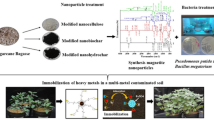
Abstract
This report shows that soil heavy metals can be totally immobilized by grinding with nano-Fe/Ca/CaO. Remediation of soils contaminated by heavy metals is a critical issue in Japan. Indeed, contaminated soils are notoriously difficult to remediate using available technologies. Major setbacks in typical immobilization techniques for heavy metals are wet conditions, forming secondary effluents and further treatment for effluents. Solidification with nano-Fe/Ca/CaO dispersion mixture is a promising treatment for the total immobilization of soil heavy metals As, Cd, Cr, Pb, and separation in dry conditions. Here, we studied the heavy metal immobilization by simple grinding with the addition of three mixtures: nano-Fe/CaO, nano-Fe/Ca/CaO, and nano-Fe/Ca/CaO/PO4. Samples were analyzed by inductively coupled plasma optical emission spectrometry (ICP-OES) and scanning electron microscopy combined with electron dispersive spectroscopy (SEM–EDS). Results show that the addition of nano-Fe/Ca/CaO immobilized 95–99 % of heavy metals, versus 65–80 % by simple grinding. After treatment, 36–45 wt% of magnetic and 64–55 wt% of nonmagnetic fractions of soil were separated. Their condensed heavy metal concentration was 85–95 % and 10–20 %, respectively. Nano-Fe/Ca/CaO treatment reduced the concentration of leachates heavy metals to values lower than the Japan soil elution standard regulatory threshold of 0.01 mg/l for As, Cd, and Pb; and 0.05 mg/l for Cr. This technology can therefore immobilize totally soil heavy metals and reduce heavy metal by separation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The remediation of contaminated soils has shown a rapid upward trend in Japan, especially after the Soil Contamination Countermeasures Law, which was proclaimed in 2002 and enforced from 2003 (Ministry of Environment Government of Japan 2002). Soils at heavy metal-contaminated sites, which pose a severe hazard to public health and the environment, are notoriously difficult to remediate. Consequently, remediation is an extremely difficult task to be accomplished using suitable technologies.
Typical remediation techniques for heavy metals in soil are extraction and immobilization (Sheppard and Thibault 1992; Glass 1999; Abramovitch et al. 2003). Soil washing by the extraction of contaminants with water is one soil remediation technology based on mineral processing technologies such as grinding and sieving (Marino et al. 1997; Orumwense 2006). Various extractants were also explored for ex situ remediation to remove heavy metals from the soil matrix by transfer to another phase. Extractive techniques might involve inorganic acids (Tessier et al. 1979; Lair et al. 2007) or organic acids and surfactants (Macauley and Hong 1995). Ex situ extractive technologies are rarely adopted because of their attendant high risks and costs related to the use of hazardous extractants, their consequent need for treating secondary wastewater, and the worsening shortage of landfill sites. However, in situ extractive technologies mainly constitute phytoextraction and electrokinetic extraction (Garbisu and Alkorta 2001; Reddy et al. 2001; Vithanage et al. 2011), but phytoextraction might require an extremely long time, and the efficiency of electrokinetic extraction might be affected strongly by the soil type and contaminant species to be removed.
Considering the limitations of the extractive techniques described above, immobilization processes are generally preferred for treating soils contaminated with heavy metals (Paff and Bosilovich 1995). Immobilization is typically performed by mixing the contaminated soil with suitable binders, which can reduce heavy metal leachability through pH to minimize their solubility, or by increasing the adsorption, ionic exchange, and precipitation of pollutants (USEPA 1982; Stegmann et al. 2001). Various binders designed to immobilize heavy metals in soils have been investigated, but they mainly comprise hydroxyapatite (Boisson et al. 1999), zeolites, calcium hydroxide (Castaldi et al. 2005), and phosphates (Patricia and Alicia 2008; Cao et al. 2003; Raicevica et al. 2005). Cement-based stabilization/solidification treatment processes for immobilizing hazardous substances that contain heavy metals are well known (USEPA 1993; Lin et al. 1998; Li et al. 2001; Polettini et al. 2001). Unfortunately, these immobilization treatment processes are also wet conditions, forming secondary effluents and necessitating their treatment, entailing additional costs, and the use of cement. Therefore, treatment under dry and water-free conditions should be considered.
