Abstract
Purpose
Currently available techniques for the evaluation of small fiber neuropathy and related sudomotor function remain suboptimal. Electrochemical skin conductance (ESC) has recently been introduced as a simple noninvasive and fast method for the detection of sudomotor dysfunction. The purpose of this review is to synthesize and appraise research using ESC measurements for sudomotor evaluation in adults.
Methods
Electronic databases including MEDLINE and Google Scholar were searched (up to March 13, 2017). The search strategy included the following terms: "electrochemical skin conductance,” “Sudoscan,” and “EZSCAN.” Evidence was graded according to defined quality indicators including (1) level of evidence; (2) use of established tests as reference tests (e.g., quantitative sudomotor axon test [QSART], sympathetic skin responses [SSR], thermoregulatory sweat test [TST], and skin biopsies to assess sudomotor and epidermal small fibers); (3) use of consecutive/non-consecutive subjects; and (4) study design (prospective/retrospective).
Results
A total of 24 studies met the inclusion criteria. These were classified into preclinical, normative, comparative/diagnostic, or interventional. ESC measurement properties, diagnostic accuracy, and similarities to and differences from established tests were examined.
Conclusions
ESC measurements expand the arsenal of available tests for the evaluation of sudomotor dysfunction. The advantages and disadvantages of ESC versus established tests for evaluating sudomotor/small fiber function reviewed herein should be used as evidence to inform future guidelines on the assessment of sudomotor function.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Small nerve fibers are thinly myelinated Aδ fibers and unmyelinated C fibers, which are further classified as autonomic (efferent) or sensory/somatic (afferent). Their dysfunction (i.e., small fiber neuropathy [SFN]) can result in autonomic symptoms, sensory symptoms, or a combination of both. SFN with mixed involvement of both sensory and autonomic fibers is the most common form, affecting millions of individuals worldwide and contributing significantly to morbidity [1,2,3,4,5,6]. SFN can be further classified into idiopathic or secondary. Among the secondary causes, cardiovascular autonomic neuropathy (CAN) caused by diabetes mellitus (DM) is a common cause of SFN [5]. CAN is present in up to 90% of patients with type 1 and 20–73% of patients with type 2 diabetes mellitus [5], depending on the diagnostic test. Medical history and physical examination, although important, are usually insufficient for establishing a diagnosis of SFN with a high level of certainty or for determining its severity. Therefore, patients with suspected SFN with autonomic dysfunction should undergo ancillary diagnostic testing to confirm the diagnosis.
Established objective autonomic function tests endorsed by the American Autonomic Society (AAS) for the evaluation of sudomotor function include the quantitative sudomotor axon test (QSART), the sympathetic skin response (SSR), and the thermoregulatory sweat test (TST) [7]. When cardiovascular autonomic failure is suspected in addition to sudomotor dysfunction, cardiovascular autonomic testing (including Valsalva maneuver, heart rate variability during paced deep breathing, and tilt-table test with continuous blood pressure readings with plethysmography) is recommended.
Additional methods for evaluating SFN include skin biopsy for the quantification of epidermal nerve fiber density (ENFD) [6] and sudomotor (sweat gland) nerve fiber density (SGNFD) [8]. ENFD is the standard test for evaluation of SFN with high diagnostic accuracy and is recommended by the European Federation of Neurological Societies (EFNS) [3]. The diagnostic accuracy of SGNFD, however, has yet to be established. A number of challenges remain in evaluating the autonomic nervous system. The main difficulty arises from the fact that most functional tests quantify the end-organ effect of the autonomic input but not the autonomic nerve function per se.
Electrochemical skin conductance (ESC) was recently introduced as a simple electrophysiological method for the functional evaluation of sweat glands and related sudomotor/small fiber dysfunction [9,10,11,12]. The rationale and methodology involving ESC is explained in detail in the “Results” section. Similar to the majority of functional autonomic tests, ESC measures end-organ responses as a surrogate marker of autonomic nerve function. There are two types of human sweat glands: eccrine and apocrine. Eccrine sweat glands cover most of the skin; they produce a clear sweat composed primarily of water and sodium chloride, and they play a major role in thermoregulation. Apocrine sweat glands are concentrated in the scalp, armpits, and groin; they produce a dense sweat released in the canals of hair follicles, and they do not participate significantly in thermoregulation. Postganglionic sympathetic cholinergic non-myelinated C fibers innervate the periglandular tissue of the eccrine sweat glands [13]. A minority of these fibers are adrenergic, but their role in thermoregulation is questionable. ESC measurements assess eccrine gland sweat production.
In this work, we performed a systematic literature review of studies using ESC. Here, we summarize their limitations, potential clinical applications, advantages and disadvantages, and comparison with other well-established tests for evaluating sudomotor function. Specifically, this review addresses two questions:
-
1.
Can ESC provide accurate measurements of sympathetic sudomotor (cholinergic) function?
-
2.
Do ESC results correlate with other established tests of sudomotor function or sweat gland morphology?
Methods
Literature search strategy
A search of the PubMed and Google Scholar (scholar.google.com) databases was performed for the period up to March 13, 2017, using the following search terms: “electrochemical skin conductance” [All Fields] OR “Sudoscan” [All Fields] OR “EZSCAN” [All Fields]. The search yielded 47 entries. Only peer-reviewed scientific articles were considered; review articles or studies with overlapping results were excluded.
Quality ratings
In order to quantify the quality of each publication, the level of evidence (Table 1) [14] and several quality indicators were extracted. Quality indicators included:
-
a.
Comparison with well-established sudomotor tests (QSART, TST, or SSR).
-
b.
Prospective versus retrospective study: if the study was not explicitly described in the methods section as prospective, it was considered retrospective, unless it was obvious from the methods that the study was prospective.
-
c.
Consecutive versus non-consecutive inclusion of patients: if the study did not specify whether consecutive patients were included, non-consecutive inclusion was assumed.
-
d.
Performance of skin biopsy for assessment of small fiber density using a methodology defined by the EFNS [3].
-
e.
Performance of standardized cardiovascular autonomic testing. Established autonomic testing was defined by AAS recommendations [7] for autonomic screening, and included heart rate variability during paced deep breathing, tilt-table test, and Valsalva maneuver, and one of the following: QSART, TST or SSR.
