Abstract
A rapid and simple analytical method for the determination of ten chlorinated priority substances (hexachloro-1,3-butadiene, pentachlorobenzene, hexachlorobenzene, hexachlorocyclohexane isomers, heptachlor, and heptachlor epoxides) in fish samples using QuEChERS extraction, dual dispersive solid-phase extraction (dSPE) clean-up, and GC analysis was developed. For the extraction, two published extraction/partitioning procedures were evaluated, and the recoveries obtained for the analytes (in range 54–98 % with RSDs ≤15 %) were in favour of the conventional QuEChERS method. The use of the dual dSPE clean-up yields cleaner extracts than in the case of single dSPE, which enables the use of ECD for the detection of the analytes and simplifies the maintenance of the GC system. The method was optimised using homogenates of chub fish that is frequently sampled for monitoring purposes. The linearity of the method was evaluated using matrix-matched calibration curves (in the range 2–50 μg kg−1), and correlation coefficients (r 2) in the range 0.9927–0.9992 and RSDs of the relative response factors (RRF) below the value of 20 % were achieved. LODs ranged from 0.5 to 1.1 μg kg−1, while LOQs ranged from 1.5 to 3.5 μg kg−1. The accuracy of the method was verified by the analysis of the NIST standard reference material SRM 1946 (Lake Superior Fish Tissue), and most of the analytes of interest presented good agreement with the certified values.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Among the requirements of the Environmental Quality Standards Directive 2008/105/EC [1], amending the European Water Framework Directive (WFD) [2] is the monitoring of biota samples for the presence of those priority substances (PS) that tend to accumulate in biota. In this directive, hexachlorobenzene and hexachloro-1,3-butadiene were selected from organic PSs as first to be monitored in biota, and environmental quality standards (EQS) have been derived for them. The EQS for hexachlorobenzene was set at 10 μg kg−1 ww (wet weight) considering the risks to humans, while hexachloro-1,3-butadiene was set at 55 μg kg−1 ww based on the protection of top predators from second poisoning. In the EC guidance document No. 25 [3], hexachlorocyclohexane (HCH) isomers and pentachlorobenzene were suggested from other chlorinated PS for sediment and biota trend monitoring. However, the EQS values for these compounds were not established yet for the biota. The very low EQS of 6.7 × 10−3 μg kg−1 ww has been derived for heptachlor and heptachlor epoxides with a goal of protecting human health via consumption of fishery products [4].
The results from the monitoring of selected PSs for compliance checking with biota EQSs have been recently published in several European studies. The presented results showed that pentachlorobenzene was not detected in the samples of eels in Scotland [5]. Hexachloro-1,3-butadiene was below the EQS value in all samples, except one, and hexachlorobenzene was below the EQS in majority of samples of different fish species caught in rivers of Scotland [5], France [6], England [7], Czech Republic [8], and Spain [9]. The exception was the catfish samples caught from the Ebro River (Spain) with total mass in range from 5.5 to 55 kg [9]. According to the 26 results from the analysis of catfish tails, the EQS was exceeded 17 times for hexachlorobenzene and only once for hexachloro-1,3-butadiene. The concentrations of HCH isomers in eels in Scotland were generally <3 μg kg−1 ww or below detection [5], while in fish from fishing grounds of the Czech Republic, the highest determined value was for lindane 1.0 μg kg−1 ww [8]. The highest exceedance (70 %) of the strict biota EQS value was obtained for heptachlor epoxide in fish survey accomplished in Denmark [10]. The maximum determined concentration of heptachlor epoxide was 23 × 10−3 μg kg−1 ww, while heptachlor was in all fish samples below LOD of 0.7 × 10−3 μg kg−1.
