Abstract
Lodging is one possible risk factor that leads to increased cereal mycotoxin contamination, but few reports have been published on the subject. We examined the effects of lodging on the level of deoxynivalenol (DON) and nivalenol (NIV) contamination in wheat, barley, and rice infected with the Fusarium graminearum species complex. Case-control and intervention studies were applied to test the hypothesis that lodging increases the level of mycotoxin contamination. A total of 66 grain samples were collected from each field in 12 Japanese prefectures from 2002 to 2006. Each sample set consisted of grains from lodged and nonlodged plants. The concentration of DON + NIV in lodged plants was significantly higher than in nonlodged plants. All samples of wheat and barley were contaminated with DON and NIV; however, most of the lodged rice samples were contaminated only with NIV. In intervention trials to investigate the effects of lodging duration, a small area of wheat inoculated with the pathogen was completely lodged by trampling. Even with 5 days of lodging, the levels of DON + NIV in wheat grain at harvest increased by 27–51% compared to nonlodged control plots. For rice, half of each plot area was completely lodged by trampling 20 days before harvest. The level of NIV in lodged rice grain was significantly higher than that in nonlodged rice at optimum and delayed harvests, because lodging significantly increased the level of Fusarium mycotoxins in the three crops. Thus, practices (e.g., rational use of fertilizers) to avoid lodging should reduce the risk of mycotoxin contamination. This is the first epidemiological study on the effect of lodging on mycotoxin production by the F. graminearum species complex in wheat, barley, and rice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fusarium head blight (FHB) is a destructive disease of small grain cereals in many warm, humid regions worldwide. The Fusarium graminearum species complex (Ward et al. 2002), once considered a single species [F. graminearum Schwabe; teleomorph: Gibberella zeae (Schwein.) Petch] is the main cause of FHB in Japan (Koizumi et al. 1991). Molecular phylogenetic analyses revealed that F. graminearum is a species complex consisting of at least nine biogeographically structured lineages (O’Donnell et al. 2000, 2004; Ward et al. 2002). Two of these, F. graminearum Schwabe sensu stricto (s. str.) and F. asiaticum O’Donnell et al., have been isolated from Japan (O’Donnell et al. 2000; Suga et al. 2008).
This disease infects spikes and reduces grain yield and quality. Moreover, the fungi produce mycotoxins such as deoxynivalenol (DON), nivalenol (NIV), and zearalenone (ZEA). These mycotoxins can be so harmful to animals and humans that protective guidelines and legislation on allowable levels are in place or under consideration in most countries. In Japan, the Food Sanitation Council of the Japanese Ministry of Health, Labor and Welfare set provisional guidelines for DON content in unpolished wheat grains as 1.1 ppm (µg/g) in 2002, while the Ministry of Agriculture, Forestry and Fisheries stipulated that the percentage of FHB-damaged grains of wheat and barley for food must be less than 0.05%. The establishment of these two standards for DON and FHB has caused confusion in wheat-producing areas in Japan.
Because Fusarium mycotoxins are produced on the crops in the field, it is crucial to understand how agricultural practices affect mycotoxin contamination in grain. Such evidence could be used to establish guidelines for good agricultural practices (GAP) to minimize the risks of mycotoxin contamination of cereal products. In Japan, lodging is common after strong winds accompanied by heavy rains near harvest time. Recently, overluxuriant growth during warm winters due to global warming has also led to increased lodging.
Lodging of cereals greatly reduces grain yield and quality (Easson et al. 1993; Fischer and Stapper 1987) because of the permanent displacement of plant shoots from their upright position (Pinthus 1973). Lodging normally results from the interactions between the crop and the wind, rain, and soil, with a subsequent increase in the moisture content of the grain. Therefore, lodging seems to be a significant risk factor in relation to mycotoxin production. However, few reports (Langseth and Stabbetorp 1996; Nicholson et al. 2003) have addressed the interaction between lodging and mycotoxin levels, and previous studies on these issues focused only on DON. In Japan, however, the NIV prpducing isolates (chemotype) of the F. graminearum species complex are widely distributed in western parts of Japan and more frequently isolated than the DON chemotype (Ichinoe et al. 1984; Nakajima and Yoshida 2007; Yoshizawa and Jin 1998). Their virulence may also be very high (Nakajima and Yoshida 2007). Additionally, NIV is more toxic to animals than is DON (Minervini et al. 2004; Ryu et al. 1988; Takahashi et al. 2007; Ueno et al. 1997).
