Abstract
The dichotomy between the two Pan species, the bonobo (Pan paniscus) and chimpanzee (Pan troglodytes) has been strongly emphasized until very recently. Given that most studies were primarily based on adult individuals, we shifted the “continuity versus discontinuity” discussion to the infant and juvenile stage. Our aim was to test quantitatively, some conflicting statements made in literature considering species differences between immature bonobos and chimpanzees. On one hand it is suggested that infant bonobos show retardation in motor and social development when compared with chimpanzees. Additionally it is expected that the weaning process is more traumatic to chimpanzee than bonobo infants. But on the other hand the development of behaviors is expected to be very similar in both species. We observed eight mother–infant pairs of each species in several European zoos. Our preliminary research partially confirms that immature chimpanzees seem spatially more independent, spending more time at a larger distance from their mother than immature bonobos. However, the other data do not seem to support the hypothesis that bonobo infants show retardation of motor or social development. The development of solitary play, environmental exploration, social play, non-copulatory mounts and aggressive interactions do not differ between the species. Bonobo infants in general even groom other group members more than chimpanzee infants. We also found that older bonobo infants have more nipple contact than same aged chimpanzees and that the weaning process seems to end later for bonobos than for immature chimpanzee. Additionally, although immature bonobos show in general more signs of distress, our data suggest that the weaning period itself is more traumatic for chimpanzees.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
For years the differences between bonobos (Pan paniscus) and chimpanzees (Pan troglodytes) have been stressed. Presumably typical bonobo characteristics such as female dominance (Parish 1996), highly promiscuous sexual interactions (De Waal 1987; Kano 1992), low levels of aggression (Kuroda 1980; de Waal 1998), higher reconciliatory tendencies (de Waal 1998) and the apparent absence of infanticide (de Waal 1998; Kano 1998) all contrasted markedly with chimpanzee features. However, as research on bonobo socio-ecology and behavior gradually adds up, the bonobo-chimpanzee dichotomy needs to be reconsidered (Fruth et al. 1999; Doran et al. 2002; Hohmann and Fruth 2002; Vervaecke et al. 2003a). More recent research, revealing non-exclusive female dominance (Vervaecke et al. 2000; Stevens et al. 2001, 2003), severe aggression (Parish 1996; Vervaecke et al. 2000; Hohmann 2001; MDL personal observation), highly individual variation in reconciliatory tendencies (Palagi et al. 2004; B. Meuleman, personal communication), infant harassment (Vervaecke et al. 2003b; personal observation), and even possible infanticide by females (Hohmann and Fruth 2002), shifted nuances and suggests that there is considerable overlap in the adaptive potential of bonobo and chimpanzee behavior.
While generally research on adult bonobos has been the focus, information on infant and juvenile behavior is lagging behind. Hardly any quantitative data or results are reported when comparing the behavior of immature bonobos and chimpanzees, and our understanding today remains primarily based on qualitative comparisons. So far only a few papers specifically reported on behavioral development in the wild (Kuroda 1989; Enomoto 1990; Hashimoto and Furuichi 1994). Even detailed information from immature captive animals, which can help to interpret the scarce data from field research, is rare (Johnson 1997; Weaver 1997). Additionally, the interpretations made are conflicting: on one hand it is suggested that bonobos are paedomorfic and show retardation in motor and social development when compared with chimpanzees (Kuroda 1989). On the other hand the similarities between the species’ development seem to far outnumber the differences (Johnson 1997). Our aim is to make the first quantitative inter-specific comparison using data on captive mother-reared individuals in order to test these conflicting statements.
First we will investigate the hypothesis that bonobos show a temporal retardation in motor and social skills (Kuroda 1989). Kuroda (1989, 2000) states that wild bonobos at 6 months of age are unable to move independently and that bonobo mobility remains poor until about 1-year old. One-year-old infants begin to play with other infants, but mothers keep them within hand reach (Kuroda 1989). Chimpanzees of the same age act much more independently as they often leave their mothers for more than 10 m (Kuroda 1989). Hence, we predict that, at least until 1 year of age, (1) bonobo infants will spend more time in ventral contact with their mother and (2) will spend less time not in contact with their mother when compared to chimpanzee infants. Additionally we predict that around 1 year of age (3) chimpanzee infants spend more time at a lager distance from their mother, (4) spend more time in solitary play and (5) environmental exploration, as well as more time socially interacting with other group members [(6) social play and (7) grooming] than bonobo infants.
