Abstract
Classic biological control is generally understood as an environmentally safe practice of insect pest management. We investigated the overwintering abilities of Trichogramma brassicae Bezd. and T. cacoeciae Marchall in Tekirdağ Turkey. Parasitized eggs of Ephestia kuehniella Zell (Lep.: Pyralidae) by T. cacoeciae and T. cacoeciae were exposed under outdoor conditions between 17 August and 9 October. Emergence of T. brassicae and T. cacoeciae for the five exposure dates occurred in the same year. An emergence of 99% was observed in the following offspring for all of the tested parasitoid species exposed on 17 September. For eggs that were parasitized later than 9 November 2003, the emergence of parasitoids was in spring, on 19 March 2004. We found that T. brassica and T. cacoeciae were able to overwinter successfully on E. kuehniella. Fecundity of T. brassicae and T. cacoeciae females that overwintered on E. kuehniella was significantly different from the fecundity of control females that were reared under optimal conditions at 25°C. Our results demonstrate that the egg parasitoids T. brassicae and T. cacoeciae are able to overwinter successfully in Turkey .
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Trichogramma is a large genus of hymenopteran parasitoids used as biological control agents against a range of agricultural pests in large numbers, leading sometimes to spectacular successes (Smith 1996). Trichogramma spp are the most widely used natural enemies in biological control worldwide; and both native and exotic species have been mass-reared and released (Hassan 1993; Li 1994). The vast majority of Trichogramma spp are known to be polyphagous, attacking a wide range of lepidopterans as well as insects belonging to other orders (Thomson and Stinner 1989).
Egg parasitoids generally overwinter as immature stages within their host eggs. Consequently, unlike many overwintering insects, they are unable to directly choose a suitable and protected overwintering location and rely instead on physiological changes to successfully survive unfavorable conditions and environmental extremes. These egg parasitoids enter a state of dormancy under winter conditions during development within the host eggs. The main factor influencing dormancy is thought to be temperature (Boivin 1994; Rundle and Hoffman 2003).
Most studies on overwintering in egg parasitoids focus on the genus Trichogramma (Hymenoptera, Trichogrammatidae) in order to improve storage conditions and the potential for mass release (Pizzol and Voegele 1998; Voegele et al. 1998; Zaslavskii and Umarova 1990; Boivin 1994; Laing and Corrigan 1995; Özder 2002; Özder 2004). Trichogramma egg parasitoids are generally used for inundative biological control. Inundative releases are done every year.
We investigated whether T. brassicae and T. cacoeciae are able to survive winter under the climatic conditions in the Thrace regions of Turkey.
Materials and methods
Parasitoids
Prior to experiments, the parasitoids were reared for four to five generations on the Mediterranean flour moth, Ephestia kuehniella, at 16 h:8 h light:dark and 25±1°C.
Host
E. kuehniella were reared on wheat bran at 25±1°C and 60–70% relative humidity (RH).
Overwintering of T. brassicae and T. cacoeciae
For each parasitoid species and parasitization date, large batches of host eggs on strips were offered to groups of T. brassicae and T. cacoeciae females in a single plastic vial (3×12 cm) covered with gauze for 1 day at 25±1°C, 60–70% RH, 16 h:8 h light:dark. After 1 day, females were removed and ten strips were exposed under outdoor conditions or reared in an environmental chamber at 25±1°C, 60–70% RH. The treatment group was kept in a shelter under outdoor conditions at the Agricultural Faculty at Tekirdağ, Turkey. To prevent desiccation, eggs were misted with water once every other week. Host eggs were offered to parasitoids first on 17 and 25 September, 2, 12 and 17 October and 9 November.
Emergence of the control group from eggs kept under outdoor conditions was checked daily. After emergence, all parasitized eggs were checked 20 days later. After hatching of progeny, all eggs were dissected to confirm the hatching of a wasp or to determine mortality. The percentage of successful emergence was analyzed by ANOVA. Treatment means were compared by Scheffe tests.
Fecundity and longevity of overwintering females
In order to evaluate the fitness of overwintering females, we measured the fecundity and longevity of 20–26 females which developed in eggs of E. kuehniella under outdoor conditions from 9 October 2003 to late March 2004, in comparison with 20 females which were also reared on E. kuehniella but kept a constant temperature of 25°C (controls).