The objectives of this study were to elucidate the effects of lodging on the level of DON and NIV contamination in wheat, barley, and rice infected with the F. graminearum species complex.
Materials and methods
Sample collection from naturally infected fields (Experiment 1)
Sixty-six sets of grain samples of wheat (31), barley (15), and rice (20) were collected from different fields in 12 Japanese prefectures from 2002 to 2006. Each sample set consisted of grains from lodged and nonlodged plots. One grain sample was harvested from the same field in which some plants had lodged naturally. The crop from an area of 3.3 m2 in one plot was harvested by hand at the optimal harvest time and dried in a greenhouse to a moisture content below 12% (w/w); the spikes were then threshed carefully. The grains were carefully mixed, and 500 g were packed in a paper bag and dried at 70°C for 1 day to stop fungal development.
Artificial lodging in an artificially infested field (Experiment 2)
This experiment was conducted in Koshi (32.5°N, 130.4°E), Kumamoto, Japan, during 2002–2004 for wheat and in 2006 for rice. The wheat cultivar Chikugoizumi and the rice cultivar Nishiaoba were used to examine the effect of artificial lodging on mycotoxin accumulation. Chikugoizumi is a leading soft wheat cultivar in Japan with medium resistance to FHB, and Nishiaoba is a newly developed forage rice cultivar with unknown resistance to FHB resistance. The wheat cultivars were sown in a field of volcanic ash soil on 25 November 2002 and 26 November 2003, in single rows 0.8 m apart and 50 m long at a seeding rate of 5 g m−2. Basal fertilizer was applied before sowing at 5 g N m−2, 5 g P2O5 m−2, and 5 g K2O m−2, and the wheat plants were again fertilized at the stem elongation stage [Zadoks growth stage (ZGS) 30–31; Zadoks et al. 1974] with ammonium sulfate at 2 g N m−2. The rice cultivar was seeded on 12 June 2006, and the four or five seedlings per hill were transplanted on 3 July 2006 with equidistant spacing of 30 × 18 cm in a 50 × 10 m paddy field. The fertilizers (N, P2O5, and K2O, each at 14 g m−2) were then applied. Basal fertilizer was applied to rice plants before transplanting at a rate of 71% and top-dressed at 29% at the early-boot stage (ZGS 40). The wheat and rice cultivars were grown in the field in accordance with standard agronomic practices until heading.
A mixture of two isolates of the F. graminearum species complex, differing in trichothecene chemotype, i.e., H-3 (F. graminearum s. str.; MAFF101551) of the DON type and NIV-2 (F. asiaticum; Nakajima and Yoshida 2007; Suga et al. 2008) of the NIV type, was used as the inoculum. In the experiment in 2002–2003, we used only isolate H-3 (DON chemotype). The inoculum was prepared according to methods described in our previous study (Yoshida et al. 2007) and stored at −20°C. On the days of inoculation, macroconidia suspensions of the two isolates were thawed and diluted to 5 × 105 spores per ml and then mixed in equal volumes. New Gramin (Sankyou-Agro Co, Tokyo, Japan) was added to a final concentration of 0.3 ml l−1 as a spreader. The heads of each wheat or rice cultivar in a 10 × 50 m field were sprayed with 50 l of spore suspension at anthesis (ZGS 64, 50% of the spikelets flowered).
Two weeks after inoculation, 50 spikes were arbitrarily selected from each plot and assessed for FHB severity on a scale of 0–100 (0, 2, 5, 10, 20, 30, 40, 50, 60, 70, 80, 90, 100%), according to a visual rating of the percentage of infected area in each spike. Using these data, FHB incidence (%), that is, the percentage of visually diseased spikes, was calculated for each plot. The frequency of perithecia formation was also investigated at harvest using 100 diseased spikes, and fungi were isolated from a single perithecium using selective medium (Togawa 1994) for F. graminearum. The surface of the perithecium was sterilized in 70% ethanol and sodium hypochlorite solution (1% available chlorine), and incubated at 25°C for 7 days under fluorescent light.
