Abstract
We investigated carbon dioxide (CO2) exchange and its environmental response during two years with contrasting climate (2006 and 2007) in a cool-temperate mixed evergreen coniferous forest dominated by Japanese cedar (Cryptomeria japonica) and Japanese cypress (Chamaecyparis obtusa). The study, which was conducted in a mountainous region of central Japan, used the eddy-covariance technique. Our results (crosschecked using the common u * approach and van Gorsel’s alternative approach) showed that annual gross primary production (GPP) and ecosystem respiration (RE) were at least 6% higher in the dry year than in the wet year, whereas net ecosystem exchange (NEE) was similar in both years. Without soil water stress, strong light stress or seasonality of plant area index during most of the study period, the forest had high metabolic activity. GPP and RE differed greatly between the two years, especially in spring (April–May) and summer (July–September), respectively. The spring GPP difference (>20%) was influenced by different winter air temperatures and snow melt timing, which controlled photosynthetic capacity in spring, and by different spring light intensities. The annual NEE differed depending on the evaluation method used, but the mean 2-year NEE estimated by the u * threshold approach [−3.39 ± 0.11 (SD) MgC ha−1 year−1] appears more reasonable in comparison with results from other forests.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Forests significantly influence the atmospheric composition and climate because they act as important sources and sinks for trace gases and energy, both locally and regionally. To predict how forests will respond to climate change, we must therefore understand how CO2 fluxes between forests and the atmosphere respond to climate variations (Misson et al. 2007). Evergreen coniferous forest is a dominant forest type in East Asia (Ito 2008). Indeed, evergreen coniferous forest dominated by Japanese cedar (Cryptomeria japonica D. Don) and Japanese cypress (Chamaecyparis obtusa Sieb. et Zucc.) account for 66% of the total plantation area and 30% of Japan’s total forested area (Japan FAO Association 1997), with the majority situated in mountainous regions (Sawano et al. 2005; Kumagai et al. 2008). Therefore, understanding CO2 exchange and its environmental response in this forest type is crucial to predicting future carbon cycling in Japan and East Asia in response to climate change.
Despite the importance of such research, few studies of CO2 exchange and its environmental responses based on methods such as the eddy-covariance technique have been reported for evergreen coniferous forest (e.g., Takanashi et al. 2005 for Japanese cypress forest), in contrast with the large number of studies of other forest types such as deciduous forests in Japan (e.g., Saigusa et al. 2002, 2005; Hirano et al. 2003; Kominami et al. 2003, 2008; Kosugi et al. 2005; Hirata et al. 2007) and in Asia (e.g., Li et al. 2005; Saitoh et al. 2005; Zhang et al. 2006; Kosugi et al. 2008).
To assess the characteristics of CO2 exchange by forest ecosystems and its climatic response, year-long measurements using the eddy-covariance technique have been performed around the world (Baldocchi 2008). The popularity of the eddy-covariance technique results from its ability to provide data on the temporal variation in fluxes from diurnal to inter-annual timescales (Baldocchi et al. 2001). However, the technique has certain problems, including underestimation of nighttime fluxes and energy imbalances. In general, this approach works better in flat terrain than in complex terrain (e.g., Lee et al. 2004). However, Asian forests usually occupy mountainous areas, requiring flux observations in complex topography (Mizoguchi et al. 2009). Nighttime data is one of the most critical sources of uncertainty at many sites, especially given the complex topography of mountainous regions, and corrections are required for important sources of error such as horizontal and vertical advection in tall canopies; however, effective protocols for data correction have not yet been established (Saigusa et al. 2008). In the present study, to avoid the effects of uncertain nighttime data on evaluations of the environmental response of CO2 exchange, as gap-filling methods we adopted the common friction velocity (u *) approach (see, e.g., Black et al. 1996; Goulden et al. 1997; Jarvis et al. 1997) and an alternative approach proposed by van Gorsel et al. (2007). In our evaluation of the environmental response of CO2 exchange, we have reported consistent results obtained with those two approaches. This type of careful evaluation will be suitable for analysis over complex topography in various regions.
In this study, we analyzed CO2 flux over a cool-temperate mixed evergreen coniferous forest in central Japan dominated by Japanese cedar and Japanese cypress, during two years (2006 and 2007) with contrasting climate, using the eddy-covariance technique. Our goal was to reduce the uncertainty about carbon cycling in evergreen coniferous forest and its environmental response in East Asia. We specifically addressed the following questions: (1) How do environmental factors control variations in net ecosystem exchange (NEE), gross primary production (GPP), and ecosystem respiration (RE) as a function of climatic seasonality in this forest? (2) How large is the carbon sequestration in this forest type in Japan? This study will further our understanding of carbon dynamics and contribute to improved prediction of their responses to climate change in East Asia.
Methods
Site description
The study was carried out in the Takayama evergreen coniferous forest (TKC; 36°08′N, 137°22′E; 800 m a.s.l.), one of the AsiaFlux forest sites (http://asiaflux.net). It is located about 13 km east of Takayama City, in the central part of Japan’s main island (Fig. 1a). This region belongs to a cool temperate zone under the influence of the Asian monsoon climate, and is characterized by mild, humid springs and autumns; hot, humid, summers; and cold, snowy winters (Lee et al. 2008). The annual average temperature and cumulative precipitation for the past 20 years are 11.2°C and 1,723 mm, respectively, and showed clear seasonal variation at the Takayama station of the Japan Meteorological Agency-Automated Meteorological Data Acquisition System (JMA-AMeDAS) at an elevation of 560 m a.s.l., which is near the TKC site. Snow covers the ground from November or December to March or April.
The study site is located at a mid-slope position in a valley (Fig. 1b). The mean local slope at the site is about 21° downhill in a north–northwest direction (334°). The study site has a 30-m tower and a 0.15-ha ecological-process research plot. Micrometeorological and soil environmental measurements have been carried out at the site since March 2005, and eddy-covariance measurements since September 2005 (Muraoka and Koizumi 2009). For the present study, we used two years of continuous data from 1 January 2006 to 31 December 2007.
