Abstract
Management of the whitefly Bemisia tabaci biotype B (Hemiptera: Aleyrodidae) has challenged scientists and famers worldwide because this insect is highly polyphagous and resistant to traditional control methods. Innovative management tools, such as the use of the ecosystem service of biological control, are necessary, especially in a century in which sustainable crop systems are emerging. We investigated how management practices and farm diversity within the property limits affect the relationship between biodiversity conservation and the biological control of B. tabaci. We determined the mortality factors of whitefly nymph cohorts on 33 small farms growing tomatoes. The sampled farms formed a gradient of increasing farm diversity and decreasing management intensity. The diversity of natural enemies increased from conventional to agroecological farms, and predation was identified as the key mortality factor of B. tabaci nymphs on all farm types except the conventional farms. Biological control provided by biodiversity components (predators, parasitoids and pathogens) was enhanced on more diverse and less intensively managed farms. Higher predator richness and abundance resulted in more effective biological control and made it less variable among farms. Different components of natural enemy biodiversity were conserved and acted in a complementary way on agroecological farms, and the enhanced ecosystem services reduced the need for insecticides.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Key messages
-
We assessed how whitefly mortality differed among farms showing different levels of plant diversity and management
-
Biological control provided by different biodiversity components (predators‚ parasitoids and pathogens) was enhanced on more diverse and less intensively managed farms
-
Predation was identified as the key mortality factor of whitefly nymphs and predator richness and abundance reduced the variability in biological control
-
Agroecological farms are suitable for sustainable whitefly control based on ecological processes rather than insecticide spraying
Introduction
Landscapes in most countries are a mosaic of fragmented natural vegetation interspersed with agricultural areas, including in tropical regions, where most of the biodiversity is concentrated. For example, Brazil is one of the main producers of food and fiber in the world and is a mega-diverse country with at least two biomes that are considered hotspots of biodiversity and are threatened by agricultural expansion (Cerrado and Atlantic Rainforest) (Myers et al. 2000; Klink and Machado 2005; Colombo and Joly 2010). Reconciling the demand for food production and biodiversity conservation is a key strategy for enhancing ecosystem services and human well-being. This can be achieved by managing agricultural landscapes based on principles of conservation biology and ecological processes instead of artificial chemical and external inputs (UN 2017).
The biological control of crop pests is an important ecosystem service that benefits agricultural production and is influenced by natural ecosystem processes and human management (Bengtsson 2015). Natural enemy diversity and abundance can be enhanced through increased habitat complexity that offers alternative prey/hosts, shelters, improved microclimatic conditions, oviposition sites and plant-provided food (pollen and nectar) (Begg et al. 2017). In addition, agricultural management practices can be adapted to benefit natural enemies. The reduction of disturbance by, for example, broad-spectrum insecticide spraying, increases the diversity and efficacy of natural enemies that provide pest control (Bengtsson 2015).
Interestingly, a positive effect on biological control is not always achieved by increasing the diversity of natural enemies (Straub et al. 2008; Letourneau et al. 2009; Macfadyen et al. 2011), and this relationship is highly variable among studies (Bengtsson et al. 2005). In some situations, key species are responsible for the control of a target pest, and the identity of the species may be more important than the diversity (e.g., Ives et al. 2005; Straub and Snyder 2006). Negative effects can occur via behavioral interactions (e.g., intraguild predation, apparent competition), and habitat structure may play a role in preventing such negative interactions (e.g., Janssen et al. 2007). Neutral effects occur when natural enemies share similar niches, and no gain in functionality is achieved by diverse natural enemy communities (Ives et al. 2005; Straub et al. 2008). An additive effect can occur due to complementarity in resource use (e.g., Macfadyen et al. 2011). However, there is a need to better understand the interactions among herbivores, natural enemies and crop management strategies at a broader scale, such as at the farm level.
Organic vegetable crop systems are suitable for studying these interactions because ecosystem services are usually utilized to reduce pest problems (Zehnder et al. 2007). In such ephemeral crops, the efficiency of pest control by natural enemies could be directly related to the species pool available at the farm, from which natural enemies could rapidly colonize a new habitat in response to an increase in herbivore populations (Tylianakis et al. 2005). The availability of habitats such as agroforestry systems (Harterreiten-Souza et al. 2014), strips of non-crop plants (Amaral et al. 2013), aromatic flowering plants (Togni et al. 2010) and non-cropped areas (Tylianakis et al. 2005) could serve as a source of these beneficial insects mitigating the need for adverse management practices, such as broad-spectrum insecticide spraying (Naranjo and Ellsworth 2009).
We predict that a more diverse and abundant community of natural enemies could be positively related to the provisioning of the ecosystem service of biological control, reducing the need for human intervention with external and artificial inputs. In addition, farms with more diverse habitats within the property limits and that use less intensive management practices are likely to be more suitable for biodiversity conservation and functionality. To test these predictions, we determined the effects of different components of biodiversity (predators, parasitoids and pathogens) on whitefly nymph mortality and evaluated the relationship between natural enemy conservation and the provisioning of whitefly biological control in small farms growing tomatoes in Brazil.