We selected three uniformly diseased plots (4 rows × 20 m) from each field (10 × 50 m). For wheat, one-quarter of the area of the each plot was completely lodged by trampling 15, 10, or 5 days before harvest. All plots were harvested at optimum maturity stage (ZGS 91; caryopsis hard, no longer dented by thumbnail) on 2 June 2003 and 4 June 2004. The experiment had a one-way randomized complete block design with four durations (0, 5, 10, 15 days) of lodging and three replications. For rice, half of each plot area was completely lodged by trampling 20 days before harvest. Each plot was harvested at the optimal maturity stage (ZGS 92; over 90% of the spikelets were ripened) on 15 October 2006 and a 10 day delayed harvest. Therefore, the experiment had a two-way split-plot randomized complete block design having three replications, with lodging as the main effect factor, and harvest timing as the second factor.
We sampled all spikes in a 2 m length of row from the center of each plot. The harvested spikes were dried immediately at 70°C for 1 day to stop fungal development. After drying, the spikes were threshed carefully so that no shriveled or small grains were lost. The grains were mixed carefully, and 1 kg was packed in a paper bag.
Toxin analysis
After all other evaluations were done, the grains from each plot were analyzed for DON and NIV content at the KM Assay Center, Kyowa Medex Co. (Nagaizumi, Shizuoka, Japan) for toxin analyses with the ELISA system, as previously described (Yoshizawa et al. 2004). In the case of rice, ZEA was also analyzed in an ELISA system at Kyowa Medex Co. The quantification limits for DON, NIV and ZEA were 0.1, 0.05, 0.08 µg/g, respectively.
Statistical analyses
To evaluate the effects of natural lodging on mycotoxin accumulation for samples of wheat, barley, and rice collected from naturally infested fields, we ran nonparametric Wilcoxon matched-pair signed-rank tests for each crop, and the statistical probability (P-value) was determined. Data analyses for artificial lodging of rice were performed based on a split-plot randomized complete block design. Wilcoxon matched-pair signed-rank tests were used to compare the level of mycotoxins in a lodged plots with those in nonlodged plots. All statistical analyses were performed with JMP statistical discovery software (SAS Institute, Cary, NC, USA). If the level of the mycotoxins in the samples was less than the quantification limit, the quantification limit was used for the calculations.
Results
Effect of natural lodging (Experiment 1)
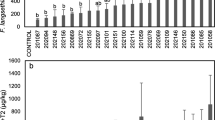
In total, 22 of 31 wheat sample sets, 10 of 15 barley sample sets, and 6 of 20 rice sample sets were positive for DON and/or NIV. The effect of natural lodging in the three crops on the level of DON and NIV is shown in Fig. 1. The concentration of DON + NIV in lodged crops was significantly higher than that in nonlodged crops, and the P-values of Wilcoxon matched-pair signed-rank tests for wheat, barley, and rice were <0.001, 0.004, and 0.031, respectively. With increasing levels of DON + NIV in the nonlodged plot, the increasing effect of lodging was considerable. This tendency was observed in every year and sample set from every district in Japan. The ratio between DON and NIV was not affected by lodging: they increased in parallel. All rice samples in the nonlodged plot were negative for DON and NIV; only six lodged samples were above the detection limit. The mean concentration of DON + NIV in contaminated rice was 0.27 mg kg−1, much lower than that of the other crops. All samples of wheat and barley were co-contaminated with DON and NIV; however, five of six lodged rice samples were contaminated only with NIV. We could not collect information as to the cause (i.e., wind, rain, cultivar, fertilization, seeding ratio, pesticide application, or diseases), duration, and degree of lodging from each field (Fig. 2).