Second we will test the hypothesis that the species have different weaning strategies. Although wild bonobo infants are weaned at approximately the same age as chimpanzees (Kuroda 1989), it has been suggested that the process may be more traumatic for chimpanzees than for bonobos: no weaning tantrums by bonobo juveniles have been reported in the wild (Kano 1992) and active rejection is not typically practiced by bonobo mothers (Kuroda 1989). In wild and captive semi-free ranging chimpanzees, infants are gradually weaned over a 2-year period and are considered completely weaned between 3.5 and 4.5 years of age (van Lawick-Goodall 1968; Clark 1977). We will test the predictions that chimpanzees and bonobos are weaned at approximately the same age as bonobos and that the weaning process is more traumatic to chimpanzees than to bonobos by inter-specifically comparing nipple contacts (8) and expression of distress (9).
Third we will investigate the hypothesis that the two species show differences in development of socio-sexual and aggressive behavior. Adult bonobos have highly promiscuous sexual interactions (de Waal 1987; Kano 1992) and are supposed to behave less aggressive (Kuroda 1980; de Waal 1998), than chimpanzees. Adult bonobos, in contrasts with chimpanzees, show a variety of non-copulatory sexual behaviors, which serve many social functions such as reducing tension among individuals, terminating agonistic interactions and forming and supporting new affiliative social relationships (de Waal 1987; Kano 1989, 1992; Hohmann and Fruth 2002). Although quantitative data on the performance of sexual behaviors of immature chimpanzees and bonobos are rare, the nature of behaviors expressed is relatively similar (Van Lawick-Goodall 1968; Hasegawa and Hiraiwa-Hasegawa 1983; de Plooij 1984; Kitamura 1989).
We will test the prediction that bonobo infants and juveniles are sexually more active and less aggressive than chimpanzees by comparing the development of sexual inspections (10), non-copulatory mounting (11) and severe aggressions (12).
Methods
Behavioral data collection
Study subjects include eight infants of each species, living in seven zoos (Tables 1, 2). Focal animal sampling on infants was conducted between January 2000 and December 2002 (for detailed information, see Table 1). Data were collected using instantaneous sampling (intervals of 15 s) (Altmann 1994). Each infant was sampled on average over five periods in 3 years. Infant age ranged from 5 to 56 months. An observation period consisted on average of six consecutive days. Each day five sessions of 30 min, randomly spread throughout the day, were carried out, resulting in 2.5 h of observation per day and 15 h per period. In total data were collected during 1,090 h. All mother–infant pairs were socially housed in multi-male multi-female social groups including animals of different age classes (except for the mother-infant pairs kept at Frankfurt Zoo, where group composition changed due to fission–fusion management and subgroups did not always contain an adult male). The bonobo group sizes ranged from 8 to 12 individuals; the chimpanzee group sizes ranged from 12 to 34 individuals. Given that our study was conducted during the course of 3 years, group compositions inevitably changed due to natural dynamics (Table 3).
All behaviors, as described below, were calculated as proportions of the observed time.
(1) Ventral contact: infant clings ventrally to the mother. (2) No contact: the infant does not touch the mother. (3) Distance far: proportion of no contact time that the infant spends at a distance of more than 5 m away from the mother. (4) Solitary play: climbing, swinging, rolling, and jumping, etc. including object play. (5) Exploration of the environment: touching, manipulating, licking, and sniffing the surrounding environment (this does not include objects). (6) Social play: mouthing, wrestling, chasing individuals other than the mother; only those bouts initiated by the infant itself usually expressing a playface or relaxed facial expression were included. (7) Grooming: manipulating (e.g. licking, nibbling, picking with the fingers) an individual’s body surface. Only grooming by the infants themselves was included.