The forest surrounding the tower consists mainly of evergreen coniferous forest (Japanese cedar and Japanese cypress). This forest covers most of the 2-km2 area surrounding the study site, but deciduous forest is also present in patches, mainly from the northwestern to northeastern side. Croplands, a few houses, and roads are also present at these locations, several hundred meters from the tower. The fetch of the evergreen coniferous forest varies with the wind direction, and ranges from 100 to 1,000 m. Based on the minimum fetch requirements to obtain reliable data, some of the flux data were eliminated (for details see the section on Eddy-covariance measurements below). The evergreen coniferous forest was planted at about 40–50 years ago, and has a continuous canopy height of about 20 m surrounding the tower. The understory was sparsely covered by shrubs, herbs, and ferns. The soil parent material is a volcanic ash deposit, and the soil type is a Dystric Cambisol with a dominant soil texture of clay loam at the ecological research plot. Lee et al. (2008) provide more detailed descriptions of the study site.
Micrometeorological and soil environment measurements
We measured several meteorological components to characterize the forest’s micrometeorological environment. Short- and long-wave downward and upward radiation were measured with solar radiation radiometers (CM-6B, Kipp & Zonen, Delft, the Netherlands) and infrared radiometers (CG3, Kipp & Zonen) installed at the top of the tower, about 30 m above ground level. At the top of the tower, downward and upward photosynthetic photon flux density (PPFD) were measured by quantum sensors (LI-190SZ, Li-Cor, Lincoln, NE) and precipitation was measured using a tipping-bucket rain gauge (RH-5A, Ikeda Keiki, Tokyo, Japan). Air temperature and humidity were measured at eight heights on the tower (30.2, 22.8, 18.4, 14.7, 10.6, 6.5, 3.6, and 1.3 m) using humidity and temperature probes (HMP45A, Vaisala, Vantaa, Finland) with ventilated radiation shields. Snow depth was measured by a sonic ranging sensor (SR50, Campbell Scientific, Logan, UT), and snow coverage on the forest floor was monitored using an Internet camera (CG-NCMIN, corega, Yokohama, Japan). On the forest floor, downward PPFD was also measured at five locations by quantum sensors (LI-190SB, Li-Cor; IKS-25 and IKS-27, Koito, Tokyo, Japan) and used to calculate the plant area index (PAI). The volumetric soil water content (θ, m3 m−3) and soil temperature were measured with a time-domain reflectometer (TDR; CS616, Campbell Scientific) and a temperature probe (Model 107, Campbell Scientific) at depths of 5, 10, 15, 20, and 40 cm at a location 10 m apart from the tower. In the present study, we represented soil moisture conditions using the soil water content (SWC; m3 m−3), which equals the θ value for the top 40 cm of the soil. All these data were collected at 5-s intervals and averaged for 1-min intervals using a CR23X datalogger (Campbell Scientific).
Eddy-covariance measurements
The flux related to NEE (F NEE) was calculated as follows:
where F NEP is the flux related to NEP (net ecosystem production), F c is the CO2 flux above the canopy estimated by the eddy-covariance method, and F s is the CO2 storage below the height of the eddy-covariance measurements. Baldocchi (2003) provides detailed explanations of these components of eddy-covariance measurements.
Fc and the fluxes of H2O, sensible heat, latent heat, and momentum above the canopy were obtained using an eddy-covariance system installed at the top of the tower. This system comprised a three-dimensional sonic anemometer (R3-50, Gill Instruments, Hampshire, UK) and an open-path CO2/H2O infrared gas analyzer (IRGA; LI-7500, Li-Cor) installed at 31 m above ground level (i.e., 10 m above the mean canopy height). The open-path IRGA was calibrated once every 2–4 months using N2 gas and 500 ppmv CO2 standard gases to calibrate the CO2 and H2O zeros and the CO2 span, respectively. The H2O span was calibrated using the H2O concentration estimated based on air temperature and relative humidity, which was measured using a humidity and temperature probe (HMP45A, Vaisala) near the open-path IRGA. Three wind components (i.e., u, v, and w for x, y, and z axis), the sonic virtual temperature, and the CO2 and H2O gas concentration time series were all sampled at 10 Hz. Eddy fluxes were calculated for 30-min runs with quality test (for details see Appendix A in Electronic Supplementary Material). In addition, as part of our flux analysis, we examined the influences of ground surfaces other than the evergreen coniferous forest and of the fetch using footprint analysis (Schuepp et al. 1990). This analysis revealed that almost all of the flux data obtained when the wind blew from the north was influenced by a surface other than that of the evergreen coniferous forest. Thus, we eliminated 30-min flux data when the wind originated between 300° and 50°. After quality tests and footprint analysis, the remaining data were used for analysis.
We calculated F s at 30-min intervals from the time change of CO2 storage in an air column within the height of the eddy-covariance measurements using vertical profile data for CO2 concentrations and air temperature. The CO2 concentration profile measurement below the height of the eddy-covariance measurements was carried out at the same eight heights as the air temperature measurements using diaphragm pumps and an auto-solenoid switching system, and the CO2 concentration was measured using a closed-path CO2 IRGA (LI-820, Li-Cor). The CO2 channels were calibrated every month using N2 gas and 500 ppmv CO2 standard gases, and all raw data were recorded with a CR10X data-logger (Campbell Scientific).
The proportions of the F NEE data that we retained after quality control were 43% (2006) and 47% (2007) in the daytime and 20% (2006) and 17% (2007) in the nighttime. Long gaps in the F NEE data (longer than 2 weeks) occurred from 8 February [day of year (DOY)39] to 22 March (DOY81) 2006 and from 7 May (DOY127) to 31 May (DOY151) 2007. Eddy fluxes including F c were missed during the former period, while F s was missed during the latter period.
Energy imbalance is a common issue in eddy-covariance measurements. Wilson et al. (2002) evaluated the energy-balance closure using eddy-flux data from 22 sites across 50 site-years. They found that the slope of the regression line of the sum of latent and sensible heat fluxes against available energy (using 30-min data) ranged from 0.53 to 0.99, with a mean of 0.79, and that the energy-balance ratio (EBR), which represents the ratio of the annual sum of latent and sensible heat flux to available energy, ranged from 0.34 to 1.69, with a mean of 0.84. The regression slope of 0.72 and the EBR of 0.84 based on our data are thus very similar to those reported by Wilson et al. (2002).
Gap-filling method
The fragmented data sets of F NEE including missing values contain sufficient information for half-hourly model fitting but to fill the data gap is needed for daily, monthly and annual sums.