A great variety of methods have been used for the extraction of organochlorine compounds from fish samples and have been combined with different methods of clean-up and analysis of the extracts. Examples of methods employing solid–liquid extraction (SLE), Soxhlet extraction, accelerated solvent extraction (ASE), supercritical fluid extraction (SFE), microwave-assisted extraction (MAE), matrix solid-phase dispersion (MSPD), and so-called QuEChERS method along with the used extracting agents, clean-up procedures, and methods of instrumental analysis are listed in Table S1 (see Electronic Supplementary Material). The presented methods involve both classic extraction methods (Soxhlet, SLE) that are usually time-consuming (extraction times up to 24 h), labour-intensive, and require large volumes of solvent (up to few hundreds of mL), and novel methods (others in Table S1) in which the duration of sample extraction can be decreased to 10–60 min and the volumes of extraction media used are substantially lower. However, the disadvantage of ASE, SFE, and MAE methods lies in the cost of equipment setup.
In the last decade, the QuEChERS (stands for quick, easy, cheap, effective, rugged, and safe), a novel low-cost sample preparation method, is gaining significant popularity in the analysis of organic analytes in food samples [11]. The original method [12] involves an acetonitrile (MeCN) salting-out extraction of a solid sample in an aqueous environment followed by dSPE to remove a majority of the matrix interferences. Few papers [13–16] (presented in Table S1) are dealing with the determination of some of the selected PS in fish using the QuEChERS sample preparation method (in its original or modified form) in combination with gas chromatography and mass spectrometry (GC–MS and GC–MS/MS). The major modifications of the original method were related to the clean-up process. In the presented works, in general, more complex clean-up procedures were applied to ensure satisfactory removal of co-extractives (mainly lipids) from the final extract. The problem of co-extractives is that they can build up in a GC injector or column resulting in poor chromatography and also can cause interferences and false positive results. Therefore, in the cases of samples with higher lipid content, the single dSPE treatment may be insufficient for the extract clean-up prior to the instrumental analysis.
The aim of the present work was the development and validation of a simple and rapid method for the determination of ten chlorinated PS in fish using QuEChERS sample preparation with dual dSPE clean-up and subsequent GC-ECD analysis.
Experimental
Standards and Reagents
Individual standards of pentachlorobenzene, hexachlorobenzene, hexachloro-1,3-butadiene, α-HCH, β-HCH, lindane, δ-HCH, heptachlor, and heptachlor-exo-epoxide at a concentration of 10 ng μL−1, and heptachlor-endo-epoxide at 100 ng μL−1 in cyclohexane, and a neat standard of 1,3,5-tribromobenzene were purchased from Dr. Ehrenstorfer (Augsburg, Germany). The purity of the standards ranged from 98.5 to 99.5 %.
Anhydrous magnesium sulphate (ReagentPlus), sodium chloride (ReagentPlus), and acetonitrile (MeCN, Chromasolv) were obtained from Sigma–Aldrich (Steinheim, Germany). Primary secondary amine (PSA) and C18 endcapped bulk sorbent were from Agilent Technologies (Santa Clara, CA, USA). Toluene and acetone Emsure grade, cyclohexane SupraSolv grade, and ethyl acetate LiChrosolv grade were purchased from Merck (Darmstadt, Germany).
Working standard mixture solutions of chlorinated PS at concentrations of 1 ng μL−1 in cyclohexane and 0.1 ng μL−1 in acetone were prepared from the individual standard solutions by dilution with the corresponding solvents. 1,3,5-tribromobenzene was dissolved in acetone and was used as an internal standard (IS) at concentration of 0.5 ng μL−1.
Fish Samples
Chub (Squalius cephalus), one of the most frequently sampled species for biota monitoring in European rivers [4], has been chosen as a source of testing matrix for the development of the present method. The chub samples were obtained during a fish survey performed in Slovak water bodies in 2011 within the project: Monitoring and assessment of water body status (see acknowledgment). The fish were collected by electrofishing and were deep frozen after catch until sample preparation. After thawing, composite samples were prepared from several pieces of the whole fish from each sampling site to give a wet weight at least 600 g. The samples were homogenized using a food chopper Eta 6078 (ETA, Hlinsko, Czech Republic) and tissue homogenizer SilentCrusher M (Heidolph Instruments, Schwabach, Germany). The fish homogenates were stored in a freezer at −20 °C until extraction and analysis.