Our recent investigations showed that the nanometallic Ca/CaO mixture was the most effective for hydrodechlorination of about 98 % of polychlorinated dibenzodioxins (PCDDs), polychlorinated dibenzofurans (PCDFs), and polychlorinated biphenyls (PCBs) in contaminated fly ash, and also, for cesium immobilization of about 96 % in soil by ball-milling treatment (Mitoma et al. 2011; Srinivasa Reddy et al. 2012). The high PCDD, PCDF, and PCB hydrodechlorination and cesium immobilization with the addition of nanometallic Ca/CaO might result from the high reduction potentials and high surface area produced by ball milling. In the case of cesium immobilization, Ca/CaO can also produce immobile salts with moisture and CO2 in atmosphere, including pozzolanic cement and hydraulic properties, which bring cesium into the immobile Ca/PO4 salts. Therefore, a CaCO3/Ca(OH)2 layer in the soil surface can be produced in the presence of moisture because of its electron sources. Consequently, the cesium on the soil surface can be enclosed or bound (Srinivasa Reddy et al. 2012).
We assumed that the addition of the nano-Fe/Ca/CaO would reduce the heavy metal leaching potential from contaminated soil because of its high reduction potential and high surface area achieved with simple grinding. The main advantage of this process is that the natural moisture content in soil can bind heavy metals in the presence of nano-Fe/Ca/CaO in a simple grinding process. Further, encapsulated nano-Fe particles in solidified soil media can also be separated by magnetic separation, this way heavy metals-contaminated soil volume can be reduced. Therefore, in the present work, the use of three different addictive (nano-Fe/CaO, nano-Fe/Ca/CaO, and nano-Fe/Ca/CaO/PO4) for the immobilization of heavy metals (As, Cd, Cr, and Pb) in contaminated soil was investigated using a simple grinding process. The degree of metal immobilization was evaluated by analyzing the leachable fraction of heavy metals obtained through the application of the Japanese soil elution standards. Furthermore, the detailed heavy metal immobilization and separation mechanism were also assessed.
Materials and methods
General experimental condition
In typical experiments, soil samples are non-polluted and commercially available mica/fibrolite soils produced in Okayama prefecture, Japan, were obtained and used. Their physical and chemical characteristics are the following particle dimensions: 75 μm–2.0 mm; chemical composition (JIS M 8856-1998)—SiO2 (74.1 %), Al2O3 (13.0 %), Na2O (3.2 %), K2O (2.7 %), Fe2O3 (1.7 %), CaO (0.86 %), TiO2 (0.20 %), MgO (0.16 %), the remainder (4.08 %) being organic material. The determined moisture content (JIS A 1203-1999) is 7.6 %. Heavy metals for the contamination of soil samples were prepared in laboratory, where sodium arsenate (HAsNa2O4), cadmium nitrate tetrahydrate [Cd (NO3)2 4H2O), chromium (III) nitrate (CrN3O9 9H2O), and lead nitrate (Pb (NO3)2] of each 0.1 g were dissolved in 5 ml water, solution was spread to 1 kg of soil in a plastic bottle, and mixed thoroughly for 24 h to obtain each 100 mg/kg of As, Cd, Cr, and Pb desired concentrations.