Comparison of the sum of deoxynivalenol (DON) and nivalenol (NIV) contamination between lodged and nonlodged grain samples of wheat, barley, and rice collected from different fields in 12 Japanese prefectures from 2002 to 2006. Significant differences in the concentration of DON + NIV between lodged and nonlodged grain samples were detected using Wilcoxon matched-pair signed-rank tests. The probability values of wheat, barley, and rice were <0.001, 0.004, and 0.031, respectively
Effect of lodging duration on the concentration of deoxynivalenol (DON) and nivalenol (NIV) at optimal harvest timing in a wheat field after artificial inoculation with the Fusarium graminearum species complex in a 2003 and b 2004. Each point is the mean of three replications; bars indicate standard errors
Effect of artificial lodging of wheat and rice (Experiment 2)
In the intervention trials on the effect of duration of lodging in a small area of wheat inoculated with an isolate of F. graminearum, DON and NIV concentrations increased with increasing duration of lodging in both years. The average levels of disease severity caused by inoculation at anthesis were 39.8 with 100% incidence in 2003 and 23.6 with 80% incidence in 2004. The total precipitation during the 15-day lodging in 2003 and 2004 was 200 and 108 mm, respectively. In the lodged plot, abundant perithecia formed on the spikes of wheat (Fig. 3). Using selective medium, we isolated the typical scarlet colony of the F. graminearum species complex from the single perithecium. At the optimal harvesting stage, the perithecia formed only in the 10 to 15-day lodged plots in 2003 and the 15-day lodged plot in 2004 (Fig. 4); however, at the 10-day delayed harvest, perithecia formed even in the nonlodged plot. Preharvest sprouting was observed in the 10 to 15-day lodged plots at the optimal harvesting stage in both years.
Artificial inoculation of rice with the F. graminearum species complex in the field was successful: the incidence and severity of FHB in the plots were uniform (Table 1). DON was not detected in three of four plots even though DON and NIV producers were included in the inoculum. The level of contamination with NIV in lodged rice grain was significantly higher than that in nonlodged grain for both harvests, but the concentration was lower than that in the experiment with wheat. In the case of ZEA, an increasing effect of lodging was seen in the delayed harvest plots, but a significant difference was not detected in the optimal harvest plots. Neither the increase in mycotoxin with delayed harvest nor the interaction effect of lodging and harvest timing was significant.
Discussion
To our knowledge, this is the first epidemiological study on the effect of lodging on mycotoxin accumulation from FHB in wheat, barley, and rice. We investigated the accumulation of both DON and NIV because they often co-occur (Placinta et al. 1999; Tanaka et al. 1988; Yoshizawa and Jin 1998). Two types of study design, case-control and intervention, were used to provide evidence that lodging increases the level of mycotoxin contamination in wheat, barley, and rice. In Experiment 1, using natural lodging, the level of DON + NIV in the lodged crop was significantly higher than that in the nonlodged crop. However, the amount of increase differed among sample sets for each crop. The increasing effect of lodging appeared to be considerable when the level of DON + NIV in the nonlodged crop was high. These results suggest that the increasing level of mycotoxins in lodged crops depends on the inoculum potential of mycotoxin-producing fungi at the time when the lodging occurred. Langseth and Stabbetorp (1996) reported an increasing effect of lodging on DON levels in barley and oats; however, they only provided averaged data among plots. Therefore, it was not possible to compare increasing patterns of mycotoxins without analyses of the one-to-one relationship, as shown in Fig. 1. Nicholson et al. (2003) provided only a fragmentary observation that when lodging occurred, DON production was very high irrespective of any fungicide treatment.
Co-contamination with DON and NIV was common in the collected wheat and barley; however, five of six lodged rice samples were only contaminated with NIV. The level of the mycotoxins in rice was lower than that in the other two crops. Tanaka et al. (2004b) reported that of 124 Japanese rice samples analyzed for DON and NIV, only 4 (3.2%) and 15 (12.1%) were positive, respectively. Mean concentrations in all rice samples tested were 0.7 (DON) and 0.6 (NIV) µg/kg, and the exposure levels estimated from these concentration were very low (Tanaka et al. 2004b). The natural occurrence of DON (0.12–2.90 µg/g), fusarenon-X (1.9 μg/g), and NIV (0.2–2.2 μg/g) on lodged and water-damaged domestic rice in Japan was recorded after a typhoon in 1998 (Tanaka et al. 2004a). The NIV chemotype produces NIV and fusarenon-X (Lee et al. 2002). These results indicated that NIV producers rather than DON-producers of F. graminearum may be associated with rice and that the risk of DON and NIV in rice is relatively low.