For the validation of the proposed method, the standard reference material SRM 1946 (Lake Superior Fish Tissue) was purchased from the National Institute of Standards and Technology (NIST, Gaithersburg, MD, USA). This SRM was a frozen fish tissue homogenate, prepared from lake trout (Salvelinus namaycush), with 10.2 % of extractable fat and 71.4 % of water.
To compensate for matrix effects, matrix-matched calibration curves were prepared by analysing blank chub composite samples (with 7.1 % of total lipids, 71.0 % of water, and no pesticides detected previously) spiked with studied analytes at concentrations of 2, 5, 10, 20, 30, and 50 μg kg−1. The IS solution was added to the extracts (at 20 ng mL−1) after finishing the clean-up step. The spiked blank fish samples were also used to perform recovery experiments.
Moisture and Lipid Determination
The moisture and lipid determination was accomplished according to our previous work [17].
Sample Preparation Method
Five mL of MeCN was added to 5 g of fish homogenate weighed in a 50-mL polypropylene centrifuge tube (Corning CentriStar, Sigma–Aldrich). The tube was closed with a stopper and shaken using vortex mixer (Stuart SA8, UK) at 800 rpm for 1 min. Then, 2 g of anhydrous MgSO4 and 0.5 g of NaCl were added, and again, the tube was shaken vigorously for 1 min. Then, it was centrifuged (centrifuge Rotina 380, Hettich, Tuttlingen, Germany) at 5000 rpm (4863 rcf) for 5 min. Next, a 2-mL aliquot of the upper organic phase was transferred into a 15-mL centrifuge tube containing 150 mg PSA, 150 mg C18, and 300 mg MgSO4. The tube was shaken for 1 min and centrifuged for 5 min at 5000 rpm. Then, the whole supernatant was transferred into a second 15-mL centrifuge tube containing 50 mg PSA, 50 mg C18, and 150 mg MgSO4. Again, the tube was shaken for 1 min and centrifuged for 5 min at 5000 rpm. Finally, a 0.5-mL aliquot of the supernatant was transferred into a 10-mL graduated tube, mixed with 1 mL of toluene and 20 μL of IS solution (1,3,5-tribromobenzene, 0.5 ng/μL), and concentrated to a volume of 0.5 mL by evaporation in a stream of air. The cleaned-up fish extract was transferred into an autosampler vial for GC analysis.
Gas Chromatographic Analysis
An Agilent Technologies 7890A GC system (Palo Alto, CA, USA) with a split/splitless injector and a micro-electron capture detector (μECD) was used for the analysis of fish homogenate extracts. The chromatograph was equipped with an HP-5 capillary column (30 m × 0.32 mm I.D. × 0.25 μm film thickness) connected to a 5 m × 0.53 mm I.D. uncoated retention gap (Agilent Technologies). The columns were connected by an Agilent Ultimate Union. Helium (purity 4.6, Messer Tatragas, Bratislava, Slovak Republic) was used as the carrier gas at constant flow of 1.8 mL min−1.
The GC oven temperature was programmed from 60 °C (initial hold time 1 min) to 170 °C at a rate of 40 °C min−1, and then to 300 °C at a rate of 10 °C min−1. The μECD detector was maintained at 300 °C, and the make-up nitrogen flow in the detector was set at 25 mL min−1. The temperature of the injector was 250 °C. Extract aliquots of 3 μL were injected into the column by means of an autosampler using a pulsed splitless injection with an injection pulse pressure at 65 psi until 1 min.
The analysis of GC-amenable co-extractives was carried out on a Hewlett-Packard 6890 GC system (Palo Alto, CA, USA) coupled to a 5972 mass selective detector (MSD). The analytes were separated on an HP-5MS capillary column (30 m × 0.25 mm I.D., film thickness 0.25 μm) from Agilent Technologies using helium as the carrier gas at a constant pressure of 11.2 psi. Temperatures were as follows: injection port 250 °C, transfer line 300 °C, and ion source 169 °C. The following oven temperature programme was employed: initial temperature 70 °C (hold time 2 min), increasing at a rate of 25 °C min−1 to 150 °C, then at a rate of 3 °C min−1 to 200 °C, and finally, at a rate of 8 °C min−1 to 300 °C (final hold time 3 min). Residues from the sample clean-up procedure were re-dissolved in ethyl acetate and injected (1 μL) into the GC column in the splitless mode (1 min). The MSD was operated in the scan mode, and the mass range was scanned from m/z 50 to 500 every 1.70 s. The identification of chromatographic peaks was done using Wiley7n mass spectral library.