FNEE equals the difference between FRE and FGPP:
where FRE represents the flux related to ecosystem respiration (i.e., CO2 release by roots, stems, branches, foliage, and microbial respiration), and FGPP represents the flux due to gross primary production (i.e., CO2 assimilation by photosynthesis). Negative values of FNEE therefore represent net CO2 uptake by the forest.
We adopted a widely used gap-filling method that involves filling F RE gaps using a yearly temperature-dependency curve for nighttime F NEE (i.e., nighttime F RE) and filling daytime gaps using a monthly light-response curve for F GPP.
We fitted F RE to the equation of Lloyd and Taylor (1994), which provides an accurate and unbiased estimate of respiration rates across a wide range of air temperatures:
where R ref is the nighttime F RE at a specified reference temperature (283.15 in absolute temperature unit, K), E 0 is the ratio of the activation energy to the ideal gas constant (K), and T is the air temperature at the height of the eddy-covariance measurements (K). We used nighttime data from January to December to determine the equation’s parameters. Nighttime data was defined according to sunrise and sunset times derived from the local time and standard routines for solar geometry (Jones 1992).
Effective protocols for nighttime data correction in the complex topography of mountainous regions have not yet been established. Accordingly, we used the common u * approach and an alternative approach proposed by van Gorsel et al. (2007) to examine and cross-check the results of the gap-filling calculations for RE, GPP and NEE. In the former approach, we chose u * = 0.35 m s−1 as a suitable threshold value for the TKC site and used this value for our estimation of RE, GPP and NEE (for details see Appendix B in Electronic Supplementary Material). The alternative approach proposed by van Gorsel et al. (2007) uses the maximum nighttime F NEE values for each night to construct a temperature-response function instead of using all the nighttime F NEE at high u * values. Further description of the nighttime estimation is provided in Appendix C in Electronic Supplementary Material.
To fill the F GPP gaps, we calculated the daytime 30-min F GPP values using Eq. 2 and fitted these values for each month using a Michaelis–Menten rectangular hyperbola (e.g., Aubinet et al. 2001; Falge et al. 2001; Owen et al. 2007; Aires et al. 2008; Kominami et al. 2008) as a function of downward PPFD (Q d) at the height of the eddy-covariance measurements:
where A max is the estimated maximum photosynthetic rate for the whole forest canopy (μmol m−2 s−1), and a s is the initial slope of the F GPP−Q d curve. Here, A max is expected to be a product of maximum photosynthetic rate at the leaf level and leaf area index. In comparison between Michaelis–Menten rectangular hyperbola and non-rectangular hyperbola equation with the constant value (0.9) of the convexity (e.g., Hirata et al. 2008), predicted F GPP was similar, while A max estimated by former equation was about 20–30% higher than that estimated by the latter equation.
To interpolate data during the long-term gaps in February and March 2006 and May 2007, we estimated the parameters in Eq. 4 using the weighted average of the F GPP−Q d curve for DOY22 to DOY38 and for DOY83 to DOY98 in 2006 for the parameters in February and March 2006, and the weighted average of the F GPP−Q d curve for DOY111 to DOY126 and for DOY152 to DOY171 2007 for the parameters in May 2007.
Plant area index
We calculated PAI based on the attenuation of PPFD in the canopy using Beer’s law (Monsi and Saeki 1953):
where Q d and Q f are the cumulative downward PPFD measured over the course of the day at the top of the tower and above the forest understory layer, respectively, and K is an extinction coefficient for diffuse radiation. Here, we assumed K to be 0.58, which is the value obtained in a coniferous forest about 45 years old (Hirata et al. 2007). To eliminate the effect of direct radiation on the estimation of PAI, we used only the data obtained on cloudy days. Here, we chose the cloudy days by defining a threshold for the ratio of diffuse radiation to solar radiation as below. We estimated diffuse radiation using the method of Spitters et al. (1986). To eliminate the effect of spatial variation in Q f, we measured Q f at five points on the forest floor, and used the average value. We then calculated daily values of PAI using Eq. 5 based on the daily cumulative Q d and Q f. Finally, we eliminated spike values of PAI, which were related to periods when snow covered the sensors at the forest floor.
Statistical analysis
We carried out Student’s t test by using original programs to assess the differences of meteorological factors and carbon budget between the two years. Regression analyses for Eqs. 3 and 4 were conducted using KyPlot 5.0 (KyensLab, Tokyo, Japan) and original programs.
Results
Microclimatic features and their differences between the two years
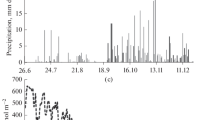
Figure 2 shows the seasonal variation in the monthly precipitation (PPT), average SWC to a depth of 40 cm, average air temperature at the height of 30.2 m, average daytime air vapor pressure deficit (VPD) at a height of 30.2 m, daily PPFD above the canopy, and PAI in 2006 and 2007. Annual PPT and average SWC were much higher in 2006 than in 2007 (t test for SWC, P < 0.001), and annual average VPD was much lower in 2006 than in 2007 (t test, P < 0.05), whereas annual PPFD and air temperature in 2006 were similar to those in 2007. In addition, there were obvious differences in the number of days of snow cover on the forest floor: 95 days in 2006 versus 74 days in 2007. These values depended on the snow-melt timing in each year. Snow cover on the forest floor disappeared in early April 2006 and late March 2007. Furthermore, there was no snow cover from late February to early March in 2007 (data not shown).
Seasonal variations in monthly averages of a precipitation (PPT), b soil water content (SWC) to a depth of 40 cm, c air temperature at a height of 30.2 m, d daytime vapor pressure deficit (VPD) at a height of 30.2 m, e daily photosynthetic photon flux density (PPFD), and f plant area index (PAI), in 2006 and 2007. Annual cumulative or average values were a 1,874 and 1,407 mm, b 0.304 and 0.257 m3 m−3, c 9.3 and 9.6°C, d 4.1 and 4.5 hPa, e 8,724 and 8,814 mol m−2, and f 4.9 and 5.2 m2 m−2 in 2006 and 2007, respectively. Error bars Standard error
Monthly PPT was almost always higher in 2006 than in 2007, especially in July. Accordingly, although the seasonal patterns of SWC were similar, SWC was significantly higher in 2006 than in 2007 throughout the year, except in July (t test, P < 0.001). The minimum monthly SWC appeared in August 2007. Monthly air temperature, daytime VPD, and PPFD exhibited clear seasonality, but daytime VPD and PPFD in July dipped suddenly due to the effect of the Asian monsoon (Baiu) in both years. Although the maximum monthly solar radiation at the top of the atmosphere appeared in June at this latitude, the maximum monthly PPFD appeared in August because of short precipitation periods in August in both years. The average VPD and PPFD in spring (April and May) differed between the two years, with values of 4.7 hPa and 29.7 mol m−2 day−1, respectively, in 2006 versus values of 6.3 hPa and 33.1 mol m−2 day−1 in 2007. Air temperatures in winter (January and February) and in September were 1.6–2.6°C higher in 2006 than in 2007. PAI exhibited little seasonality, and PAI was slightly higher in 2007 than in 2006 throughout the year. As a result, the microclimate in 2006 can be characterized as relatively wet throughout the year, with late snow-melt, lower PPFD, and more humid air in spring and lower air temperature in winter compared with 2007.
