Abstract
Mineralization defects like amelogenesis imperfecta are often of hereditary origin. This article reviews the diagnostic findings and summarizes the suggested treatment approaches. Currently, there are no defined therapy recommendations available for patients suffering from amelogenesis imperfecta. The mentioned therapies are more or less equal but no comprehensive therapy recommendation is evident. When treating patients suffering from amelogenesis imperfecta, a comprehensive therapy of almost every dental discipline has to be considered. The earlier the diagnosis of amelogenesis imperfecta is confirmed, the better the outcome is. Optimal treatment approaches consist of early diagnosis and treatment approach and frequent dental recall appointments to prevent progressive occlusal wear or early destruction by caries. Full-mouth prosthetic treatment seems to be the best treatment option.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mineralization defects of dental hard tissues may affect dentin and/or enamel. Diseases with a low number of altered teeth like molar-incisor-hypomineralization (MIH) have a prevalence ranging from 2.8 to 25 % depending on country and region [1]. The aetiology of MIH is still unclear, but some specific diagnosis are often evident-like deprivation of oxygen at birth or during post-natal period, chronic obstructive pulmonary disorder, the presence of dioxins or polichlorinated biphenyl (PCB) in the mother’s milk and/or infectious diseases during young childhood and lack of mineral fixing [1]. A characteristic finding associated with MIH is the alteration of the enamel ranging from a diffuse opacity to hypoplasia and/or hypomineralization of enamel and dentin. Usually, the most often affected teeth are the first permanent molars and permanent incisors [1]. Diseases with an ubiquitous alteration of the teeth are in general of genetic, i.e. hereditary origin. Hereditary alterations of dental hard tissues are specific diseases like amelogenesis imperfecta (AI), dentinogenesis imperfecta (DI) and dentin dysplasia (DD) [2]. Additionally, dental alterations could be accompanied as symptoms of syndromes [3] like osteogenesis imperfecta [4] or Tricho-odonto-onycho-dermal syndrome [3, 5]. When considering dental mineralization defects besides enamel, also solitary affection of dentin is possible. Mineralization defects of dental hard tissues have different characteristics. In general, hypomineralization, hypoplasia/aplasia, malformation and discolouration are possible.
The types of alterations may reach from mild hypomineralization with moderate discolouration caused by mineralization defects that alter the optical characteristics to hypoplastic alterations [6] with excessive loss of dental hard tissues, especially of enamel. In the following, the different defects of dental hard tissues are presented briefly.
Dental hard tissue defects
Formation of dentin and enamel
The formation of highly mineralized dental hard tissues like enamel with its highly organized hydroxyapatite crystals is a rigorously controlled process [6]. The process of mineralization is an epithelial-derived process, which is entirely guided under genetic instruction [7]. Different hereditary and non-hereditary alterations of dental hard tissues have been described.
Alterations of the mineralization sequences lead to similar mineralization errors in hard tissues, i.e. osteogenic defects can also cause defects in dentin [8]. In contrast to dentin, bone underlies a steady balance between resorption and apposition [8]. The formation of dentin starts in utero and does not show continuous resorption like bone, so dentin displays a continuous growing process [8] that occurs in intervals and an incremental manner [9]. During the thickening of dentin, collagenous fibres extend from the process of the odondoblasts to the dentino-enamel-junction and when mineralization occurs the process retreats and further predentin is formed [9]. In general, mineralization begins when a wide zone of predentin has been elaborated and mineralization cores spread around in the collagenous fibres [9]. Later, ageing of dentin is characterized by an increased sclerosing, which can lead to obliteration of the dentinal tubules [10]. However, the formation of enamel is different compared to the formation of bone and dentin. Enamel is a tissue that consists of a high grade of crystallites and does not contain fibres or proteins. Amelogenesis as well as dentinogenesis is genetically controlled [7]. Differentiated ameloblasts secrete right before the basal lamina disintegrates small amounts of enamel proteins [7]. When the basal lamina dissolves, the mineralization of dentin starts and the superficial collagen fibrils associate with the apical surface of the ameloblasts [7]. In the beginning of the mineralization, secretory phase enamel is only aprismatic [7] and, at that time, a new cell extension of ameloblasts is formed for secretion of prismatic enamel [7].
Molar-incisor-hypomineralization (MIH)
The term MIH has been suggested by Weerheijm et al. [11]. MIH is an enamel developmental defect of systemic origin of the first permanent molars, frequently associated with similar defects of the first incisors [11] (Fig. 1). Histologically, demarcated opacities and extensive hypomineralized enamel [12] and a distinct border demarcating the altered enamel from enamel of normal formation have been described [12]. The prevalence of MIH ranges from 2.8 to 25 % [1, 13–15].
a Molar-incisor-hypomineralization upper occlusal view. Notably, the malformed and discoloured enamel of the tooth 16 and the yellow to whitish circumscribed opacities of the upper first incisors. b Molar-incisor-hypomineralization lower occlusal view. Noteworthy the brown discoloured enamel of tooth 36 with an extensive loss of enamel on the buccal, occlusal and mesial aspect
Amelogenesis imperfecta (AI)
Amelogenesis imperfecta (AI) is a genetically determined hereditary disease. The first classification was published in 1945 [16]. The authors divided AI into 2 groups: hypoplastic and hypocalcified [16]. During the following years, the classification was modified as new diagnostics such as gene sequencing emerged. A further classification by Witkop [17] divided the alterations of enamel into four main groups [17].
Classification of AI by Witkop [17]:
- Type I::
-
hypoplastic
- IA::
-
hypoplastic, pitted autosomal dominant.
- IB::
-
hypoplastic, local autosomal dominant.
- IC::
-
hypoplastic, local autosomal recessive.
- ID::
-
hypoplastic, smooth autosomal dominant.
- IE::
-
hypoplastic, smooth X-linked dominant.
- IF::
-
hypoplastic, rough autosomal dominant.
- IG::
-
enamel agenesis, autosomal recessive.
- Type II::
-
hypomaturation
- IIA::
-
hypomaturation, pigmented autosomal recessive.
- IIB:
-
hypomaturation, X-linked recessive.
- IID::
-
snow-capped teeth, autosomal dominant?
- Type III::
-
hypocalcified autosomal genetic trait
- IIIA::
-
autosomal dominant.
- IIIB::
-
autosomal recessive.
- Type IV::
-
mixed hypomaturation and hypoplastic in combination with taurodontism
- IVA::
-
hypomaturation–hypoplastic with taurodontism, autosomal dominant.
- IVB::
-
hypoplastic–hypomaturation with taurodontism, autosomal dominant.