(8) Nipple contact: the infant holds his mother’s nipple in his mouth. (9) Distress: scratching (repeated movement of the hand or foot during which the fingertips are drawn across the hair), pouting, whimpering and tantrum. (10) Sexual inspections: inspecting (i.e. smelling, licking, touching) the genitals of a group member excluding the mother. (11) Non-copulatory mounts: all mounts or genito-genital contacts between the infant and the other group members, excluding the mother, which were initiated by the infant. (12) Aggression: all brusque movements, full speed pursuits and/or physical attacks often accompanied by high-pitched vocalizations; only interactions initiated by the infant with other group members, excluding the mother, were included.
Statistics
We conducted ancovas, to investigate species differences; proportions of behaviors are the dependent variable, infant age is a covariate. We tested the main effects “species” and “infant sex” and focused on the interaction effect “species*infant age” and “species*sex infant”. Because we sampled the infants several times during 3 years, a repeated measures design was used with a random statement and the Kenwardroger option (Verbeke and Molenberghs 1997). The data were transformed to meet the requirements for parametric testing (Sokal and Rohlf 1981). All tests were two-tailed, performed in SAS (Version 8.0), and the alpha value was set at 0.05.
Results
The statistical results for the main effects species, infant age and infant sex and the significant interactions are included in Table 4.
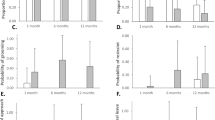
From the 12 behaviors tested, only four showed a significant “species” effect: distance far (3), grooming (7), distress (9), and sexual inspections (10). Chimpanzee infants in general spend more time at a larger distance from their mother then bonobo infants [F (1,25)=23.63, P<0.0001] (Fig. 1). They spend less time grooming other group members then bonobo infants [F (1,11)=14.77, P<0.01] (Fig. 3) and they show in general less signs of distress compared to bonobo infants [F (1,43)=9.87, P<0.01]. However, from 40 months of age onward chimpanzee infants start showing more signs of distress than bonobo infants (significant interaction species*infant age effect: F (1,41)=5.03, P<0.05) (Fig. 4). Chimpanzee infants spend more time sexually inspecting other group members than bonobo infants [F (1,25)=9.12, P<0.01]. This latter species difference is mainly due to a gender difference [significant interaction species*infant sex: F (1,24)=5.40, P<0.05]. Chimpanzee male infants spend more time sexually inspecting other group members than male bonobo infants [F (1,23)=14.30, P<0.001] (Fig. 5); the behavior does not differ between the female infants of the species [F (1,16)=0.27, NS].
For all other behaviors—ventral contact (1) (Fig. 1), no contact (2) (Fig. 2), solitary play (4) (Fig. 5), environmental exploration (5) (Fig. 6), social play (6) (Fig. 7), non-copulatory mounts (11) (Fig. 12) and aggression (12) (Fig. 13)—no significant species effect or interaction effect with infant age or infant sex was found. Only for nipple contact did we find a significant interaction effect between “species” and “infant age” [F (1,52)=4.31, P<0.05]. From 40 months of age infant bonobos have more nipple contact than chimpanzee infants (Fig. 4).
Discussion
Our aim was to test quantitatively, two conflicting statements made in literature considering species differences between immature bonobos and chimpanzees. On one hand it is suggested that bonobos show retardation in motor and social development when compared with chimpanzees (Kuroda 1989), but on the other hand the development of behaviors is expected to be very similar (Johnson 1997).
Our preliminary results, based on data from captivity, partially confirm the first hypothesis tested. Immature chimpanzees seem to spend more time at a larger distance from their mother than immature bonobos, but time spent in ventral contact and time spent not in contact with the mother do not differ between the species. Given the similarity in development of solitary play and environmental exploration, behaviors with a high-activity level, our results give reasons to doubt that bonobos show retardation of motor development. Initiating social play also does not differ between the species. Our results also indicate that immature bonobos groom more than immature chimpanzees. Grooming creates bonds that directly affect group cohesion and serves other important social functions such as stress reduction and reconciliation, which have been described as central features in bonobo society (de Waal 1989). Our results suggest that grooming may be a more important social tool for immature bonobos than chimpanzees. Hence, these results indicate that our study does not fully support the hypothesis that bonobo infants show retardation of motor and social development.