Seasonal variations in RE, GPP, NEE, and A max
Figure 3 shows seasonal variations in monthly RE, GPP, NEE, and A max in 2006 and 2007 estimated by the u * threshold approach (left panels) and the van Gorsel approach (right panels). RE exhibited clear seasonality in both years and both approaches, reaching its maximum value in August, when the air temperature was the highest (Fig. 2c). RE in summer (June–September) accounted for more than 50% of annual RE, whereas RE in winter (January, February and December) accounted for 11% or less of annual RE. GPP also exhibited clear seasonality, with the highest monthly values (more than 300 gC m−2 month−1) in August. GPP did not decrease to zero even in winter, although the winter GPP accounted for less than 10% of annual GPP. The summer GPP accounted for more than 50% of annual GPP.
Based on these two results, NEE exhibited large negative values (net carbon uptake by the forest) in spring or early summer of both years, caused by a steeper increase in GPP (Fig. 3b, f) than in RE from winter to early summer (Fig. 3a, e). In contrast, NEE had a small absolute value in late summer because, at that time, GPP was nearly balanced by RE. A max exhibited clear seasonality in both years and with both approaches (Fig. 3d, h), independent of PAI, which showed small seasonality throughout the year (Fig. 2f). The maximum values of A max were greater than 40 μmol m−2 s−1 in July and August. Annual average A max was about 25 μmol m−2 s−1.
Differences in the carbon balance between wet and dry years
Table 1 shows annual RE, GPP and NEE in 2006 and 2007 estimated by u * threshold and van Gorsel approach. Although absolute values of annual RE, GPP and NEE differed between the two approaches, annual RE and GPP in 2007 were much higher than in 2006, and NEE was almost constant in both years regardless of the evaluation method.
To better understand the differences in RE, GPP, NEE, and A max between the two years, we divided each year into four periods (Table 2). Table 2 shows the differences in cumulative RE, GPP, and NEE and in average A max for four periods between 2006 and 2007 for the u * threshold and van Gorsel approaches. Annual GPP and RE were significantly higher (by at least 6%) in 2007 in both approaches, whereas NEE and A max did not differ significantly between the two years. The significant difference in RE between 2006 and 2007 appeared mainly in summer (June–September): RE was more than 8% higher in 2007 in both approaches. This difference accounted for more than 68% of the annual difference in RE between the two years. The difference in GPP between the two years was more than 20% in spring (April–May), and the difference was significant. This difference accounted for more than 48% of the annual difference in GPP between the two years. GPP in winter and early spring (January–March) also differed significantly between the two years. From October to December, the differences in RE and GPP between the two years were consistently small (less than 3%).
NEE differed significantly between the two years in spring and summer. NEE in spring was much lower in 2007 (by more than 98%), mainly because of the large difference in GPP at this time. On the other hand, NEE was much higher in summer (by more than 84%) in 2007, mainly because of the difference in summer RE between the two years. As a result, the seasonal variation in NEE was larger in 2007 (standard deviations of 32–39 gC m−2 month−1) than in 2006 (23–24 gC m−2 month−1). A max in spring was significantly higher in 2007 (by more than 17%). However, NEE data was missing for most of May 2007. To ensure that the difference in A max in May was real, we estimated \( A_{{\max }}^{\prime } \) in May by means of regression analysis between Q d and \( F_{{GPP }}^{\prime } \), which was calculated from the observed F c (i.e., F GPP without accounting for F s), and compared the results between the two years. The \( A_{{\max }}^{\prime } \) in May estimated based on this regression analysis also differed significantly between 2006 and 2007: the values were 24.4 ± 1.2 (estimated value ± SE) in 2006 and 27.8 ± 1.1 in 2007 using the u * threshold approach (P < 0.05), and 23.8 ± 0.9 in 2006 and 26.1 ± 0.8 in 2007 using the van Gorsel approach (P < 0.05). During the other seasons, A max did not differ significantly between the two years, except in August (Fig. 3d, h).
Discussion
Characteristics of seasonal variation in RE, GPP, NEE and A max
Our study showed that the cool-temperate evergreen coniferous forest maintains photosynthetic CO2 assimilation even in winter, although GPP in winter is less than 10% of annual GPP. Actually, canopy images provided by the Phenological Eyes Network (http://www.pheno-eye.org/) in addition to the internet camera noted in Methods indicated that the forest canopy was not continuously covered by snow during winter. NEE exhibited large negative values during spring and early summer, but small absolute values in late summer, reflecting differences in the seasonal patterns of GPP and RE. Those results may suggest that high GPP in late summer was canceled out by high growth respiration and high soil respiration, depending mainly on the seasonal progression of soil temperature (Lee et al. 2008). In a comparison of results from 11 Asian forest sites, including two evergreen coniferous forests, the seasonal pattern of NEE at the TKC site was similar only to that of a Japanese cypress forest in the Kiryu experimental watershed (KEW; Saigusa et al. 2008), despite the fact that the annual average temperature was at least 5°C higher at the KEW site than at the TKC site. This seasonal pattern may therefore be one of the characteristics of evergreen coniferous forest dominated by Japanese cedar and cypress. Further long-term and multi-site research under variable environmental conditions, which will be provided by altitudinal and/or latitudinal gradients and climate change, will enable us to reveal the ecosystem mechanisms underlying the temporal change and environmental responses of GPP, RE and NEE of these forests.