The whitefly Bemisia tabaci biotype B Gennadius (Hemiptera: Aleyrodidae) is a highly polyphagous herbivore species that uses more than 500 plant species as host plants worldwide (Byrne and Bellows Jr. 1991; Oliveira et al. 2001). It has several well-known natural enemies, including predators, parasitoids and pathogens that can potentially interfere with its population dynamics (Gerling et al. 2001). This species is one of the most important pests of tomatoes in Brazil and around the world (Oliveira et al. 2013). Tomato growers in Brazil, especially small farmers, use different farm designs and management strategies that vary from monocultures with intensive broad-spectrum insecticide spraying to organic and agroecological farms with highly diversified crops and natural pest control. Given the characteristics of B. tabaci and these farm traits, the system used in this study is relevant for seeking sustainable management options for combatting this worldwide pest.
Our specific aim was to identify how farm management practices and farm diversity within the property limits affect the role of biodiversity in the mortality of B. tabaci populations. To accomplish this, we addressed the following questions: (1) Are less intensively managed and more diversified farms more suitable for the conservation of natural enemies of B. tabaci? (2) Do farm characteristics and the biodiversity of natural enemies affect the abundance of B. tabaci adults? (3) What is the role of biodiversity as a mortality factor among B. tabaci nymphs, and how does it vary according to farm management strategies and diversity? (4) Is there a relationship between biodiversity conservation and biological control?
Materials and methods
Location
The study was conducted on 33 small farms growing tomatoes in the Brazilian Federal District, Brazil (Fig. 1). The farms were located in the core of the Cerrado biome, the Brazilian tropical savanna. The climate of the region is Cwa semi-humid based on the Köppen–Geiger classification. The mean temperature ranges from 22 to 27 °C, the average rainfall is 1200 mm per year, and the altitude ranges from 1000 to 1200 masl (Klink and Machado 2005). There are two well-defined seasons throughout the year. The wet season usually occurs from November to March, whereas the dry season occurs from May to September. The experiments were conducted during the dry season in 2013 and 2014, and there was no rainfall during the experimental period.
Farm characterization
At each small farm sampled, we evaluated the plant and habitat diversity at the farm level as well as the management practices used (Methods S1 and Table S1). The sampled farms in the Brazilian Federal District, Brazil, are characterized by the employment of family labor, diversity of farm designs and use of different types of pest management strategies. To classify the sampled farms according to the intensity of management and farm plant diversity, the following criteria were used (Table S1 and Fig. S1): (a) the presence and composition of windbreaks, (b) the presence, location and approximate age of agroforests, (c) the identity of the species other than tomatoes grown during the experimental period, (d) the identity of the crops surrounding the tomato plot, (e) the diversity of the plants within the tomato plot (crops and non-crop plants), (f) the management of non-crop plants (within and surrounding the tomato crop), (g) irrigation management, (h) the presence and type of soil cover, (i) fertilization (inorganic or organic, including the type of fertilizers used) and (j) the pest control strategy and frequency of pesticide spraying (botanical, biological or chemical pesticides).
Based on these variables, the farms were classified and grouped into four categories (Fig. 1 and Fig. S1): (1) conventional (n = 10)—high-intensity management and very low plant and habitat diversity, (2) transition (n = 9)—intermediate between conventional agriculture and an organic system (transition phase) but without meeting all criteria to be certified as organic, with moderate management intensity and low plant and habitat diversity, (3) organic (n = 8)—a certified organic farm with low management intensity and moderate plant and habitat diversity and (4) agroecological (n = 6)—an organic farm using mainly natural processes for farm management instead of external inputs with very low management intensity and high plant and habitat diversity.
Insect sampling
Yellow sticky traps (15 cm × 25 cm) were used to evaluate how adult whitefly population levels (abundance) and the richness and abundance of whitefly arthropod natural enemies varied among the different farm categories. Five traps were placed on each farm; they were spaced at least 5.0 m apart at the height of the tomato plant canopy and between two tomato plants. The traps were maintained in the field for five days between the second and third weeks after the tomato plants were transplanted, when plants are more susceptible to whitefly infestation (Oliveira et al. 2013). Using the same traps, we measured the abundance of whiteflies and the abundance and richness of arthropod natural enemies. We identified only the natural enemy species that are known as whitefly predators and parasitoids and were attached to these traps (see details on the species selected for sampling in Methods S1). Using this method, we were able to sample whitefly parasitoids in addition to predators using the same sampling procedure. All species were identified directly on the traps or were removed from them and sent to a taxonomist when needed. We performed the nymph mortality experiment over the same period, as described below. After this period, the traps were removed, and the number of whitefly adults per trap was counted. The whiteflies were identified as B. tabaci biotype B based on the testing of 10 individuals per farm using random amplified polymorphic DNA (RAPD) markers. The numbers of species and the abundance of whitefly natural enemies on the traps were also evaluated.
Nymph mortality factor experiment
In this experiment, we aimed to determine how the farm category influenced the mortality factors of B. tabaci. Groups of twenty adult whiteflies were randomly collected from a reared colony and transferred to clip cages (for details, see Methods S1) using a manual entomological aspirator. A sex ratio tending toward females was assumed (2:1) (Villas-Bôas et al. 2002); thus, no sex differentiation was conducted.