Our investigation in Experiment 1 was only a case-control study. This type of study requires less effort for collecting data and reflects actual contamination in a field, but confounding factors cannot be controlled in a field with natural lodging. The cause (i.e., wind, rain, cultivar, fertilization, seeding ratio, pesticide application, or diseases such as eyespot of wheat), duration and degree of lodging, incidence or severity of FHB, and mycotoxin productivity of the pathogen in the collected sample sets were unknown in Experiment 1. These factors might affect the conditions during the lodging period, and the subsequent mycotoxin concentration would differ depending on the cause of lodging.
Hence, Experiment 2 was conducted as an intervention trial to investigate the effect of the duration of lodging in a small area of wheat inoculated with the pathogen. The degree of lodging, incidence and severity of FHB, and mycotoxin productivity of the pathogen were nearly uniform through the use of artificial lodging by trampling and inoculation with isolates of the F. graminearum species complex to control confounding factors. Even after 5 days in lodging plots in which preharvest sprouting was not observed, the level of DON + NIV in wheat grain at harvest increased by 51 and 27% compared to nonlodged control plots in 2003 and 2004, respectively. In both years, we had adequate precipitation during the experiments. Although precipitation during lodging might increase the level of DON and NIV, short-term lodging even for 5 days would be a risk factor for mycotoxin contamination because strong wind accompanied by heavy rain are the main causes of lodging.
We observed abundant perithecia of the F. graminearum species complex on spikes of lodged wheat at harvest (Figs. 3, 4). The moisture conditions in the lodged wheat spikes seemed to be ideal for perithecia formation. Perithecia are considered to be important for long-term survival of F. graminearum in fields (Atanasoff 1920). These results indicate that lodging would increase the inoculum potential in the following year. New infections of the wheat heads by mycotoxin-producing fungi from the soil might occur because some wheat spikes had been in contact with the soil surface during lodging. Not only Fusarium mycotoxins (DON, NIV, and ZEA), but also Aspergillus or Penicillium mycotoxins (e.g., aflatoxin, sterigmatocystin, citrinin, ochratoxins) might occur and increase after lodging. Fusarium species were isolated at significantly higher frequencies in lodged cereals in Denmark, but species of Aspergillus and Penicillium were not correlated with the degree of lodging (Welling 1975). Further work is required to investigate the new infection by mycotoxin-producing fungi from the soil during lodging periods.
In the case of the artificial inoculation trial with rice, the level of DON was mostly below the detection limit, even though a mixture of DON and NIV producers was inoculated (Table 1). The level of NIV in lodged rice grain was significantly higher than that in nonlodged grain at optimal harvest timing and delayed harvesting (Table 1). Although the concentration of mycotoxins was lower than that in Experiment 2 for wheat, the increasing effect of lodging on mycotoxin levels in rice was confirmed. Delayed harvesting resulted in higher DON levels in wheat (Farrer et al. 2006) and maize (Lauren et al. 1996), but mycotoxin (DON, NIV, ZEA) levels were not affected by delayed harvesting in pearl millet (Wilson et al. 1995). The effect of delayed harvesting in rice was not clear, but the detection of ZEA was noteworthy. Few surveys have examined contamination by ZEA in rice (Park et al. 2005). Because the F. graminearum species complex isolates obtained from the western part of Japan had high ZEA productivity (Nakajima and Yoshida 2007), ZEA accumulation should be studied further.