The seasonal progression of monthly A max and that of monthly air temperature were close at the TKC site, exhibiting a highly linear relationship (R 2 = 0.92). This result suggests that air temperature did not reach the turning point where the reduction of maximum leaf photosynthesis occurs, even in August with the highest average daily maximum temperature of 27.9°C. In construct, at the KEW site, a peak value of A max and reduction in A max appeared in late June and August, respectively (Hirata et al. 2008). Seasonal progression of A max was almost independent of plant area index at the TKC site, while previous studies reported that A max depended not only on air temperature but also on leaf area index in a cool-temperate deciduous broadleaf forest (Saigusa et al. 2002) and in a cool-temperate deciduous coniferous forest (Hirata et al. 2007). The fact that the seasonal progression of A max depends only on air temperature at the TKC site may be understood as a general characteristics of a cool-temperate evergreen coniferous forest without soil water stress, strong light stress or seasonality of PAI during most of the year.
Environmental response of carbon balance in wet and dry years
It is expected that the difference in precipitation between the two study years (2006 and 2007) influences carbon flux. Summer RE (June–September) accounted for more than 50% of annual RE, and the difference in summer RE between the two years explained more than 60% of the difference in annual RE. On the other hand, although summer GPP accounted for more than 50% of annual GPP, the difference in summer GPP between the two years was not large. The difference in GPP between the two years appeared mainly in spring; spring GPP in the drier year (2007) was significantly higher (by more than 20%) than that in 2006. This higher spring GPP in the dry year occurred as a result of enhancement of both photosynthetic capacity (A max) and radiation. The significantly higher A max in spring of the dry year (Fig. 3d, h) can be attributed to the earlier snow melt and higher air temperature in the winter of that year (Fig. 2c). The difference in snow melt timing between the two years depended mainly on differences in the maximum snow depth and the cumulative daily winter air temperature, with values of 0.4 m and −172 day degrees, respectively, in 2006 versus values of 0.2 m and −47 day degrees in 2007. The importance of the effect of winter and spring conditions on annual GPP (or NEE) of forests has been suggested in previous studies: Black et al. (2000) reported that CO2 uptake by a boreal aspen forest was increased by a warm spring, and Saigusa et al. (2008) noted that CO2 uptake by two Asian evergreen coniferous forests was increased by warm conditions at the beginning of the year, probably due to enhanced photosynthetic activity. Our results support previous ideas about the importance of environmental conditions at the beginning of the year for determining the carbon balance of evergreen coniferous forests (e.g., Saigusa et al. 2008).
A significant difference in A max between the two years appeared not only in spring but also in August (Fig. 3d, h). The difference in A max in August may have been influenced by soil dryness, because the lowest SWC values were observed in August 2006 (Fig. 2b). Our results suggest that soil water stress appeared only in August 2006 during the observation period.
Comparison with annual GPP, RE and maximum A max in other Asian forests
Annual carbon fluxes (GPP and RE) at the TKC site are compared with those of previous reports in Asia (Hirata et al. 2008; Kato and Tang 2008) in Fig. 4. The annual GPP and RE at the TKC site in 2006 and 2007 were greater than those predicted by the regression lines at the same annual average temperature in both estimation approaches. These results suggest that the forest at the TKC site exhibited higher metabolic activity than other Asian forests under the same temperature conditions: the forest absorbed and released large amounts of CO2 by photosynthesis and respiration. This characteristic may be attributed to two factors: (1) Japanese cedar and cypress have a high potential for plant growth, and (2) afforestation in appropriate sites where Japanese cedar and cypress can maintain high plant growth rate without soil water- or strong light-stress.
Relationship between annual carbon fluxes and annual mean temperature at the TKC site and at other Asian Flux sites. The solid and dashed lines indicate the results of linear regressions between GPP or RE and the annual air temperature at the Asian Flux sites obtained by Hirata et al. (2008) and Kato and Tang (2008). The linear regressions between GPP and T a were both significant and positive: GPP = 0.97 T a + 8.4(R 2 = 0.92) in Hirata et al. (2008); GPP = 0.78 T a + 6.6(R 2 = 0.92) in Kato and Tang (2008). The results of the regression analysis between RE and T a were also significant and positive: \( {\text{RE}} = 14.47{\text{e}}^{{24.99/R*\left[ {1/56.02 - 1/\left( {T_{\text{a}} + 46.02} \right)} \right]}} , \) where R* is the ideal gas constant (J mol−1 K−1; R 2 = 0.89; Hirata et al. 2008); \( {\text{RE}} = 4.90{\text{e}}^{{0.06671\,T{\text{a}}}} \) (R 2 = 0.71; Kato and Tang 2008)
In a comparison among the results of subarctic (TUR and SKT sites), cool temperate (LSH, TSE, TMK, TKY, and FJY sites) and temperate forest sites (KEW site) in Asia (see Table 1 in Hirata et al. 2008), the maximum A max of about 40 μmol m−2 s−1 at the TKC site was close to cool-temperate forest sites, after correcting for the 20–30% difference in the estimation equation of A max by using the Michaelis–Menten rectangular hyperbola (this study) and non-rectangular hyperbola (Hirata et al. 2008) as noted in Methods. In contrast, the maximum A max at the TKC site was considerably higher than those at the subarctic and temperate forest sites reported by Hirata et al. (2008), e.g., the 4-year average value of maximum A max (23.6 μmol m−2 s−1) in a temperate Japanese cedar forest at the KEW site. The absence of environmental stresses in the summer period in a cool temperature region may maintain higher maximum A max compared to those of subarctic forest sites with low temperature and temperate forest sites with high temperature and VPD.