-
f.
For interventional studies, whether the intervention was randomized and the patients/investigators were blinded to the intervention [14].
Results
Twenty-two studies fulfilled the inclusion criteria. Figure 1 shows a PRISMA flow diagram [15]. The selected publications included preclinical/mechanism-of-action studies, normative diagnostic or correlation studies (using a reference test), and interventional studies.
Preclinical and mechanism-of-action studies
Two studies analyzing preclinical data were retrieved. The measurement of ESC is an active electrophysiological method based on two physical principles [9, 10, 12]: (1) low-voltage electrical activation of sweat glands and (2) reverse iontophoresis to capture the flow of chloride ions produced by activated sweat glands. ESC measures the flow of chloride ions induced by electric current. According to a theoretical model, electric current directly stimulates the sudomotor fibers, innervating the sweat glands. Subsequently, the glands produce sweat, which is composed of chloride ions. The chloride ions are consumed at the anode, mimicking the corrosion process using principles of reverse electrophoresis. In direct iontophoresis, ions move in a medium driven by an electric current [16]. In reverse iontophoresis, also called electro-osmotic flow, the molecules are extracted from the medium. In the case of ESC, subepidermal chloride ions are moved to the skin surface. Human skin has relatively high electrical capacitance due to its dense lipid layers, and therefore the ions cannot penetrate the stratum corneum at low voltage [12, 17]. However, ions driven by electric current can move in an electric field via sweat duct pores. In vitro electrochemical studies [18] evaluating nickel and stainless steel electrode behavior in sweat-mimicking solutions showed that the concentration of chloride had a strong influence on the anodic potential. The chloride ion flow produced by sweat glands is proportional to the level of stimulus and sweat gland function. Since the stimulus is electrical, it is possible to exactly define the intensity of the stimulation level, which may be a key factor in reproducibility.
During ESC measurement, subjects place both palms and soles on large-area stainless-steel electrodes. An incremental low direct voltage (< 4 V) is applied for 15 s. The voltage applied on skin electrodes generates a current between the anode and the cathode that is measured. For example, the right-foot electrode is used as an anode, and voltage of 1 V is applied for 1 s. This induces voltage on the cathode, i.e., the left-foot electrode, and a current travels through the body towards the cathode. The voltage occurring on the cathode is measured using a voltmeter, and the current between the electrodes is measured using a Wheatstone bridge. The voltage applied on the anode is then increased to 1.2 V over 1 s, and a new measurement is performed. The voltage continues to be increased in increments of 0.2 V per second until a maximum voltage of 3.8 V is reached (15 steps in all). The same 15 incremental increases in voltage are applied on the left-foot electrode used as anode, while the right-foot electrode is used as cathode. Finally, the same 15 steps are applied twice on the hand electrodes, with each electrode used alternately as an anode or a cathode.
The ESC is calculated from the generated current, which is expressed as follows: (1) current as a function of the anodic potential (so-called E); (2) current as a function of the cathodic potential after applying an incremental voltage at the anode (so-called V); and (3) current as a function of U = E + V. Quantitative results are expressed as ESC, i.e., a ratio of the current generated and the direct voltage stimulus in microsiemens (μS). Two ESC values are usually provided separately for the average of the two hands and the two feet.
Clinical studies
Table 2 shows a summary of the clinical studies reviewed, along with their quality assessment. Only one study had level-1 quality (higher quality standard), and only one had level-2 quality. The others were all of level-3 or worse quality.
Studies describing the properties of ESC measurements
Safety and tolerability
The measurement of ESC is consistently safe and tolerable, as no discomfort or adverse events were reported in 1376 tests performed on healthy participants [19].
Symmetry
There were no differences between the left and right sides, with a high correlation of ESC values between the right and left hands (r = 0.96, p < 0.0001) and the right and left feet (r = 0.97, p < 0.0001) among 1350 healthy participants [19].
Ethnicity, body mass index, sex, and age effect
ESC values appear to be lower in African American, Indian, and Chinese subjects than in whites [19]. There was no significant effect of body mass index on ESC measurements [19]. No differences were observed in ESC measured in hands and feet between men and women [9, 19]. In a study evaluating 1350 participants, ESC in the hands and feet decreased significantly with increasing age (R = −0.17, p < 0.0001; feet: R = –0.19, p < 0.0001) [19]. Thus, studies using ESC should include age as a covariate.
Reproducibility
When ESC measurements were performed in the same patient twice during the same day, the coefficients of variation were 7% for the hands and 5% for the feet of patients with cardiovascular risk, and 15% and 7%, respectively, for the hands and feet of patients with diabetes [9].
Diagnostic, comparative, or association studies
These studies were designed to study ESC measurements in different disorders, all characterized by defective sudomotor function. Some studies also determined potential associations with established markers of autonomic, sensory, or motor dysfunction.
Cystic fibrosis
Cystic fibrosis is a genetic disorder characterized by defective electrolyte transport associated with increased sweat chloride concentration [20]. ESC was measured in 41 adult patients with classical cystic fibrosis and 20 healthy controls [12]. ESC on the hands and feet was increased in patients with cystic fibrosis compared to controls (feet: 75 ± 10 vs. 62 ± 13 μS; hands: 73 ± 14 vs. 61 ± 15 μS; p < 0.01 in both cases). This is consistent with the mechanism of action of the ESC technique, which is based on chloride iontophoresis.
Diabetic neuropathy
Most studies are based on the premise that sudomotor dysfunction occurs due to chronic hyperglycemia and related metabolic perturbations in patients with diabetes or abnormal glucose metabolism. In a study performed in China, Zhu et al. [21] used ESC as an early biomarker for metabolic syndrome. The study included 1078 participants, 567 of whom had metabolic syndrome. Participants with metabolic syndrome had reduced ESC (70.6 ± 13 μS [cardiac risk score 20–29] and 63.2 ± 16.6 μS [cardiac risk score ≥ 30]) compared to controls (78.1 ± 10.1 μS, p < 0.0001).