The second hypothesis, that bonobo and chimpanzee infants are weaned at approximately the same age, seems not supported by our data. First, it is striking that nipple contact increases until about 3 years of age in both species, and even continues to increase beyond this age in bonobos. Second, the four juvenile bonobos, observed at an age older then 4 years, were still not fully weaned. In contrast, two out of four chimpanzee juveniles (observed at an age older then 3.5 years) were already in the final stage of the weaning process and their mothers gave birth in the following months. The later weaning of bonobo infants in captivity is also reflected in the length of the interbirth intervals. Bonobo females in captivity have on average longer interbirth intervals than chimpanzee females (De Lathouwers and Van Elsacker 2005). In contrast with reports from the wild, active rejection by bonobo mothers does occur in captivity. Bonobo mothers even reject their infants more than chimpanzee mothers (De Lathouwers and Van Elsacker 2004), which may correspond with the general higher expression of distress in bonobo infants. Juvenile chimpanzees however, show markedly more signs of distress from 35 months of age onward than bonobos, which corresponds with the onset of weaning.
Hence, our results seem to indicate that the weaning period itself seems more stressful for chimpanzee infants when being compared to the distress levels of younger un-weaned conspecifics and bonobos. More detailed analyses are needed to confirm whether the weaning bouts are indeed accompanied with simultaneous higher distress levels.
Finally, we tested the third prediction that bonobo infants are sexually more active and less aggressive than chimpanzee infants. We found no species difference regarding aggressive interactions initiated by the infants. This suggests that if species differences in aggression occur at the adult level, they will develop at a later stage than the developmental time frame investigated in this study. Contrary to the prediction regarding sexual activity we found that chimpanzee infants spend more time sexually inspecting other group members than bonobo infants, but this difference is more apparent when comparing only male infants. Our results indicate that differences in sexual behavior may already become apparent from an early developmental stage, in contrast with earlier studies (Hashimoto and Furuichi 1994), when gender differences are included. Further research is needed to investigate whether these early developmental differences are related to species differences in sexual competition between adult males.
The dichotomy between the two Pan species has been strongly emphasized until very recently. Given that most studies were primarily based on adult individuals, we shifted the “continuity versus discontinuity” discussion to the infant and juvenile stage. Our preliminary research reveals that the species indeed seem to resemble in their development of solitary play, environmental exploration, social play, initiated non-copulatory mounts and aggressive interactions, but it also emphasizes that species differences among infants and juveniles do occur. Bonobos and chimpanzees seem to differ in their development of spatial independence, nipple contact, grooming, expression of distress, and sexual inspections (Table 4). Development of behavior is influenced by a large number of variables. Sample size and other statistical restrictions did not allow us to correct for all, possibly influential, variables such as group size, age and experience of the mothers, presence of peers. Hence, we remain cautious with our conclusions. But we do hope that our findings may stimulate further investigation of species differences in development of behavior. It may prove to reveal striking differences and equalities that may aid us to understand species differences in association patterns, sociality, development of sexual behavior and temperament.