Validity of annual NEE
Our estimation of average annual NEE over the two years at the TKC site showed a large gap between the two estimation approaches: the values averaged −3.39 ± 0.11 (average ± SD) and −0.67 ± 0.4 MgC ha−1 year−1 for the u * threshold and van Gorsel approaches, respectively. To assess the validity of absolute NEE values estimated by these approaches, we calculated the ratio of the eddy-covariance net ecosystem production (NEP = −NEE) to the living biomass increment (LBI) and compared the results with those from other forests. Although NEP measured by the eddy-covariance method is generally comparable with the NEP obtained by biometric methods, the biometric NEP contains a potentially large uncertainty in the estimation of fine root production and heterotrophic respiration (Gough et al. 2008). Therefore, we have instead chosen to discuss the absolute value of the eddy-covariance NEP estimated by the u * threshold and van Gorsel approaches based on the ratio of the eddy-covariance NEP to LBI. The average annual LBI over the two years was 4.9 MgC ha−1 year−1 at the TKC ecological research plot (Y.Y., unpublished data). The average annual NEP values estimated for the two years using the u * threshold and van Gorsel approaches in the present study were approximately 70% and 14% of annual LBI, respectively. The ratio of the eddy-covariance NEP to LBI calculated from previous reports conducted in different forest types and forest ages was higher than 50% (Granier et al. 2000; Barford et al. 2001; Curtis et al. 2002; Ehman et al. 2002; Kolari et al. 2004; Schelhaas et al. 2004; Black et al. 2007; Ohtsuka et al. 2007, 2008; Gough et al. 2008; Kominami et al. 2008), except for a young evergreen coniferous forest in which the value was 22% (Kolari et al. 2004).
Furthermore, annual average net primary production and soil respiration estimated at the ecological research plot at the TKC site were 7.70 and 6.83 MgC ha−1 year−1 (Yashiro et al. 2010). Hanson et al. (2000) reviewed that root respiration can account for 20–90% of soil respiration, with a mean of 45.8% in most forest sites. If we assume that heterotrophic respiration ranges from 10% to 80% (i.e., root respiration ranged from 90% to 20%) of soil respiration, predicted NEE can range from −7.02 to −2.24 MgC ha−1 year−1.
Our estimation of annual NEE using the u * threshold approach (−3.39 ± 0.11 MgC ha−1 year−1) is also supported by a previous report of annual NEE in a Japanese cypress forest at the KEW, one of the AsiaFlux sites: −4.8 MgC ha−1 year−1 reported by Takanashi et al. (2005), but ranging from −4.3 to −5.4 MgC ha−1 year−1 over a period of four years (Hirata et al. 2008). Judging from these results, the NEE of −3.39 ± 0.11 MgC ha−1 year−1 estimated by the u * threshold approach is more reasonable than the value of −0.67 ± 0.4 MgC ha−1 year−1 estimated by the van Gorsel approach at the TKC site.
Conclusion
We investigated seasonal changes in carbon fluxes, such as RE, GPP and NEE, and their environmental responses during two years with contrasting climate (wet and dry years) in a cool-temperate planted mixed evergreen coniferous forest dominated by Japanese cedar and Japanese cypress. Our estimation of mean 2-year NEE (−3.39 ± 0.11 MgC ha−1 year−1) is thus available for one sample of carbon stock, accounting for post Kyoto protocol as knowledge on planted forest from flux studies. Our environmental response analyses found that (1) spring GPP differed between the two years due to the different winter air temperatures, snow melt timing and spring light intensity; and (2) the forest had high metabolic activity depending on stressless and suitable environmental conditions (i.e., without soil water stress, strong light stress or seasonality of PAI during most of the study period).
The finding that snow melt timing was one of the most important environmental factors suggests that the monitoring of snow-cover by remote sensing and treatment of snow-cover in modeling analysis are important in any study of cool-temperate evergreen coniferous forest. Considering the altitudinal dependency of snow melt timing over mountainous regions, a high spatial resolution analysis using remote sensing and modeling may be needed for studies of carbon budget in Asian regions.
References
Aires MLI, Pio CA, Pereira JS (2008) Carbon dioxide exchange above a Mediterranean C3/C4 grassland during two climatologically contrasting years. Glob Change Biol 14:539–555
Aubinet M, Chermanne B, Vandenhaute M, Longdoz B, Yernaux M, Laitat E (2001) Long term carbon dioxide exchange above a mixed forest in the Belgian Ardennes. Agric For Meteorol 108:293–315
Baldocchi DD (2003) Assessing the eddy covariance technique for evaluating carbon dioxide exchange rates of ecosystems: past, present and future. Glob Change Biol 9:479–492
Baldocchi DD (2008) ‘Breathing’ of the terrestrial biosphere: lessons learned from a global network of carbon dioxide flux measurement systems. Aust J Bot 56:1–26
Baldocchi DD, Falge E, Gu LH, Olson R, Hollinger D, Running S, Anthoni P, Bernhofer C, Davis K, Evans R, Fuentes J, Goldstein A, Katul G, Law B, Lee XH, Malhi Y, Meyers T, Munger W, Oechel W, Paw UKT, Pilegaard K, Schmid HP, Valentini R, Verma S, Vesala T, Wilson K, Wofsy S (2001) FLUXNET: A new tool to study the temporal and spatial variability of ecosystem-scale carbon dioxide, water vapor, and energy flux densities. Bull Am Meteorol Soc 82:2415–2434
Barford CC, Wofsy SC, Goulden ML, Munger JW, Pyle EH, Urbanski SP, Hutyra L, Saleska SR, Fitzjarrald D, Moore K (2001) Factors controlling long- and short-term sequestration of atmospheric CO2 in a mid-latitude forest. Science 294:1688–1691