A study by Freedman et al. [22] evaluated 205 African Americans with type 2 diabetes mellitus, 93 African American non-diabetic controls, 185 European Americans with type 2 diabetes mellitus, and 73 European American non-diabetic controls. The authors found that ESC was lower in subjects with type 2 diabetes mellitus and also in African Americans. After adjusting for age, sex, BMI, and HbA1c, ESC decreased with decreasing renal function (estimated glomerular filtration rate [eGFR]) in African Americans with type 2 diabetes mellitus. Another study [23] examined the relationship between ESC and other parameters of renal function (eGFR and albumin-to-creatinine ratio in urine) in 202 European Americans and 181 African Americans with type 2 diabetes mellitus. Specific attention was paid to patients with diabetic kidney disease, as defined by an eGFR below 60/ml/min/1.73 m2. ESC decreased with decreasing renal function in both ethnic groups, but was lower in patients with type 2 than type 1 diabetes mellitus [24]. Another study including 2833 Chinese adults with type 2 diabetes mellitus showed that lower ESC scores were associated with chronic kidney disease, with moderate sensitivity (76.7%) and specificity (63.4%) [25]. A cross-sectional study including 36 patients with type 2 diabetes mellitus and coronary artery disease and 20 age- and sex-matched controls showed that ESC was significantly lower in patients than in controls, and that lower ESC results were associated with higher C-reactive protein levels, higher HbA1c, and lower ankle-brachial index, a marker of peripheral artery disease [26]. The accuracy and reproducibility of ESC measurements were tested in 133 patients with type 2 diabetes mellitus and 41 controls. The sensitivity and specificity of low ESC measurements (cutoff point of < 50% of the EZSCAN scale) for the diagnosis of type 2 diabetes mellitus were 75% and 100%, respectively [27]. In a study including Asian Indian subjects [28], low ESC measurements (cutoff point of < 50% of EZSCAN scale) demonstrated sensitivity of 75% for diagnosing type 2 diabetes mellitus, 70% for diagnosing impaired glucose tolerance, and 84% for detecting metabolic syndrome. A similar study in Chinese subjects found similar sensitivity and low specificity for detecting diabetes mellitus or impaired glucose tolerance [29]. In a study including white subjects, Casellini and colleagues [30] measured ESC in 83 patients with type 1 or type 2 diabetes and 210 controls. In receiver operating characteristic (ROC) curve analysis, ESC measurements (feet ESC cutoff 77 μS) showed sensitivity of 78% and specificity of 92% for detecting diabetic neuropathy using the Neuropathy Impairment Score-Lower Limbs as a reference. Another study in 394 Chinese subjects with type 2 diabetes mellitus showed that ESC values decreased with increasing scores of the Neurological Symptom Score (R = −124, p < 0.05) and Neuropathy Disability Score (R = −0.3, p < 0.01) [31].
Selvarajah and colleagues [32] evaluated 45 participants with type 1 diabetes mellitus and 25 healthy controls. Assessments included evaluation for the presence of diabetic peripheral neuropathy [33] and cardiac autonomic neuropathy with cardiovascular autonomic testing. The authors found that ESC in the feet was lower in subjects with diabetic peripheral neuropathy (53.5 ± 25.1 µS) than in patients without peripheral neuropathy (77.0 ± 7.9 µS) or in healthy controls (77.1 ± 14.3 µS, p < 0.001). Similarly, subjects with cardiac autonomic neuropathy had lower ESC results (55.0 ± 28.2 µS) than patients with no cardiac autonomic neuropathy (72.1 ± 12.2 µS) or healthy controls (77.1 ± 12.2 µS, p < 0.001).
Sheshah and colleagues [34] studied 296 Saudi patients with type 2 diabetes mellitus using the Neuropathy Symptom/Neuropathy Disability score. For values of ESC < 70 µS, the sensitivity/specificity for diagnosis of diabetic neuropathy was 67.5/58.9%. ESC values increased with increasing amplitude and velocity of peroneal and sural nerves (p < 0.005 in all cases).
Yajnik and colleagues [35] assessed ESC and its correlation with measures of peripheral and cardiac neuropathy in 265 Indian patients with type 2 diabetes mellitus. Lower ESC at the feet was associated with both increasing symptoms of peripheral neuropathy and worse physical disability scores. Lower ESC at the feet was also significantly associated with lower vibration sense perception (p < 0.01). Patients with abnormal cardiovascular autonomic testing had lower ESC (p < 0.05). Another study [36] including 232 patients with diabetes found that lower ESC values predicted abnormal cardiac autonomic responses with 92% sensitivity and 49% specificity. Ang and colleagues [37] assessed ESC in a longitudinal study of 37 patients with type 2 diabetes mellitus and 40 age-matched healthy controls. At baseline, there were no differences in ESC between patients with type 1 diabetes and controls, whereas heart rate variability and Valsalva ratios were significantly lower in the diabetic patients. Twelve months later, the patients with type 1 diabetes had experienced a significant decline in ESC (p = 0.0006 at hands and p = 0.023 at feet). However, no associations were found between ESC and other measures.
Non-diabetic neuropathy
Smith and colleagues [38] evaluated ESC in patients with symptoms consistent with distal symmetric polyneuropathy (22 patients with diabetes, two patients with pre-diabetes, and 31 patients with idiopathic neuropathy) as well as 42 controls. Patients were also assessed using QSART, nerve conduction studies, and skin biopsies. ESC was reduced in patients, and there was no difference between diabetic and non-diabetic subjects. Lower ESC values were associated with higher frequency of neuropathy signs using the Utah Early Neuropathy Scale (UENS R = 0.388, p < 0.004) and with symptoms using the Michigan Neuropathy Screening Instrument (MNSI R = –0.398, p < 0.005). Higher ESC values were associated with higher intra-epidermal fiber density (IENFD) at the proximal leg (R = 0.373, p < 0.012) but not the distal leg (R = 0.263). ESC results were significantly associated with the results of QSART at the foot (R = 0.348, p < 0.015). ESC also correlated with sural amplitude (R = 0.337, p < 0.019) but not with peroneal motor amplitude (p < 0.068) or peroneal motor conduction velocity (p < 0.081). ESC demonstrated sensitivity of 77% and specificity of 67% in detecting distal symmetric neuropathy using UENS as a reference.
