Abstract
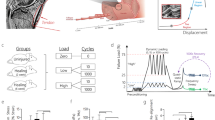
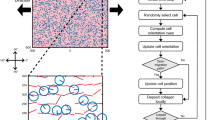
The importance of fluid-flow-induced shear stress and matrix-induced cell deformation in transmitting the global tendon load into a cellular mechanotransduction response is yet to be determined. A multiscale computational tendon model composed of both matrix and fluid phases was created to examine how global tendon loading may affect fluid-flow-induced shear stresses and membrane strains at the cellular level. The model was then used to develop a quantitative experiment to help understand the roles of membrane strains and fluid-induced shear stresses on the biological response of individual cells. The model was able to predict the global response of tendon to applied strain (stress, fluid exudation), as well as the associated cellular response of increased fluid-flow-induced shear stress with strain rate and matrix-induced cell deformation with strain amplitude. The model analysis, combined with the experimental results, demonstrated that both strain rate and strain amplitude are able to independently alter rat interstitial collagenase gene expression through increases in fluid-flow-induced shear stress and matrix-induced cell deformation, respectively.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adeeb S, Ali A, Shrive N, Frank C and Smith D (2004). Modelling the behaviour of ligaments: a technical note. Comput Methods Biomech Biomed Eng 7: 33–42
Archambault JM, Elfervig-Wall MK, Tsuzaki M, Herzog W and Banes AJ (2002). Rabbit tendon cells produce MMP-3 in response to fluid flow without significant calcium transients. J Biomech 35: 303–309
Arnoczky SP, Lavagnino M, Whallon JH and Hoonjan A (2002a). In situ cell nucleus deformation in tendons under tensile load; a morphological analysis using confocal laser microscopy. J Orthop Res 20: 29–35
Arnoczky SP, Tian T, Lavagnino M, Gardner K, Schuler P and Morse P (2002b). Activation of stress-activated protein kinases (SAPK) in tendon cells following cyclic strain: the effects of strain frequency, strain magnitude, and cytosolic calcium. J Orthop Res 20: 947–952
Arnoczky SP, Tian T, Lavagnino M and Gardner K (2004). Ex vivo static tensile loading inhibits MMP-1 expression in rat tail tendon cells through a cytoskeletally based mechanotransduction mechanism. J Orthop Res 22: 328–333
Arnoczky SP, Lavagnino M and Egerbacher M (2007). The response of tendon cells to changing loads: implications in the etiopathogenesis of tendinopathy. In: Woo, SL, Renstrom, P, and Arnoczky, SP (eds) Tendinopathy in athletes, pp 46–59. Blackwell, Oxford
Ateshian GA, Likhitpanichkul M and Hung CT (2006). A mixture theory analysis for passive transport in osmotic loading of cells. J Biomech 39: 464–475
Ateshian GA, Costa KD and Hung CT (2007). A theoretical analysis of water transport through chondrocytes. Biomech Model Mechanobiol 6: 91–101
Atkinson TS, Haut RC and Altiero NJ (1997). A poroelastic model that predicts some phenomenological responses of ligaments and tendons. J Biomech Eng 119: 400–405
Banes AJ, Tsuzaki M, Yamamoto J, Fischer T, Brigman B, Brown T and Miller L (1995). Mechanoreception at the cellular level: the detection, interpretation, and diversity of responses to mechanical signals. Biochem Cell Biol 73: 349–365
Belkoff SM and Haut RC (1992). Microstructurally based model analysis of gamma-irradiated tendon allografts. J Orthop Res 10: 461–464
Brown RA, Prajapati R, McGrouther DA, Yannas IV and Eastwood M (1998). Tensional homeostasis in dermal fibroblasts: mechanical responses to mechanical loading in three-dimensional substrates. J Cell Physiol 175: 323–332
Butler SL, Kohles SS, Thielke RJ, Chen C and Vanderby R (1997). Interstitial fluid flow in tendons or ligaments: a porous medium finite element simulation. Med Biol Eng Comput 35: 742–746
Chen CT, Malkus DS and Vanderby R (1998). A fiber matrix model for interstitial fluid flow and permeability in ligaments and tendons. Biorheology 35: 103–118
Diamant J, Keller A, Baer E, Litt M and Arridge RG (1972). Collagen; ultrastructure and its relation to mechanical properties as a function of ageing. Proc R Soc Lond B Biol Sci 180: 293–315
Frank EH and Grodzinsky AJ (1987). Cartilage electromechanics—II. A continuum model of cartilage electrokinetics and correlation with experiments. J Biomech 20: 629–639
Giori NJ, Beaupre GS and Carter DR (1993). Cellular shape and pressure may mediate mechanical control of tissue composition in tendons. J Orthop Res 11: 581–591