References
Altmann J (1974) Observational study of behavior: sampling methods. Behaviour 49:227–267
Clark CB (1977) A preliminary report on weaning among chimpanzees of the Gombe National Park, Tanzania. In: Chevalier-Skolnikoff S, Poirier FE (eds) Primate bio-social development: biological, social and ecological determinants. Garland Publishing Inc, New York, pp 235–260
De Plooij FX (1984) The behavioural development of free-living chimpanzee babies and infants. ABLEX Publishing Corporation, Norwood, NJ
De Waal FBM (1987) Tension regulation and nonreproductive functions of sex in captive bonobos (Pan paniscus). Nat Geogr Res 3(3):318–335
De Waal FBM (1989) Behavioral contrasts between bonobo and chimpanzee. In: Heltne PG, Marquardt LA (eds) Understanding chimpanzees. Harvard University Press, Cambridge, pp154–175
De Waal FBM (1998) Reply to Stanford CB (1998) The social behavior of chimpanzees and bonobos: empirical evidence and shifting assumptions. Curr Anthropol 39(4):407–408
De Lathouwers M, Van Elsacker L (2004) Comparing maternal styles in bonobos (Pan paniscus) and chimpanzees (Pan troglodytes). Am J Primatol 64:411–423
De Lathouwers M, Van Elsacker L (2005) Reproductive parameters of female Pan paniscus and P. troglodytes: quality versus quantity. Int J Primatol 26(1):55–71
Doran DM, Jungers WL, Sugiyama Y, Fleagle JG, Heesy CP (2002) Multivariate and phylogenetic approaches to understanding chimpanzee and bonobo behavioral diversity. In: Boesch C, Hohmann G, Marchant LF (eds) Behavioural diversity in chimpanzees and bonobos. Cambridge University Press, New York, pp14–34
Enomoto T (1990) Social play and sexual behavior of the bonobo (Pan paniscus) with special reference to flexibility. Primates 31(4):469–480
Fritz J, Howell SM, Hogan H, Nankivell B, Nash L (1991) Weaning in captive chimpanzees (Pan troglodytes): as assessed by an easily collected measure. Lab primates Newsl 30(4):13–16
Fruth B, Hohmann G, McGrew WC (1999) The Pan species. In: Dolhinow P, Fuentes A (eds) The nonhuman primates. Mayfield Publishing Company, London Toronto, Essay 7, pp 64–72
Fruth F, Hohmann G (2002) How bonobos handle hunts and harvests: why share food? In: Boesch C, Hohmann G, Marchant LF (eds) Behavioural diversity in chimpanzees and bonobos. Cambridge University Press, Cambridge, pp 231–243
Furuichi T (1997) Agonistic interactions and matrifocal dominance rank of wild bonobos (Pan paniscus) at Wamba. Int J Primatol 18(6):855–875
Hasegawa T, Hiraiwa-Hasegawa M (1983) Opportunistic and restrictive matings among wild chimpanzees in the Mahale Mountains, Tanzania. J Ethol 1:75–85
Hashimoto C, Furuichi T (1994) Social role and development of noncopulatory sexual behavior of wild bonobos. In: Wrangham RW, McGrew WC, de Waal FBM (eds) Chimpanzee cultures. Harvard University Press, Cambridge, MA, pp155–168
Hohmann G (2001) Association and social interactions between strangers and residents in bonobos (Pan paniscus). Primates 42(1):91–99
Hohmann G, Fruth B (2002) Dynamics in social organization of bonobos (Pan paniscus). In: Boesch C, Hohmann G, Marchant LF (eds) Behavioural diversity in chimpanzees and bonobos. Cambridge University Press, New York, pp138–150
Idani G (1991) Social relationships between immigrant and resident bonobo (Pan paniscus) females at Wamba. Folia Primatol 57:83–95
Johnson MJ (1997) Juvenile Bonobo’s (Age 2–5 years). In: Mills J, Reinartz G, De Bois H, Van Elsacker L, Van Puijenbroeck B (eds) The care and management of bonobos (Pan paniscus) in captive environments: a husbandry manual jointly developed for the bonobo species survival Plan and European endangered species program. Zool Soc Milw Cty Milw 2(9):1–9
Kano T (1989) The sexual behavior of pygmy chimpanzees. In: Heltne PG, Marquardt LA (eds) Understanding chimpanzees. Harvard University Press, Cambridge, MA, pp176–183
Kano T (1992) The last Ape: Pygmy chimpanzee behavior and ecology. Stanford University Press, Stanford
Kano T (1996) Male rank order and copulation rate in a unit-group of bonobos at Wamba, Zaïre. In: McGrew WC, Marchant LF, Nishida T (eds) Great Ape societies. Cambridge University Press, Cambridge, pp 92–103