In a prospective blinded study, Novak [39] investigated the association between ESC at the feet and ENFD and SGNFD at the distal leg using skin biopsies. The author also studied the association between ESC and QSART as well as symptom scales (neuropathy, pain, and autonomic). Eighty-one consecutive patients with small fiber neuropathy were included. ESC adjusted for weight was reduced in subjects with abnormally low ENFD (ENFD normal/abnormal, ESC = 1.17 ± 0.27/0.87 ± 0.34 μS/kg, p < 0.0008) and abnormally low SGNFD (SGNFD normal/abnormal ESC = 1.09 ± 0.34/0.78 ± 0.3 μS/kg, p < 0.0003). ESC adjusted for weight correlated with ENFD (ρ = 0.73, p = 0.0001) and SGNFD (ρ = 0.64, p = 0.0001). ESC did not correlate with symptom scales. Castro and colleagues [40] studied ESC in 133 subjects with the Val30Met mutation in the transthyretin familial amyloid polyneuropathy (TTR-FAP) gene, divided into asymptomatic and symptomatic stage 1 (walking unaided), which were compared with 37 healthy controls. ESC at the feet showed 76% sensitivity and 85% specificity (feet ESC cutoff 66 μS) for detection of autonomic dysfunction, defined as a score of 1 or above on the Compound Autonomic Dysfunction Test [41], which is a questionnaire evaluating symptoms of dysautonomia.
Interventional studies
Two studies used ESC to monitor the effect of therapeutic interventions. The first [42] evaluated the effect of disease-modifying anti-rheumatic drugs on ESC in 42 patients with rheumatoid arthritis, 18 with ankylosing spondylitis, and 30 aged-matched controls. In addition to ESC measurements, the authors performed sympathetic (blood pressure response to standing and handgrip test) and parasympathetic function tests (heart rate response to paced deep breathing and to the Valsalva maneuver). After a 12-week open-label period of administration of disease-modifying anti-rheumatic medication, parasympathetic and ESC measurements in the patient groups increased. The second study [43] evaluated the impact of bariatric surgery on cardiac autonomic tone, ESC, and sensory function in obese subjects with and without type 2 diabetes mellitus. Patients were evaluated at baseline and 4, 12, and 24 weeks after vertical sleeve gastrectomy or Roux-en-Y gastric bypass. Subjects were assessed using ESC, time- and frequency-domain heart rate variability analysis, Neuropathy Impairment Score of the Lower Legs (NIS-LL), quantitative sensory testing, and sural nerve conduction studies. Seventy subjects completed up to 24 weeks of follow-up (24 without type 2 diabetes mellitus, 29 with pre-diabetes, and 17 with type 2 diabetes mellitus). ESC of the feet was significantly higher in type 2 diabetic subjects (baseline vs. 12-week vs. 24-week, p < 0.005). Heart rate variability also increased significantly only in patients with type 2 diabetes. Basal heart rate, weight, body mass index, percentage of body fat, waist circumference, and high-density lipoprotein improved in all groups. These two studies suggest that ESC measurements can detect changes after specific therapeutic interventions, opening the door to its use as a potential outcome measure in clinical trials of diabetic and non-diabetic patients with sudomotor dysfunction.
Discussion
This systematic review was undertaken to evaluate studies using ESC measurements and to provide an evidence base to inform the use of this technique for the evaluation of sudomotor dysfunction.
ESC reflects the flow of chloride ions provoked by electric current. A study that included patients with cystic fibrosis [20] showed that ESC values were increased compared to those of controls. Because the sweat chloride concentration in patients with cystic fibrosis is increased [39, 44, 45], this ESC result is consistent with the assumption that ESC reflects changes in the concentration of sweat chloride. Thus, ESC values appear to reflect sweat gland dysfunction, which can be caused by both neurological and non-neurological disorders. Chronic denervation also results in atrophy of the sweat glands. Sweat glands are prone to atrophy and hypertrophy as a result of several non-neurological disorders [46]. Therefore, reduced ESC measurements may be caused by several mechanisms, including loss of sudomotor fibers, sweat gland atrophy, reduced number of sweat glands, or glandular dysfunction caused by toxic, metabolic, or other disorders.
Only one study in this review was found to have level-1 quality. This study demonstrated that ESC measurement correlated with sweat gland nerve fiber density [39], but also with the density of epidermal small fibers [39] unrelated to sweat production. The latter correlation may be due to a common offending factor causing both reduced sweat gland and epidermal nerve density. This study thus strongly suggests that ESC measurements could be used to monitor the severity of small fiber neuropathy, provided that non-neurological disorders of the sweat glands have been ruled out. In any case, further confirmatory studies are needed.
The studies included in this review that used ESC in patients with diabetes were of level-3 and level-4 quality. Many of the studies reported associations between lower ESC measurements and higher severity of kidney disease [22, 23, 25] or metabolic syndrome [21, 28]. There are two potential explanations for this. The most apparent is that the sudomotor small nerve dysfunction measured by ESC and the presence of metabolic or end-target organ damage are all comorbid consequences of the same disorder (i.e., diabetes), and that all are expected to progress in parallel. Therefore, in patients with diabetes, worse renal function (or the presence of metabolic syndrome) might be associated with lower ESC values, because both are surrogate markers of disease progression in patients with diabetes. The other—perhaps less likely—explanation is that, in these patients, reduced ESC measurements reflect sweat gland dysfunction due to metabolic derangement rather than sudomotor small fiber dysfunction. In this case, ESC would be directly activating the sweat gland but not the sudomotor fibers innervating the sweat glands. At this time, the evidence is not sufficient to provide a conclusive answer to this question, as the studies measuring ESC, kidney disease, and metabolic syndrome in patients with diabetes did not measure sudomotor function with an established test.
A theoretical model suggests that ESC does stimulate small fibers directly [9, 18, 47], and there is experimental evidence that electric current directly stimulates the small fibers, at least in part. In isolated monkey eccrine sweat glands with preserved innervation [48], atropine administration reduced sweat secretion induced by electric current in 70% of sweat glands, proving that sudomotor cholinergic innervation plays a dominant role in electrical stimulation. The remaining (30%) sweat glands continued to secrete at a lower rate (15% of maximum), which suggests the presence of non-cholinergic transmission (i.e., adrenergic) or non-neural effects of electrical stimulation. The latter observation would suggest that even if an electric field stimulates the sweat gland directly, the effect is likely a minor compared to the effect due to stimulation of the sudomotor cholinergic nerves.
