Abstract
Egg size is a critical life-history trait in which maternal investment is optimized to maximize maternal fitness. The adaptive significance of variable egg size among spawning groups of Ayu (Plecoglossus altivelis) landlocked in the Lake Biwa system was examined through field investigations and rearing experiments. Observed egg size variations were explained by the water temperature around spawning grounds established near the mouths of inlet streams. Two typical streams with different incubation temperatures showed similar maternal body sizes and hatchling sizes, but eggs attached to the stream bed were larger in the colder stream. An experiment that used eggs from a single clutch showed that a smaller hatchling size was obtained with a lower incubation temperature, indicating that the effect of differences in egg size on hatchling size can be canceled out by variations in incubation temperature. In general, larvae that are less than a certain threshold of effective body size are not expected to be assured of early success among conspecifics competing for foods. It is proposed that environments in which the incubation temperature varies favor variability in egg size to ensure that sufficient food is accessible to larvae.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Egg size is one of the most important life history traits, reflecting maternal investment and affecting offspring fitness (Roff 1992; Stearns 1992). Smith and Fretwell (1974) demonstrated that the tradeoff between size and number of offspring is optimized when offspring size maximizes the female’s total fitness return. Their model predicts that the optimum egg size within a population will vary if the relationship between egg size and offspring survivorship fluctuates with the local environment together with female characters. Egg size variation has been the subject of extensive research, much of which has focused on mechanisms that maintain variation within and between populations (Quinn et al. 1995). However, our understanding of the specific adaptability of variable egg size is still relatively poor, resulting in the need for empirical approaches for each target species.
The Ayu, Plecoglossus altivelis, is a common freshwater fish with an annual life history. The amphidromous and landlocked (Lake Biwa system) forms of the species differ in mean egg size (Azuma 1964; Iguchi and Yamaguchi 1994). Further, the egg size of the amphidromous form varies latitudinally, as observed among the 13 populations that cover its principal distribution area from 28 to 44°N (Iguchi 1993). The observed geographic variation in egg size can be plausibly explained by adaptation to different climatic conditions by different local populations on the basis of genetic divergence (Iguchi and Takeshima 2006). In the case of the landlocked form, the egg size variation occurs among spawning groups that comprise fish without a distinct genetic structure (Iguchi et al. 2002). Despite the fact that the spawning sites are located inside a narrow range of latitudes within the Lake Biwa system, the landlocked form of the fish show strong variability in egg size comparable to that seen for the amphidromous form (Iguchi and Kuwahara 1999). The adaptive significance of the egg size variation in the landlocked form has not yet been clarified.
Fish egg size is determined by the ability of females to enhance offspring survivorship, by changing environments, which affects early survivorship or interactions between the female ability and changing environments (Sargent et al. 1987). A previous study detected a negative correlation between stream length and mean egg size in the landlocked form of Ayu; mature females that ascended shorter streams appeared to lay larger eggs (Iguchi and Kuwahara 1999). Because spawning grounds that are established in lower courses close to the mouths of inlet streams give fish no opportunity to survey the entire stream, stream length cannot be responsible for determining egg size. Stream water temperature is a function of the distance from the source (Kitano et al. 1995). Since water temperature often affects early survivorship (Beacham and Murray 1990), changing water temperature can be regarded as a candidate for an environmental factor that explains the egg size variation in the landlocked form. It has been hypothesized that the landlocked form of Ayu lay larger eggs at lower water temperatures.
In general, larger hatchlings have advantages, such as superior competitive ability (Chapman 1962), decreased predator vulnerability (Folkvord and Hunter 1986), and more effective feeding (Knutsen and Tilseth 1985). Early survival after hatching should depend upon hatchling size but not upon egg size if egg size does not translate linearly into hatchling size (Coleman and Galvani 1998). The work described in this study aimed to determine the adaptive significance of egg size variation in the landlocked form. Egg size and larval size were investigated in inlet streams of the Lake Biwa system in relation to water temperature at the spawning sites. Using clutches of rearing fish, the eggs were incubated experimentally to check the hatchling sizes at different water temperatures.
Materials and methods
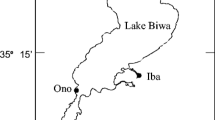
Ten streams along the west coast of Lake Biwa (the Shiotsuoh, Ohura, Chinai, Ishida, Imazu, Ado, Kamo, Ukawa, Hachiyado and Wani Rivers; Fig. 1) were selected to carry out synchronized investigations of water temperature and egg size on 20/21 September 1993/1994. The median maximum and minimum water temperatures were determined by monitoring the temperatures over 24 h with a maximum–minimum thermometer to estimate the incubation temperature around the spawning grounds. Pre-eyeing-stage eggs attached to substrates at the spawning grounds were collected by hand and fixed in 10% formalin for subsequent measurement. The average diameter of 100 randomly chosen eggs was regarded as the egg size of the spawning group.