Clip cages were individually placed on twenty randomly selected tomato plants on each farm. Each clip cage contained 20 whitefly adults. All leaves were examined with a 15 × magnifier to check for any whitefly eggs left by local populations before placing the clip cages. The clip cages were left in the field for 48 h to allow female oviposition. Using this method, it was possible to standardize the cohort age independently from the local population densities. Subsequently, the clip cages were removed, and each leaf containing a leaflet with eggs was marked with a flagging tape. After 15–20 days, we counted the number of nymphs per plant at each farm using a 15 × magnifier. This period corresponds to the average time between egg deposition and the fourth instar stage. Moreover, whitefly nymphs are generally sessile; only first instar nymphs are mobile, but they move only a few centimeters after hatching (Byrne and Bellows Jr. 1991). Taking into account that the importance of mortality factors differs as individuals age, we counted the number of living nymphs of the same age on each leaflet. Five days after the first nymph sampling in the field, the infested leaflets were cut and taken to the laboratory to identify the mortality factors for each cohort. We identified the mortality factors only for the fourth instar nymphs, which is the stage that is the most susceptible to the different mortality factors affecting whitefly development (Naranjo and Ellsworth 2005; Karut and Naranjo 2009). Moreover, only during the fourth instar it is possible to identify parasitized individuals. We also recorded the number of live whitefly nymphs on the leaflets taken to the laboratory.
Five different mortality factors were analyzed based on the descriptions of Naranjo and Ellsworth (2005). We considered that an individual was dead because of predation when an empty, transparent and wrinkled cuticle (sucking predators) or partially intact cadaver (chewing predators) was observed. Parasitized individuals presented either the displacement of mycetomes or a parasitoid larva or pupa inside the host. Nymphs presenting an evident color change or hyphal growth were considered dead by pathogens. Dislodgement was estimated by subtracting the number of nymphs counted on the leaflets taken to the laboratory from the number of nymphs counted in the field. We assumed that dislodgment could be caused by several unspecific causes, including knockdown by insecticide spraying. Finally, all non-evident causes of death were classified as unknown.
Statistical analyses
The species richness and diversity of the natural enemies found on the sticky traps were compared among the farm categories using rarefaction curves and the diversity profiles obtained using the Rényi function (Ricotta 2003).
To evaluate whether the abundance of natural enemies was affected by the different farm categories, we fitted a linear mixed-effect model (LME). The abundance of natural enemies per farm was used as the response variable, farm category was used as the explanatory variable, and year and location (i.e., different landscapes) were used as random factors. The model significance was assessed using an F-test. We then compared the full model with the null model. If significant differences from the null model were found, we accepted the final model. When differences in the abundance of natural enemies were found, the factor levels were compared using post hoc analysis by grouping factor levels (Crawley 2007). Finally, an analysis of the model residuals was performed.
The abundance of whiteflies collected on the traps was compared among the farm categories using the same procedure described for the abundance of natural enemies. The number of adult whiteflies per trap was used as the response variable, and the farm category was used as the explanatory variable. Year of collection and farm location were used as random factors.
To investigate whether the abundance and variation in the abundance of whitefly adults among farms were related to natural enemy richness and abundance, we fitted a linear model. The variation in the abundance of whiteflies among farms was measured using the coefficient of variation (CV) for each farm. The CV for each farm was calculated by dividing the standard deviation of the number of whiteflies per farm by the mean number of whiteflies per farm.
To analyze the nymph mortality factors, it was necessary to consider that there is no obvious sequence of mortality causes because one cause of death can obscure the action of another (Naranjo and Ellsworth 2005). To avoid this methodological bias, the observed (or apparent) mortality of each mortality factor was transformed into marginal rates of mortality (Buonaccorsi and Elkinton 1990). The latter value was transformed into k-values, which are standardized values that are additive across mortality factors (Varley and Gradwell 1960).
We evaluated whether the overall mortality rate from all mortality factors (total K = Σk) was affected by the farm category by fitting a LME as described above. Each mortality factor (represented by a k-value) was also evaluated separately to determine whether it was affected by the farm category. Key mortality factors were quantitatively identified using the method in Podoler and Rogers (1975), which consists of regressing individual k-values against the total K. The mortality factor with the largest slope was considered to be the key mortality factor (for details, see Methods S1).
Death due to biological control agents (pathogens + parasitoids + predators) was identified as an important mortality factor and was then analyzed separately. To accomplish this, we evaluated the influence of the farm categories on the overall mortality due to biological control by summing the individual k-values for death by predation, parasitism and pathogens, resulting in a partial total k-value. Then, a LME was fitted using the overall mortality of nymphs due to biological control as the response variable, farm category as the explanatory variable, and year and farm identity as random factors.
Because predation was identified to be the key mortality factor at most farms, and its importance varied among the farm categories, we investigated whether the richness and abundance of predators could affect the mortality rates and the reliability of the mortality rates by predation at all farms. The reliability of the mortality rates was assessed by calculating the CV of the predation rates per farm, assuming that lower CV values are related to a higher reliability (i.e., lower variance) of predation among farms. We excluded the parasitoid data from our analysis and fitted a linear model between the richness of the predator species collected per farm and the average abundance of predators per farm using the average predation rates at each farm separately. Subsequently, we fitted another linear model between the richness of the predator species collected per farm and the average abundance of predators per farm using the CV of the predation rates per farm. All analyses were performed using the R software (R Core Team 2017).