On the other hand, the level of electrical stimulation required to stimulate sweat production in humans appears to be high enough to also activate nociceptive afferent fibers, i.e., electrical stimulation in humans does not produce sweat unless the stimulation is sufficient to cause pain [49]. The voltage and intensity of ESC measurements are too low to be detected by fine touch or nociceptive fibers. A potential explanation for this discordant observation is that the ESC apparatus uses a direct current of variable intensity, whereas the above-mentioned study in humans [49] used repetitive stimulation.
Therefore, whether ESC stimulates sudomotor fibers or sweat glands directly is not absolutely clear. Designing a study using anticholinergic medications to confirm that ESC stimulates sudomotor fibers (i.e., ESC will be reduced) and not sweat glands (ESC should be unchanged) should be relatively straightforward. This design was already used to demonstrate that electrical stimulation of the lacrimal gland in rabbits activated small fibers (both efferent and afferent) and not the lacrimal gland per se, since the stimulation effect was eliminated with cholinergic muscarinic blockers [50]. It would also be useful to assess ESC in patients with congenital absence of sweat glands [51] or congenital absence of cholinergic sudomotor nerves (e.g., congenital insensitivity to pain and anhidrosis) [52].
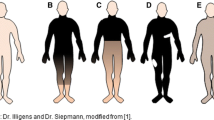
Even if ESC stimulates the sweat glands directly and not small fibers, or if it stimulates both, it still may be a useful test: sweat glands are an end-organ of small cholinergic fibers, and degeneration of small fibers can result in sweat gland atrophy [46], affecting ESC. Furthermore, it is important to establish how clinical improvement in patients with small fiber neuropathies and re-innervation of sweat glands affects ESC. In either scenario, prospective studies are required to improve our interpretation of ESC measurements. Figure 2 summarizes the potential mechanisms of ESC.
Principles of ESC measurements. There are two proposed targets of stimulation. The sympathetic sudomotor fibers (cholinergic and adrenergic, with the majority cholinergic) are stimulated by electric current (1). The activated fibers stimulate sweat glands that release NaCl. Alternatively, the sweat glands are stimulated directly (2). Cl is then extracted by the anode, and the chloride ion flow is measured. Ach acetylcholine, NE norepinephrine
Similarities to and differences from other available techniques
This systematic review shows that there was considerable heterogeneity in the use of reference tests for comparison with ESC, and in many studies established sudomotor tests were not used. Table 3 summarizes the features of well-established techniques for measuring sudomotor functions.
Comparison with QSART
Both QSART and ESC use sweat glands as a proxy for small fiber dysfunction. QSART measures the sweat volume and uses acetylcholine as a mediator, which stimulates postganglionic fibers via muscarinic receptors [53]. ESC measures chloride ion flow in the skin and uses the electric current as a stimulus. In addition to the different measurement techniques, a major difference between QSART and ESC is that QSART uses chemical while ESC uses electrical stimuli. ESC appears to generate an exact level of electrical stimulation via the feedback of electrical drivers. This is in contrast to the QSART, where the number of molecules of acetylcholine delivered and measured may vary from measurement to measurement, since the iontophoretic driver used for acetylcholine delivery does not use a chemical feedback control. Another difference between QSART and ESC, although of unclear clinical significance, is the fact that QSART uses a pure cholinergic stimulus (acetylcholine), whereas the electrical stimulation used in ESC, in theory, could stimulate both cholinergic and adrenergic sudomotor fibers, even if the latter are a minority [13, 54]. Furthermore, ESC provides measurements at the palms and soles, which are under emotional control of sweating [55, 56], whereas QSART mainly assesses thermoregulatory sweating at the proximal and distal limbs [53]. Surprisingly, studies thus far have shown little or no correlation between ESC and QSART [39, 44]. The differences in measurement principles between tests as discussed above may play a role. Another unexplained difference between QSART and ESC is that QSART is much more sensitive to age and gender. Further studies may help provide greater clarity regarding the relationship between these two tests.
Comparison with SSR
Both SSR [57] and ESC rely on electrical recordings from the surface of the skin. The SSR response can be triggered by an inspiratory gasp, forceful expiration, or electrical stimulation. SSR is believed to be generated by a reflex activation of sweat glands via cholinergic sudomotor fibers. The SSR is a multi-synaptic reflex, and therefore depends on the integrity of both the peripheral and central nervous systems. Abnormal SSR could result from abnormal autonomic efferent fibers, abnormal sensory afferent fibers, or atrophy of effector sweat glands after chronic denervation [58]. SSR has a number of limitations: it is qualitative in nature, with poor reproducibility, is clearly inferior to QSART for the evaluation of sudomotor function [54,55,56,57, 59], and may be normal in patients with absent sweat glands [60]. As a screening tool for sudomotor dysfunction in the context of a small fiber neuropathy, ESC appears to be superior to SSR, as ESC is reproducible, fully quantitative, without habituation, requires minimal patient cooperation, and is more sensitive than SSR for the diagnosis of small fiber neuropathy (sensitivity/specificity 49.4/92.5% for ESC vs. 33.3/77.6% for SSR) [61].
Comparison with TST
TST measures the integrity of both the central and peripheral nervous systems [57]. A major disadvantage of TST is the requirement for a complex setup. However, TST is unique in that it provides sweating topography. ESC and TST have different objectives, and it is not expected that ESC will replace TST, as ESC measurements are restricted to hands and feet, and do not assess the integrity of the CNS.
Comparison with skin biopsies
A prospective blinded study showed a correlation between ESC and both SGNFD and ENFD [39]. Another study showed a correlation between higher ESC values at the feet and higher ENFD at the thigh, but not ENFD at the calf [44]. The direct functional–pathological correlation (ESC-SGNFD), although moderate (R = 0.64, p = 0.0001) [39], supports the notion that ESC reflects sudomotor fiber dysfunction. This correlation was obtained after adjusting the ESC by weight, indicating that weight or related metrics (BMI, size of the measured area, etc.) may be a confounder. Clinicians should exercise care when interpreting interventional studies if patient weight changes over time.