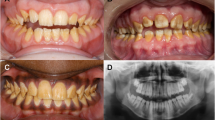
The subtypes of the hypoplastic type (HP) summarize teeth with different rough dental surfaces [18, 19] caused by defects in enamel formation and including also enamel agenesis [3, 17] (Fig. 2). All alterations are possible in combination with different genetic traits. A normal radiographic contrast between enamel and dentin is evident [17], while the thickness of enamel is markedly reduced [17, 20–22] (Fig. 2c).
a Hypoplastic amelogenesis imperfecta upper occlusal view. The hypoplastic enamel is characterized by a rough and irregular pitted surface. b Hypoplastic amelogenesis imperfecta lower occlusal view. Noticeable are the vertical grooves on the lower anterior incisors and the reduced enamel layer, especially on the buccal an occlusal aspects of the teeth. c Orthopantomogram of a patient with hypoplastic Amelogenesis imperfecta showing the representative thin enamel layer
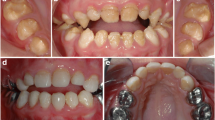
The hypomaturated type (HM) summarizes alterations of enamel with normal thickness but softer consistency [3]. The altered enamel has a mottled, discoloured appearance [23] (Fig. 3). Hypomaturated enamel can be easily chipped away [17]. The radiopacity of this hypomaturated enamel is similar to that of dentin [17] (Fig. 3c). An autosomal and an x-linked genetic trait have been described [17].
a Hypomaturated amelogenesis imperfecta upper occlusal view. Notice the brown yellow discoloured opaque enamel and the wear of the cusps of the molars and premolars. b Hypomaturated amelogenesis imperfecta lower occlusal view. The teeth show the distinguishing yellow opaque discolouration and marked wear of the cusps. c Orthopantomogram of a patient with hypomaturated amelogensis imperfecta revealing an almost equal radioopacity of enamel and dentin
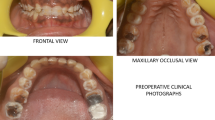
The third type distinguishes the autosomal trait from hypocalcified (HC) alterations of enamel [17]. The malformed enamel is of normal thickness but discoloured orange-yellow [3] and consists of a poorly calcified matrix (Fig. 4), resulting in a lower radiopacity compared to dentin [17]. The last type is characterized by an alteration of enamel in combination with taurodontism [17]. Histologically, the malformed enamel shows a mixed appearance of hypomaturated and hypocalcified enamel with a radiographically slightly lower translucency than dentin [17] (Fig. 5). The hypoplastic type is the most frequent form of AI (61.2 %) followed by the hypomaturated type (32.2 %) and both the hypocalcified and mixed hypomaturated/hypoplastic type (3.2 %) [24].
a Hypocalcified amelogenesis imperfecta upper occlusal view. Notice the yellowish discoloured enamel and the fracture of enamel on the palatal aspect of the first permanent molars. b Hypocalcified amelogenesis imperfecta (lower occlusal view) with the distinctive yellowish discoloured enamel and occlusal wear on the buccal aspects of the lower first molars
The classification of alterations which can be found in an autosomal recessive genomic trait has been revisited by Nusier et al. [25]. The authors found some recessive forms of AI which cannot be classified according to the classification of Witkop [17].
Dentinogenesis imperfecta (DI)
The primary and the permanent dentition are affected by each type of DI. Ubiquitous obliteration, pulp stones and thistle tube like pulp configuration are evident [4]. Dentinogenesis Imperfecta can be seen in patients with osteogenesis imperfecta and also solitary as an isolated disorder [4]. Dentinogenesis Imperfecta was first described in 1973 [4] and three different types have been suggested:
Type I is characterized by teeth showing an amber translucency and the covering enamel is often chipped away (Fig. 7). Radiographically, short roots in both dentitions and obliteration of the pulp chamber (Fig. 6) even shortly after eruption, possibly due to hypertrophy of the “mantle” dentin, are evident. Although DI Type I affects both dentitions, the alterations are more severe in the primary one. In contrast to the other types of DI, Type I only occurs in combination with Osteogenesis Imperfecta in an autosomal dominant trait [4].
Type II is characterized by opalescent dentin and the incidence ranges from 1:6000 to 1:8000 [26] (Fig. 8). Type II shows more clinical variabilities as type I and both dentitions are affected equally [4].
Type III shows multiple pulp exposures in the deciduous dentition. Shape and colouration of teeth seem to be more variable as in types I and II (Fig. 7) [4].
Search of the literature
The aim of this article was to review the diagnostic findings of AI and to summarize the currently suggested treatment approaches of these hereditary mineralization defects.
The PubMed and Web of Science databases were searched for full papers and reviews using the following MeSH terms: “AI, mineralization defects”. In addition, the following keywords were also used: “dental, dentin, dysplasia, treatment, hypoplasia, hypomaturation”. After an initial screening of the abstracts, relevant full-length peer-reviewed articles were selected. The reference lists of the articles were also screened for additional relevant articles.
In the attempt to include the most recent publications, a hand search of articles published online, “in-press” and “early view” was performed for the Journal of Dental Research, Journal of Prosthetic Dentistry and Quintessence International. The date of the last search was June 20th 2016. The reference lists of those articles included were checked for additional articles of relevance.
Diagnosis
In general diagnosis of mineralization defects, especially AI in the primary as well as in the permanent dentition is easy due to characteristic clinical findings [6, 27, 28]. Every hereditary disorder of teeth affects both dentitions due to the genetic origin of the formation of dental hard tissues. However, the severity can differ markedly between the primary and the permanent dentition as for instance in DI type I [4]. AI is characterized by an alteration of enamel in both dentitions [6, 28]. Similar but only localized alterations of incisors and first molars are evident in MIH [1, 11–15]. In contrast to AI, the aetiology of MIH in the permanent dentition is not fully understood [11].
Besides malformations of enamel also hereditary alterations of dentin are possible, but clinically these diseases show typical findings such as chipping of enamel in DI [4]. Radiographic findings are also helpful to differentiate AI, DI and DD. In DD, often obliterations of the pulp space are evident [4, 17]. Clinically, the primary dentition of DD type II shows a similar appearance as teeth affected by DI type I [4, 17].
Regular enamel has a higher radiopacity than dentin [29] and, therefore, the different diseases can be differentiated radiologically when comparing the opacities of enamel and dentin. Enamel and dentin of subjects with hypomaturated AI have an almost equal radiopacity [17, 30, 31], while in hypocalcified forms of AI the enamel has a lower radiopacity than dentin [32, 33]. Both in hypomaturated and hypocalcified forms of AI, chipping of enamel has been reported as a typical finding [34].
Besides the clinical examination, genetic investigations are also beneficial for diagnosis. Due to many known single nucleotide polymorphisms (SNP) of the different hereditary disorders of dental hard tissues, a more precise classification by detection of the responsible SNP is possible. SNP are known in different genes like FAM83H, KLK4, MMP20, WDR72, ENAM, AMELX, AMBN, DLX3 [3, 6, 7, 20, 35–38]. These are associated with the formation of enamel and can cause different clinical manifestations.
Clinical findings
The main complaints of patients who are affected by AI are increased sensibility to hot and cold [3, 18, 21–24, 27, 30, 32, 34, 36, 39–62], reduced aesthetic appearance and discolouration of mandibular and maxillary anterior teeth [3, 22, 24, 27, 30–32, 34, 36, 39, 41–44, 46, 48–58, 60–74] and masticatory problems [24, 30–32, 34, 43, 44, 46, 50, 51, 53–55, 57, 63, 66, 73–76]. Parekh et al. [68] investigated in 2014 the impact of the alterations of AI on the quality of life of patients (61 patients, response rate 61 %) suffering from AI. For about 90 % of all patients, the main complaint was the discolouration of their teeth and about 77 % asked for an improvement of their smile. For nearly the same percentage (74 %), reduction of sensitivity was the most important reason to seek for dental treatment [68], while improvement of the tooth size was important for 60 % of all patients [68].