Results
Diversity and abundance of natural enemies
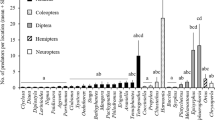
A total of 2512 individuals known to be natural enemies of B. tabaci were collected across all 33 farms and classified into seven orders, 15 families and 37 species or species groups (Table S2). More species per sample were obtained from agroecological farms, followed by transition and organic farms. The rarefaction curves for conventional farms rapidly stabilized, indicating lower diversity on these farms than in the other farm categories (Fig. 2a). The assemblages of natural enemies on agroecological farms were more diverse than those on farms of other categories (Fig. 2b).
The mean abundance of natural enemies per farm was affected by the farm management practices (F = 14.88, d.f. = 3, P < 0.0001). Fewer individuals were trapped on conventional farms than on transition farms. Organic and agroecological farms had the highest abundance of natural enemies (Fig. 3).
Mean number (± SE) of natural enemies of Bemisia tabaci per trap collected on farms growing tomatoes in conventional, transitional to organic, organic and agroecological cropping systems in the Brazilian Federal District. Means followed by the same letter did not significantly differ (P > 0.05) according to a post hoc analysis conducted by grouping factor levels
Whitefly population levels
The population levels of adult whiteflies found on the sticky traps on tomatoes did not differ among the farm categories (F = 1.156, d.f. = 3, P = 0.345) (Fig. 4). We also did not find differences in the CV among the farm categories (F = 3.66, d.f. = 3, P = 0.0691) (Fig. 4). There were no relationships between the adult whitefly population levels and the richness (R2 = − 0.025, F = 0.264, d.f. = 29, P = 0.611) or abundance of natural enemies per farm (R2 = 0.037, F = 1.114, d.f. = 29, P = 0.299). Similarly, the CV of the whitefly population levels at each farm was not related to the abundance (R2 = − 0.034, F = 0.001, d.f. = 29, P = 0.970) or richness (R2 = − 0.011, F = 0.691, d.f. = 29, P = 0.413) of natural enemies.
Nymph mortality factors
The average cohort mortality of B. tabaci nymphs was back-transformed from k-values and was estimated as 28.2% for conventional farms, 47.6% for transition farms, 57.6% for organic farms and 70.2% for agroecological farms. The overall mortality of B. tabaci nymphs (K) was significantly affected by the farm category (F = 31.59, d.f. = 3, P = < 0.0001). Agroecological farms presented the highest mortality rates (k-values) (Fig. 5). From conventional farms to transition and organic farms, the overall mortality of nymphs approximately tripled. The overall mortality of nymphs doubled from these categories to agroecological farms (Fig. 5).
Mean levels of mortality factors expressed as k-values affecting the total mortality of Bemisia tabaci nymphs on farms growing tomatoes in conventional, transitional to organic, organic and agroecological cropping systems in the Brazilian Federal District. Means followed by the same letter did not significantly differ (P > 0.05) in terms of overall mortality (∑k-values) in each farm category according to a post hoc analysis conducted by grouping factor levels
Decomposing the overall mortality to specific causes of death, we observed that predation (F = 15.83, d.f. = 3, P < 0.001), parasitism (F = 10.78, d.f. = 3, P = 0.0001), dislodgement (F = 4.06, d.f. = 3, P = 0.016) and pathogen attack (F = 20.60, d.f. = 3, P < 0.0001) were affected by farm diversity and the intensity of management, while unknown causes were not (F = 2.36, d.f. = 3, P = 0.093). Comparing these causes of death within farm categories using post hoc analysis by grouping the factor levels, predation was lower on conventional than on transition (Likelihood ratio = 13.31, d.f. = 6, P = 0.0003), organic (L-ratio = 11.93, d.f. = 6, P < 0.0001) and agroecological farms (L-ratio = 26.21, d.f. = 6, P < 0.0001). However, predation presented intermediate and similar values on transition and organic farms (L-ratio = 0.06, d.f. = 6, P = 0.799), which were lower than those on agroecological farms (L-ratio = 7.28, d.f. = 6, P = 0.007) (Fig. 5). Parasitism did not differ on conventional and transition farms (L-ratio = 3.45, d.f. = 6, P < 0.0001) and was lower than that on organic (L-ratio = 13.63, d.f. = 6, P = 0.0002) and agroecological farms (L-ratio = 17.54, d.f. = 6, P < 0.0001). However, death by parasitism did not differ between the organic and agroecological farms (L-ratio = 0.89, d.f. = 6, P < 0.344). Dislodgment was lowest on conventional and transition farms, and the dislodgement values did not differ on these two farm types (L-ratio = 0.13, d.f. = 6, P = 0.714). Although the values were higher than those in these categories (L-ratio = 4.76, d.f. = 6, P = 0.029), death by dislodgment was similar on organic and agroecological farms (L-ratio = 0.69, d.f. = 6, P = 0.407). Death by pathogen attack did not differ among conventional, transition and organic farms (L-ratio = 4.82, d.f. = 6, P = 0.0899), but it was higher on agroecological farms (L-ratio = 21.39, d.f. = 6, P < 0.0001) (Fig. 5).
