Abstract
In order to clarify how vegetation types change along the environmental gradients in a cool temperate to sub-alpine mountainous zone and the determinant factors that define plant species richness, we established 360 plots (each 4 × 10 m) within which the vegetation type, species richness, elevation, topographic position index (TPI), slope inclination, and ground light index (GLI) of the natural vegetation were surveyed. Mean elevation, TPI, slope inclination, and GLI differed across vegetation types. Tree species richness was negatively correlated with elevation, whereas fern and herb species richness were positively correlated. Tree species richness was greater in the upper slope area than the lower slope area, whereas fern and herb species richness were greater in the lower slope area. Ferns and trees species richness were smaller in the open canopy, whereas herb species richness was greater in the open canopy. Vegetation types were determined firstly by elevation and secondary by topographic configurations, such as topographic position, and slope inclination. Elevation and topography were the most important factors affecting plant richness, but the most influential variables differed among plant life-form groups. Moreover, the species richness responses to these environmental gradients greatly differed among ferns, herbs, and trees.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding how biodiversity is structured across various spatiotemporal scales is a great concern of ecologists and conservationists. Multiple spatial scales of environmental gradients are considered primary factors determining biodiversity gradients (Field et al. 2009). Species richness is one of the most fundamental descriptors of biodiversity (Zechmeister and Moser 2001).
Species richness can be measured by counting individual species in the field. However, information concerning inventories and distributions of species is often incomplete or absent for most regions and groups of organisms (Hortal and Lobo 2005). Detailed surveys of flora are in many cases limited or even impossible due to constraints on time, research funds, and/or the availability of researchers with adequate professional knowledge (Jiang et al. 2007). Then, maps of biodiversity surrogates can be interpolated from even a few well-characterised sites (Hortal and Lobo 2005), and the effects of spatial and temporal variability of vegetation due to environmental gradients are well established. Therefore, environmental gradients, such as elevation or topography, can serve as good indicators for predicting species richness.
In mountainous regions, several studies have reported decreasing patterns and non-monotonic or mid-altitudinal bulging patterns of species richness with increasing elevation (Bhattarai and Vetaas 2003; Desalegn and Beierkuhnlein 2010; Sánchez-González and López-Mata 2005; Wangda and Ohsawa 2006). Furthermore, mountainous environments are characterised by local or micro-scale environmental gradients mediated by topographic configuration (Tsujino et al. 2006). However, whether topographic gradients play a major role among these multiple coexistence mechanisms remains unclear. In addition, local species richness is typically correlated not only with local topographic complexity (Enoki 2003; Hofer et al. 2008), but also with light conditions (e.g., Aiba et al. 2004; Masaki et al. 2005). Vegetation types along altitudinal and environmental gradients also provide basic ecological information for better conservation and management of mountain forest ecosystems.
The objectives of the present study were to examine the responses of vegetation types and plant species richness to environmental gradients in natural or minimally disturbed (by humans) forests around Mt. Naeba in the Akiyama region of central Japan. Our primary questions were as follows:
-
1.
How do vegetation types change along the environmental gradients?
-
2.
What are the determinant factors that define plant species richness?
Methods
Study area
The Akiyama region (138.66°N, 36.85°E) includes various human settlements (~730–970 m a.s.l.) along the Nakatsugawa River valley between Mt. Naeba (2,145 m a.s.l.) and Mt. Torikabuto (2,037 m a.s.l.) at the boundary between Nagano and Niigata prefectures in central Japan (Inoue 2002). The local people of Akiyama have historically used the surrounding forests for hunting and gathering, slash-and-burn cultivation, and/or timber production. They have also conserved several forests since at least the middle of the Edo period (ca. 18th century; Sirouzu 2011).