Results and Discussion
GC-ECD Analysis Method
The GC oven temperature program for the analysis of selected PS was adopted from Agilent Technologies application note [18]. Because of the possibility of injection of larger volumes of sample and for obtaining better analytical performance (to improve sensitivity and protect the analytical column contamination), the 5 m × 0.53 mm I.D. uncoated retention gap was installed before the analytical capillary column. For injection of sample extracts, pulsed splitless injection mode was selected to maximize the transfer of the analytes into the column without the risk of backflash and to minimize their degradation in the injector liner. The parameters of pressure pulse and injected sample volume have been tested for optimum analytical performance. For the pressure pulse, the optimum values for injection pressure and pulse time obtained were 65 psi and 1 min, respectively. The maximum volume of injected sample was 3 μL, because for the higher volumes, the peak distortion of early eluting analytes occurred.
Co-extractives
Large amounts of co-extractives present in the sample extract can cause problems in instrumental analysis, which were mentioned in the introduction. To avoid these problems, it has been estimated that a fat residue of less than 0.25 mg mL−1 is required for the GC-ECD analysis [19]. Therefore, a clean-up procedure employing dual dSPE [16] was involved into the developed method and was optimised. For the clean-up, a mixture of PSA and C18 sorbents and anhydrous MgSO4, which has been proven effective in removing co-extracted lipids from fatty animal matrices [20, 21], was chosen.
In the first experiment, 10 g aliquots of fish homogenate were processed by extraction/partitioning using 10 mL of MeCN, 4 g of anhydrous MgSO4, and 1 g of NaCl. After centrifugation, 2 mL aliquots of the supernatant solution were subjected to clean-up by the first dSPE with various amounts of anhydrous MgSO4, PSA, and C18 sorbent (see Table 1). Next, 1 mL extract aliquots obtained after dSPE clean-up and centrifugation were transferred into pre-weighed glass tubes and evaporated to dryness under the stream of air. The weights of the co-extractives residues and co-extractives’ removal efficiency for the tested dSPE mixtures are presented in Table 1. According to the presented results, the mixture with 300 mg of anhydrous MgSO4, 150 mg of PSA, and 150 mg of C18 was chosen for the 1st dSPE clean-up step.
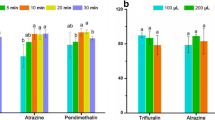
In the second experiment, the whole supernatant after the first dSPE clean-up step with optimal sorbent mixture was subjected to the second dSPE clean-up. Similarly, as in the first experiment, different amounts of PSA, C18, and anhydrous MgSO4 were tested to obtain a higher degree of co-extractives removal and a cleaner final extract. The result of the optimisation of the fish homogenate clean-up procedure is presented in Fig. 1. In Fig. 1a, total ion chromatograms from GC–MS analysis of co-extractives residues after single dSPE clean-up and after dual dSPE clean-up with different amounts of sorbent mixtures are shown. It can be seen that when in the second dSPE clean-up step 150 mg MgSO4, 25 mg PSA, and 25 mg C18 had been used, fatty acids, glycerides, and cholesterol were still present in the extract, similarly as in the case of single dSPE clean-up. The third chromatogram in Fig. 1a revealed that the optimal clean-up mixture for the second dSPE was composed of 150 mg MgSO4, 50 mg PSA, and 50 mg C18. Fatty acids were almost complete, and glycerides and cholesterol were partially removed with this mixture. In the chromatograms from the GC-ECD analysis (Fig. 1b) of the same extracts can be clearly seen the difference in the co-extractives removal efficiency when using single dSPE clean-up and dual dSPE clean-up, respectively. The optimised dual dSPE clean-up was sufficient for the analysed fish samples with lipid content up to 10 %.