Predation was identified as the key mortality factor on all farm types except conventional farms (Table 1). On transition and organic farms, dislodgement was also a prominent mortality factor for B. tabaci nymphs. On conventional farms, dislodgement was identified as the key mortality factor, but predation also played a role, while parasitism was almost negligible (Table 1). Unknown causes of death were relatively consistent among the different farm categories (Table 1).
Role of biological control
The overall mortality due to biological control agents (predators + parasitoids + pathogens), back-transformed from k-values, represented 35, 71.6, 69.32 and 77.3% of the overall mortality observed among all cohorts on conventional, transition, organic and agroecological farms, respectively. Death by biological control was affected by the farm category (F = 19.27, d.f. = 3, P < 0.0001). Conventional farms presented mortalities that were similar to those on transition farms (L-ratio = 0.02, d.f. = 6, P = 0.8898), differing from organic (L-ratio = 7.52, d.f. = 6, P = 0.023) and agroecological farms (L-ratio = 30.22, d.f. = 6, P < 0.0001). The mortality due to biological control was higher on agroecological farms than organic farms (L-ratio = 14.61, d.f. = 6, P = 0.0001).
As mortality by predation caused the most nymph mortality, we investigated whether the mean predator abundance and richness per farm could have affected the predation rates and the CV (i.e., reliability) of the predation rates on all farms. Farms with a higher abundance and richness of whitefly predator species presented higher predation rates (Fig. 6a, b) and a lower CV of predation rates (Fig. 6c, d).
Relationships between the mean mortality by predation (k-values) and the species richness of predators per farm (a), mean mortality by predation (k-values) and the mean abundance of predators per farm (b), coefficient of variation (CV) and the species richness of predators per farm (c) and coefficient of variation (CV) and the mean abundance of predators per farm (d) on 33 small farms growing tomatoes in the Brazilian Federal District
Discussion
Our study demonstrated a positive relationship between biodiversity conservation and the provisioning of the ecosystem service of biological control on small farms in a tropical biome threatened by agricultural expansion. Less intensively managed and more diverse farms are more suitable for biodiversity conservation and sustainable pest management. Therefore, biological control is provided by the combined effects of human and natural capital and can be managed by varying the input from each counterpart (Bengtsson 2015). Such an approach is also in line with the United Nations recommendations for a reduction in pesticide use via the promotion of agroecology and with the previous Convention on Biological Diversity (UN 2017).
Biological control agents increased in their importance as mortality factors of the worldwide pest B. tabaci from conventional to agroecological farms. Among these, predation was the most important mortality factor, with predator diversity favoring biological control and increasing its reliability (i.e., by being less variable) among farms.
More diverse assemblages of natural enemies were found on agroecological farms. However, the natural enemy abundance reached maximum values on the organic farms, with no differences from those on agroecological farms. As a general trend, habitat diversity associated with organic management positively affects the diversity and abundance of natural enemies (Letourneau et al. 2011; Isbell et al. 2017). The mechanisms underlying these benefits on the more diverse farms could be related to habitat diversity and complexity. For example, agroforestry systems occurring near horticultural crops serve as a high-quality source of generalist predators that colonize nearby vegetable crops (Harterreiten-Souza et al. 2014). Similarly, non-crop habitats provide a great number of parasitoid (Macfadyen et al. 2011) and predator species (Amaral et al. 2015). These farm traits can favor the coexistence of more species that use different types of resources in the same habitat but not necessarily their abundance (Bianchi et al. 2006). Conversely, pesticides and intense management tend to reduce natural enemy biodiversity, resulting in more homogeneous communities on conventional farms (Geiger et al. 2010; Crowder et al. 2010).
In contrast to those of natural enemies, the population levels of adult whiteflies were not affected by farm diversity and management. Adult mobile polyphagous insects, such as whiteflies, are less affected by crop diversification because they can use a wide variety of hosts for feeding but not necessarily for reproduction (Bernays 1999). This was previously observed in organic and conventional tomato crops planted in monoculture or intercropped with coriander in Brazil (Togni et al. 2009). Natural enemies also did not affect the population levels of adult whiteflies. This might have occurred because there are a few species known to be natural enemies of adult whiteflies (Gerling et al. 2001). In our sample, there were only two arthropod groups (Dolichopodidae flies and spiders) that potentially prey upon adult whiteflies. Therefore, knowing natural enemy species traits is imperative in applied research focusing on the use of biodiversity in ecological pest management (Kremen 2005; Perovic et al. 2017).