In conclusion, our experiments provide strong evidence that lodging significantly increased Fusarium mycotoxin levels in grains of wheat, barley, and rice. Therefore, appropriate management measures (e.g., lodging-resistant cultivars, seed sowing rates, rational use of fertilizers, trampling before wintering, soil top dressing and application of plant growth regulators) should be used to reduce lodging and thus the risk of mycotoxin contamination. In addition, the lodged part of a field should be harvested separately, especially if symptoms of FHB are visible.
References
Atanasoff D (1920) Fusarium-blight (scab) of wheat and other cereals. J Agric Res 20:1–32
Easson DL, White EM, Pickles SJ (1993) The effects of weather, seed rate and cultivar on lodging and yield in winter wheat. J Agric Sci 121:145–156
Farrer D, Weisz R, Heiniger R, Murphy JP, Pate MH (2006) Delayed harvest effect on soft red winter wheat in the southeastern USA. Agron J 98:588–595
Fischer RA, Stapper M (1987) Lodging effects on high-yielding crops of irrigated semi-dwarf wheat. Field Crops Res 17:245–248
Ichinoe M, Hagiwara H, Kurata H (1984) Distribution of trichothecene-producing fungi in barley and wheat fields in Japan. In: Kurata H, Ueno Y (eds) Toxigenic fungi: their toxins and health hazard. Elsevier, Tokyo, pp 190–198
Koizumi S, Kato H, Yoshino R, Hayashi N, Ichinoe M (1991) Distribution of causal Fusaria of wheat and barley scab in Japan. Ann Phytopath Soc Jpn 57:165–173
Langseth W, Stabbetorp H (1996) The effect of lodging and time of harvest on deoxynivalenol contamination in barley and oats. J Phytopathol 144:241–245
Lauren DR, Jensen DJ, Smith WA, Dow BW, Sayer ST (1996) Mycotoxins in New Zealand maize: a study of some factors influencing contamination levels in grain. NZ J Crop Horticult Sci 24:13–20
Lee T, Han Y-K, Kim K-H, Yun S-H, Lee Y-W (2002) Tri13 and Tri7 determine deoxynivalenol- and nivalenol-producing chemotypes of Gibberella zeae. Appl Environ Microbiol 68:2148–2154
Minervini F, Fornelli F, Flynn KM (2004) Toxicity and apoptosis induced by the mycotoxins nivalenol, deoxynivalenol and fumonisin B1 in a human erythroleukemia cell line. Toxicol In Vitro 18:21–28
Nakajima T, Yoshida M (2007) Mycotoxin productivity and virulence of Fusarium graminearum species complex causing Fusarium head blight on wheat and barley in the western part of Japan. Jpn J Phytopathol 73:106–111 (in Japanese with English abstract)
Nicholson P, Turner JA, Jenkinson P, Jennings P, Stonehouse J, Nuttall M, Dring D, Weston G, Thomsett M (2003) Maximising control with fungicides of Fusarium ear blight (FEB) in order to reduce toxin contamination of wheat. In Project Report No. 297, HGCA, London, pp 1–85
O’Donnell K, Kistler HC, Tacke BK, Casper HH (2000) Gene genealogies reveal global phylogeographic structure and reproductive isolation among lineages of Fusarium graminearum, the fungus causing wheat scab. Proc Natl Acad Sci USA 97:7905–7910
O’Donnell K, Ward TJ, Geiser DM, Kistler HC, Aoki T (2004) Genealogical concordance between the mating type locus and seven other nuclear genes supports formal recognition of nine phylogenetically distinct species within the Fusarium graminearum clade. Fungal Genet Biol 41:600–623
Park JW, Choi SY, Hwang HJ, Kim YB (2005) Fungal mycoflora and mycotoxins in Korean polished rice destined for humans. Int J Food Microbiol 103:305–314
Pinthus MJ (1973) Lodging in wheat, barley and oats: The phenomenon, its causes and preventative measures. Adv Agron 25:209–263
Placinta CM, D’Mello JBF, MacDonald AMC (1999) A review of world contamination of cereal grains and animal feeds with Fusarium mycotoxins. Anim Feed Sci Technol 78:21–37