Overwintering and mated females less than 12-h old were individually transferred to glass vials (60.0×1.5 cm) provided with an egg card (about 50 eggs of E. kuehniella per card) and a small droplet of honey. After 24 h, the egg cards were removed and placed in a new vial, which was incubated under laboratory conditions (25±1°C, 60–70% RH, 16 h:8 h light:dark) until the emergence of parasitoid offspring. Fresh host eggs were provided daily; and the number of eggs offered was more than the maximum daily female capacity for parasitism. For each combination of parasitoids, the number of eggs parasitized was determined by counting the number of eggs which had turned black on the egg card. After hatching of progeny, all black eggs were dissected to confirm the hatching of a wasp or to determine mortality based on dead Trichogramma pupae. Fecundity and longevity of overwintering females and those developing under laboratory conditions were compared using the Mann–Whitney U-test.
Results
Overwintering of T. brassicae and T. cacoeciae
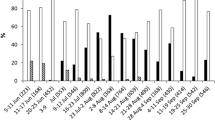
Emergence of T. brassicae from parasitized eggs kept under optimal laboratory conditions (controls) was determined at 99.80% for all parasitization dates. From the eggs parasitized on the first five dates and exposed outdoors, adult wasps emerged during the same autumn. Eggs parasitized after 17 October 2003 did not emerge during the same year. Emergence of T. brassicae in spring was from 19 March. Emergence of T. brassicae in the following offspring from eggs parasitized on 17 and 25 September, 2, 12 and 17 October and 9 November was determined (respectively, 99.60, 98.80, 99.20, 95.20, 92.40, 87.80%; Table 1). Successful emergence decreased from the first to the sixth exposure dates (ANOVA, F5,59=4.27, P<0.01). In fact, emergence from eggs exposed on the sixth date was significantly lower, compared with emergence from eggs exposed on the first date (Scheffe test, P<0.05). A difference was also found between the emergence of the controls in the laboratory at 25°C and the outdoor exposure (ANOVA, F1,19=7.74, P<0.01).
For T. cacoeciae, similarly, from eggs parasitized on the first five dates and exposed outdoors, the adult wasps emerged during the same autumn. For eggs parasitized on 9 November, the T. cacoeciae emerged in spring, on 19 March. Successful emergence decreased from the first to the sixth exposure dates (Table 1; ANOVA, F5,59=46.65, P<0.01) and a significant difference was observed between the first and sixth exposure dates (Scheffe test, P<0.05). A difference was also found between the emergence of the control in the laboratory at 25°C and the outdoor exposure (ANOVA, F1,19=67.83, P<0.01).
Fecundity and longevity of overwintering females
We investigated to what degree T. brassicae and T. cacoeciae females emerging from E. kuehniella eggs after approximately 5 months were able to successfully parasitize host eggs. The fecundity of T. brassicae females which survived the outdoor winter conditions was 40.46±22.80 eggs while the fecundity of females kept under controlled environmental conditions was 69.40±11.73 eggs. A significant difference was found between these two groups (Mann–Whitney U-test, U20,26=72, P<0.001) The fecundity of T. cacoeciae females which survived the outdoor winter conditions was 37.93±13.55 eggs while the fecundity of females kept under controlled environmental conditions was 67.60±9.19 eggs (Mann–Whitney U-test, U20,27=24, P<0.001). The longevity of females of T. brassicae (Mann–Whitney U-test, U20,26=118, P<0.01) and T. cacoeciae (Mann–Whitney U-test, U20,27=102, P<0.01) which survived the outdoor winter conditions declined relative to the controls (Table 2) .
Discussion
Our data indicate that T. brassicae and T. cacoeciae were able to overwinter in Tekirdağ in the Thrace parts of Turkey, on eggs of E. kuehniella. Successful emergence decreased from the first to the sixth exposure date. Emergence was, in our study, high compared with other studies on trichogrammatids (Babendreier et al. 2002). Weather conditions were good during the winter. In fact, temperatures did not drop below −9°C on 14 days and fell to −9°C only on 1 day during the winter. Overwintering lasted for 132 days for the sixth exposure date. These results are similar to those for other species: T. evanescens Westw. overwintered for 99 days in Egypt (Zaki 1996), while T. brassicae and T. evanescens were known to withstand temperatures as low as −20°C to −37°C (Boivin 1994; Babendreier et al. 2002). In our study, successful emergence was 87.80% and 64.40% from E. kuehniella eggs parasitized by T. brassicae and T. cacoeciae on 9 November. Winter mortality was, in our study, low compared with other studies on trichogrammatids (Burbutis et al. 1976; Babendreier et al. 2002).