The Ukawa and Ado Rivers (Fig. 1), which differ in scale and therefore potential water temperatures at spawning sites, were selected for a comparison of maternal size, egg size, and hatchling size of Ayu on 21 September 1993. Diel changes in water temperature recorded by a maximum–minimum thermometer at that time ranged between 15.4 and 16.8°C and between 15.3 and 18.8°C around the spawning grounds of the Ukawa and Ado Rivers, respectively. Adult females ascending the streams to spawn were collected using a cast net and immediately measured for standard length to the nearest 0.1 cm. Pre-eyeing-stage eggs attached to substrates at the spawning grounds were collected by hand and fixed in 10% formalin for subsequent measurement of diameter to the nearest 0.001 mm using a profile projector. Newly hatched larvae drifting in the lower course of the spawning site were collected using a drift net and fixed in 10% formalin for subsequent measurement of notochord length to the nearest 0.001 mm using a profile projector. The average values obtained by measuring 100 adult standard lengths, 100 egg diameters, and 100 larval notochord lengths (all chosen randomly) were regarded as the estimates for maternal size, egg size, and hatchling size for the spawning group, respectively.
Six-month-old Ayu were collected from Lake Biwa in May 1993. They were kept in outdoor ponds (1 × 5 × 0.5 m) at the Ueda Station, National Research Institute of Aquaculture, Nagano Prefecture, and fed assorted feed until maturation in order to obtain fish for experimental use. In late September, as the spawning season approached, pebbles were placed near the upper end of each pond with a 100 cm/s current to induce ovulation at the simulated spawning ground (Iguchi and Yamaguchi 1994). The mean temperature of the rearing water measured at 1000 hours changed monthly: 13.4, 17.8, 17.0, 18.0, 15.9, and 12.1°C in May, June, July, August, September, and October, respectively. On 26 October, three females that ovulated concurrently with a sufficient number of good-quality eggs were chosen for experiments investigating early development. After measuring the average diameters (in μm) of 100 water-hardened eggs from each clutch, the clutches were labeled A, B, and C, respectively. Eggs fertilized with milt from live males were randomly divided into three groups and incubated under a natural photoperiod in aerated water at 14, 17, and 20°C from 26 October. Just-hatched larvae were fixed in 10% formalin for subsequent measurements. Fifty randomly chosen larvae had their notochord lengths measured, and the average was taken to represent the estimate of hatchling size for each incubation category.
Results
Egg size variation was observed at the spawning grounds of ten inlet streams along the west coast of Lake Biwa in different sampling years, ranging from 796.0 to 863.1 μm in 1993 and from 805.6 to 854.6 μm in 1994 (Fig. 2; two-way ANOVA, streams, F 9,1980 = 88.852, P < 0.001; years, F 1,1980 = 6.041, P = 0.014). Water temperatures around the spawning grounds were not constant but differed among streams, ranging from 16.1 to 17.8°C in 1993 and from 18.0 to 22.7°C in 1994. The data from 1993 were only suggestive of a negative relationship between water temperature and egg size, probably due to the small difference in water temperature between the spawning grounds, whereas the data from 1994 presented a significant relationship, with comparatively large fluctuations in water temperature (Fig. 3; 1993, r = −0.572, n = 10, P = 0.084; 1994, r = −0.819, n = 10, P = 0.004). Egg size changed with the variation in water temperature between years (ANCOVA, water temperatures, F 1,17 = 12.802, P = 0.002; years, F 1,17 = 11.310, P = 0.004).
In the Ukawa River, as well as in the Ado River, adult females were the dwarf type of landlocked Ayu (so-called “Koayu”; Azuma 1964). The body size of this dwarf type was approximately uniform and showed no significant difference between the two streams (Fig. 4a; t test, t = 0.721, df = 198, P = 0.472). Females in the Ukawa River laid larger eggs on the stream bed than those in the Ado River (Fig. 4b; t = 18.575, df = 198, P < 0.001). In the same evening, drifting larvae caught in both the Ukawa and Ado Rivers hatched with similar body sizes (Fig. 4c; t = −0.459, df = 198, P = 0.646).
For all three clutches examined, it took 9, 13, and 20 days for the larvae to hatch in water maintained at 14, 17, and 20°C, respectively. An interaction between clutch and incubation temperature was detected (two-way ANOVA, F 4,441 = 62.121, P < 0.001), whereas, in each clutch, lower incubation temperature resulted in smaller hatchling size (Fig. 5; ANOVA, clutch A, F 2,147 = 90.966, P < 0.001; clutch B, F 2,147 = 47.108, P < 0.001; clutch C, F 2,147 = 75.965, P < 0.001).