Nano-Fe/Ca/CaO preparation and heavy metal-contaminated soil treatment
Nano-Fe/CaO and Fe/Ca/CaO were prepared with Fe and CaO/and or Fe/metallic Ca/CaO through planetary ball mill (Retsch PM-100). Granular particles of metallic calcium were purchased (99 %, 2.0–2.5 mm particle size distribution, 0.43–0.48 m2 g−1 surface area; Kishida Chemical). Fine grade CaO was also obtained commercially with 98 % purity (Kishida Chemical). Similarly, iron (Fe) powder (size 0.15 mm) and sodium phosphate (NaH2PO4) were also obtained from Kishida Chemical. Room temperature Ar atmosphere, Fe/metallic Ca/CaO (825 °C for 2 h) composition (Fe/CaO = 2/5; Fe/Ca/CaO = 2/2/5) were individually introduced into the planetary ball mill (20 stainless steel balls; 10 mm diameter). Stirring was conducted for 1 h at 600 rpm to a rotation-to-revolution ratio of 1:2. These Fe/Ca/CaO composition, balls-to-powder ratio, and rotation-to-revolution ratio conditions were established after performing several experiments to ascertain the optimal conditions. After stirring, samples were collected in glass bottles and Ar was filled and stored for use in further treatment experiments.
Heavy metal-contaminated soil alone and mixtures of soil and individual nano-Fe/CaO (10:1), nano-Fe/Ca/CaO (10:1), and nano-Fe/Ca/CaO/PO4 (10:0.5:0.5 (NaH2PO4)) mixers were grinded with a ceramic tumbling mill (500 ml ceramic pot along with 10 ceramic balls; 10 mm diameter) for 2 h at 100–150 rpm under an air atmosphere. At the end of each treatment interval untreated soils, mixing time equal to 0, were sampled to be analyzed. Further, after treatment of magnetic and nonmagnetic fractions, soils were separated with laboratory magnetic stick.
Heavy metals analysis
Initially, total heavy metal content concentrations in soil were measured using the method described by Baker and Amacher (1982), and this method is used to analyze the total contents of As, Cd, Cr, and Pb, which involves the digestion of soil samples in a mixture of HF-HNO3-HClO4-H2SO4 in Teflon beakers placed on a hot plate. However, after treatment with and without the addition of nano-Fe/CaO, nano-Fe/Ca/CaO, and nano-Fe/Ca/CaO/PO4, the soil mixture was cooled to room temperature and soil elution tests were performed according to Japanese standard methods (Ministry of Environment Government of Japan 2003).
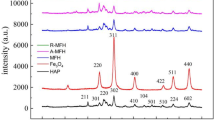
Heavy metal concentrations in eluted solutions were measured using inductively coupled plasma optical emission spectrometry (ICP-OES, 720-ES; Varian). These experiments were repeated three times, yielding similar results. To elucidate the immobilization mechanisms and to verify whether soil alterations occurred during the treatment, the following analyses were conducted. Scanning electron microscope combined with energy dispersive X-ray spectrometer (SEM–EDS) microanalysis and semiquantitative analysis were performed [JSM6510A equipped with a Si(Li)] probe with 138 eV resolution; JEOL) to analyze As, Cd, Cr, and Pb distribution within the solid matrix. Generally, EDS was able to detect the fluorescent X-rays from the surface layer to 10 μm depth.
Results and discussion
Heavy metals immobilization and separation
The total concentrations of As, Cd, Cr, and Pb in the solution, after acid digestion of untreated soil, were 3.15, 2.83, 3.21, and 2.95 mg/l. The concentrations of heavy metals in the eluted solution after treatment by grinding and soil samples ground with nano-Fe/CaO, nano-Fe/Ca/CaO, and nano-Fe/Ca/CaO/PO4 are presented in Table 1. All values presented in the table are the averages of three replications. By simple grinding, As, Cd, Cr, and Pb eluted concentrations decrease from 3.15, 2.83, 3.21, and 2.95 mg/l to 0.696, 1.02, 0.97, and 0.987 mg/l, respectively. On the other hand, when nano-Fe/CaO, nano-Fe/Ca/CaO, and nano-Fe/Ca/CaO/PO4 were added during grinding, significantly leaching heavy metal concentration decreased (Table 1), while these eluted concentration of heavy metals corresponds to intrinsic heavy metal immobilization efficiencies of about 95–99 % with nano-Fe/Ca/CaO grinding treatment.