The study area was established in a 6 × 4-km area at approximately 700–2,145 m a.s.l., extending from the bank of the Nakatsugawa River to the top of Mt. Naeba, including the two settlements of Koakazawa and Uenohara. According to data (1979–2000) from the Nozawa-Onsen Local Meteorological Observatory (138.44°N, 36.92°E) located at 576 m a.s.l. about 20 km from the study area, the mean annual temperature at the observatory is 10.5 °C, and mean annual rainfall is 1,899.9 mm (Japan Meteorological Agency, http://www.jma.go.jp/jma/indexe.html). Given that the lapse rate is 0.6 °C per 100 m in elevation and we adopt temperature data of the nearest observatory, the mean annual temperature can be estimated as 9.2 °C around the settlements (~800 m a.s.l.) and 1.1 °C at the top of Mt. Naeba (2,145 m). Substantial snowfall occurs in winter, to a maximum depth of about 3–4 m (Wang et al. 2008).
The vegetation of the study area exhibits vertical variation (Ishizawa et al. 2003). Below 1,400 m, the forests consist of natural and secondary deciduous broad-leaved forests and conifer plantations of sugi cedar Cryptomeria japonica (L.f.) D. Don or the deciduous conifer Larix kaempferi (Lamb.) Carrière. Forests located at 1,300–1,550 m are characterised as mixed conifer and broad-leaved forests. Abies-dominated forests occur from 1,500 to 1,900 m, and dwarf forests characterise altitudes from 1,700 to 2,000 m. A high plain (~4 km2) of grassland and/or wetland vegetation occurs near the top of Mt. Naeba consisting mainly by herbs or dwarf bamboo (Sasa kurilensis (Rupr.) Makino et Shibata) of which height was around 50-cm accompanied by dwarf trees or shrubs. This grassland does not harbour typical alpine vegetation, as indicator plant species of alpine vegetation are not apparent. To examine the relationship between environmental gradients and plant species richness, the study site included a wide altitudinal gradient from approximately 700–2,145 m a.s.l., various topographic positions, and several vegetation types.
Field survey
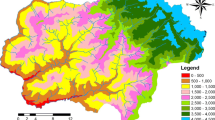
The field studies were conducted from late July to September in 2007, 2008, and 2009. In the study area, 360 transect plots (each 4 m wide × 10 m long) were established (Fig. 1). To maximise the variation in environmental gradients, plots were placed along forest tracking paths and in the forest at various elevational and topographical gradients in vegetation minimally or undisturbed by humans. In this study area, main forest disturbance factors were slash-and-burn cultivation, forest logging and plantation of conifers. However slash-and-burn cultivation had been ceased around the 1960s (Sekido 2012), and the vegetation of abandoned cultivation fields had been shifted to typically Betula platyphylla Sukaczev var. japonica (Miq.) H.Hara tree forests or young broad-leaved trees forests lacking large or old trees. As for forest logging, we could find several logging stamps, if the forest logging had been conducted in the near past. Thus, highly disturbed, secondary forests, or plantation forests were not included in this study. However, we examined a few plots on the lower slope positions at the high altitudinal zone, since there were few valley or lower slope topographic positions at the high altitudinal zone and it was difficult to access the steep mountain.
Vascular plant species richness was surveyed in each plot (4 × 10 m). In each plot, we recorded all living fern, herb, and tree species within 4 × 10 m horizontal area from the ground level to the canopy layer. Analyses of species richness associations with environmental gradients should focus on different components of species groups because responses to environmental gradients are not always similar among different taxonomic or life-form groups (Desalegn and Beierkuhnlein 2010). Thus we grouped species into three life-form groups, namely ferns, herbs and trees, based on life-form and species distribution within the canopy profile. After that, we divided herbs into annual (including biennium) herbs and pellenial herbs, and trees into lianas, shrubs and tall trees. After all plants were identified, we counted the species numbers of each life-form group for each plot. All nomenclature followed that of Yonekura and Kajita (2003).
The location of each plot was estimated using GPS (Garmin GPS 60CSx) and a 1/25,000 topographic map published by the Geological Survey of Japan. In addition, environmental parameters such as elevation above sea level, topography, slope inclination, light levels at ground level, and forest type were also investigated in the field. The elevation of the study plots was determined from values on the topographic map.