Guilak F and Mow VC (2000). The mechanical environment of the chondrocyte: a biphasic finite element model of cell–matrix interactions in articular cartilage. J Biomech 33: 1663–1673
Gupta T and Haut Donahue TL (2006). Role of cell location and morphology in the mechanical environment around meniscal cells. Acta Biomater 2: 483–492
Han S, Gemmell SJ, Helmer KG, Grigg P, Wellen JW, Hoffman AH and Sotak CH (2000). Changes in ADC caused by tensile loading of rabbit achilles tendon: evidence for water transport. J Magn Reson 144: 217–227
Han Y, Cowin SC, Schaffler MB and Weinbaum S (2004). Mechanotransduction and strain amplification in osteocyte cell processes. Proc Natl Acad Sci USA 101: 16689–16694
Hannafin JA and Arnoczky SP (1994). Effect of cyclic and static tensile loading on water content and solute diffusion in canine flexor tendons: an in vitro study. J Orthop Res 12: 350–356
Hannafin JA, Arnoczky SP, Hoonjan A and Torzilli PA (1995). Effect of stress deprivation and cyclic tensile loading on the material and morphologic properties of canine flexor digitorum profundus tendon: an in vitro study. J Orthop Res 13: 907–914
Hansen KA, Weiss JA and Barton JK (2002a). Recruitment of tendon crimp with applied tensile strain. J Biomech Eng 124: 72–77
Haridas B, Butler DL, Malaviya P, Boivin G, Awad H, Smith F (1999) Transversely isotropic poroelastic finite element simulations of the intact and surgically translocated rabbit flexor tendon. Proceedings of the 1999 ASME Summer Bioengineering Conference
Haut RC (1985). The effect of a lathyritic diet on the sensitivity of tendon to strain rate. J Biomech Eng 107: 166–174
Haut TL and Haut RC (1997). The state of tissue hydration determines the strain-rate-sensitive stiffness of human patellar tendon. J Biomech 30: 79–81
Helmer KG, Nair G, Cannella M and Grigg P (2006). Water movement in tendon in response to a repeated static tensile load using one-dimensional magnetic resonance imaging. J Biomech Eng 128: 733–741
Hewitt J, Guilak F, Glisson R and Vail TP (2001). Regional material properties of the human hip joint capsule ligaments. J Orthop Res 19: 359–364
Hurschler C, Loitz-Ramage B and Vanderby R (1997). A structurally based stress-stretch relationship for tendon and ligament. J Biomech Eng 119: 392–399
Ingber DE (1997). Tensegrity: the architectural basis of cellular mechanotransduction. Annu Rev Physiol 59: 575–599
Iwasaki H, Eguchi S, Ueno H, Marumo F and Hirata Y (2000). Mechanical stretch stimulates growth of vascular smooth muscle cells via epidermal growth factor receptor. Am J Physiol Heart Circ Physiol 278: H521–529
Jin G, Sah RL, Li YS, Lotz M, Shyy JY and Chien S (2000). Biomechanical regulation of matrix metalloproteinase-9 in cultured chondrocytes. J Orthop Res 18: 899–908
Johnson GA, Livesay GA, Woo SL and Rajagopal KR (1996). A single integral finite strain viscoelastic model of ligaments and tendons. J Biomech Eng 118: 221–226
Kastelic J, Palley I and Baer E (1980). A structural mechanical model for tendon crimping. J Biomech 13: 887–893
Kwan MK and Woo SL (1989). A structural model to describe the nonlinear stress-strain behavior for parallel-fibered collagenous tissues. J Biomech Eng 111: 361–363
Lai WM, Mow VC and Roth V (1981). Effects of nonlinear strain-dependent permeability and rate of compression on the stress behavior of articular cartilage. J Biomech Eng 103: 61–66
Lambert CA, Soudant EP, Nusgens BV and Lapiere CM (1992). Pretranslational regulation of extracellular matrix macromolecules and collagenase expression in fibroblasts by mechanical forces. Lab Invest 66: 444–451
Lanir Y, Salant EL and Foux A (1988). Physico-chemical and microstructural changes in collagen fiber bundles following stretch in-vitro. Biorheology 25: 591–603
Lavagnino M and Arnoczky SP (2005). In vitro alterations in cytoskeletal tensional homeostasis control gene expression in tendon cells. J Orthop Res 23: 1211–1218
Lavagnino M, Arnoczky SP, Tian T and Vaupel Z (2003). Effect of amplitude and frequency of cyclic tensile strain on the inhibition of MMP-1 mRNA expression in tendon cells: an in vitro study. Connect Tissue Res 44: 181–187
Lavagnino M, Arnoczky SP, Frank K and Tian T (2005). Collagen fibril diameter distribution does not reflect changes in the mechanical properties of in vitro stress-deprived tendons. J Biomech 38: 69–75