Although no effect on adult abundance was observed, nymph mortality differed among the farm categories. Predation was the key mortality factor of nymphs in all farm types except conventional farms. Predation was also identified as a key mortality factor of third and fourth instar nymphs of B. tabaci in cotton fields in the USA (Naranjo and Ellsworth 2005) and in Turkey (Karut and Naranjo 2009) and was found to play a significant role in mortality in cassava fields in Uganda (Asiimwe et al. 2007). In terms of parasitism, a farm solely using organic practices and reducing the use of insecticides (synthetic or not) was sufficient to maintain the ecosystem service of nymph parasitism. Pathogens presented an additive effect on nymph mortality on agroecological farms. This could have occurred due to the better microclimatic conditions, availability of perennial habitats such as agroforestry systems and less intensive soil management where many species of pathogens live (Klingen et al. 2002). Furthermore, if the ecosystem service of biological control compensates for the need for artificial and intensive pest control, as in our study, conserving it is a suitable management practice for whitefly control among organic growers.
Dislodgment also played an important role in mortality, being the key mortality factor of nymphs on conventional farms. Dislodgment can be caused by intrinsic or extrinsic factors, such as when a newly formed nymph fails to reinsert its mouthparts into the plant tissue (Walker and Perring 1994) or due to abiotic conditions (e.g., wind speed, temperature, relative humidity) (Naranjo and Ellsworth 2005). Water drops from overhead sprinkler irrigation—an extrinsic factor—reduces adult establishment in crops because they tend to move between plants more often for feeding and reproduction (Castle et al. 1996), disrupting insect–plant interactions. This could be true for most farms, but the conventional farmers used only drip irrigation. On conventional farms, we observed growers applying insecticides directly over the tomato plants using manual spraying. We hypothesized that the drops of insecticide during spraying or the knockdown effect on nymphs could be the major factor responsible for nymph dislodgement on such farms. This remains to be tested because it is difficult to disentangle the factors that specifically cause dislodgment. The mortality caused by insecticides may also explain the evenness in the abundance of adults despite the differences in nymph mortality among the farm categories.
Increasing the abundance and richness of predators leads to higher rates of B. tabaci nymph predation. Interestingly, the relationship between biodiversity and ecosystem service functioning could be context dependent, in which key species can control pests rather than assemblages of multiple predators in some cases (Straub et al. 2008), while natural enemy diversity can positively impact biological control in others (Snyder et al. 2006; Macfadyen et al. 2011). Our study supports the hypothesis that biodiversity may be directly related to pest control, likely due to complementarities in the resource use of different species of predators in tropical agroecosystems. Moreover, a more diverse and abundant assemblage of whitefly predators resulted in a more reliable (lower CV) and constant mortality of whiteflies. Similarly, Macfadyen et al. (2011) found that parasitoid richness resulted in lower variation in pest control over time on organic farms but not on conventional farms. Therefore, a higher richness and abundance of predators is also related to the reliability of whitefly control, mainly in organic crops.
In conclusion, more diversified and less intensively managed farms are more suitable for the conservation of natural enemies of B. tabaci, which are the main causes of nymph but not adult death. However, solely using organic practices has a limited impact on B. tabaci mortality. It is necessary to manage farms in order to facilitate the natural provisioning of biological control, which reduces the need for human intervention. Brazil is one of the main users of pesticides in the world (Bombardi 2017), and it was demonstrated here that a worldwide pest, B. tabaci, can be managed without the need for such products. The Cerrado is the second largest biome in Brazil and is the biome that is most threatened by agricultural expansion (Myers et al. 2000; Klink and Machado 2005). In this sense, our results provide evidence that there are several strategies that can be used to reconcile agricultural production with biodiversity conservation and ecosystem service functioning.
Author contribution
PHBT, MV and ERS conceived the study and designed the methodology. PHBT, LMS and JPCRS collected the data. PHBT and ERS analyzed the data. PHBT led the writing of the manuscript. All authors contributed critically to the drafts and gave approval for the final version.
References
Amaral DSSL, Venzon M, Duarte MVA, Sousa FF, Pallini A, Harwood JD (2013) Non-crop vegetation associated with chili pepper agroecosystems promote the abundance and survival of aphid predators. Biol Control 64:338–346. https://doi.org/10.1016/j.biocontrol.2012.12.006