Regarding the intraoral examination, several characteristic findings have been reported. Often wear of enamel with exposed dentin areas are evident especially at the occlusal aspects of posterior teeth [22, 30–32, 39, 41, 47, 50, 52, 54, 55, 57–59, 61, 62, 65, 66, 77–82]. Due to loss or hypoplastic enamel, the lack of proximal contacts is typical [50, 54, 57, 60, 63–65, 67, 77, 79, 83–85]. A further consequence of the loss/lack of enamel is the loss of the vertical dimension [18, 19, 22, 32, 39, 41, 42, 47, 49, 50, 54, 55, 58, 60, 64, 66, 67, 73, 80, 83, 86] and functional problems like decreased masticatory efficiency [20, 31, 32, 43, 44, 46, 50, 51, 54, 55, 57, 73, 76]. Besides alterations of dental hard tissues, also craniofacial alterations of patients with AI have been reported, like anterior open bite (AOB) [3], posterior open bite (POB), negative overjet and altered vertical jaw relationship [20]. Some authors described a greater number of unerupted teeth [63, 74].
Treatment concepts
Dahash et al. stated in 2013 [87] that no randomised controlled studies to survey the outcome of dental treatment in patients with AI are available up to now. Thus, the currently available evidence regarding the treatment approaches of patients with AI is very limited and levels of evidence ranging from 1a [systematic reviews (with homogeneity) of randomized controlled trials] to 2a [systematic reviews (with homogeneity) of cohort studies], as proposed by the Oxford Centre for Evidence-based Medicine [88], are not available yet.
In 2015, Lundgren et al. [85] published a split mouth study on the long-term outcome of full-crown restoration in patients with AI using different crown materials [85]. Besides this clinical trial, only very few studies comparing different treatment procedures and assessing treatment outcome in subjects with AI have been published [24, 48, 85, 89], while several other studies only focused on the description of clinical findings [68, 90–93] or described genetic surveys [37]. The majority of available publications are mere case reports [18, 19, 21–23, 27, 30–34, 39, 42–44, 46, 47, 49–67, 69–71, 74–81, 83, 94–97]. Besides the aforementioned typical clinical findings, these authors mentioned a relation between Amelogenesis and anterior open bite malocclusion [3, 24, 27, 35–37, 42, 44, 51, 65, 71, 75, 83, 93].
Furthermore, the surface of the affected teeth can be rougher than normal and the surface hardness is reduced [6]. Typically, the still existing enamel shows marked discolouration [6, 17]. In subjects with hypoplastic AI, the characteristic appearance of the affected teeth is a rough pitted surface with grooves and also a reduced thickness of the enamel layer due to deficiencies in building of the enamel [6, 17, 28]. In the case of hypocalcified AI, the hardness of enamel is reduced [82, 98] but the enamel layer is of normal thickness and the enamel is often yellow-brownish discoloured [3, 6, 17, 28, 82]. The enamel layer in subjects with hypomaturated forms of AI is of normal thickness but the enamel is slightly softer and is often chipped away [3, 17].
Adequate treatment concepts of patients with AI must be based on an interdisciplinary approach [48].
Restorative treatment
In general, the longevity of dental restorations in patients with AI is considerably reduced and this correlates with the severity of AI [89]. In comparison with a control group of patients with unaltered enamel (80 %), the survival rate after 5 years of coronal restoration in patients with AI was only 50 % [89]. The rate of replacement of defective restorations was about 2.5 times higher than in unaffected patients [89]. Comparison of the longevity of restorations with regard to the different types of AI revealed that survival rates were higher in patients with hypoplastic types of AI than in patients with hypomaturated or hypocalcified types of AI [89].
The main reason of failure of restorations in patients with AI was fracture of the restoration or of the tooth, while in the control group without AI the main reason of restorations failure was recurrent caries [89]. Compared with a control group (27 %), this risk of either fracture of the restoration or fracture of hard tissue was substantially increased to 60–69 % [89]. This finding corresponds with the failure pattern of patients with hypomaturated AI (21 %) [89]. When comparing hypomaturated AI with hypoplastic AI, it was evident that the rate of recurrent caries was lower in the hypoplastic group [89].
The reduced longevity of adhesive restorations can be explained by the reduced amount and quality of the existing enamel [89]. The hypoplastic type of AI is in contrast to the other types of AI characterized by a reduced quantity of enamel [3, 64], although the quality of enamel is almost identical compared with unaltered enamel [64, 89]. Therefore, the reduced longevity is probably due to the inferior etching pattern of the enamel resulting in reduced bond strengths and so retention of the restoration in mainly provided by bonding of the resin to the dentin surface which is less reliable [6, 17, 99], especially in patients with hypoplastic AI [89]. Additionally, nanoleakage [100] of widespread dentin surfaces will lead to an early failure of the adhesive bond. The shear bond strength of resin to dentin is significantly lower compared to enamel [99, 101, 102].
Only few authors recommended the use of direct composite restorations in patients with AI [23, 27, 30, 40, 42, 48, 55, 62, 67, 69, 70, 89, 103]. Treatment with direct composite restorations was mostly performed as a temporary therapy to gain some time and to perform the definitive restoration in an older stage of life [23, 24, 27, 39, 42, 48, 55, 61, 66, 70, 83, 89]. Few authors performed complete direct composite restorations due to financial reasons claimed by the patient [62, 69].
When performing composite restorations, differences in longevity depending on the severity and type of AI have been reported [24, 89]. Especially direct composite restorations in patients with hypocalcified AI have a higher failure rate than in patients with other types of AI [24]. Direct composite restorations in patients with hypomaturated and hypocalcified AI are solely of temporary character [104] due to a markedly increased risk of breakdown of altered enamel [6]. Koruyucu et al. suggested [24] that a higher amount of proteins in the enamel may be responsible for the reduced bonding strength. Therefore, Sönmez et al. [105] tried to improve the adhesive bond strength by deproteination of the hypocalcified enamel and they reported a slight improvement of the marginal integrity of direct composite restorations in a split mouth study (left/right) [105].
Even root canal treatment was required in some cases [49, 50, 58, 66, 67, 74, 81, 95] and severe cases have been published reporting that every tooth of the dentition required root canal treatment [32, 47, 77].
Prosthetic treatment
Besides the clinically relevant problem of structural alterations of dentin, the age of the patients has to be taken into consideration for adequate treatment. Today AI is usually diagnosed in an earlier stage; thus, treatment of younger patients suffering from AI requires proper concepts [19–21, 23, 24, 27, 34, 36, 39, 41, 42, 45, 48, 49, 51, 55, 56, 60–62, 67, 68, 70, 78, 83, 85, 87, 89]. When considering the treatment of younger patients, the extension of the pulp chamber with prominent pulp horns requires attention [62, 106]. Hence, the increased risk of iatrogenic exposure of the pulp chamber during preparation and irreversible irritation of the pulp tissue due to the preparation trauma must be anticipated. Therefore, radiographs (e.g. bitewings) are mandatory for adequate assessment of the extension of the pulp chamber prior to preparation. Preparation of the teeth should be as minimal invasive as clinically possible.