On the other and, after treatment with nano-Fe/CaO, nano-Fe/Ca/CaO, and nano-Fe/Ca/CaO/PO4, about 36, 44, and 45 wt% of magnetic fraction soil were separated (Fig. 1), while only mixing, without any addition of addictives, soil magnetic separation was not possible due to the absence of Fe particles. Results indicating that Fe/Ca and NaH2PO4 presence showed better magnetic separation. Further, the condensed total concentrations of As, Cd, Cr, and Pb in the solution, after acid digestion of magnetic and nonmagnetic fractions soil was about 80 and 20 %, respectively, from initial total heavy metals concentration (Fig. 1). This fact indicating that the above-mentioned magnetic separated fraction (about 44 wt% with nano-Fe/Ca/CaO) contains highly condensed heavy metals, while nonmagnetic fraction (about 66 wt%) soil contains very low concentrations of heavy metals. By this way, high heavy metals-contaminated soil volume can be reduced. Furthermore, it may also clearly seen from Table 1 that the leachable heavy metal concentrations in nonmagnetic soil fraction are much lower than the Japan soil elution standard regulatory threshold, <0.01 mg/l for As, Cd, and Pb, and 0.05 mg/l for Cr, respectively (Ministry of Environment Government of Japan 1991). These results appear to be very promising, and the addition of nano-Fe/Ca/CaO mixture with simple grinding technique may be considered potentially applicable for the remediation and volume reduction of contaminated soil by heavy metals.
Magnetic and nonmagnetic separated fraction soil and its condensed heavy metals, As, Cd, Cr, and Pb, after treatment with various additives. After treatment with nano-Fe/Ca/CaO dispersion mixture, significantly, high and low percent of condensed heavy metal were separated with magnetic and nonmagnetic fractions soil. These condensed heavy metal percent was calculated from initial total heavy metals concentration. Each value is the mean of 3 measurements
Immobilization and separation mechanism of heavy metals
Semiquantitative SEM–EDS analysis, for heavy metals-contaminated soil before and after nano-Fe/CaO, Fe/Ca/CaO, and Nano-Fe/Ca/CaO/PO4 treatment was presented in Table 2. Semiquantitative SEM–EDS analysis clarified that the surface amounts of As, Cd, Cr, and Pb in the soil surface decrease after nano-Fe/Ca/CaO treatment (Table 2), while these decreased mass percent corresponds to 99 % Cr, 100 % As, 99.4 % Cd, and 99.7 % for Pb. On the other hand, the Ca and Fe mass percent increased about 70 and 7 %, respectively. The reduction in As, Cd, Cr, and Pb amounts on the soil particle surface and is a possible explanation for the increase in immobilization efficiency with nano-Fe/Ca/CaO treatment, by an enclosed/bound soil surface with Ca-associated compounds.
The most probable mechanisms for the enhanced heavy metal immobilization capacity with nano-Fe/Ca/CaO treatment might be due to the adsorption and entrapment of heavy metals into newly formed aggregates because of the aggregation of soil particles and enclosure/binding with Ca-associated immobile salts and its separation, as portrayed schematically in Fig. 2. It is possible to assume that when soil is contaminated, heavy metals are adsorbed onto soil particles through a surface coordination process (Fig. 2a). By simple grinding, coarser sand or gravel soil particles decreases to fine clay or silts particles; hence, the particle surface area increased and the heavy metal mobility decreased because of adsorption area increase (Orumwense 2006). On the other hand, grinding with nano-Fe/Ca/CaO additives, soil particles are subjected to collisions that might promote aggregation and breakage phenomena (Montinaro et al. 2007). When aggregation occurs in the presence of nano-Fe/Ca/CaO, the amount of heavy metal adsorbed onto the surface of two overlapping particles might be entrapped within the newly formed aggregates. However, nano-Fe/Ca/CaO/PO4 can produce immobile salts with moisture and CO2 in atmosphere, including pozzolanic cement and hydraulic property. As a result, heavy metals (As, Cd, Cr, and Pb) would be brought into the immobile Ca/PO4 salts. Therefore, the soil surfaces might enclose/bind with Ca-associated [CaCO3/Ca(OH)2] salts (Fig. 2b) (Srinivasa Reddy et al. 2012; Nakayama and Itoh 2003; Ganjidoust et al. 2009).