Amaral DSSL, Venzon M, Perez AL, Schmidt JM, Harwood JD (2015) Coccinellid interactions mediated by vegetation heterogeneity. Entomol Exp Appl 156:160–169. https://doi.org/10.1111/eea.12319
Asiimwe P, Ecaat JS, Otim M, Gerling D, Kyamanyawa S, Legg JP (2007) Life-table analysis of mortality factors affecting populations of Bemisia tabaci on cassava in Uganda. Entomol Exp Appl 122:37–44. https://doi.org/10.1111/j.1570-7458.2006.00487.x
Begg GS, Cook SM, Dye R, Ferrante M, Franck P, Lavigne C, Lövei GL, Mansion-Vaquie A, Pell JK, Petit S, Quesada N, Ricci B, Wratten SD, Birch ANE (2017) A functional overview of conservation biological control. Crop Prot 97:145–158. https://doi.org/10.1016/j.cropro.2016.11.008
Bengtsson J (2015) Biological control as an ecosystem service: partitioning contributions of nature and human inputs to yield. Ecol Entomol 40:45–55. https://doi.org/10.1111/een.12247
Bengtsson J, Ahnström J, Weibull AC (2005) The effects of organic agriculture on biodiversity abundance: a meta-analysis. J Appl Ecol 42:261–269. https://doi.org/10.1111/j.1365-2664.2005.01005.x
Bernays EA (1999) When host choice is a problem for a generalist herbivore: experiments with the whitefly, Bemisia tabaci. Ecol Entomol 24:260–267. https://doi.org/10.1046/j.1365-2311.1999.00193.x
Bianchi FJJA, Booij CJH, Tscharntke T (2006) Sustainable pest regulation in agricultural landscapes: a review on landscape composition, biodiversity and natural pest control. Proc R Soc Lond B 273:1715–1727. https://doi.org/10.1098/rspb.2006.3530
Bombardi LM (2017) Geografia do uso de agrotóxicos no Brasil e conexões com a união europeia. FFLCH-USP, São Paulo
Buonaccorsi JP, Elkinton JS (1990) Estimation of contemporaneous mortality factors. Popul Ecol 32:151–171. https://doi.org/10.1007/BF02512596
Byrne DN, Bellows TS Jr (1991) Whitefly biology. Ann Rev Entomol 36:431–457. https://doi.org/10.1146/annurev.en.36.010191.002243
Castle SJ, Henneberry TJ, Toscano NC (1996) Suppression of Bemisia tabaci (Homoptera: Aleyrodidae) infestations in cantaloupe and cotton with sprinkler irrigation. Crop Prot 15:657–663. https://doi.org/10.1016/0261-2194(96)00037-3
Colombo AF, Joly CA (2010) Brazilian Atlantic Forest lato sensu: the most ancient Brazilian forest, and a biodiversity hotspot, is highly threatened by climate change. Braz J Biol 70:697–708. https://doi.org/10.1590/S1519-69842010000400002
Core Team R (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Crawley MJ (2007) The R book. Wiley, Chichester
Crowder DW, Northfield TD, Strand MR, Snyder WE (2010) Organic agriculture promotes evenness and natural pest control. Nature 466:109–113. https://doi.org/10.1038/nature09183
Geiger F, Bengtsson J, Berendse F, Weisser WW, Emmerson M, Morales MB, Ceryngier P, Liira J, Tscharntke T, Winqvist C, Eggers S, Bommarco R, Pärt T, Bretagnolle V, Plantegenest M, Clement LW, Dennis C, Palmer C, Onate JJ, Guerrero I, Hawro V, Aavik T, Thies C, Flohre A, Hänke S, Fischer C, Goedhart PW, Inchausti P (2010) Persistent negative effects of pesticides on biodiversity and biological control potential on European farmland. Basic Appl Ecol 11:97–105. https://doi.org/10.1016/j.baae.2009.12.001
Gerling D, Alomar O, Arnó J (2001) Biological control of Bemisia tabaci using predators and parasitoids. Crop Prot 20:779–799. https://doi.org/10.1016/S0261-2194(01)00111-9
Harterreiten-Souza ES, Togni PHB, Pires CSS, Sujii ER (2014) The role of integrating agroforestry and vegetable planting in structuring communities of herbivorous insects and their natural enemies in the Neotropical region. Agrofor Syst 88:205–219. https://doi.org/10.1007/s10457-013-9666-1
Isbell F, Adler PR, Eisenhauer N, Fornara D, Kimmel K, Kremen C, Letourneau DK, Liebman M, Polley HW, Quijas S, Scherer-Lorenzen M (2017) Benefits of increasing plant diversity in sustainable agroecosystems. J Ecol 105:871–879. https://doi.org/10.1111/1365-2745.12789
Ives AR, Cardinale BJ, Snyder WE (2005) A synthesis of subdisciplines: predator–prey interactions, and biodiversity and ecosystem functioning. Ecol Lett 8:102–116. https://doi.org/10.1111/j.1461-0248.2004.00698.x
Janssen A, Sabelis MW, Magalhães S, Montserrat M, van der Hammen T (2007) Habitat structure affects intraguild predation. Ecology 88:2713–2719. https://doi.org/10.1890/06-1408.1
Karut K, Naranjo SE (2009) Mortality factors affecting Bemisia tabaci population on cotton in Turkey. J Appl Entomol 133:367–374. https://doi.org/10.1111/j.1439-0418.2008.01369.x
Klingen I, Eilenberg J, Meadow R (2002) Effects of farming system, field margins and bait insect on the occurrence of insect pathogenic fungi in soils. Agr Ecosyst Environ 91:191–198. https://doi.org/10.1016/S0167-8809(01)00227-4
Klink CA, Machado RB (2005) The conservation of the Brazilian Cerrado. Conserv Biol 19:707–713. https://doi.org/10.1111/j.1523-1739.2005.00702.x
Kremen C (2005) Managing ecosystem services: what do we need to know about their ecology? Ecol Lett 8:468–479. https://doi.org/10.1111/j.1461-0248.2005.00751.x