Extensive coronal restorations of anterior and posterior teeth are often indicated [18, 19, 22, 24, 32, 34–36, 43, 44, 46, 48–54, 57–59, 64–66, 71–75, 80, 81, 83, 85, 86, 93, 95, 97, 103]. Besides full metal crowns especially in the posterior region [19, 22, 36, 41, 49, 58], also porcelain-fused-to-metal crowns and bridges [18, 32, 43, 44, 46, 48–50, 52–54, 57, 58, 64, 66, 71, 73, 74, 97] and recently all-ceramic crowns have been shown to allow sufficient long-term restoration of the affected teeth [18, 44, 51, 52, 65, 67, 72, 75, 81, 83, 85, 89, 95]. Depending on the age of the patient, also other materials like composite crowns [34, 41, 42, 61, 84, 103], composite veneers [23, 39, 48, 67] and in younger patients even stainless steel crowns [3, 21, 27, 34, 41, 55, 86] may be beneficial.
In complex cases of definitive full-mouth rehabilitation, a diagnostic wax-up is required, as due to the vertical loss the vertical dimension of occlusion has to be restored [18, 22, 47, 51, 57, 59, 65, 66, 71–73, 75, 80, 83, 94]. However, some authors reported the rise of the vertical dimension of occlusion without a wax-up [30, 32, 49, 54, 58, 60, 103] and splints [58, 60, 64] and instead direct composite restorations [103] or provisional restorations [30, 47, 49] were used to assess the increased vertical dimension. Due to the loss of the vertical dimension and the interocclusal relationship, the centric relation of the temporomandibular joint was taken as reference in most reports of full-mouth rehabilitation [18, 19, 22, 30, 46, 51, 52, 54, 65, 66, 75, 80, 93].
In advance of the prosthetic treatment, the short clinical crowns sometimes require to perform crown lengthening or to perform gingivectomy or gingivoplasty [30, 74] to ensure adequate preparation for fixed crowns and bridges [32, 50, 53, 54, 57, 59, 62, 71–73].
Periodontal treatment
Besides coronal restoration and reconstruction of the affected teeth, it is of outmost importance to establish an intensive care and a proper oral hygiene to prevent further loss of dental hard tissues and restorations by caries [24, 30, 47, 48, 50]. Due to the increased sensitivity of teeth affected by AI, the patients often perform poor oral hygiene [3, 19, 23, 27, 31, 32, 46, 48, 49, 51, 55, 57, 59, 61, 64, 66, 71, 76, 107] resulting in generalized gingivitis. Persistent gingivitis [23, 24, 48, 50, 51, 55, 59, 61, 66, 71, 83, 85, 103] and marginal parodontitis [61, 71] are also evident due to the increased accumulation of plaque [3, 59] because of the rough tooth surfaces, especially in the hypoplastic forms of AI [50, 51, 82]. Therefore, improvement of oral hygiene and reduction of the degree of gingivitis are essential to ensure optimal longevity of restorations and to improve treatment outcome [22, 27, 34–36, 47, 49, 55, 71, 85, 103]. Periodontal treatment usually consists of scaling and root planning [23, 40, 50, 53, 64, 67, 71, 73, 76, 103].
Orthodontic treatment
About 22 % [24] to 64 % [83] of all patients with AI show AOB [24, 83]. Especially patients with hypoplastic and hypocalcified forms of AI have a higher incidence for AOB [24, 108, 109]. Rowley et al. [109] reported that vertical dysgnathia occurred frequently in association with AI, most often in association with the hypocalcified type of AI. These alterations of the vertical dimension were diagnosed more often in female than in male subjects [35, 109]. Due to the tenderness of teeth to hot and cold caused by the lack of enamel and increased tooth wear [18, 20, 32, 41, 42, 49, 50, 54, 55, 58, 60, 64, 66, 73, 80, 83, 86], the malposition of the tongue due to the altered vertical relation may inhibit the compensation of the vertical malocclusion by the vertical alveolar growth so that the AOB can persist [35, 93, 109]. Besides differences in the vertical growth pattern, also Class II and III malocclusions have been reported [43, 49, 52, 54, 55, 57, 63, 65, 71, 95].
Thus, the most severe orthodontic problem of patients with AI is the AOB [3, 35]. When treating the AOB, the high relapse rate has to be taken into consideration, so treatment by orthodontic surgery has to be performed depending of the severity of the existing craniofacial alterations [35, 51, 70, 94]. Other malocclusions besides the AOB can usually be corrected by regular orthodontic treatment [18, 24, 42, 43, 52, 54, 55, 60, 70, 72, 94].
Surgical treatment
Especially in subjects with anterior open bite orthodontic surgery may be necessary [34, 51, 54, 63, 83, 93, 94], often in combination with extraction of some teeth [21, 24, 34, 47, 50, 54, 57, 63, 97].
Discussion
Dental treatment of patients with AI is a challenge, requiring a detailed interdisciplinary treatment approach based on the correct diagnosis [48]. To differentiate the multiple forms of malformation of dental hard tissues, it is mandatory to elucidate if family members show equal alterations to assess if a hereditary malformation of dental hard tissues is evident [6, 17]. In this context, it is of importance to consider that normally hereditary malformations occur in both the primary and the permanent dentition and affect all existing teeth [6, 17].
When a suspected diagnosis of AI is confirmed, an early treatment has to be considered to prevent further destructions due to occlusal wear [18, 27, 49, 61, 78] or disturbance of the vertical growth [39, 41, 47]. Frequent appointments and a frequent recall are required to assess the gingival and periodontal status to obtain an optimal periodontal health [23, 24, 27, 34–36, 49, 83, 85] and to improve the individual oral hygiene to prevent destruction of the erupted teeth in both dentitions by carious lesions and periodontal disease [24, 27, 36, 49, 63, 71, 83, 85]. The reduced longevity of direct composite restorations especially in the hypomaturated and hypomineralized types of AI [89, 93] has to be considered when treatment is required in early childhood, often requiring recurring replacement of the restorations [89] until definitive prosthetic restorations can be performed [70, 83, 84, 89]. In the case of extensive destructions of primary teeth, stainless steel [27, 34, 36, 41, 55] or composite crowns [42, 103] are indicated to avoid loss of the vertical dimension [55, 86] which is evident in this state [42, 55, 86]. Besides timely renewal of failed adhesive restorations, only few further treatment procedures are necessary in childhood. However, orthodontic treatment is often required due to an anterior open bite [20, 23, 27, 34–37, 42, 44, 51, 63, 65, 70, 71, 75, 81, 83, 93, 94], as AOB is associated with AI in 22 % to up to 39 % of cases [108, 109]. Besides AOB, other mal-occlusions like cross-bite, Class I [18, 22, 65, 70, 75] or Class III occlusion [52, 54, 65, 71, 95], and constriction of the maxilla [18, 34, 42, 63] may require orthodontic treatment. Additionally, generalized proximal spacing has been reported [50, 60, 63, 65, 77, 79] due to the lack of enamel and the reduced thickness of enamel [17, 20–22]. AOB and loss of proximal contacts [50, 57, 65, 83] are often evident in patients with hypoplastic AI.