The topography of mountainous areas consists of various micro-topographic units. Each plot was classified into three topographic positions (upper slope area, middle slope area and lower slope area), according to the land surface, soil, and slope inclination (Hara et al. 1996; Nagamatsu and Miura 1997; Tsujino and Yumoto 2007). The upper slope area consisted of a ridge with a gentle and convex slope, and the ridge vicinity with a steeper downward slope adjacent to the ridge area. The lower slope consisted of a valley with a gentle and concave slope, and the valley vicinity with a steeper upward slope adjacent to the valley area. We also included six plots of pond-side fens into the lower slope area. The middle slope area was defined by an intermediate slope between the upper and lower slope area. According to above criteria, we estimated topographic position in the field with a topographic map. To evaluate topography, topographic index (TPI) of the upper, middle, and lower slopes were defined as TPI 1, TPI 2 and TPI 3, respectively. Slope inclinations of each plot were measured using a clinometer (211-0016, Kamiyama-seisakusyo, Tokyo). Since this study area was located on the western part of a mountain, lacking east-facing slope, we did not analyse slope direction.
To determine light levels on the ground, the ground light index (GLI) was defined by assessing forest canopy openness. When the forest canopy of a focal plot was open and the forest floor was well lit, the GLI was defined as 0. In the case of a single canopy layer, the GLI was defined as 1. The GLI was defined as 2 if the canopy had two or more layers (e.g., a canopy layer and a sub-canopy layer). Intermediate canopy covers were defined as GLI values of 0.5 and 1.5. According to above criteria, we estimated GLI in the field.
Vegetation types were classified into five categories, according to Ishizawa et al. (2003) and Fukushima and Iwase (2005). When the canopy trees consisted mainly of deciduous broad-leaved canopy trees, such as Fagus crenata Blume, Quercus crispula Blume, Acer pictum subsp. mayrii, Aesculus turbinate, or Pterocarya rhoifolia Siebold et Zucc., and the forest was minimally disturbed by humans, the forest was defined as a low-disturbed deciduous broad-leaved forest. A forest with a canopy composed primarily of deciduous broad-leaved canopy trees (e.g., Fagus crenata) and evergreen coniferous canopy trees [e.g., Tsuga diversifolia (Maxim.) Mast., Thuja standishii (Gordon) Carrière, and Pinus parviflora Siebold et Zucc. var. pentaphylla (Mayr) A. Henry] was defined as a mixed forest. In the case of a forest canopy composed of evergreen coniferous trees, mainly Abies mariesii Mast., often accompanied by Betula ermanii Cham., the forest was defined as an evergreen conifer forest. When the forest canopy trees, typically Abies mariesii and Betula ermanii, were severely dwarfed (heights of a few metres) by heavy snowfall or wind, but sometimes accompanied by few emergent trees, the forest was defined as a dwarf tree forest. Finally, if no trees were apparent and the vegetation was covered mainly by herbs, forbs, or dwarfed Sasa kurilensis (usually the height of dwarfed Sasa kurilensis was less than 0.5 m) accompanied by dwarf trees or shrubs, the vegetation was defined as grassland vegetation. Wet grasslands were also included in the grassland category.
Statistical analysis
The relationships between numbers of plant species of ferns, herbs, trees, and all vascular plants combined and major environmental gradients were described in the form of generalised additive models (GAM) using stepwise procedures from full models. A GAM is built using smoothed functions derived from the explanatory variables, instead of pre-establishing a parametric model. This approach allows for testing whether a response curve is bulging and also permits direct comparisons between the additive effect of each explanatory variable and its predicted response curves. In this analysis, we included all plots from 700 to 2,145 m a.s.l. Poisson distributions, log link and spline based smoothing for elevation and slope inclination were assumed for model fitting to data of numbers of plant species. Four factors represented the number of species (num): (1) elevation (elev), smoothed by spline function, (2) topographic position index (TPI), (3) slope inclination (incl), smoothed by spline function, and (4) GLI of the focal plot.
TPI and GLI are categorical variables and s() indicates smoothed variables. We used the gam function in mgcv package of R for windows ver. 2.15.0 (R Development Core Team 2012). The amount of smoothing (i.e., degrees of freedom) for each continuous explanatory variable was estimated automatically by using generalised cross-variation. Akaike’s information criterion (AIC) was used for model selection, with the minimum AIC as the best-fit estimator.