Schematic representation of possible mechanisms, which may determine the remediation, and separation of heavy metal-contaminated soils after simple mixing in dry conditions: a soil before treatment heavy metals adsorption and nano-Fe/Ca/CaO addition, b soil surface covered/enclosed/heavy metals immobilization, and c high and low concentrated heavy metals soil separation by magnet
Further more, the controlling mechanism is a function of the standard redox potential of the contaminant metal in the presence of nano iron (Fe). Heavy metals are either reduced at the nano-Fe surface, Cu2+, Ag2+ or directly adsorbed to the nano-Fe surface where they are rendered immobile, Zn2+, Cd2+, Cr6+, Cu2+, Pb2+, As3+ etc. (Boparai et al. 2011; Uzum et al. 2009; Kanel et al. 2005; Jegadeesan et al. 2005). The standard redox potential of zero-valent iron is (−0.41 V) very close to that of Cd2+ (−0.40 V), Cr3+ (−0.42 V), Pb2+ (−0.13 V), and As3+ (−0.23 V); thus the removal of As, Cd, Cr, and Pb ions by nano-Fe is due to sorption (Li and Zhang 2007; Boparai et al. 2011; Kanel et al. 2005; Jegadeesan et al. 2005). Moreover, the magnetic properties of nano-Fe facilitate the rapid separation of nano-Fe from soil, via a magnetic field (Yavuz et al. 2006; Liu et al. 2009). By this way, encapsulated nano-Fe particles in solidified (small/finer and bigger/aggregate) soil fractions can also be separated (Fig. 2c). Consequently, the grinding treatment for contaminated soil with nano-Fe/Ca/CaO can reduce heavy metal leachability with the minimization of their solubility by the reduction in heavy metals, or fixing by adsorption, and precipitation of heavy metals, or the decrease of contact change of solution to heavy metals by soil aggregation or covering of its surface, or by all three factors. The amount of heavy metals exposed to leaching is reduced; thereby, enhancing high immobilization and volume reduction (by its separation) of heavy metal in contaminated soil was achieved with nano-Fe/Ca/CaO treatment.
Conclusion
This study was conducted to evaluate a nano-Fe/Ca/CaO dispersion mixture as an immobilization treatment for heavy metal (As, Cd, Cr, and Pb)-contaminated soils and its separation. With simple grinding, 65–80 % heavy (As, Cd, Cr, and Pb) metal immobilization can be achieved in soil. Grinding can achieve about 95–99 % of heavy metal immobilization with a nano-Fe/Ca/CaO dispersion mixture. After treatment, about 36–45 wt% of magnetic and 64–55 wt% of nonmagnetic fraction soils were separated, and its condensed heavy metals concentration was about 80 and 20 %, respectively.
After nano-Fe/Ca/CaO treatment, the leachable heavy metal concentrations were reduced to a concentration lower than the Japan soil elution standard regulatory threshold. SEM semiquantitative analysis revealed that the amounts of As, Cd, Cr, and Pb that were detectable on soil particle surfaces decrease after nano-Fe/Ca/CaO treatment. The most probable mechanism for heavy metal immobilization is the minimization of the through reduction of heavy metals and the decrease in the surface of the solution to heavy metals by soil aggregation or covering of its surface and its separation by grinding with nano-Fe/Ca/CaO. The results obtained in this study suggest that the nano-Fe/Ca/CaO mixture is a suitable treatment for the gentle immobilization and its separation of heavy metals in contaminated soil. The addition of nano-Fe/Ca/CaO that enhanced heavy metal immobilization and its separation in soil in normal moisture conditions is an innovative approach for the remediation of soils polluted with heavy metals.