Following the completion of the skeletal growth, full-mouth rehabilitation is generally recommended [18, 19, 22, 24, 30, 32, 34, 43, 44, 46–54, 57–59, 64–66, 71–75, 80, 81, 83, 86, 93, 95, 97, 103]. Due to the aesthetic demands especially all-ceramic or porcelain-fused-to-metal restorations are best suited [18, 22, 24, 30, 32, 43, 44, 46–48, 50, 51, 53, 54, 57, 64–66, 71–75, 80, 81, 83, 85, 95, 97]. Full-crown restorations in patients with AI possess a nearly equal longevity as in patients with unaltered enamel [85]. When performing full-crown restorations, endodontic problems may arise [85] due to the lack of regular enamel in the hypoplastic type and the reduced hardness of enamel in hypomaturated and hypocalcified types resulting in excessive loss of hard tissue during preparation [98]. To date, no success rates of root canal treatment in patients with hereditary enamel alterations have been published. The risk of extensive loss of dental hard tissue during preparation is considerably increased in patients with hypoplastic AI due to the fact that the existing enamel layer is thinner than normal [17, 20–22]. Possible occlusal wear especially of the cusps has to be taken into consideration and, thus, complete coverage of the entire occlusal surface is usually required [22, 32, 39, 41, 47, 65, 66, 75, 77, 78, 80, 81]. For prosthetic treatment, the existing proximal spacing seems to be beneficial [85] as less hard tissues must be removed during preparation [85]. Newer, more aesthetic materials for full-crown restorations like lithium disilicate offer a further clinically relevant advantage as these materials require a reduced reduction of dental hard tissues during preparation. Lithium disilicate all-ceramic crowns require only a thickness of approximately 0.8 mm in the cervical region, and an occlusal reduction of only 1.5–2.0 mm is sufficient [110]. Physiologically, the thickness of the enamel layer reaches from nearly absent near the cemento-enamel-junction to about 1.5–2 mm in the region of the cusps and the incisal edge [111–114]. So in absence of enamel and the corresponding proximal spacing [50, 54, 57, 60, 63–65, 67, 77, 79, 83–85] and the loss of vertical dimension [18, 20, 22, 32, 39, 41, 42, 47, 49, 50, 54, 55, 58, 60, 64, 66, 67, 73, 80, 83, 86], these newer ceramic materials allow a sufficient preparation of the affected teeth without an increased risk of iatrogenic irritation of the pulp tissue [85]. In the case the proximal spacing in hypoplastic types of AI is not evident it has to be considered if an orthodontic pre-treatment to widen the interapproximal space may be indicated prior to prosthetic therapy.
Besides the strong recommendation of full-cover crowning of all affected teeth, the loss of the vertical dimension is another aspect that requires adequate treatment. In general, the protection of the temporomandibular joint during and after treatment is important [115] and, therefore, the determination of the centric position of the mandibular condyle in the temporomandibular joint is beneficial as a reference prior to full-mouth rehabilitation [115]. When the centric relation is determined, a testing of the new occlusion plane should be performed to prevent the patients from suffering pain caused by alterations in the temporomandibular joint due to the prosthetic therapy [116]. This can be performed by a full wax-up and simulation of the future definitive reconstruction using provisional crowns, direct composite fillings or splints [22, 39, 47, 49–51, 58, 59, 61, 66, 83, 94, 117]. Moreover, the application of non-removable therapeutic appliances for increase of the vertical dimension seems to be beneficial [116, 118]. The non-removable tooth shaped splint is a useful help to establish and test physiological mastication prior to prosthetic full-mouth rehabilitation.
Conclusion
In summary, AI is a disorder that should be diagnosed in an early stage [18, 27, 49, 61, 71, 78] to minimize the destructions caused by this mineralization disorder. Frequent dental appointments to optimize the gingival and periodontal status, to establish an optimal oral hygiene and to screen for progressive destructions are required [23, 35, 36, 49]. Especially for patients with hypoplastic forms of AI, orthodontic treatment to gain the best possible status for further prosthetic treatment is beneficial. As soon as the completion of the skeletal growth is finished, a prosthetic full-mouth reconstruction is indicated for all patients suffering from AI to prevent further attrition and destruction of the teeth and to avoid loss of the vertical dimension.
References
Mast P, Rodriguez Tapia MT, Daeniker L, Krejci I. Understanding MIH: definition, epidemiology, differential diagnosis and new treatment guidelines. Eur J Paediatr Dent. 2013;14:204–8.
Cabay R. An overview of molecular and genetic alterations in selected benign odontogenic disorders. Arch Pathol Lab Med. 2014;138:754–8.
Seow WK. Developmental defects of enamel and dentine: challenges for basic science research and clinical management. Aust Dent J. 2014;59(Suppl 1):143–54.
Shields ED, Bixler D, El-Kafraw AM. A proposed classification for heritable human dentine defects with a description of a new entity. Arch Oral Biol. 1973;18:543–53.
Al-Batayneh OB. Tricho-dento-osseous syndrome: diagnosis and dental management. Int J Dent. 2012. doi:10.1155/2012/514692.
Crawford PJM, Aldred M, Bloch-Zupan A. Amelogenesis imperfecta. Orphanet J Rare Dis. 2007;2:17.
Hu JCC, Chun Y-HP, Al Hazzazzi T, Simmer JP. Enamel formation and amelogenesis imperfecta. Cells Tissues Organs. 2007;186:78–85.
Vital SO, Gaucher C, Bardet C, Rowe PS, George A, Linglart A, Chaussain C. Tooth dentin defects reflect genetic disorders affecting bone mineralization. Bone. 2012;50:989–97.
Bevelander C, Nakahara H. The formation and mineralization of dentin. Anat Rec. 1966;156:303–23.
Nalbandian J, Gonzales F, Sognnaes RF. Sclerotic age changes in root dentin of human teeth as observed by optical, electron, and x-ray microscopy. J Dent Res. 1960;39:598–607.
Weerheijm KL, Jälevik B, Alaluusua S. Molar-incisor hypomineralisation. Caries Res. 2001;35:390–1.
Jälevik B, Norén J. Enamel hypomineralization of permanent first molars: a morphological study and survey of possible aetiological factors. Int J Paediatr Dent. 2000;10:278–89.
Gotler M, Ratson T. Molar incisor hypomineralization (MIH)—a literature review. Refuat Hapeh Vehashinayim. 1993;2010(27):10–8 (abstract).
Allazzam SM, Alaki SM, El Meligy OAS. Molar incisor hypomineralization, prevalence, and etiology. Int J Dent. 2014. doi:10.1155/2014/234508.
Jälevik B, Klingberg G, Barregård L, Norén JG. The prevalence of demarcated opacities in permanent first molars in a group of Swedish children. Acta Odontol Scand. 2001;59:255–60.