Lujan TJ, Underwood CJ, Henninger HB, Thompson BM and Weiss JA (2007). Effect of dermatan sulfate glycosaminoglycans on the quasi-static material properties of the human medial collateral ligament. J Orthop Res 25: 894–903
Mochitate K, Pawelek P and Grinnell F (1991). Stress relaxation of contracted collagen gels: disruption of actin filament bundles, release of cell surface fibronectin, and down-regulation of DNA and protein synthesis. Exp Cell Res 193: 198–207
Mullender M, El Haj AJ, Yang Y, Burger EH, Klein-Nulend J and Duin MA (2004). Mechanotransduction of bone cells in vitro: mechanobiology of bone tissue. Med Biol Eng Comput 42: 14–21
Ng CP, Hinz B and Swartz MA (2005). Interstitial fluid flow induces myofibroblast differentiation and collagen alignment in vitro. J Cell Sci 118: 4731–4739
Puxkandl R, Zizak I, Paris O, Keckes J, Tesch W, Bernstorff S, Purslow P and Fratzl P (2002). Viscoelastic properties of collagen: synchrotron radiation investigations and structural model. Philos Trans R Soc Lond B Biol Sci 357: 191–197
Redaelli A, Vesentini S, Soncini M, Vena P, Mantero S and Montevecchi FM (2003). Possible role of decorin glycosaminoglycans in fibril to fibril force transfer in relative mature tendons—a computational study from molecular to microstructural level. J Biomech 36: 1555–1569
Ritty TM, Roth R and Heuser JE (2003). Tendon cell array isolation reveals a previously unknown fibrillin-2-containing macromolecular assembly. Structure (Camb) 11: 1179–1188
Robinson PS, Lin TW, Jawad AF, Iozzo RV and Soslowsky LJ (2004). Investigating tendon fascicle structure-function relationships in a transgenic-age mouse model using multiple regression models. Ann Biomed Eng 32: 924–931
Scott JE (2003). Elasticity in extracellular matrix ‘shape modules’ of tendon, cartilage, etc. A sliding proteoglycan-filament model. J Physiol 553: 335–343
Stouffer DC, Butler DL and Hosny D (1985). The relationship between crimp pattern and mechanical response of human patellar tendon–bone units. J Biomech Eng 107: 158–165
Voet A (1997). A comparison of finite element codes for the solution of biphasic poroelastic problems. Proc Inst Mech Eng 211: 209–211
Wang JH (2006). Mechanobiology of tendon. J Biomech 39: 1563–1582
Wang JL, Parnianpour M, Shirazi-Adl A and Engin AE (1997). Failure criterion of collagen fiber: viscoelastic behavior simulated by using load control data. Theory Appl Fracture Mechs 27: 1–12
Weiss JA and Maakestad BJ (2006). Permeability of human medial collateral ligament in compression transverse to the collagen fiber direction. J Biomech 39: 276–283
Wellen J, Helmer KG, Grigg P and Sotak CH (2005). Spatial characterization of T1 and T2 relaxation times and the water apparent diffusion coefficient in rabbit Achilles tendon subjected to tensile loading. Magn Reson Med 53: 535–544
Wilson A, Shelton F, Chaput C, Frank C, Butler D and Shrive N (1997). The shear behaviour of the rabbit medial collateral ligament. Med Eng Phys 19: 652–657
Wilson W, van Donkelaar CC, van Rietbergen B, Ito K and Huiskes R (2004). Stresses in the local collagen network of articular cartilage: a poroviscoelastic fibril-reinforced finite element study. J Biomech 37: 357–366
Woo SL, Gomez MA, Woo YK and Akeson WH (1982). Mechanical properties of tendons and ligaments. II. The relationships of immobilization and exercise on tissue remodeling. Biorheology 19: 397–408
Woo SL, Livesay GA, Runco TJ and Young EP (1997). Structure and function of tendons and ligaments. In: Mow, VC and Hayes, WC (eds) Basic orthopaedic biomechanics, pp 209–251. Lippincott-raven, Philadelphia
Yasuda K and Hayashi K (1999). Changes in biomechanical properties of tendons and ligaments from joint disuse. Osteoarthritis Cartilage 7: 122–129
Yin L and Elliott DM (2004). A biphasic and transversely isotropic mechanical model for tendon: application to mouse tail fascicles in uniaxial tension. J Biomech 37: 907–916
You J, Yellowley CE, Donahue HJ, Zhang Y, Chen Q and Jacobs CR (2000). Substrate deformation levels associated with routine physical activity are less stimulatory to bone cells relative to loading-induced oscillatory fluid flow. J Biomech Eng 122: 387–393
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lavagnino, M., Arnoczky, S.P., Kepich, E. et al. A finite element model predicts the mechanotransduction response of tendon cells to cyclic tensile loading. Biomech Model Mechanobiol 7, 405–416 (2008). https://doi.org/10.1007/s10237-007-0104-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-007-0104-z