Results
We found 11 families and 38 species of ferns, 49 families and 202 species of herbs, 52 families and 162 species of trees, and 93 families and 402 species of all vascular plants combined. Common canopy trees were Fagus crenata, Acer pictum subsp. mayrii, Abies mariesii, Thuja standishii, and Aesculus turbinata Blume.
Forest types and environmental gradients
We surveyed 360 plots located at various elevational, topographic, and slope inclination positions, i.e., from 690 to 2,140 m a.s.l., from the ridge top to the valley bottom, and from 0 to 60°, respectively (Fig. 2). The GLIs of the five forest types varied widely (Fig. 2).
Relationships between environmental gradients and vegetation types within study plots (4 × 10 m). DBL, BCM, EC, DWF, and GRA indicate deciduous broad-leaved forests, broad-leaved-conifer mixed forests, evergreen conifer forest, dwarf tree forest, and grassland, respectively. In figures for topography and the ground light index (GLI), areas of each rectangle indicate the portion of study plots. In the boxplots of elevation and slope inclination, the lowest observation (lowest line), first quartile (bottom line of the box), median (thick line in the box), third quartile (top line of the box), highest observation (highest line), and outliers (dots) are shown
By comparing the distribution of vegetation types with elevation in our study area (Fig. 2), we observed a distinct altitudinal pattern of vegetation types. The forests below 1,300 m were dominated by deciduous broad-leaved forest. The forests at 1,300–1,500 m included three types, deciduous broad-leaved forest, mixed forest, or evergreen conifer forest. Because this altitudinal zone was the upper limit of deciduous broad-leaved forest and the lower limit of evergreen conifer forest, these forests were naturally mixed into the deciduous broad-leaved forest type in this vegetation transition zone. Although these three vegetation types coexisted within the same altitudinal zone, their topographic positions and slope inclinations exhibited some differentiation. Deciduous broad-leaved forests were located at various topographic and slope inclination positions, i.e., from the ridge top to the valley bottom and from gentle to steep slopes. In contrast, mixed forests and evergreen conifer forests were relatively aggregated within more upper slope and gentler slope areas. Forests at 1,500–1,900 m consisted of two vegetation types, evergreen conifer and dwarf tree forests. Although the main canopy trees in this altitudinal zone were Abies mariesii and Betula ermanii, the environmental locations of these two vegetation types were quite different. Both evergreen conifer and dwarf tree forests were located on mainly upper slope areas. However, evergreen conifer forests were found at a relatively wide range of topographic positions compared to dwarf tree forests, i.e., from upper to lower slope areas, whereas dwarf tree forests were aggregated within upper slope areas. In addition, dwarf tree forests were located at steeper slope positions compared to evergreen conifer forests. The vegetation above 1,900 m was mainly dominated by grassland, accompanied by evergreen conifer forests.
Number of plant species, environmental gradients, and forest types
The GAM analyses of the relationships between the number of fern, herb, and tree species and environmental gradients showed that the full model was selected with a minimum AIC value (Table 1; Figs. 3, 4). For all vascular plant species combined, the best-fit model was selected with a minimum AIC value (2,273.9; full model AIC = 2,274.4) (Table 1; Figs. 3, 4).
Relationship between numbers of plant species found in a 4 × 10-m plot and environmental variables such as elevation (m), topography position (TPI), slope inclination (°), and the ground light index (GLI). In the topography figures, values indicate the topographic position of upper slope areas (1), middle slope areas (2), lower slope (3). In the GLI figures, values indicate canopy gap (0), canopy closure (1), and canopy closure with lower trees (2), and 0.5 and 1.5 represent intermediate grades between 0 and 1 and 1 and 2, respectively
Generalized additive model smoothing curves for significant parameters on fern, herb, tree and all vascular plant combined species richness in a 4 × 10-m plot. Hatch lines represent SE ranges. The degrees of freedom for each smoothing curve are indicated on the y-axis. Vertical dashes on the x-axis represent the distribution of the explanatory variable
In the GAM analyses, increases in ΔAIC values for the s(elev)-excluded or TPI-excluded models were greater than those for models excluding the other variables, but not for s(elev)-excluded model of lianas and TPI-excluded model of trees (Table 1). These results indicated that elevation and topography were the most important variables among selected variables. Inclination or GLI were less important, even though these variables were selected by AIC-based model selection.
