agric Anim Husb 15:123–128
Licznar–Małańczuk M, Sosna I (2005) Evaluation of several apricot cultivars and clones in the Lower Silesia climatic conditions. Part II: vigor, health and mortality. J Fruit Ornam Plant Res 13:49–57
Marschner H (1995) Mineral nutrition of higher plants. Academic Press, London.
Mengel K, Kirkby EA, Kosegarten H, Appel T (2001) Principles of plant nutrition. Kluwer Academic Publisher, Dordrecht, p 848
Milosevic T, Milosevic N (2011a) Seasonal changes in micronutrients concentrations in leaves of apricot trees influenced by different interstocks. Agrochimica 55:1–14
Milošević T, Milošević N (2011b) Diagnose apricot nutritional status according to foliar analysis. Plant Soil Environ 57:301–306
Monastra F, Salvador FRD (1995) Apricot: present and future. Acta Hortic 384:401–414
Monney P, Evéquoz N, Christen D (2010) Alternative to Myrobalan rootstock for apricot cultivation. Acta Hortic 862:381–384
Montañés L, Heras L, Abadía J, Sanz M (1993) Plant analysis interpretation based on a new index: deviation from optimum percentage (DOP). J Plant Nutr 16:1289–1308
Moreno MA, Montañés LL, Tabuenca MC, Cambra R (1996) The performance of Adara as a cherry rootstock. Sci Hortic 65:58–91
Palk J (1984) Ploomija kirsipuude kasvatamine. Valgus, Tallinn. (in Estonian)
Prieto P, Pineda M, Aguilar M (1999) Spectrophotometric quantification of antioxidant capacity through the formation of a phosphomolybdenum complex:specific application of vitamin E. Anal Biochem 269:337–341
Restrepo-Díaz H, Melgar JC, Lombardini L (2010) Ecophysiology of horticultural crops: an overview. Agron Colomb 28:71–79
Rosati A, DeJong TM, Southwick SM (1997) Comparison of leaf mineral content, carbon assimilation and stem water potential of two apricot (Prunus armeniaca L.) cultivars grafted on ‘Citation’ and ‘Marianna 2624’ rootstocks. Acta Hortic 451:263–268
Sánches-Rodrígues AR, Cañasveras JC, del Campillo MC, Barrón V, Torrent J (2013) Iron chlorosis in field grown olive as affected by phosphorus fertilization. Eur J Agron 51:101–107
Scalzo J, Politi A, Pellegrini N, Mezzetti B, Battino M (2005) Plant genotype affects total antioxidant capacity and phenolic contents in fruit. Nutrition 21:207–213
Shackel KA, Ahmadi H, Biasi W, Buchner R, Goldhamer D, Gurusinghe S, Hasey J, Kester D, Krueger B, Lampinen B, McGourty G, Micke W, Mitcham E, Olson B, Pelletrau K, Philips H, Ramos D, Schwankl L, Sibbett S, Snyder R, Southwick S, Stevenson M, Thorpe M, Weinbaum S, Yeager J (1997) Plant water status as an index of irrigation need in deciduous fruit trees. HortTechnology 7:23–29
Shear CB (1975) Calcium-related disorders of fruits and vegetables. HortScience 10:361–365
Singleton V, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic–phosphotungstic acid reagents. Am J Enol Vitic 16:144–158
Sitarek M, Bartosiewicz B (2011) Influence of a new seedling rootstocks on the growth, yield and fruit quality of apricot trees. J Fruit Ornam Plant Res 19:81–86
Son L, Küden A (2003) Effects of seedling and GF-31 rootstocks on yield and fruit quality of some table apricot cultivars grown in Mersin. Turk J Agric For 27:261–267
Sosna I, Licznar–Małańczuk M (2012) Growth, yielding and tree survivability of several apricot cultivars on Myrobalan and ‘Wangenheim prune’ seedlings. Acta Sci Pol Hortoru 11:27–37
Southwick SM, Weis KG (1998) Selecting and propagating rootstocks to produce apricots. HortTechnology 8:164–170
Southwick SM, Yeager JT (1999) Effect of rootstock, cultivar and orchard system on apricot production. Acta Hortic 488:483–488
Taylor KB, Goubran HF (1975) The phosphorus nutrition of the apple tree. I. Influence of rate of application of superphosphate on the performance of young trees. Aust J Agr Res 26:843–853
Taylor MA, Rabe E, Dodd MC, Jacobs G (1993) Influence of sampling date and position in the tree on mineral nutrients, maturity and gel breakdown in cold stored ‘Songold’ plums. Sci Hortic 54:131–141
Tisdale SL, Nelson WL (1966) Soil fertility and fertilisers. The Macmillan Company, New York
Vachůn Z (1983) Influence of interstock apricot cultivars on the growth and crop yield of the Velikopavlovicka apricot. Acta Hortic 121:289–294
Vachůn Z (2001) Yield potential of new apricot cultivars and hybrids within the first eight years after planting. Hortic Sci 28:41–46
Vachůn Z (2003) Variability of 21 apricot (Prunus armeniaca L.) cultivars and hybrids in selected traits of fruit and stone. Hortic Sci 30:90–97
Velemis D, Almaliotis D, Bladenopoulou S, Karayannis I (1999) Growth and nutritional status of twelve apricot varieties grafted on two rootstocks. Acta Hortic 488:489–494
Webster AD (1995) Rootstock and interstock effects on deciduous fruit tree vigour, precocity and yield productivity. New Zeal J Crop Hort 23:373–382
Acknowledgements
This work is part of the 31,064 project supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Milošević, T., Milošević, N. & Glišić, I. Apricot Vegetative Growth, Tree Mortality, Productivity, Fruit Quality and Leaf Nutrient Composition as Affected by Myrobalan Rootstock and Blackthorn Inter-Stem. Erwerbs-Obstbau 57, 77–91 (2015). https://doi.org/10.1007/s10341-014-0229-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10341-014-0229-z