Weinmann JP, Svoboda JF, Woods RW. Hereditary disturbances of enamel formation and calcification. J Am Dent Assoc. 1945;32:397–418.
Witkop CJ. Amelogenesis imperfecta, dentinogenesis imperfecta and dentin dysplasia revisited: problems in classification. J Oral Pathol. 1988;17:547–53.
Kumar S, Gupta S. The restoration of function and esthetics of a patient with amelogenesis imperfecta using a combination of orthodontic and prosthodontic treatment: a case report. J Contemp Dent Pract. 2009;10:E079–85.
Malone W, Bazola FN. Early treatment of amelogenesis imperfecta. J Prosthet Dent. 1966;16:540–4.
Pavlič A, Battelino T, Podkrajšek KT, Ovsenik M. Craniofacial characteristics and genotypes of amelogenesis imperfecta patients. Eur J Orthod. 2011;33:325–31.
Hunter L, Stone D. Supraoccluding cobalt-chrome onlays in the management of amelogenesis imperfecta in children: a 2-year case report. Quintessence Int. 1997;28:15–9.
Chan KHC, Ho EHT, Botelho MG, Pow EHN. Rehabilitation of amelogenesis imperfecta using a reorganized approach: a case report. Quintessence Int. 2011;42:385–91.
Horowitz RA, Gautam DK, Karol S, Kumari B. Periodontal management and restoration of an amelogenesis imperfecta patient: a case report. Compend Contin Educ Dent. 2014;35:e6–11.
Koruyucu M, Bayram M, Tuna EB, Gencay K, Seymen F. Clinical findings and long-term managements of patients with amelogenesis imperfecta. Eur J Dent. 2014;8:546–52.
Nusier M, Yassin O, Hart TC, Samimi A, Wright JT. Phenotypic diversity and revision of the nomenclature for autosomal recessive amelogenesis imperfecta. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2004;97:220–30.
Maciejewska I, Chomik E. Hereditary dentine diseases resulting from mutations in DSPP gene. J Dent. 2012;40:542–8.
Marquezin MC, Zancopé BR, Pacheco LF, Gavião MB, Pascon FM. Aesthetic and functional rehabilitation of the primary dentition affected by amelogenesis imperfecta. Case Rep Dent. 2015. doi:10.1155/2015/790890.
Aldred MJ, Savarirayan R, Crawford PJ. Amelogenesis imperfecta: a classification and catalogue for the 21st century. Oral Dis. 2003;9:19–23.
Pekkan G, Pekkan K, Hatipoglu MG, Tuna SH. Comparative radiopacity of ceramics and metals with human and bovine dental tissues. J Prosthet Dent. 2011;106:109–17.
Akin H, Tasveren S, Yeler DY. Interdisciplinary approach to treating a patient with amelogenesis imperfecta: a clinical report. J Esthet Restor Dent. 2007;19:131–5.
Ghodsi S, Rasaeipour S, Vojdani M. Oral rehabilitation of a patient with amelogenesis imperfecta using removable overlay denture: a clinical report. J Contemp Dent Pract. 2012;13:227–31.
Sholapurkar AA, Joseph RM, Varghese JM, Neelagiri K, Acharya SR, Hegde V, Pai KM, Bhat M. Clinical diagnosis and oral rehabilitation of a patient with amelogenesis imperfecta: a case report. J Contemp Dent Pract. 2008;9:92–8.
Witkop CJ Jr, Kuhlmann W, Sauk J. Autosomal recessive pigmented hypomaturation amelogenesis imperfecta. Report of a kindred. Oral Surg Oral Med Oral Pathol. 1973;36:367–82.
Millet C, Duprez JP. Multidisciplinary management of a child with severe open bite and amelogenesis imperfecta. J Contemp Dent Pract. 2013;14:320–6.
Alachioti XS, Dimopoulou E, Vlasakidou A, Athanasiou AE. Amelogenesis imperfecta and anterior open bite: etiological, classification, clinical and management interrelationships. J Orthod Sci. 2014;3:1–6.
Cogulu D, Becerik S, Emingil G, Hart PS, Hart TC. Oral rehabilitation of a patient with amelogenesis imperfecta. Pediatr Dent. 2009;31:523–7.
Kida M, Ariga T, Shirakawa T, Oguchi H, Sakiyama Y. Autosomal-dominant hypoplastic form of amelogenesis imperfecta caused by an enamelin gene mutation at the exon-intron boundary. J Dent Res. 2002;81:738–42.
Stephanopoulos G, Garefalaki ME, Lyroudia K. Genes and related proteins involved in amelogenesis imperfecta. J Dent Res. 2005;84:1117–26.
Ağaçkiran E, Tümen EC, Celenk S, Bolgül B, Atakul F. Restoring aesthetics and function in a young boy with hypomature amelogenesis imperfecta: a case report. ISRN Dent. 2011. doi:10.5402/2011/586854.
Ayna E, Celenk S, Kadiroglu ET. Restoring function and esthetics in 2 patients with amelogenesis imperfecta: case report. Quintessence Int. 2007;38:51–3.
Bouvier D, Duprez JP, Pirel C, Vincent B. Amelogenesis imperfecta—a prosthetic rehabilitation: a clinical report. J Prosthet Dent. 1999;82:130–1.
de Souza JF, Fragelli CM, Paschoal MA, Campos EA, Cunha LF, Losso EM, Cordeiro RC. Noninvasive and multidisciplinary approach to the functional and esthetic rehabilitation of amelogenesis imperfecta: a pediatric case report. Case Rep Dent. 2014. doi:10.1155/2014/127175.
Doruk C, Ozturk F, Sari F, Turgut M. Restoring function and aesthetics in a class II division 1 patient with Amelogenesis Imperfecta: a clinical report. Eur J Dent. 2011;5:220–8.
Gokce K, Canpolat C, Ozel E. Restoring function and esthetics in a patient with amelogenesis imperfecta: a case report. J Contemp Dent Pract. 2007;8:95–101.
Hoppenreijs TJ, Voorsmit RA, Freihofer HP, van ‘t Hof MA. Open bite deformity in amelogenesis imperfecta. Part 2: Le Fort I osteotomies and treatment results. J Craniomaxillofac Surg. 1998;26:286–93.
Kar SK, Tripathi A, Singh SV. Full mouth rehabilitation of hypomaturation type amelogenesis imperfecta: a clinical report. J Oral Biol Craniofac Res. 2012;2:213–6.
Kamble VD, Parkhedkar RD. Multidisciplinary approach for restoring function and esthetics in a patient with amelogenesis imperfecta: a clinical report. J Clin Diagn Res. 2013;7:3083–5.
Markovic D, Petrovic B, Peric T. Case series: clinical findings and oral rehabilitation of patients with amelogenesis imperfecta. Eur Arch Paediatr Dent. 2010;11:201–8.
Mathew L, Hegde AM, Shetty YR. Oral rehabilitation of a case of amelogenesis imperfecta with multiple periapical cysts. Int J Clin Pediatr Dent. 2008;1:25–31.
Mete JJ, Dange SP, Khalikar AN, Vaidya SP. Functional and esthetic rehabilitation of mutilated dentition associated with amelogenesis imperfecta. J Indian Prosthodont Soc. 2012;12:94–100.