Fecundity and longevity of overwintering female parasitoids decreased, compared with the control. Successful overwintering of biocontrol agents is of crucial importance for classic biological control (Overholt and Smith 1990). We conclude that T. brassicae and T. cacoeciae are well adapted to the environmental conditions in the Thrace parts of Tekirdağ and have the potential to establish outside their release fields. But, parasitoid overwintering success will also depend on the abundance of appropriate hosts (Tian et al. 1998).
References
Babendreier D, Kuske S, Bigler F (2002) Overwintering of the egg parasitoid Trichogramma brassicae (Hym.: Trichogrammatidae) in Northern Switzerland. Biocontrol 48:261–273
Boivin G (1994) Overwintering strategies of egg parasitoids. In: Wajnberg E, Hassan SA (eds) Biological control with egg parasitoids. CAB International, Wallingford, pp 219–244
Burbitis PP, Curl GD, Davis CD (1976) Overwintering of Trichogramma nubilale in Delaware. Environ Entomol 5:888–890
Hassan SA (1993) The mass rearing and utilization of Trichogramma to control lepidopterous pests: achievements and outlook. Pestic Sci 37:387–391
Laing JF, Corrigan JE (1995) Diapause induction and post-diapause emergence in Trichogramma minutum Riley (Hymenoptera: Trichogrammatidae): the role of host species, temperature and photoperiod. Can Entomol 127:103–110
Li LY (1994) Worldwide use of Trichogramma for biological control on different crops: a survey. In: Wajnberg E, Hassan SA (eds) Biological control with egg parasitoids. CAB International, Wallingford, pp 37–53
Overhold WA, Smith JW Jr (1990) Colonization of six exotic parasites hymenoptera against Diatraea grandiosella (Lep.: Pyralidae) in corn. Environ Entomol 19:1889–1902
Özder N (2002) Parasitization performance of Trichogramma cacoeciae, T. evanescens and T. brassicae (Hym. Trichogrammatidae) reared on the embryos of Ephestia kuhniella Zell. (Lep. Pyralidae) killed by freezing. Gt Lakes Entomol 35:107–111
Özder N (2004) Effect of different cold storage periods on parasitization performance of Trichogramma cacoeciae (Hymenoptera, Trichogrammatidae) on eggs of Ephestia kuhniella (Lepidoptera, Pyralidae). Biocontrol Sci Technol 14:441–447
Pizzol J, Voegele J (1988) The diapause of Trichogramma maidis Pintureau and Voegele in relation to some characteristics of its alternative host Ephestia kuehniella Zell. Colloq INRA 48:93–94
Rundle BJ, Hoffmann AA (2003) Overwintering of Trichogramma funiculatum carver (Hymenoptera: Trichogrammatidae) under semi-natural conditions. Popul Ecol 32:290–298
Smith SM (1996) Biological control with Trichogramma: advances, successes, and potential of their use. Annu Rev Entomol 41:375–406
Thomson MS, Stinner RE (1989) Trichogramma spp (Hymenoptera: Trichogrammatidae): field hosts and multiple parasitism in North Carolina. J Entomol Sci 24:232–240
Tian KF, Yang RX, Jiang YL, Tian KF, Yang RX, Jiang YL (1989) Study of the overwintering dynamics of Trichogramma closterae in nature. Nat Enemies Insects 20:106–111
Voegele J, Pizzol J, Babi A (1998) The overwintering of some Trichogramma species. Colloq INRA 43:275–282
Zaki FN (1996) Overwintering of Trichogramma evanescens Westw. in Egypt (Trichogrammatidae, Hymen.). Anz Schaedlingsk Pflanzens Umweltschutz 69:44–45
Zaslavskii VA, Umarova YaT (1990) Environmental and endogenous control of diapause in Trichogramma species. Entomophaga 35:23–29
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Özder, N., Sağlam, Ö. Overwintering of the egg parasitoids Trichogramma brassicae and T. cacoeciae (Hymenoptera: Trichogrammatidae) in the Thrace region of Turkey. J Pest Sci 78, 129–132 (2005). https://doi.org/10.1007/s10340-005-0083-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-005-0083-8