Potential advantages
Because of the high reproducibility of ESC, potential advantages include the capacity for grading of abnormalities and the use of ESC values as longitudinal measurements for assessing disease progression. Two interventional studies—one investigating bariatric surgery in patients with obesity [43] and the other examining disease-modifying anti-rheumatic drugs in patients with arthritis [62]—showed improvement in ESC abnormalities post-treatment. However, neither study performed established tests of sweat function to validate the ESC results. Also, future studies are needed to clarify whether ESC is able to detect sudomotor re-innervation.
Recommendations
Although a burgeoning body of evidence suggests that ESC can accurately measure sudomotor function, and that it correlates with SGNFD and ENFD, most studies thus far are of limited quality. In this review, only one study was blinded, and all but two failed to include consecutive patients or to use other well-established sudomotor function tests as reference. Furthermore, studies evaluating the effect of medication that can affect sudomotor function—particularly anticholinergic medication—are lacking. Thus future studies should (1) recruit consecutive patients, (2) perform the analysis and interpretation of data blinded to the suspected diagnosis, and (3) include established tests of sudomotor function and/or skin biopsy. Cardiovascular autonomic testing (heart rate variability during paced deep breathing, Valsalva maneuver, and tilt-table test) should be performed in patients with suspected cardiovascular autonomic failure [7]. ESC must never replace cardiovascular autonomic testing under any circumstances, as each technique measures different outcomes. Most of the ESC studies were funded by the device manufacturer, raising concerns about potential industry bias. Non-industry-sponsored studies would be ideal.
Conclusions
A growing body of evidence suggests that ESC measurements have the potential for use in evaluating sudomotor function. The European Network for TTR-FAP (ATTReuNET) recommends evaluation of asymptomatic carriers for the presence of small fiber damage using ESC measurements as an alternative to SSR or QSART [63]. Our review of this evidence can now be used to inform recommendations for sudomotor testing. Fully quantitative, noninvasive, and simple methods are needed for the detection of SFN and related autonomic dysfunction. A number of studies indicate the clinical potential of ESC. The association between ESC and SGNFD, along with the high reproducibility of ESC measurements, is particularly promising. At the same time, clinicians using ESC measurements must be aware of some caveats. ESC provides an index of sudomotor activity, and should in no way be considered a replacement for established gold-standard cardiovascular autonomic tests (heart rate variability during paced deep breathing, Valsalva maneuver, and tilt-table testing) for the assessment of cardiovascular autonomic function. Although the ESC correlates with skin biopsies assessing small fibers, the correlation is not high enough to justify replacement of skin biopsies by ESC. Moreover, ESC measurements may be abnormal in patients with abnormal sweat composition (e.g., cystic fibrosis) but otherwise normal sudomotor function. Although existing studies suggest that ESC is superior to SSR, important knowledge gaps remain, as the quality of the evidence regarding the relationship between ESC and QSART or SSR needs to be clarified.
References
Hovaguimian A, Gibbons CH (2011) Diagnosis and treatment of pain in small-fiber neuropathy. Curr Pain Headache Rep 15:193–200. doi:10.1007/s11916-011-0181-7
England JD, Gronseth GS, Franklin G et al (2009) Practice Parameter: evaluation of distal symmetric polyneuropathy: role of autonomic testing, nerve biopsy, and skin biopsy (an evidence-based review). Report of the American Academy of Neurology, American Association of Neuromuscular and Electrodiagnostic Medicine, and American Academy of Physical Medicine and Rehabilitation. Neurology 72:177–184. doi:10.1212/01.wnl.0000336345.70511.0f
Lauria G, Hsieh ST, Johansson O et al (2010) European Federation of Neurological Societies/Peripheral Nerve Society Guideline on the use of skin biopsy in the diagnosis of small fiber neuropathy. Report of a joint task force of the European Federation of Neurological Societies and the Peripheral Nerve Society. Eur J Neurol 17(903–912):e44–e49. doi:10.1111/j.1468-1331.2010.03023.x
Peters MJH, Bakkers M, Merkies ISJ et al (2013) Incidence and prevalence of small-fiber neuropathy: a survey in the Netherlands. Neurology 81:1356–1360. doi:10.1212/WNL.0b013e3182a8236e
Dimitropoulos G, Tahrani AA, Stevens MJ (2014) Cardiac autonomic neuropathy in patients with diabetes mellitus. World J Diabetes 5:17–39. doi:10.4239/wjd.v5.i1.17
Devigili G, Tugnoli V, Penza P et al (2008) The diagnostic criteria for small fibre neuropathy: from symptoms to neuropathology. Brain J Neurol 131:1912–1925. doi:10.1093/brain/awn093
14 Autonomic Testing Policy v001.pdf. https://www.aan.com/uploadedFiles/Website_Library_Assets/Documents/3.Practice_Management/1.Reimbursement/1.Billing_and_Coding/5.Coverage_Policies/14%20Autonomic%20Testing%20Policy%20v001.pdf Accessed 3/18/2017
Gibbons CH, Illigens BMW, Wang N, Freeman R (2009) Quantification of sweat gland innervation: a clinical-pathologic correlation. Neurology 72:1479–1486. doi:10.1212/WNL.0b013e3181a2e8b8
Ayoub H, Calvet JH, Lair V et al (2012) Electrochemical basis for EZSCAN/SUDOSCAN: a quick, simple, and non-invasive method to evaluate sudomotor dysfunctions. In: Chun JH (ed) Developments in electrochemistry. ISBN 978-953-51-0851-1. https://www.intechopen.com/books/developments-in-electrochemistry/electrochemical-basis-for-ezscansudoscan-a-quick-simple-and-non-invasive-method-to-evaluate-sudomot