a Total ion chromatograms from the GC–MS analysis of co-extractives residues obtained after first dSPE clean-up and after dual dSPE clean-up with different amounts of sorbent mixtures. The identified compounds: 1 hexadecenoic acid, Z-11-; 2 palmitic acid; 3 linoleic acid; 4 2-palmitoylglycerol; 5 2-oleoylglycerol; 6 cholesterol. b Chromatograms from the GC-ECD analysis of the same extracts after single dSPE clean-up and optimised dual dSPE clean-up
Exchange of Solvent
Direct injection of MeCN extract is problematic for the GC analysis because of poor focusing of chromatographic peaks due to the high polarity of MeCN and limited injection volume due to its high-expansion coefficient. In addition, MeCN strips the deactivation and can damage the column film [22]. To overcome these drawbacks, toluene was chosen as an exchange solvent. The toluene enables obtaining good chromatographic resolution of the analytes on a non-polar GC column and using higher injection volumes in the splitless mode. For the replacement of MeCN by toluene, it is advantageous that the toluene is of low volatility (often used as a keeper solvent), miscible with MeCN, and that the binary mixture of 80 % MeCN and 20 % toluene forms a positive azeotrope with boiling point of 81.4 °C [23].
For solvent exchange, an aliquot of 0.5 mL of MeCN extract was transferred into a 10 mL graduated tube, mixed with 1 mL of toluene, and evaporated to the final volume of 0.5 mL under a stream of air. During the evaporation process, the lower boiling binary azeotrope is removed preferentially, leading to MeCN-free toluene, which was added in excess.
The solvent exchange procedure was tested using an MeCN solution fortified with the studied analytes, each at 50 ng mL−1. The recovery of the analytes was evaluated by comparing their concentration in the resulting toluene solution to the spiking concentration determined by the GC-ECD method. The procedure was carried out with four replicates, and the average recoveries were in the range from 94.0 ± 2.7 % for hexachloro-1,3-butadiene to 106.8 ± 6.3 % for heptachlor. Thus, the solvent exchange procedure was performed without a significant loss of analytes of interest.
Evaluation of Two Extraction/Partitioning Procedures
Extraction/partitioning procedures, adopted from the works of Anastassiades et al. [12] and Liu et al. [24], were evaluated for the determination of selected organochlorine compounds in fish matrix. In both the cases, half the amounts of sample, extracting agents, and inorganic salts as compared with the original works were used. In the first procedure (conventional QuEChERS method), 5 g of fish homogenate, 5 mL of MeCN, 2 g of MgSO4, and 0.5 g of NaCl were used. In the second procedure, everything was the same as in the first, with the exception that the NaCl was substituted for chloroform (1 mL). As the authors of the modified QuEChERS method [24] state, the addition of chloroform helps to remove water from MeCN phase, thus leading to decreasing the co-extraction of polar matrix components. In the experiment, the whole procedure (described in “Sample preparation method”) involving extraction/partitioning, dual dSPE clean-up, and exchange of solvent was employed. The blank chub composite samples were spiked with the analytes at 30 μg kg−1. To calculate the recoveries, the matrix-matched standards prepared by spiking the blank fish extracts at the concentration of the analytes equal to that in the samples were analysed.
The results from the recovery experiment are presented in Fig. 2. Average recoveries and relative standard deviations (RSDs) for the six replicate analyses indicate that the extraction/partitioning procedure of the conventional QuEChERS method gave better recovery and similar repeatability results as compared to the procedure of the modified QuEChERS method. Thus, the procedure originally modified to obtain a better analytical selectivity with higher recoveries for polar analytes as compared to the conventional QuEChERS procedure did not bring an improvement of analytical performance for non-polar organochlorine pesticides. Therefore, the first procedure was involved in the sample preparation method.