Ryu JC, Ohtsubo K, Izumiyama N, Nakamura K, Tanaka T, Yamamura H, Ueno Y (1988) The acute and chronic toxicities of nivalenol in mice. Fund Appl Toxicol 11:38–47
Suga H, Karugia GW, Ward T, Gale LR, Tomimura K, Nakajima T, Miyasaka A, Koizumi S, Kageyama K, Hyakumachi M (2008) Molecular characterization of the Fusarium graminearum species complex in Japan. Phytopathology 98:159–166
Takahashi M, Shibutani M, Sugita-Konishi Y, Aihara M, Inoue K, Woo GH, Fujimoto H, Hirose M (2007) A 90-day subchronic toxicity study of nivalenol, a trichothecene mycotoxin, in F344 rats. Food Chem Toxicol 46:125–135
Tanaka K, Kobayashi H, Nagata T, Manabe M (2004a) Natural occurrence of trichothecenes on lodged and water-damaged domestic rice in Japan. J Food Hyg Soc Jpn 45:63–66
Tanaka T, Hasegawa A, Yamamoto S, Lee US, Sugiura Y, Ueno Y (1988) Worldwide contamination of cereals by the Fusarium mycotoxins nivalenol, deoxynivalenol, and zearalenone. 1. Survey of 19 countries. J Agric Food Chem 36:979–983
Tanaka T, Yoshizawa T, Tanaka T, Sugiura Y, Takatori K, Kumagai S (2004b) Contamination of staple cereals with deoxynivalenol and nivalenol in Japan. In: Yoshizawa T (ed) New horizon of mycotoxicology for assuring food safety. Bikohsha, Takamatsu, pp 83–88
Togawa M (1994) Selective medium for isolation of Fusarium graminearum. Soil Microorg 44:77–88 (in Japanese with English abstract)
Ueno Y, Aikawa Y, Okumura H, Sugiura Y, Nakamura K, Masuma R, Tanaka T, Young CJ, Savard ME (1997) Trichothecenes produced by Fusarium species Fn 2B. Mycotoxins 45:25–31
Ward TJ, Bielawski JP, Kistler HC, Sullivan E, O’Donnell K (2002) Ancestral polymorphism and adaptive evolution in the trichothecene mycotoxin gene cluster of phytopathogenic Fusarium. Proc Natl Acad Sci USA 99:9278–9283
Welling B (1975) Lodging and cereal quality. Investigation on germination capacity and fungus flora at harvest and after storage of lodged cereal crop. Tidsskrift Planteavl 79:243–253
Wilson JP, Casper HH, Wilson DM (1995) Effect of delayed harvest on contamination of pearl millet grain with mycotoxin-producing fungi and mycotoxins. Mycopathologia 132:27–30
Yoshida M, Kawada N, Nakajima T (2007) Effect of infection timing on Fusarium head blight and mycotoxin accumulation in open- and closed-flowering barley. Phytopathology 97:1054–1062
Yoshizawa T, Jin YZ (1998) Trichothecene occurrence in Japanese wheat and barley—its characteristics. Mycotoxins 47:15–18
Yoshizawa T, Kohno H, Ikeda K, Shinoda T, Yokohama H, Morita K, Kusada O, Kobayashi Y (2004) A practical method for measuring deoxynivalenol, nivalenol, and T-2 + HT-2 toxin in foods by an enzyme-linked immunosorbent assay using monoclonal antibodies. Biosci Biotechnol Biochem 68:2076–2085
Zadoks JC, Chang TT, Konzak CF (1974) A decimal code for the growth stages of cereals. Weed Res 14:415–421
Acknowledgments
The authors thank everyone who helped collect samples and the staff of the research team for FHB control at KONARC for technical assistance. This study was conducted as part of the Integrated Research Program for Functionality and Safety of Food toward an Establishment of Healthy Diet. Funding was provided by the Ministry of Agriculture, Forestry and Fisheries of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakajima, T., Yoshida, M. & Tomimura, K. Effect of lodging on the level of mycotoxins in wheat, barley, and rice infected with the Fusarium graminearum species complex. J Gen Plant Pathol 74, 289–295 (2008). https://doi.org/10.1007/s10327-008-0103-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10327-008-0103-7