Letourneau DK, Jedlicka JA, Bothwell SG, Bothwell SG, Moreno CR (2009) Effects of natural enemy biodiversity on the suppression of arthropod herbivores in terrestrial ecosystems. Ann Rev Ecol Evol Syst 40:573–592. https://doi.org/10.1146/annurev.ecolsys.110308.120320
Letourneau DK, Armbrecht I, Rivera BS, Lerma JM, Carmona EJ, Daza MC, Escobar S, Galindo V, Gutiérrez C, López SD, Mejía JL, Rangel AMA, Rangel JH, Rivera L, Saavedra CA, Torres AM, Trujillo AR (2011) Does plant diversity benefit agroecosystems? A synthetic review. Ecol Appl 21:9–21. https://doi.org/10.1890/09-2026.1
Macfadyen S, Crazem PG, Polaszek A, van Achterberg K, Memmott J (2011) Parasitoid diversity reduces variability in pest control services across time on farms. Proc R Soc Lond B 278:3387–3394. https://doi.org/10.1098/rspb.2010.2673
Myers N, Mittermeier RA, Mittermeier CG, Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858. https://doi.org/10.1038/35002501
Naranjo SE, Ellsworth PC (2005) Mortality dynamics and population regulation in Bemisia tabaci. Entomol Exp Appl 116:93–108. https://doi.org/10.1111/j.1570-7458.2005.00297.x
Naranjo SE, Ellsworth PC (2009) The contribution of conservation biological control to integrated control of Bemisia tabaci in cotton. Biol Control 51:458–470. https://doi.org/10.1016/j.biocontrol.2009.08.006
Oliveira MRV, Henneberry TJ, Anderson P (2001) History, current status, and collaborative research projects for Bemisia tabaci. Crop Prot 20:709–723. https://doi.org/10.1016/S0261-2194(01)00108-9
Oliveira CM, Auad AM, Mendes SM, Frizzas MR (2013) Economic impact of exotic insect pests in Brazilian agriculture. J Appl Entomol 137:1–15. https://doi.org/10.1111/jen.12018
Perovic DJ, Gámez-Virués S, Landis DA, Wäckers F, Gurr G, Wratten SD, You MS, Desneux N (2017) Managing biological control services through multi-trophic trait interactions: review and guidelines for implementation at local and landscape scales. Biol Rev 93:306–321. https://doi.org/10.1111/brv.12346
Podoler H, Rogers D (1975) A new method for identification of key factors from life-table data. J Anim Ecol 44:85–114. https://doi.org/10.2307/3853
Ricotta C (2003) On parametric evenness measures. J Theor Biol 222:189–197. https://doi.org/10.1016/S0022-5193(03)00026-2
Snyder WE, Snyder GB, Finke DL, Straub C (2006) Predator biodiversity strengthens herbivores suppression. Ecol Lett 9:789–796. https://doi.org/10.1111/j.1461-0248.2006.00922.x
Straub CS, Snyder W (2006) Species identity dominates the relationship between predator biodiversity and herbivore suppression. Ecology 87:277–282. https://doi.org/10.1890/05-0599
Straub CS, Finke DL, Snyder WE (2008) Are conservation of natural enemy biodiversity and biological control compatible goals? Biol Control 45:225–237. https://doi.org/10.1016/j.biocontrol.2007.05.013
Togni PHB, Frizzas MR, Medeiros MA, Nakasu EYT, Pires CSS, Sujii ER (2009) Dinâmica populacional de Bemisia tabaci biótipo B em tomate monocultivo e consorciado com coentro sob cultivo orgânico e convencional. Hortic Bras 27:183–188. https://doi.org/10.1590/S0102-05362009000200011
Togni PHB, Cavalcante KR, Langer LF, Gravina CS, Medeiros MA, Pires CSS, Fontes EMG, Sujii ER (2010) Conservação de inimigos naturais (Insecta) em tomateiro orgânico. Arq Inst Biol 77:669–679
Tylianakis JM, Klein AM, Tscharntke T (2005) Spatiotemporal variation in the diversity of Hymenoptera across a tropical hábitat gradient. Ecology 86:3296–3302. https://doi.org/10.1890/05-0371
UN - United Nations. (2017). Report of the special rapporteur on the right for food. General assembly: Human rights council
Varley GC, Gradwell GR (1960) Key factors in population studies. J Anim Ecol 29:399–401. https://doi.org/10.2307/2213
Villas-Bôas GL, França FH, Macedo N (2002) Potencial biótico da mosca-branca Bemisia argentifolii a diferentes plantas hospedeiras. Hortic Bras 20:71–79. https://doi.org/10.1590/S0102-05362002000100014
Walker GP, Perring TM (1994) Feeding and oviposition behavior of whiteflies (Homoptera: Aleyrodidae) interpreted from AC electronic feeding monitor waveforms. Ann Entomol Soc Am 87:263–374. https://doi.org/10.1093/aesa/87.3.363
Zehnder G, Gurr GM, Kühne S, Wade MR, Wratten SD, Wyss E (2007) Arthropod pest management in organic crops. Annu Rev Entomol 52:57–80. https://doi.org/10.1146/annurev.ento.52.110405.091337
Acknowledgements
We are grateful to all the farm owners who kindly allowed us to collect field data on their lands. Technicians from EMATER-DF offices supported us in the first contact with farm owners. This study was supported by research grants and fellowships to the authors from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by A.R. Horowitz.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Togni, P.H.B., Venzon, M., Souza, L.M. et al. Biodiversity provides whitefly biological control based on farm management. J Pest Sci 92, 393–403 (2019). https://doi.org/10.1007/s10340-018-1021-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-018-1021-x