Ayoub H, Lair V, Griveau S et al (2012) Electrochemical characterization of stainless steel as a new electrode material in a medical device for the diagnosis of sudomotor dysfunction. Electroanalysis 24:1324–1333. doi:10.1002/elan.201200058
Vinik AI, Nevoret M-L, Casellini C (2015) The new age of sudomotor function testing: a sensitive and specific biomarker for diagnosis, estimation of severity, monitoring progression, and regression in response to intervention. Front Endocrinol 6:94. doi:10.3389/fendo.2015.00094
Hubert D, Brunswick P, Calvet J-H et al (2011) Abnormal electrochemical skin conductance in cystic fibrosis. J Cyst Fibros Off J Eur Cyst Fibros Soc 10:15–20. doi:10.1016/j.jcf.2010.09.002
Shibasaki M, Crandall CG (2010) Mechanisms and controllers of eccrine sweating in humans. Front Biosci 2:685–696
García Villar C (2011) Evidence-based radiology for diagnostic imaging: what it is and how to practice it. Radiologia 53:326–334. doi:10.1016/j.rx.2011.02.009
Shamseer L, Moher D, Clarke M et al (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ 349:g7647
Leboulanger B, Guy RH, Delgado-Charro MB (2004) Reverse iontophoresis for non-invasive transdermal monitoring. Physiol Meas 25:R35–R50
Chizmadzhev YA, Indenbom AV, Kuzmin PI et al (1998) Electrical properties of skin at moderate voltages: contribution of appendageal macropores. Biophys J 74:843–856. doi:10.1016/S0006-3495(98)74008-1
Brunswick P, Mayaudon H, Albin V et al (2007) Use of Ni electrodes chronoamperometry for improved diagnostics of diabetes and cardiac diseases. Conf Proc Annu Int Conf IEEE Eng Med Biol Soc IEEE Eng Med Biol Soc Annu Conf 2007:4544–4547. doi:10.1109/IEMBS.2007.4353350
Vinik AI, Smith AG, Singleton JR et al (2016) Normative values for electrochemical skin conductances and impact of ethnicity on quantitative assessment of sudomotor function. Diabetes Technol Ther 18:391–398. doi:10.1089/dia.2015.0396
Hubert D, Brunswick P, Calvet J-H et al (2011) Abnormal electrochemical skin conductance in cystic fibrosis. J Cyst Fibros Off J Eur Cyst Fibros Soc 10:15–20. doi:10.1016/j.jcf.2010.09.002
Zhu L, Zhao X, Zeng P et al (2016) Study on autonomic dysfunction and metabolic syndrome in Chinese patients. J Diabetes Investig 7:901–907. doi:10.1111/jdi.12524
Freedman BI, Bowden DW, Smith SC et al (2014) Relationships between electrochemical skin conductance and kidney disease in type 2 diabetes. J Diabetes Complic 28:56–60. doi:10.1016/j.jdiacomp.2013.09.006
Freedman BI, Smith SC, Bagwell BM et al (2015) Electrochemical skin conductance in diabetic kidney disease. Am J Nephrol 41:438–447. doi:10.1159/000437342
Calvet JH, Dupin J, Winiecki H, Schwarz PEH (2013) Assessment of small fiber neuropathy through a quick, simple and non invasive method in a German diabetes outpatient clinic. Exp Clin Endocrinol Diabetes Off J Ger Soc Endocrinol Ger Diabetes Assoc 121:80–83. doi:10.1055/s-0032-1323777
Luk AOY, Fu W-C, Li X et al (2015) The clinical utility of SUDOSCAN in chronic kidney disease in Chinese patients with type 2 diabetes. PLoS One 10:e0134981. doi:10.1371/journal.pone.0134981
Chahal S, Vohra K, Syngle A (2017) Association of sudomotor function with peripheral artery disease in type 2 diabetes. Neurol Sci Off J Ital Neurol Soc Ital Soc Clin Neurophysiol 38:151–156. doi:10.1007/s10072-016-2742-3
Mayaudon H, Miloche P-O, Bauduceau B (2010) A new simple method for assessing sudomotor function: relevance in type 2 diabetes. Diabetes Metab 36:450–454. doi:10.1016/j.diabet.2010.05.004
Ramachandran A, Moses A, Shetty S et al (2010) A new non-invasive technology to screen for dysglycaemia including diabetes. Diabetes Res Clin Pract 88:302–306. doi:10.1016/j.diabres.2010.01.023
Chen X, Chen L, Ding R et al (2015) A preliminary investigation of EZSCAN™ screening for impaired glucose tolerance and diabetes in a patient population. Exp Ther Med 9:1688–1694. doi:10.3892/etm.2015.2358
Casellini CM, Parson HK, Richardson MS et al (2013) Sudoscan, a noninvasive tool for detecting diabetic small fiber neuropathy and autonomic dysfunction. Diabetes Technol Ther 15:948–953. doi:10.1089/dia.2013.0129
Mao F, Liu S, Qiao X et al (2016) Sudoscan is an effective screening method for asymptomatic diabetic neuropathy in Chinese type 2 diabetes mellitus patients. J Diabetes Investig. doi:10.1111/jdi.12575
Selvarajah D, Cash T, Davies J et al (2015) SUDOSCAN: a simple, rapid, and objective method with potential for screening for diabetic peripheral neuropathy. PLoS One 10:e0138224. doi:10.1371/journal.pone.0138224
England JD, Gronseth GS, Franklin G et al (2005) Distal symmetric polyneuropathy: a definition for clinical research: report of the American Academy of Neurology, the American Association of Electrodiagnostic Medicine, and the American Academy of Physical Medicine and Rehabilitation. Neurology 64:199–207. doi:10.1212/01.WNL.0000149522.32823.EA
Sheshah E, Madanat A, Al-Greesheh F et al (2015) Electrochemical skin conductance to detect sudomotor dysfunction, peripheral neuropathy and the risk of foot ulceration among Saudi patients with diabetes mellitus. J Diabetes Metab Disord 15:29. doi:10.1186/s40200-016-0252-8
Yajnik CS, Kantikar VV, Pande AJ, Deslypere JP (2012) Quick and simple evaluation of sudomotor function for screening of diabetic neuropathy. ISRN Endocrinol 2012:103714. doi:10.5402/2012/103714
Yajnik CS, Kantikar V, Pande A et al (2013) Screening of cardiovascular autonomic neuropathy in patients with diabetes using non-invasive quick and simple assessment of sudomotor function. Diabetes Metab 39:126–131. doi:10.1016/j.diabet.2012.09.004