Kano T (1998) Reply to Stanford CB (1998) The social behavior of chimpanzees and bonobos: empirical evidence and shifting assumptions. Curr Anthropol 39(4):410–411
Kitamura K (1989) Genito-genital contacts in the pygmy chimpanzee (Pan paniscus). Afr Study Monogr 10(2):49–67
Kuroda S (1980) Social behavior of the pygmy chimpanzees. Primates 21:181–197
Kuroda S (1989) Developmental retardation and behavioral characterisitcs of Pygmy chimpanzees. In: Heltne PG, Marquardt AE (eds) Understanding chimpanzees. Chicago Academy of Sciences, Chicago, pp184–193
Kuroda S (2000) Developmental retardation in early infancy of Pygmy Chimpanzees in Wamba. Behavioural diversity in chimpanzees and bonobos. Abstract Book 11–17 June, Seeon, pp 16
Muniz RS (1984) Nipple contact in captive black-faced chimpanzees (Pan t. troglodytes Blumenbach, 1779). Zoo Biol 3(3):267–271
Palagi E, Paoli T, Tarli SB (2004) Reconciliation and consolation in captive bonobos (Pan paniscus). Am J Primatol 62(1):15–30
Parish AR (1996) Female relationships in bonobos (Pan paniscus): evidence for bonding, cooperation, and female dominance in a male-philopatric species. Hum Nat 7:61–96
Sokal RR, Rohlf FJ (1981) Biometry. 2nd edn. Freeman, NY
Stanford CB (1998) The social behavior of chimpanzees and bonobos: empirical evidence and shifting assumptions. Curr Anthropol 39(4):399–420
Stevens J, Vervaecke H, Van Elsacker L (2001) Sexual strategies in Pan paniscus: implications of female dominance. Primate Rep Spec issue 60(1):42–43
Stevens J, Vervaecke H, Van Elsacker L (2003) Mating harassment in captive bonobos (Pan paniscus). Primates Eye 80:22–23
Van Lawick-Goodall J (1967) Mother-offspring relationships in free-ranging chimpanzees. In: Morris D (ed) Primate ethology. Weidenfeld and Nicolson, London, pp 287–346
van Lawick-Goodall J (1968) The behaviour of free-living chimpanzees in the Gombe Stream Reserve. An Behav Mono 1:165–311
Verbeke G, Molenberghs G (1997) Linear mixed models in practice: a SAS-oriented approach. Spinger-Verlag, New York
Vervaecke H, De Vries H, Van Elsacker L (2000) Dominance and its behavioral measures in a captive group of bonobos (Pan paniscus). Int J Primatol 21:47–68
Vervaecke H, Stevens J, Van Elsacker L (2003a) Bonobos on the bandwagon. Abstract Book International Anthropological Congress Anthropology and society, May 22–24, Prague
Vervaecke H, Stevens J, Van Elsacker L (2003b) Interfering with others: female-female reproductive competition in Pan paniscus. In: Jones CB (eds) Sexual selection and reproductive competition in primates: new perspectives and directions. American Society of Primatologists, pp 231–253
Weaver ACF (1997) Bonobo infant development in the first year compared to the other Apes. In: Mills J, Reinartz G, De Bois H, Van Elsacker L, Van Puijenbroeck B (eds) The care and management of bonobos (Pan paniscus) in captive environments: a husbandry manual jointly developed for the Bonobo species survival plan and European endangered species program. Zool Soc Milw Cty Milw 2(8):1–15
Acknowledgements
We kindly thank the curators, staff members, assistants, and keepers of the following institutions for their kind co-operation: Animal Park Amersfoort, Burgers’ Zoo, Edinburgh Zoo, Frankfurt Zoo, Primate Park Apenheul, Wild Animal Park Planckendael, Wuppertal Zoo. The assistance of the members of the Center for Research and Conservation is much appreciated. We thank the Flemish Government for the structural support to the Center for Research and Conservation (CRC) of the Royal Zoological Society of Antwerp (RZSA). The first author received a BOF-Dehousse grant (October 2000–September 2001) from the University of Antwerp and a Dehousse grant (February 2002–January 2004) from the Royal Zoological Society of Antwerp in association with the University of Antwerp, Belgium.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
De Lathouwers, M., Van Elsacker, L. Comparing infant and juvenile behavior in bonobos (Pan paniscus) and chimpanzees (Pan troglodytes): a preliminary study. Primates 47, 287–293 (2006). https://doi.org/10.1007/s10329-006-0179-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10329-006-0179-7