References
Abramovitch RA, Qing LC, Hicks E, Sinard J (2003) In situ remediation of soils contaminated with toxic metal ions using microwave energy. Chemosphere 53:1077–1085
Baker DE, Amacher MC (1982) Nickel, copper, zinc, cadmium. In: Miller RH, Keeney DR (eds) Methods of Soil analysis, part 2. ASA-SSSA, Madison, WI, pp 323–334
Boisson J, Ruttens A, Mencha M, Vangronsveld J (1999) Evaluation of hydroxyapatite as a metal immobilizing soil additive for the remediation of polluted soils. Part 1. Influence of hydroxyapatite on metal exchangeability in soil, plant growth
Boparai HK, Joseph M, Carroll DMO (2011) Kinetics and thermodynamics of cadmium ion removal by adsorption onto nano zerovalent iron particles. J Hazard Mater 186:458–465
Cao RX, Ma LQ, Chen M, Singh SP, Harris WG (2003) Phosphate induced metal immobilization in a contaminated site. Environ Pollut 122:19–28
Castaldi P, Santona L, Melis P (2005) Heavy metal immobilization by chemical amendments in a polluted soil and influence on white lupin growth. Chemosphere 60:365–371
Ganjidoust H, Hassan A, Ashkiki AR (2009) Cement base solidification/stabilization of heavy metal contaminated soils with objective of achieving high compressive strength of the final matrix. Trans A Civil Engin 16:107–115
Garbisu C, Alkorta I (2001) Phytoextraction: a cost-effective plant- based technology for the removal of metals from the environment. Bioresour Technol 77:229–236
Glass DJ (1999) Economic potential of phytoremediation. In: Raskin I, Ensley BD (eds) Phytoremediation of toxic metals: using plants to clean up the environment. Wiley, New York, pp 15–31
Jegadeesan G, Mondal K, Lalvani SB (2005) Arsenate remediation using nanosized modified zerovalent iron particles. Environ Prog 24:289–296
JIS M (1998) Methods for chemical analysis of high-alumina raw material for ceramics, Japanese Industrial Standard Committee, Tokyo. 8856-1998
JIS A (1999) Test method for water content of soils, Japanese Industrial Standard Committee, Tokyo. 1203-1999
Kanel SR, Manning B, Charlet L, Choi H (2005) Removal of arsenic(III) from groundwater by nanoscale zero valent iron. Environ Sci Technol 39:1291–1298
Lair GJ, Gerzabek MH, Haberhauer G (2007) Sorption of heavy metals on organic and inorganic soil constituents. Environ Chem Lett 5:23–27
Li XQ, Zhang WX (2007) Sequestration of metal cations with zerovalent iron nanoparticles-a study with high resolution X-ray photoelectron spectroscopy (HR-XPS). J Phys Chem C 111:6939–6946
Li XD, Poon CS, Sun H, Lo IMC, Kirk DW (2001) Heavy metal speciation and leaching behaviors in cement based solidified/stabilized waste materials. J Hazard Mat 82:215–230
Lin CF, Lo SS, Lin HY, Lee YC (1998) Stabilization of cadmium contaminated soils using synthesized zeolite. J Hazard Mat 60:217–226
Liu QY, Bei Y, Zhou F (2009) Removal of lead(II) from aqueous solution with amino-functionalized nanoscale zero-valent iron. Cent Eur J Chem 7:79–82
Macauley E, Hong A (1995) Chelation extraction of lead from soil using pyridine-2,6-dicarboxylic acid. J Hazard Mater 40:257–270
Marino MA, Brica RM, Neale CN (1997) Heavy metal soil remediation: the effects of attrition scrubbing on wet gravity concentration process. Environ Prog 16:208–214
Ministry of Environment Government of Japan (1991) Environmental Quality Standards for Soil, Notification No. 46. http://www.env.go.jp/en/water/soil/sp.html
Ministry of Environment Government of Japan (2002) Soil Contamination Countermeasures Law Enforcement Regulation, Ordinance no. 29