Ang L, Jaiswal M, Callaghan B et al (2017) Sudomotor dysfunction as a measure of small fiber neuropathy in type 1 diabetes. Auton Neurosci Basic Clin. doi:10.1016/j.autneu.2017.03.001
Smith AG, Lessard M, Reyna S et al (2014) The diagnostic utility of Sudoscan for distal symmetric peripheral neuropathy. J Diabetes Complic 28:511–516. doi:10.1016/j.jdiacomp.2014.02.013
Novak P (2016) Electrochemical skin conductance correlates with skin nerve fiber density. Front Aging Neurosci 8:199. doi:10.3389/fnagi.2016.00199
Castro J, Miranda B, Castro I et al (2016) The diagnostic accuracy of Sudoscan in transthyretin familial amyloid polyneuropathy. Clin Neurophysiol Off J Int Fed Clin Neurophysiol 127:2222–2227. doi:10.1016/j.clinph.2016.02.013
Denier C, Ducot B, Husson H et al (2007) A brief compound test for assessment of autonomic and sensory-motor dysfunction in familial amyloid polyneuropathy. J Neurol 254:1684–1688. doi:10.1007/s00415-007-0617-5
Syngle A, Verma I, Krishan P et al (2015) Disease-modifying anti-rheumatic drugs improve autonomic neuropathy in arthritis: DIANA study. Clin Rheumatol 34:1233–1241. doi:10.1007/s10067-014-2716-x
Casellini CM, Parson HK, Hodges K et al (2016) Bariatric surgery restores cardiac and sudomotor autonomic C-Fiber dysfunction towards normal in obese subjects with type 2 diabetes. PLoS One 11:e0154211. doi:10.1371/journal.pone.0154211
Smith AG, Lessard M, Reyna S et al (2014) The diagnostic utility of Sudoscan for distal symmetric peripheral neuropathy. J Diabetes Complic 28:511–516. doi:10.1016/j.jdiacomp.2014.02.013
Casellini CM, Parson HK, Richardson MS et al (2013) Sudoscan, a noninvasive tool for detecting diabetic small fiber neuropathy and autonomic dysfunction. Diabetes Technol Ther 15:948–953. doi:10.1089/dia.2013.0129
Sato K, Ohtsuyama M, Samman G (1991) Eccrine sweat gland disorders. J Am Acad Dermatol 24:1010–1014
Brunswick P, Bocquet N (2014) United States Patent: 8918170—Electrophysiological analysis system
Vitale GI, Quatrale RP, Giles PJ, Birnbaum JE (1986) Electrical field stimulation of isolated primate sweat glands. Br J Dermatol 115:39–47
Sommer P, Kluschina O, Schley M et al (2011) Electrically induced quantitative sudomotor axon reflex test in human volunteers. Auton Neurosci Basic Clin 159:111–116. doi:10.1016/j.autneu.2010.09.004
Brinton M, Chung JL, Kossler A et al (2016) Electronic enhancement of tear secretion. J Neural Eng 13:016006. doi:10.1088/1741-2560/13/1/016006
Zonana J (1993) Hypohidrotic (anhidrotic) ectodermal dysplasia: molecular genetic research and its clinical applications. Semin Dermatol 12:241–246
Norcliffe-Kaufmann L, Katz SD, Axelrod F, Kaufmann H (2015) Norepinephrine deficiency with normal blood pressure control in congenital insensitivity to pain with anhidrosis. Ann Neurol 77:743–752. doi:10.1002/ana.24377
Low PA, Caskey PE, Tuck RR et al (1983) Quantitative sudomotor axon reflex test in normal and neuropathic subjects. Ann Neurol 14:573–580. doi:10.1002/ana.410140513
Wang N, Gibbons CH, Freeman R (2011) Novel immunohistochemical techniques using discrete signal amplification systems for human cutaneous peripheral nerve fiber imaging. J Histochem Cytochem 59:382–390. doi:10.1369/0022155410396931
Henderson LA, Stathis A, James C et al (2012) Real-time imaging of cortical areas involved in the generation of increases in skin sympathetic nerve activity when viewing emotionally charged images. NeuroImage 62:30–40. doi:10.1016/j.neuroimage.2012.04.049
Asahina M, Poudel A, Hirano S (2015) Sweating on the palm and sole: physiological and clinical relevance. Clin Auton Res Off J Clin Auton Res Soc 25:153–159. doi:10.1007/s10286-015-0282-1
Claus D, Schondorf R (1999) Sympathetic skin response. The International Federation of Clinical Neurophysiology. Electroencephalogr Clin Neurophysiol Suppl 52:277–282
Vetrugno R, Liguori R, Cortelli P, Montagna P (2003) Sympathetic skin response: basic mechanisms and clinical applications. Clin Auton Res Off J Clin Auton Res Soc 13:256–270. doi:10.1007/s10286-003-0107-5
Shimada H, Kihara M, Kosaka S et al (2001) Comparison of SSR and QSART in early diabetic neuropathy–the value of length-dependent pattern in QSART. Auton Neurosci Basic Clin 92:72–75. doi:10.1016/S1566-0702(01)00287-9
Illigens BMW, Gibbons CH (2009) Sweat testing to evaluate autonomic function. Clin Auton Res Off J Clin Auton Res Soc 19:79–87. doi:10.1007/s10286-008-0506-8
Lefaucheur J-P, Wahab A, Planté-Bordeneuve V et al (2015) Diagnosis of small fiber neuropathy: a comparative study of five neurophysiological tests. Neurophysiol Clin Clin Neurophysiol 45:445–455. doi:10.1016/j.neucli.2015.09.012
Syngle A, Verma I, Krishan P et al (2015) Disease-modifying anti-rheumatic drugs improve autonomic neuropathy in arthritis: DIANA study. Clin Rheumatol 34:1233–1241. doi:10.1007/s10067-014-2716-x
Obici L, Kuks JB, Buades J et al (2016) Recommendations for presymptomatic genetic testing and management of individuals at risk for hereditary transthyretin amyloidosis. Curr Opin Neurol 29(Suppl 1):S27–S35. doi:10.1097/WCO.0000000000000290
Acknowledgements
The author thanks Phillipe Brunswick, Impeto Medical, for clarification of principles for ESC measurements.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The Sudoscan® device was donated to the author by Impeto Medical. Dr. Novak has received funding support from Impeto Medical.
Rights and permissions
About this article
Cite this article
Novak, P. Electrochemical skin conductance: a systematic review. Clin Auton Res 29, 17–29 (2019). https://doi.org/10.1007/s10286-017-0467-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10286-017-0467-x