Along with elevational gradient, the species richness of ferns species showed a mid-bulging pattern, whereas that of herb species showed increasing pattern with a small peak around 1,800 m a.s.l., trees and all vascular plant species combined exhibited decreasing pattern with small peaks around 1,500 m a.s.l. (Figs. 3, 4). The number of pellenial herb species along elevation showed quite similar pattern to all herb species combined, while that of annual herb species showed several peaks (Figs. 5, 6). The number of liana and tall tree species along elevation showed similar pattern to all tree species combined, while that of shrub species showed decreasing pattern with peaks around 1,500 and 2,000 m a.s.l (Figs. 5, 6). These plant life-form groups showed completely different species richness responses on the elevation.
Generalized additive model smoothing curves for elevation and slope inclination on the number of annual herbs, pellenial herbs, lianas, shrubs and tall trees species in a 4 × 10-m plot. Hatch lines represent SE ranges. The degrees of freedom for each smoothing curve are indicated on the y-axis. Vertical dashes on the x-axis represent the distribution of the explanatory variable
Along with topographic gradient, the species richness of ferns and herbs, including annual and pellenial herbs, was greater in middle or lower slope areas than in upper slope areas (Figs. 3, 5). In contrast, tree species richness was greater in the upper slope area than in the lower slope area, though there were some differences among lianas, shrubs and tall trees (Figs. 3, 5). Fagus crenata, Quercus crispula, Thuja standishii, Pinus parviflora var. pentaphylla, and Tsuga diversifolia were distributed mainly within upper slope areas, whereas Aesculus turbinata and Pterocarya rhoifolia primarily occurred in lower slope areas. Acer pictum subsp. mayrii, Abies mariesii, and Betula ermanii did not exhibit topography-specific distribution patterns. For all vascular plant species combined, the species richness was slightly greater in middle and lower slope areas compared to upper slope areas.
Along with the slope inclination gradient, the species richness of ferns species showed increasing pattern in the GAM analyses (Figs. 3, 4). The species richness of herb species also showed increasing pattern with some fluctuations (Figs. 3, 4). However the species richness of trees and all vascular species combined showed bulging pattern (Figs. 3, 4). The number of pellenial herb species along slope inclination showed similar pattern to all herb species combined, while that of annual herb species showed decreasing pattern (Figs. 5, 6). The number of liana, shrub and tall tree species along slope inclination showed similar pattern to all tree species combined (Figs. 5, 6).
As for GLI, fern and tree species richness, including sub-categories, at GLI = 0 condition were fewer than rather dark conditions, such as GLI = 0.5–2 (Figs. 3, 5). Herb species richness, including sub-categories, was greater in open canopy conditions than in dark conditions (Figs. 3, 5).
Discussion
Distribution pattern of vegetation types along environmental gradients
In this study, vegetation types were clearly primarily determined by elevation, and secondarily by topographic configuration, such as topographic position, and slope inclination. The temperature-elevation gradient is probably one of the most continuous ecological gradients on a macro-scale (Vetaas 2000). However the zones at 1,300–1,500 m and 1,500–1,900 m consisted of several vegetation types.
The distribution of deciduous broad-leaved forests, mixed forests, and evergreen conifer forests were segregated by topography even in a same elevational zone at 1,300–1,500 m, since two types of mixed-conifer or conifer-dominated forests around the deciduous broad-leaved forest zone are usually distributed along ridges or upper slope areas in Japan (Fukushima and Iwase 2005). One type is Abies and Tsuga-dominated forest, which is primarily distributed on rather gentle ridges or upper slope areas. The other type is mixed forest dominated by Chamaecyparis, Thuja, Pinus, or Sciadopitys, which is mainly distributed on ridge tops or rocky locations (Fukushima and Iwase 2005).