Millet C, Duprez JP, Khoury C, Morgon L, Richard B. Interdisciplinary care for a patient with amelogenesis imperfecta: a clinical report. J Prosthodont. 2014;24:424–31.
Ozturk N, Sari Z, Ozturk B. An interdisciplinary approach for restoring function and esthetics in a patient with amelogenesis imperfecta and malocclusion: a clinical report. J Prosthet Dent. 2004;92:112–5.
Ranganath V, Nichani AS, Soumya V. Amelogenesis imperfecta: a challenge to restoring esthetics and function. J Indian Soc Periodontol. 2010;14:195–7.
Robinson FG, Haubenreich JE. Oral rehabilitation of a young adult with hypoplastic amelogenesis imperfecta: a clinical report. J Prosthet Dent. 2006;95:10–3.
Sabatini C, Guzmán-Armstrong S. A conservative treatment for amelogenesis imperfecta with direct resin composite restorations: a case report. J Esthet Restor Dent. 2009;21:161–70.
Saroğlu I, Aras S, Oztaş D. Effect of deproteinization on composite bond strength in hypocalcified amelogenesis imperfecta. Oral Dis. 2006;12:305–8.
Shetty YB, Shetty A. Oral rehabilitation of a young adult with amelogenesis imperfecta: a clinical report. J Indian Prosthodont Soc. 2010;10:240–5.
Sreedevi S, Sanjeev R, Ephraim R, Joseph M. Interdisciplinary full mouth rehabilitation of a patient with amelogenesis imperfecta: a case report with 8 years follow-up. J Int Oral Health. 2014;6:90–3.
Storie DQ, Cheatham JL. Management of amelogenesis imperfecta by periodontal and prosthetic therapy. J Prosthet Dent. 1970;24:608–15.
Suchancova B, Holly D, Janska M, Stebel J, Lysy J, Thurzo A, Sasinek S. Amelogenesis imperfecta and the treatment plan—interdisciplinary team approach. Bratisl Lek Listy. 2014;115:44–8.
Trentesaux T, Rousset MM, Dehaynin E, Laumaillé M, Delfosse C. 15-year follow-up of a case of amelogenesis imperfecta: importance of psychological aspect and impact on quality of life. Eur Arch Paediatr Dent. 2013;14:47–51.
Turkün LS. Conservative restoration with resin composites of a case of amelogenesis imperfecta. Int Dent J. 2005;55:38–41.
Apaydin A, Sermet B, Ureturk S, Kundakcioglu A. Correction of malocclusion and oral rehabilitation in a case of amelogenesis imperfecta by insertion of dental implants followed by Le Fort I distraction osteogenesis of the edentulous atrophic maxilla. BMC Oral Health. 2014. doi:10.1186/1472-6831-14-116.
Canger EM, Celenk P, Yenísey M, Odyakmaz SZ. Amelogenesis imperfecta, hypoplastic type associated with some dental abnormalities: a case report. Braz Dent J. 2010;21:170–4.
Ergun G, Kaya BM, Egilmez F, Cekic-Nagas I. Functional and esthetic rehabilitation of a patient with amelogenesis imperfecta. J Can Dent Assoc. 2013;79:d38.
Mittal S, Tewari S, Goel R. Esthetic and functional rehabilitation of mutilated dentition and loss of vertical dimension due to amelogenesis imperfecta. Indian J Dent. 2014;5:102–6.
Oliveira IK, Fonseca Jde F, do Amaral F, Pecorari VG, Basting RT, França FM. Diagnosis and esthetic functional rehabilitation of a patient with amelogenesis imperfecta. Quintessence Int. 2011;42:463–9.
Parekh S, Almehateb M, Cunningham SJ. How do children with amelogenesis imperfecta feel about their teeth? Int J Paediatr Dent. 2014;24:326–35.
Rada RE, Hasiakos PS. Current treatment modalities in the conservative restoration of amelogenesis imperfecta: a case report. Quintessence Int. 1990;21:937–42.
Ramos AL, Pascotto RC, Iwaki Filho L, Hayacibara RM, Boselli G. Interdisciplinary treatment for a patient with open-bite malocclusion and amelogenesis imperfecta. Am J Orthod Dentofacial Orthop. 2011;139:S145–53.
Sadighpour L, Geramipanah F, Nikzad S. Fixed rehabilitation of an ACP PDI class III patient with amelogenesis imperfecta. J Prosthodont. 2009;18:64–70.
Savi A, Turillazzi O, Crescini A, Manfredi M. Esthetic treatment of a diffuse amelogenesis imperfecta using pressed lithium disilicate and feldspathic ceramic restorations: 5-year follow up. J Esthet Restor Dent. 2014;26:363–73.
Thompson GA, Schwartz JM. Oral rehabilitation of a patient with amelogenesis imperfecta. J Prosthodont. 1997;6:257–64.
Toksavul S, Ulusoy M, Türkün M, Kümbüloğlu O. Amelogenesis imperfecta: the multidisciplinary approach. A case report. Quintessence Int. 2004;35:11–4.
Auster P. Conquering a difficult case, part 2. Lithium disilicate as a restorative solution for amelogenesis imperfecta. Dent Today. 2013;32:61–4.
Greenfield R, Iacono V, Zove S, Baer P. Periodontal and prosthodontic treatment of amelogenesis imperfecta: a clinical report. J Prosthet Dent. 1992;68:572–4.
Assunção WG, Barão VA, Kanno CM, Saito CT, Delben JA. Overdenture as a restorative option for hypocalcified-hypoplastic amelogenesis imperfecta: a case report. J Contemp Dent Pract. 2009;10:67–73.
Patel RR, Hovijitra S, Kafrawy AH, Bixler D. X-linked (recessive) hypomaturation amelogenesis imperfecta: a prosthodontic, genetic, and histopathologic report. J Prosthet Dent. 1991;66:398–402.
Preissner S, Kostka E, Blunck U. A noninvasive treatment of amelogenesis imperfecta. Quintessence Int. 2013;44:303–5.
Rajesh P, Prasad M, Haldal S. Full mouth rehabilitation of a patient with amelogenesis imperfecta: a case report. J Int Oral Health. 2014;6:76–9.
Siadat H, Alikhasi M, Mirfazaelian A. Rehabilitation of a patient with amelogenesis imperfecta using all-ceramic crowns: a clinical report. J Prosthet Dent. 2007;98:85–8.
Toller PA. A clinical report on six cases of amelogenesis imperfecta. Oral Surg Oral Med Oral Pathol. 1959;12:325–33.
Gisler V, Enkling N, Zix J, Kim K, Kellerhoff NM, Mericske-Stern R. A multidisciplinary approach to the functional and esthetic rehabilitation of amelogenesis imperfecta and open bite deformity: a case report. J Esthet Restor Dent. 2010;22:282–93.
Lamb DJ. The treatment of amelogenesis imperfecta. J Prosthet Dent. 1976;36:286–91.
Lundgren GP, Vestlund GIM, Trulsson M, Dahllöf G. A Randomized controlled trial of crown therapy in young individuals with amelogenesis imperfecta. J Dent Res. 2015;94:1041–7.