Method Performance Characteristics
Once the sample preparation procedure and GC-ECD analysis conditions have been optimised, the performance characteristics of the whole analytical method were determined. The recovery and repeatability (RSD) of the method were evaluated from six repeated analyses of a blank chub composite sample spiked at 5 and 50 μg kg−1 levels, respectively. From the results presented in Table 2, it can be seen that the recovery for most of the analytes falls within the ideal acceptable recovery range of 70–120 % with an RSD ≤20 % [25]. The lowest recovery of 54 % that was obtained for hexachlorobenzene, the most lipophilic analyte tested, is in line with recoveries obtained by employing the QuEChERS method for the analysis of fatty food matrixes [26]. The recoveries for HCH isomers, heptachlor, and heptachlor epoxide are similar, as presented in [13, 14, 27] on the analysis of organochlorine compounds in fish using QuEChERS methodology. The lower recoveries for these compounds were obtained in the work of Molina-Ruiz et al. [16].
Response linearity for the studied analytes was evaluated by matrix-matched calibration standards at six concentrations levels in the range of 2–50 μg kg−1. To evaluate the linearity of the calibration curves, correlation coefficients of the curves (r 2) as well as RSDs of the relative response factors (RRF) were investigated. The RRFs were calculated for the analytes relative to the internal standard at each concentration level. For all the analytes, the calibration curves were linear with r 2 values in the range of 0.9927–0.9992 and RSDs of the RRFs below the value of 20 % (see Table 2).
The limits of the method were determined by analysing seven replicates of the blank chub composite sample spiked at 2 μg kg−1. The limits of detection (LOD) and quantification (LOQ) were calculated as three and ten times the standard deviation of the determination, respectively. As can be seen in Table 2, the LOQs for all the investigated organochlorine compounds are in the range 1.5–3.5 μg kg−1. The LOQs for hexachloro-1,3-butadiene and hexachlorobezene are below 30 % of the EQS, while the LOQs for heptachlor and heptachlor epoxides, similarly as in the cited works, do not meet the requirement for the EQS (6.7 × 10−3 μg kg−1) set by the WFD.
The method’s accuracy was confirmed by the analysis of selected chlorinated PS in a NIST SRM 1946 standard fish tissue reference material prepared from lake trout. Figure 3 presents a comparison between measured and certified concentrations (means from four replicate analyses) of four organochlorine compounds in the SRM. Good agreement with the certified values was found for hexachlorobenzene, α-HCH, and lindane with achieved recoveries in the range 85–117 %. The recovery outside the acceptable range with value of 163 % was obtained only for heptachlor epoxide, which was probably due to matrix enhancement effect.
The presented characteristics show a satisfactory performance of the proposed method.
Conclusions
A rapid and simple analytical method for the determination of organochlorine compounds in fish samples using QuEChERS extraction, dual dSPE clean-up, and GC analysis was developed. For the extraction, two extraction/partitioning procedures were evaluated, and the recoveries obtained for the studied analytes were in favour of the conventional QuEChERS method. The use of the dual dSPE clean-up yields cleaner extracts than in the case of single dSPE, which enables the use of ECD for the detection of the analytes and simplifies the maintenance of the GC system. The solvent exchange from MeCN to toluene in the final extract allows the injection of larger volumes of sample into the GC (lowering the LODs). Furthermore, the toluene extract is more compatible with a non-polar coating of the GC column that enables obtaining a good chromatographic resolution and peak shape of the analytes.
The method was optimised using samples of chub fish that belongs to the most frequently sampled species for biota monitoring. The obtained performance characteristics show that the method is applicable to the analysis of the studied analytes at environmentally relevant concentrations. Problematic is only the EQS requirement for heptachlor and heptachlor epoxides that is not achievable with the employed approach and instrumentation.
The method was verified by the analysis of the standard fish tissue reference material, and most of the analytes of interest presented good agreement with the certified values.