Although Abies mariesii has the potential to grow over 30 m in height and often inhabits cold climates and shallow to deep snow regions (Uemura 1989), this species is dwarfed under heavy snow regimes (Mori and Mizumachi 2009). Therefore the vegetation overlap between the evergreen conifer forests and the dwarf tree forests, both of which were dominated by Abies mariesii and/or Betura ermanii, at 1,500–1,900 m had been partly caused by the fluctuation of snowfall level along the slope inclination gradient.
Even though the timberline in the study area occurs around 2,600 m a.s.l., vegetation above 1,900 m mainly consisted of grasslands. Forests were less likely to occur in the high plain located at gentle upper slope positions of the study area. One reason for the lack of forest at these elevations is that this high plateau is generally poorly drained and thus characterised by wet grassland, Sphagnum bogs, and pools (Ishizawa et al. 2003; Shimazu and Sekizawa 2003).
Overall, the results of this study indicate that the elevational gradient was the most influential, but not the only factor determining vegetation type in the study area. Topography and snowfall may also create important environmental gradients.
Responses of plant species richness along environmental gradients
Altitude can serve as a proxy variable for water-energy dynamics in mountain ecosystems (O’Brien et al. 2000). The elevation gradient also affected plant species richness on a macro spatial scale in tandem with micro-scale environmental gradients, such as topographic configuration and ground light levels. In the present study, elevation was an important determinant of species richness at the macro-scale. However the specific responses differed among plant life-form groups. The functional attributes of species (e.g., physiological, life history, and ecological) are directly related to the characteristics of physical environmental gradients (Desalegn and Beierkuhnlein 2010). Furthermore the observed altitudinal patterns did not show monotonically increase or decrease with increases in elevation; instead, the relationships displayed bulges or minor peaks.
Previous studies have documented non-linear mid-altitude bulging patterns of diversity along elevational gradients (Bhattarai and Vetaas 2003; Jiang et al. 2007). In the present study, peaks at around 1,500 m a.s.l. for tree species have occurred partly because the 1,400–1,500-m zone represented a vegetation transition zone from deciduous broad-leaved forest to evergreen conifer forest, and this zone contained three different vegetation types, which allowed various kinds of trees coexist. On the other hand, a peak at around 1,800 m a.s.l. for herb species have occurred partly because 1,700–1,900-m zone represented an another vegetation transition zone from evergreen conifer forest with dark ground condition to dwarf tree forest or grassland with rather bright ground condition, which allowed various kinds of herb species coexist. These conditions allowed diverse plant species from both the upper and lower altitudinal vegetation zones to coexist within this narrow altitudinal zone.
Topographic variation has another potential to enhance local plant species richness. Previous studies have documented that tree species composition changes along topographic gradients from ridges to valleys (e.g., Enoki 2003 Masaki et al. 1992; Suzuki 2011; Tsujino et al. 2006; Tsujino and Yumoto 2007). In the present study, we observed clear relationships between topographic position and plant species richness (Figs. 3, 5). Our results indicate that most ferns and herbs preferred middle or lower slope areas. Middle or lower slope areas tend to be rather moderate or wet and thus serve as suitable habitats for these generally drought-intolerant species, like herbs and ferns. Lwanga et al. (1998) demonstrated that fern communities tend to be abundant on fertile soils, particularly in lower slope areas.
Canopy gaps often facilitate the establishment and growth of many tree species (Denslow 1987; Schnitzer and Carson 2001). Contradictory, according to the observed number of tree species, tree species richness dropped clearly at open canopy position, while herb species richness was greater at open positions than at dark position. However, since the canopy status of forested vegetation is often closed, the data of open canopy positions (GLI = 0) had been mostly grassland vegetation (73 %) at high plateau, where were quite few fern and tree species richness than the other forested vegetation types. If grassland vegetation data were excluded from the data of GLI = 0, the mean number of species at GLI = 0 would have increased from 1.4 to 2.4 for ferns, from 9.7 to 12.2 for herbs and from 5.6 to 11.3 for trees. Thus the response patterns to the GLI will be a gentle decreasing pattern for ferns and a flat pattern for trees.