Gibbard PD. The management of children and adolescents suffering from amelogenesis imperfecta and dentinogenesis imperfecta. J Oral Rehabil. 1974;1:55–66.
Dashash M, Yeung CA, Jamous I, Blinkhorn A. Interventions for the restorative care of amelogenesis imperfecta in children and adolescents. Cochrane Database Syst Rev. 2013;6:CD007157. doi:10.1002/14651858.CD007157.pub2.
Oxford Centre for Evidence-based Medicine—levels of evidence. 2009. http://www.cebm.net/oxford-centre-evidence-based-medicine-levels-mach-2009/. Accessed 02 June 2016.
Lundgren GP, Dahllöf G. Outcome of restorative treatment in young patients with amelogenesis imperfecta. a cross-sectional, retrospective study. J Dent. 2014;42:1382–9.
Bäckman B, Adolfsson U. Craniofacial structure related to inheritance pattern in amelogenesis imperfecta. Am J Orthod Dentofac Orthop. 1994;105:575–82.
Gjørup H, Haubek D, Hintze H, Haukali G, Løvschall H, Hertz JM, Poulsen S. Hypocalcified type of amelogenesis imperfecta in a large family: clinical, radiographic, and histological findings, associated dento-facial anomalies, and resulting treatment load. Acta Odontol Scand. 2009;67:240–7.
Hashem A, Kelly A, O’Connell B, O’Sullivan M. Impact of moderate and severe hypodontia and amelogenesis imperfecta on quality of life and self-esteem of adult patients. J Dent. 2013;41:689–94.
Hoppenreijs TJ, Voorsmit RA, Freihofer HP. Open bite deformity in amelogenesis imperfecta. Part 1: an analysis of contributory factors and implications for treatment. J Craniomaxillofac Surg. 1998;26:260–6.
Kinzer GA, COMMENTARY. A multidisciplinary approach to the functional and esthetic rehabilitation of amelogenesis imperfecta and open bite deformity: a case report. J Esthet Restor Dent. 2010;22:294–6.
Patil PG, Patil SP. Amelogenesis imperfecta with multiple impacted teeth and skeletal class III malocclusion: complete mouth rehabilitation of a young adult. J Prosthet Dent. 2014;111:11–5.
Roquebert D, Champsaur A, del Real PG, Prasad H, Rohrer MD, Pintado M, Heo Y, Koutlas IG. Amelogenesis imperfecta, rough hypoplastic type, dental follicular hamartomas and gingival hyperplasia: report of a case from Central America and review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;106:92–8.
Sari T, Usumez A. Restoring function and esthetics in a patient with amelogenesis imperfecta: a clinical report. J Prosthet Dent. 2003;90:522–5.
Faria-e-Silva AL, De Moraes RR, Menezes Mde S, Capanema RR, De Moura AS, Martelli H Jr. Hardness and microshear bond strength to enamel and dentin of permanent teeth with hypocalcified amelogenesis imperfecta. Int J Paediatr Dent. 2011;21:314–20.
Fusayama T, Nakamura M, Kurosaki N, Iwaku M. Non-pressure adhesion of a new adhesive restorative resin. J Dent Res. 1979;58:1364–70.
Strobel S, Hellwig E. The effects of matrix-metallo-proteinases and chlorhexidine on the adhesive bond. Swiss Dent J. 2015;125:134–45.
Öztürk E, Bolay Ş, Hickel R, Ilie N. Shear bond strength of porcelain laminate veneers to enamel, dentine and enamel-dentine complex bonded with different adhesive luting systems. J Dent. 2013;41:97–105.
Shimada Y, Iwamoto N, Kawashima M, Burrow MF, Tagami J. Shear bond strength of current adhesive systems to enamel, dentin and dentin-enamel junction region. Oper Dent. 2003;28:585–90.
DeSort KD. Amelogenesis imperfecta: the genetics, classification, and treatment. J Prosthet Dent. 1983;49:786–92.
Chen CF, Hu JC, Estrella MR, Peters MC, Bresciani E. Assessment of restorative treatment of patients with amelogenesis imperfecta. Pediatr Dent. 2013;35:337–42.
Sönmez IS, Aras S, Tunç ES, Küçükeşmen C. Clinical success of deproteinization in hypocalcified amelogenesis imperfecta. Quintessence Int. 2009;40:113–8.
Paewinsky E, Pfeiffer H, Brinkmann B. Quantification of secondary dentine formation from orthopantomograms–a contribution to forensic age estimation methods in adults. Int J Legal Med. 2005;119:27–30.
Poulsen S, Gjørup H, Haubek D, Haukali G, Hintze H, Løvschall H, Errboe M. Amelogenesis imperfecta—a systematic literature review of associated dental and oro-facial abnormalities and their impact on patients. Acta Odontol Scand. 2008;66:193–9.
Ravassipour DB, Powell CM, Phillips CL, Hart PS, Hart TC, Boyd C, Wright JT. Variation in dental and skeletal open bite malocclusion in humans with amelogenesis imperfecta. Arch Oral Biol. 2005;50:611–23.
Rowley R, Hill FJ, Winter GB. An investigation of the association between anterior open-bite and amelogenesis imperfecta. Am J Orthod. 1982;81:229–35.
Gehrt M, Wolfart S, Rafai N, Reich S, Edelhoff D. Clinical results of lithium-disilicate crowns after up to 9 years of service. Clin Oral Investig. 2012;17:275–84.
Gillings B, Buonocore M. An investigation of enamel thickness in human lower incisor teeth. J Dent Res. 1961;40:105–18.
Gillings B, Buonocore M. Thickness of enamel at the base of pits and fissures in human molars and bicuspids. J Dent Res. 1961;40:119–33.
Grine FE, Stevens NJ, Jungers WL. An evaluation of dental radiograph accuracy in the measurement of enamel thickness. Arch Oral Biol. 2001;46:1117–25.
Schwartz GT. Enamel thickness and the helicoidal wear plane in modern human mandibular molars. Arch Oral Biol. 2000;45:401–9.
Discacciati JA, de Souza EL, Vasconcellos WA, Costa SC, Barros Vde M. Increased vertical dimension of occlusion: signs, symptoms, diagnosis, treatment and options. J Contemp Dent Pract. 2013;14:123–8.
Abduo J, Lyons K. Clinical considerations for increasing occlusal vertical dimension: a review. Aust Dent J. 2012;57:2–10.
Luvizuto ER, Queiroz TP, Betoni-Júnior W, Sonoda CK, Panzarini SR, de Castro JC, Boeck EM. Full-mouth rehabilitation using all-ceramic restorations. Compend Contin Educ Dent. 2015;36:588–91.
el-Attar MS. Periodontal and prosthodontic treatment of amelogenesis imperfecta: a clinical report. J Prosthet Dent. 1993;70:100–1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Sabandal, M.M.I., Schäfer, E. Amelogenesis imperfecta: review of diagnostic findings and treatment concepts. Odontology 104, 245–256 (2016). https://doi.org/10.1007/s10266-016-0266-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10266-016-0266-1