Black TA, Hartog GD, Neumann HH, Blanken PD, Yang PC, Russell C, Nesic Z, Lee X, Chen SG, Staebler R, Novak MD (1996) Annual cycles of water vapour and carbon dioxide fluxes in and above a boreal aspen forest. Glob Change Biol 2:219–229
Black TA, Chen WJ, Barr AG, Arain MA, Chen Z, Nesic Z, Hogg EH, Neumann HH, Yang PC (2000) Increased carbon sequestration by a boreal deciduous forest in years with a warm spring. Geophys Res Lett 27:1271–1274
Black K, Bolger AT, Davis AP, Nieuwenhuis AM, Reidy B, Saiz AG, Tobin AB, Osborne AB (2007) Inventory and eddy covariance-based estimates of annual carbon sequestration in a Sitka spruce (Picea sitchensis (Bong.) Carr.) forest ecosystem. Eur J For Res 126:167–178
Curtis PS, Hanson PJ, Bolstad P, Barford C, Randolph JC, Schmid HP, Wilson KB (2002) Biometric and eddy-covariance based estimates of annual carbon storage in five eastern North American deciduous forests. Agric For Meteorol 113:3–19
Ehman JL, Schmid HP, Grimmond CSB, Randolph JC, Hanson PJ, Wayson CA, Cropley FD (2002) An initial intercomparison of micrometeorological and ecological inventory estimates of carbon sequestration in a mid-latitude deciduous forest. Glob Change Biol 8:575–589
Falge E, Baldocchi D, Olson R, Anthoni P, Aubinet M, Bernhofer C, Burba G, Ceulemans R, Clement R, Dolman H, Granier A, Gross P, Grunwald T, Hollinger D, Jensen NO, Katul G, Keronen P, Kowalski A, Lai CT, Law BE, Meyers T, Moncrieff J, Moors E, Munger JW, Pilegaard K, Rannik U, Rebmann C, Suyker A, Tenhunen J, Tu K, Verma S, Vesala T, Wilson K, Wofsy S (2001) Gap filling strategies for defensible annual sums of net ecosystem exchange. Agric For Meteorol 107:43–69
Gough CM, Vogel CS, Schmid HP, Su H-B, Curtis PS (2008) Multi-year convergence of biometric and meteorological estimates of forest carbon storage. Agric For Meteorol 148:158–170
Goulden ML, Daube BC, Fan S-M, Sutton DJ, Bazzaz A, Munger JW, Wofsy SC (1997) Physiological responses of a black spruce forest to weather. J Geophys Res 102:28987–28996
Granier A, Ceschia E, Damesin C, Dufrene E, Epron D, Gross P, Lebaube S, Dantec VL, Goff NL, Lemoine D, Lucot E, Ottorini JM, Pontailler JY, Saugier B (2000) The carbon balance of a young beech forest. Funct Ecol 14:312–325
Hanson PJ, Edwards NT, Garten CT, Andrews JA (2000) Separating root and soil microbial contributions to soil respiration: a review of methods and observations. Biogeochemistry 48:115–146
Hirano T, Hirata R, Fujinuma Y, Saigusa N, Yamamoto S, Harazono Y, Takada M, Inukai K, Inoue G (2003) CO2 and water vapor exchange of a larch forest in northern Japan. Tellus B 55:244–257
Hirata R, Hirano T, Saigusa N, Fujinuma Y, Inukai K, Kitamori Y, Yamamoto S (2007) Seasonal and inter-annual variations in carbon dioxide exchange of a temperate larch forest. Agric For Meteorol 147:110–124
Hirata R, Saigusa N, Yamamoto S, Ohtani Y, Ide R, Asanuma J, Gamo M, Hirano T, Kondo H, Kosugi Y, Li S-G, Nakai Y, Takagi K, Tani M, Wang H (2008) Spatial distribution of carbon balance in forest ecosystems across East Asia. Agric For Meteorol 148:761–775
Ito A (2008) The regional carbon budget of East Asia simulated with a terrestrial ecosystem model and validated using AsiaFlux data. Agric For Meteorol 148:738–747
Japan FAO Association (1997) Forests and forestry in Japan, 2nd edn. Japan FAO Association, Tokyo
Jarvis PG, Massheder JM, Hale SE, Moncrieff JB, Rayment M, Scott SL (1997) Seasonal variation of carbon dioxide, water vapor, and energy exchanges of a boreal black spruce forest. J Geophys Res 102:28953–28966
Jones HG (1992) Plants and microclimate, 2nd edn. Cambridge University Press, Cambridge
Kato T, Tang Y (2008) Spatial variability and major controlling factors of CO2 sink strength in Asian terrestrial ecosystems: evidence from eddy covariance data. Glob Change Biol 14:1–16
Kolari P, Pumpanen J, Rannik U, Ilvesniemi H, Hari P, Berninger F (2004) Carbon balance of different aged Scots pine forests in Southern Finland. Glob Change Biol 10:1106–1119
Kominami Y, Miyama T, Tamai K, Nobuhiro T, Goto Y (2003) Characteristics of CO2 flux over a forest on complex topography. Tellus B 55:313–321
Kominami Y, Jomura M, Dannoura M, Goto Y, Tamai K, Miyama T, Kanazawa Y, Kaneko S, Okumura M, Misawa N, Hamada S, Sasaki T, Kimura H, Ohtani Y (2008) Biometric and eddy-covariance-based estimates of carbon balance for a warm-temperate mixed forest in Japan. Agric For Meteorol 148:723–737
Kosugi Y, Tanaka H, Takanashi S, Matsuo N, Ohte N, Shibata S, Tani M (2005) Three years of carbon and energy fluxes from Japanese evergreen broad-leaved forest. Agric For Meteorol 132:329–343
Kosugi Y, Takanashi S, Ohkubo S, Matsuo N, Tani M, Mitani T, Tsutsumi D, Nik AR (2008) CO2 exchange of a tropical rain forest at Pasoh in Peninsular Malaysia. Agric For Meteorol 148:439–452
Kumagai T, Tateishi M, Shimizu S, Otsuki K (2008) Transpiration and canopy conductance at two slope positions in a Japanese cedar forest watershed. Agric For Meteorol 148:1444–1455
Lee X, Finnigan J, Paw UKT (2004) Coordinate systems and flux bias error. In: Lee X, Massman W, Law B (eds) Handbook of micrometeorology: a guide for surface flux measurement and analysis Kluwer, Dordrecht, The Netherlands, pp 33–66
Lee M-S, Lee J-S, Koizumi H (2008) Temporal variation in CO2 efflux from soil and snow surfaces in a Japanese cedar (Cryptomeria japonica) plantation, central Japan. Ecol Res 23:777–785
Li S-G, Asanuma J, Kotani A, Eugster W, Davaa G, Oyunbaatar D, Sugita M (2005) Year-round measurements of net ecosystem CO2 flux over a montane larch forest in Mongolia. J Geophys Res 110:D09303. doi:10.1029/2004JD005453
Lloyd J, Taylor JA (1994) On the temperature dependence of soil respiration. Funct Ecol 8:315–323