The most important variables that determine plant richness were elevation and topographic gradients, but the responding patterns to the variables differed among plant life-form groups. Thus, a clear understanding of the mechanisms that generate and maintain diversity at different environmental gradients requires analysis at the level of plant life-form group components.
Abbreviations
- AIC:
-
Akaike’s information criterion
- TPI:
-
Topographic position index
- GLI:
-
Ground light index
- GAM:
-
Generalized additive model
References
Aiba S, Kitayama K, Takyu M (2004) Habitat associations with topography and canopy structure of tree species in a tropical montane forest on Mount Kinabalu, Borneo. Plant Ecol 174:147–161
Bhattarai KR, Vetaas OR (2003) Variation in plant species richness of different life forms along a subtropical elevation gradient in the Himalayas, east Nepal. Glob Ecol Biogeogr 12:327–340
Denslow JS (1987) Tropical rainforest gaps and tree species diversity. Ann Rev Ecol Syst 18:431–451
Desalegn W, Beierkuhnlein C (2010) Plant species and growth form richness along altitudinal gradients in the southwest Ethiopian highlands. J Veg Sci 21:617–626
Enoki T (2003) Microtopography and distribution of canopy trees in a subtropical evergreen broad-leaved forest in the northern part of Okinawa Island, Japan. Ecol Res 18:103–113
Field R, Hawkins BA, Cornell HV, Currie DJ, Diniz-Filho JAF, Guegan JF, Kaufman DM, Kerr JT, Mittelbach GG, Oberdorff T, O’Brien EM, Turner JRG (2009) Spatial species-richness gradients across scales: a meta-analysis. J Biogeogr 36:132–147
Fukushima T, Iwase T (2005) Illustrated: Vegetation in Japan. Asakura Publishing, Tokyo, 153 pp
Hara M, Hirata K, Fujihara M, Oono K (1996) Vegetation structure in relation to micro-landform in an evergreen broad-leaved forest on Amami Ohshima Island, south-west Japan. Ecol Res 11:325–337
Hofer G, Wagner HH, Herzog F, Edwards PJ (2008) Effects of topographic variability on the scaling of plant species richness in gradient dominated landscapes. Ecography 31:131–139
Hortal JN, Lobo JM (2005) An ED-based protocol for optimal sampling of biodiversity. Biodiv Cons 14:2913–2947
Inoue T (2002) Changing activities of utilization of wild edible plants: case study of wild edible plants and mushrooms in Akiyama-go, Sakae village, Nagano Prefecture [Henka suru Yasei-shokuyou-shokubutu no riyou-katsudou: Nagano-ken Sakae-mura Akiyama-go ni okeru sansan kinoko nado no jirei kara]. Ecosophia 10:77–100 (in Japanese)
Ishizawa S, Sato M, Iiduka H (2003) Flowering Mt. Naeba [Hanakaoru Naebasan]. Hoozuki Shoseki, Japan, (in Japanese)
Jiang Y, Kang Y, Zhu Y, Xu G (2007) Plant biodiversity patterns on Helan Mountain, China. Acta Oecologica 32:125–133
Lwanga J, Balmford A, Badaza R (1998) Assessing fern diversity: relative species richness and its environmental correlates in Uganda. Biodiv Consv 7:1387–1398
Masaki T, Suzuki W, Niiyama K, Iida S, Tanaka H, Nakashizuka T (1992) Community structure of a species-rich temperate forest, Ogawa Forest Reserve, central Japan. Vegetation 98:97–111
Masaki T, Osumi K, Takahasi K, Hozhshizaki K (2005) Seedling dynamics of Acer mono and Fagus crenata: an environmental filter limiting their adult distributions. Plant Ecol 177:189–199
Mori AS, Mizumachi E (2009) Within-crown structural variability of dwarfed mature Abies mariesii in snowy subalpine parkland in central Japan. J For Res 14:155–166
Nagamatsu D, Miura O (1997) Soil disturbance regime in relation to micro-scale landforms and its effects on vegetation structure in hilly area in Japan. Plant Ecol 133:191–200
O’Brien EM, Field R, Whittaker RJ (2000) Climatic gradients in woody plant (tree and shrub) diversity: water-energy dynamics, residual variation, and topography. Oikos 89:588–600