References
European Commission (2008) Directive 2008/105/EC of the European Parliament and of the Council on environmental quality standards in the field of water policy, amending and subsequently repealing Council Directives 82/176/EEC, 83/513/EEC, 84/156/EEC, 84/491/EEC, 86/280/EEC and amending Directive 2000/60/EC. Off J Eur Commun L 348:84–97
European Union (2000) Directive 2000/60/EC of the European Parliament and of the Council, establishing a framework for community action in the field of water policy. Off J Eur Union L 327:1–72
European Union (2010) Common implementation strategy for the Water Framework Directive (2000/60/EC): guidance document no. 25 on chemical monitoring of sediment and biota under the Water Framework Directive. Office for Official Publications of the European Communities, Luxembourg
European Union (2014) Common implementation strategy for the Water Framework Directive (2000/60/EC): guidance document no. 32 on biota monitoring (the implementation of EQSBIOTA) under the Water Framework Directive. Office for Official Publications of the European Communities, Luxembourg
Macgregor K, Oliver IW, Harris L, Ridgway IM (2010) Environ Poll 158:2402–2411
Miège C, Peretti A, Labadie P, Budzinski H, Le Bizec B, Vorkamp K, Tronczyński J, Persat H, Coquery M, Babut M (2012) Anal Bioanal Chem 404:2721–2735
Jürgens MD, Johnson AC, Jones KC, Hughes D, Lawlor AJ (2013) Sci Tot Environ 461–462:441–452
Cerveny D, Zlabek V, Velisek J, Turek J, Grabic R, Grabicova K, Fedorova G, Rosmus J, Lepic P, Randak T (2014) Ecotox Environ Safe 109:101–109
Lava R, Majoros LI, Dosis I, Ricci M (2014) Trends Anal Chem 59:103–111
Vorkamp K, Bossi R, Bester K, Bollmann UE, Boutrup S (2014) Sci Tot Environ 470–471:459–468
Rejczak T, Tuzimski T (2015) Open Chem 13:980–1010
Anastassiades M, Lehotay SJ, Štajnbaher D, Schenck FJ (2003) J AOAC Int 86:412–431
Norli HR, Christiansen A, Deribe E (2011) J Chromatogr A 1218:7234–7241
Kalachova K, Pulkrabova J, Cajka T, Drabova L, Stupak M, Hajslova J (2013) Anal Bioanal Chem 405:7803–7815
Sapozhnikova Y (2014) J Agric Food Chem 62:3684–3689
Molina-Ruiz JM, Cieslik E, Cieslik I, Walkowska I (2015) Environ Sci Pollut Res 22:369–378
Tölgyessy P, Miháliková Z (2016) Food Control 60:44–49
Meng C-K (2011) The GC/MS/MS analyzer and the pesticides and environmental pollutants MRM database. Application note, Agilent Technologies, Santa Clara, CA, USA, PN 5990-9453EN
Gillespie AM, Daly SL, Gilvydis DM, Schneider F, Walters SM (1995) J AOAC Int 78:431–437
Castillo M, Gonzáles C, Miralles A (2011) Anal Bioanal Chem 400:1315–1328
Sapozhnikova Y, Lehotay SJ (2013) Anal Chim Acta 758:80–92
Pesticide Analyzer Reference (2009) Thermo Fisher Scientific, Austin, TX, USA, PN 120390-0001
INEOS (2007) Acetonitrile. Safe storage and handling guide. http://www.ineos.com/Global/Nitriles/SHE/2007_acetonitrile_brochure.pdf. Accessed 20 Jan 2016
Liu G, Rong L, Guo B, Zhang M, Li S, Wu Q, Chen J, Chen B, Yao S (2011) J Chromatogr A 1218:1429–1436
Document No SANCO/12495/2011 (2011) Method validation and quality control procedures for pesticides residues analysis in food and feed. EU, Brussels
Lehotay SJ, Maštovská K, Yun SJ (2005) J AOAC Int 88:630–638
Barriada-Pereira M, Gonzalez-Castro MJ, Muniategui-Lorenzo S, Lopez-Mahia P, Prada-Rodriguez D (2010) J AOAC Int 93:992–998
Acknowledgments
Financial support from the EU Cohesion Funds within the project Monitoring and assessment of water body status (No. 24110110001, 2008–2012 and No. 24110110158, 2012-2015 Phase II) is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All applicable international, national, and institutional guidelines for the care and use of animals were followed.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tölgyessy, P., Miháliková, Z. & Matulová, M. Determination of Selected Chlorinated Priority Substances in Fish using QuEChERS Method with Dual dSPE Clean-up and Gas Chromatography. Chromatographia 79, 1561–1568 (2016). https://doi.org/10.1007/s10337-016-3160-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-016-3160-7