Misson L, Baldocchi DD, Black TA, Blanken PD, Brunet Y, Curiel Yuste JC, Dorsey JR, Falk M, Granier A, Irvine MR, Jarosz N, Lamaud E, Launiainen S, Law BE, Longdoz B, Loustau D, McKay M, Paw UKT, Vesala T, Vickers D, Wilson KB, Goldstein AH (2007) Partitioning forest carbon fluxes with overstory and understory eddy-covariance measurements: a synthesis based on FLUXNET data. Agric For Meteorol 144:14–31
Mizoguchi Y, Miyata A, Ohtani Y, Hirata R, Yuta S (2009) A review of tower flux observation sites in Asia. J For Res 14:1–9
Monsi M, Saeki T (1953) Über den Lichtfaktor in den Pflanzengesellschaften und seine Bedeutung für die Stoffproduktion. Jpn J Bot 14:22–52
Muraoka H, Koizumi H (2009) Satellite Ecology (SATECO)-linking ecology, remote sensing and micrometeorology, from plot to regional scale, for the study of ecosystem structure and function. J Plant Res 122:3–20
Ohtsuka T, Mo W, Satomura T, Inatomi M, Koizumi H (2007) Biometric based carbon flux measurements and Net Ecosystem Production (NEP) in a temperate deciduous broad-leaved forest beneath a flux tower. Ecosystems 10:324–334
Ohtsuka T, Lee M-S, Yashiro Y, Negishi M, Koizumi H (2008) Net primary production and carbon allocation patterns in red pine, Japanese cedar, and broadleaved forests beneath flux towers. In: Proceedings of 2nd International Symposium of 21st Century COE Program “Satellite Ecology”. Gifu University, pp 61–64
Owen KE, Tenhunen J, Reichstein M, Wang Q, Falge E, Geyer R, Xiao X, Stoy P, Ammann C, Arain A, Aubinet M, Aurela M, Bernhofer C, Chojnicki BH, Granier A, Gruenwald T, Hadley J, Heinesch B, Hollinger D, Knohl A, Kutsch W, Lohila A, Meyers T, Moors E, Moureaux C, Pilegaard K, Saigusa N, Verma S, Vesala T, Vogel C (2007) Linking flux network measurements to continental scale simulations: ecosystem CO2 exchange capacity under non-water-stressed conditions. Glob Change Biol 13:734–760
Saigusa N, Yamamoto S, Murayama S, Kondo H, Nishimura S (2002) Gross primary production and net ecosystem exchange of a cool-temperate deciduous forest estimated by the eddy covariance method. Agric For Meteorol 112:203–215
Saigusa N, Yamamoto S, Murayama S, Kondo H (2005) Inter-annual variability of carbon budget components in an AsiaFlux forest site estimated by long-term flux measurements. Agric For Meteorol 134:4–16
Saigusa N, Yamamoto S, Hirata R, Ohtani Y, Ide R, Asanuma J, Gamo M, Hirano T, Kondo H, Kosugi Y, Li S-G, Nakai Y, Takagi K, Tani M, Wang H (2008) Temporal and spatial variations in the seasonal patterns of CO2 flux in boreal, temperate, and tropical forests in East Asia. Agric For Meteorol 148:700–713
Saitoh TM, Kumagai T, Sato Y, Suzuki M (2005) Carbon dioxide exchange over a Bornean tropical rainforest. J Agric Meteorol 60:553–556
Sawano S, Komatsu H, Suzuki M (2005) Differences in annual precipitation amounts between forested area, agricultural area, and urban area in Japan (in Japanese with English abstract). J Jpn Soc Hydrol Water Resour 18:435–440
Schelhaas MJ, Nabuurs GJ, Jans W, Moors E, Sabate S, Daamen WP (2004) Closing the carbon budget of a Scots pine forest in the Netherlands. Clim Change 67:309–328
Schuepp PH, Leclerc MY, MacPherson JI, Desjardins RL (1990) Footprint prediction of scalar fluxes from analytical solutions of the diffusion equation. Boundary Layer Meteorol 50:355–373
Spitters CJT, Toussaint HAJM, Goudriaan J (1986) Separating the diffuse and direct components of global radiation and its implications for modeling canopy photosynthesis part I. Components of incoming radiation. Agric For Meteorol 38:217–229
Takanashi S, Kosugi Y, Tanaka Y, Yano M, Katayama T, Tanaka H, Tani M (2005) CO2 exchange in a temperate Japanese cypress forest compared with that in a cool-temperate deciduous broad-leaved forest. Ecol Res 20:313–324
van Gorsel E, Leuning L, Cleugh HA, Keith H, Suni T (2007) Nocturnal carbon efflux: reconciliation of eddy covariance and chamber measurements using an alternative to the u *-threshold filtering technique. Tellus B 59:397–403
Wilson K, Goldsten A, Falge E, Aubinet M, Baldocchi D, Berbigier P, Ceulenmans R, Dolman H, Field C, Grelle A, Ibrom A, Law BE, Lowalski A, Meyers T, Moncrieff J, Monson R, Oechel W, Tenhinen J, Valentini R, Verma S (2002) Energy balance closure at FLUXNET sites. Agric For Meteorol 113:223–243
Yashiro Y, Lee N-Y, Ohtsuka T, Shizu Y, Saitoh TM, Koizumi H (2010) Biometric based estimation of net ecosystem production (NEP) in a mature Japanese cedar (Cryptomeria japonica) plantation beneath a flux tower. J Plant Res (submitted in this special issue)
Zhang JH, Han SJ, Yu GR (2006) Seasonal variation in carbon dioxide exchange over a 200-year-old Chinese broad-leaved Korean pine mixed forest. Agric For Meteorol 137:150–165
Acknowledgments
We thank Mr. K. Kurumado and Mr. Y. Miyamoto of the River Basin Research Center, Gifu University, for their support at the Takayama Field Station. Thanks are also due to Prof. T. Ohtsuka of Gifu University, Dr. H. Kondo of AIST, and anonymous reviewers for thoughtful suggestions. We also thank the forest owners at the TKC site for their permission to construct the flux tower and install various measurement systems. This work was supported by the JSPS 21st Century COE program “Satellite Ecology” at Gifu University and the JSPS-KOSEF-NSFC A3 Foresight Program. T.M.S. is grateful for the financial support received from the Ministry of Education, Culture, Sports, Science and Technology of Japan, Grant-in-Aid for Young Scientists (B), no. 18780113 to T.S. and Grant-in-Aid for Scientific Research (A), no. 21241009 to H.K.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Saitoh, T.M., Tamagawa, I., Muraoka, H. et al. Carbon dioxide exchange in a cool-temperate evergreen coniferous forest over complex topography in Japan during two years with contrasting climates. J Plant Res 123, 473–483 (2010). https://doi.org/10.1007/s10265-009-0308-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-009-0308-7