Sánchez-González A, López-Mata L (2005) Plant species richness and diversity along an altitudinal gradient in the Sierra Nevada, Mexico. Divers Distrib 11:567–575
Schnitzer SA, Carson WP (2001) Treefall gaps and the maintenance of species diversity in a tropical forest. Ecology 82:913–919
Sekido A (2012) Rise and fall of slush-and-burn cultivation and Common Forest use [Yakihata no seisui to iriai-rinya no riyou]. In: Shirouzu T (ed) New record of Akiyama journey [Shin Akiyama Kikou], Kohshi shoin, Tokyo, pp 58–73 (in Japanese)
Shimazu M, Sekizawa K (2003) Geological Guide of “Akiyama-go” [“Akiyama-go” no chigaku annai], Nojima shuppan, Niigata (in Japanese)
Sirouzu S (2011) Transformation of mountainous regions in the early-modern period and conflicts over forest conservation—why were the forests in the Akiyama region preserved? [Kinse sanson no henbou to sinrin hozen wo meguru kattou—Akiyama no shizen ha naze mamorareta ka], In: Shirouzu S, Ikeya K, Yumoto T (eds) Environmental history of mountains and forests [Yamato mori no kankyoshi], Bunichi-sogo shuppan, Tokyo, pp 77–92 (in Japanese)
Suzuki M (2011) Effects of the topographic niche differentiation on the coexistence of major and minor species in a species-rich temperate forest. Ecol Res 26:317–326
R Development Core Team (2012) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0. URL: http://www.R-project.org/
Tsujino R, Yumoto T (2007) Spatial distribution patterns of trees at different life stages in a warm temperate forest. J Plant Res 120:687–695
Tsujino R, Takafumi H, Agetsuma N, Yumoto T (2006) Variation in tree growth, mortality and recruitment among topographic positions in a warm temperate forest. J Veg Sci 17:281–290
Uemura S (1989) Snowcover as a factor controlling the distribution and speciation of forest plants. Vegetation 82:127–137
Vetaas OR (2000) Comparing species temperature response curves: population density versus second-hand data. J Veg Sci 11:659–666
Wang Q, Iio A, Tenhunen J, Kakubar Y (2008) Annual and seasonal variations in photosynthetic capacity of Fagus crenata along an elevation gradient in the Naeba Mountains, Japan. Tree Physiol 28:277–285
Wangda P, Ohsawa M (2006) Gradational forest change along the climatically dry valley slopes of Bhutan in the midst of humid eastern Himalaya. Plant Ecol 186:109–128
Yonekura H, Kajita T (2003) BG plants: index of Japanese names and scientific names (YList). URL: http://bean.bio.chiba-u.jp/bgplants/ylist_main.html
Zechmeister HG, Moser D (2001) The influence of agricultural land-use intensity on bryophyte species richness. Biodiv Consr 10:1609–1625
Acknowledgments
We were permitted to conduct a series of field researches by Koakazawa settlement, Uenohara settlement, Sakaemura village office and Department of Hoku-Shin Forestry Office. We would like to thank Drs. A. Imamura, D. Kawase, R. Koda, and Mr/Ms. K. Nakura, S. Sakaguchi, J. Takahashi, N. Tsujimura who helped our field studies. We are also grateful to Prof. T. Yahara of Kyushu University for showing us transect survey. Members at Hist-Chubu working group of research project D-02 of Research Institute for Humanity and Nature (RIHN) offered me helpful suggestions and comments. We were financially assisted by research project D-02 and “Survivability and Autonomy in Southeast Asia: Perspectives from Land Use Changes and Resource Chains” of RIHN and the Environmental Research and Technology Development Fund (S9) of the Ministry of the Environment, Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Nomenclature: Yonekura and Kajita (2003).
Rights and permissions
About this article
Cite this article
Tsujino, R., Yumoto, T. Vascular plant species richness along environmental gradients in a cool temperate to sub-alpine mountainous zone in central Japan. J Plant Res 126, 203–214 (2013). https://doi.org/10.1007/s10265-012-0520-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-012-0520-8