Ministry of Environment Government of Japan (2003) Matters providing for measurement method of soil elution test, Notification no. 18
Mitoma Y, Miyata H, Egashira N, Simion A, Kakeda M, Simion C (2011) Mechanochemical degradation of chlorinated contaminants in fly ash with a calcium-based degradation reagent. Chemosphere 83:1326–1330
Montinaro S, Concas A, Pisu M, Cao G (2007) Remediation of heavy metals contaminated soils by ball milling. Chemosphere 67:631–639
Nakayama S, Itoh K (2003) Immobilization of strontium by crystalline zirconium phosphate. J European Ceramic Soc 23:1047–1052
Orumwense AO (2006) Effect of media type on regrinding with stirred mills. Minerals Metall Proces 23:40–44
Paff SW, Bosilovich BE (1995) Use of lead reclamation in secondary lead smelters for the remediation of lead contaminated sites. J Hazard Mater 40:139–164
Patricia M, Alicia FC (2008) Phosphates for Pb immobilization in soils: a review. Environ Chem Lett 6:121–133
Polettini A, Pomi R, Sirini P, Testa F (2001) Properties of Portland cement-stabilized MSWI fly ashes. J Hazard Mater 88:123–138
Raicevica S, Radoicicb TK, Zouboulisc AI (2005) In situ stabilization of toxic metals in polluted soils using phosphates: theoretical prediction and experimental verification. J Hazard Mater 117:41–53
Reddy KR, Xub CY, Chinthamreddy S (2001) Assessment of electrokinetic removal of heavy metals from soils by sequential extraction analysis. J Hazard Mater 84:27–296
Sheppard MI, Thibault DH (1992) Desorption and extraction of selected heavy metals from soil. Soil Sci Soc Am 56:415–423
Srinivasa Reddy M, Mitoma Y, Okuda T, Sakita S, Kakeda M (2012) High immobilization of soil cesium using ball milling with nano-metallic Ca/CaO/NaH2PO4: implications for the remediation of radioactive soils. Environ Chem Lette 10:201–207
Stegmann R, Brunner G, Calmano W, Matz G (2001) Treatment of contaminated soil fundamentals, analysis, applications. Springer, Berlin
Tessier A, Campbell PGC, Bisson M (1979) Sequential extraction procedure for the speciation of particulate trace metals. Anal Chem 51:844–851
USEPA (1982) Guide to the disposal of chemically stabilized and solidified waste, SW-872. Office of water and waste management, Washington DC
USEPA (1993) Technology resource document-solidification/stabilization and its application to waste materials. EPA/530/R- 93/012
Uzum C, Shahwan T, Eroglu AE, Hallam KR, Scott TB, Lieberwirth I (2009) Synthe- sis and characterization of kaolinite-supported zero-valent iron nanoparticles and their application for the removal of aqueous Cu2+ and Co2+ ions. Appl Clay Sci 43:172–181
Vithanage M, Dabrowska BB, Mukherjee AB, Sandhi A, Bhattacha P (2011) Arsenic uptake by plants and possible phytoremediation applications: a brief overview. Environ Chem Lett. doi:10.1007/s10311-011-0349-8
Yavuz CT, Mayo JT, Yu WW, Prakash A, Falkner JC, Yean S, Cong LL, Shipley HJ, Kan A, Tomson M, Natelson D, Colvin VL (2006) Low-field magnetic separation of monodisperse Fe3O4 nano crystals. Science 314:964–967
Acknowledgments
Authors are thankful to the New Energy and Industrial Technology Development Organization (NEDO) Program (Project ID: 09B35003a) for providing financial support for this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mallampati, S.R., Mitoma, Y., Okuda, T. et al. Total immobilization of soil heavy metals with nano-Fe/Ca/CaO dispersion mixtures. Environ Chem Lett 11, 119–125 (2013). https://doi.org/10.1007/s10311-012-0384-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-012-0384-0