Box plots of the notochord lengths of hatchlings incubated at different experimental temperatures (14, 17, and 20°C) within clutches A, B and C. Boxes encompass 50% of the data from the 25th to the 75th percentiles. The horizontal line indicates the sample median, bars encompass 90% of data from the 10th to 90th percentiles, and open circles indicate outliers. Bars with the same letters are not different at P = 0.05 (according to Games–Howell tests)
Discussion
Iguchi and Kuwahara (1999) demonstrated that the landlocked form of Ayu in the Lake Biwa system lay larger eggs in longer inlet streams even though the females that ascend the streams to spawn never know the full length of the stream. The present study confirmed a negative relationship between water temperature around the spawning ground and the size of eggs attached to substrates. The earlier prediction that stream scale (or distance from the source) affects the water temperature around the spawning grounds established near the stream mouth successfully interprets the former observation as resulting from a relationship between water temperature and egg size. Therefore, the opening hypothesis that the landlocked form lays larger eggs at lower water temperatures is verified. However, we have no idea how and when females control their egg size. Further study is needed to clarify the mechanism employed to produce landlocked Ayu eggs with a certain volume. The trend for larger individuals to develop in low-temperature environments holds almost universally in animals, including invertebrates (Atkinson 1994), and has been shown to be applicable to the eggs of the landlocked form of Ayu. Intrapopulation egg size variation in Ayu is explained by the body sizes of females that share a spawning ground (Iguchi and Yamaguchi 1994). In the present study, the influence of maternal size on egg size is negligible because of the similarity in body size of females belonging to the dwarf type, or “Koayu” (Azuma 1964).
Captive Ayu reared under common conditions preserve differences in egg size between the amphidromous and landlocked forms, which suggests the importance of a genetic influence on egg size at the higher intraspecific level (Iguchi and Yamaguchi 1994). If we consider only the landlocked form that dwells in native habitats, however, there must be another mechanism that determines egg size because each spawning group lacks a distinct genetic structure (Iguchi et al. 2002). This supported by the finding that geographic distance (distance between the mouths of inlet streams) inadequately explains differences in egg size between spawning groups (Iguchi and Kuwahara 1999). Ayu in the Lake Biwa system are only found in habitats within a narrow range of latitudes, but environmental conditions such as water temperature vary considerably between the spawning grounds of different streams. Species that disperse passively through water currents find it difficult to predict the environmental conditions that their offspring will encounter (Einum and Fleming 2002). Thus, in contrast to a genetic control of egg size, determining egg size through individual plastic response might be a more advantageous way to enhance offspring fitness in a changing environment.
If all conditions are the same, egg size will be parallel to hatchling size. The occurrence of dissimilar egg sizes and similar hatchling sizes in the Ukawa and Ado Rivers implies different environmental condition regimes during the early development of Ayu. The larger eggs obtained in the colder running water of the Ukawa River suggest a process that restrains hatchling size at lower incubation temperatures. The experiment that used eggs from a single clutch of landlocked Ayu showed diminishing hatchling size as the incubation temperature decreased, which explains the mismatch in the link between egg size and hatchling size observed simultaneously in the two streams. Experimental use of amphidromous Ayu results in the detection a tendency similar to that seen in the present study for landlocked fish (Iguchi, unpublished data). In addition, a trend for increasing egg size with decreasing incubation temperature has been observed for discrete oocyte batches from a particular individual Ayu, whether amphidromous or landlocked (Iguchi and Yamaguchi 1994). Thus, it is likely that larger eggs are counterbalanced by a lower water temperature, so that larvae will not hatch with larger body sizes but with ordinary body sizes. In general, the evolution of egg size variation is driven by selection favoring offspring with different performances in different environments (Fox and Czesak 2000). In this context, egg size selection in the landlocked form seems rather distinctive, since the goal for eggs that experience different environments is to produce offspring that are not different but equivalent in performance.
Organisms living in a given environment should be adaptive, allocating their available resources into eggs of an optimum size (Smith and Fretwell 1974). It is notable that the production of larger eggs leads to lower fecundity. However, the advantage of a larger egg size for offspring fitness is capable of compensating for this potential loss of fecundity. In the landlocked form of Ayu, just-hatched larvae start drifting downstream, which is followed by mass recruitment into a common habitat that occurs across Lake Biwa (Azuma 1970). Larvae of Ayu have to depend upon zooplankton (Sakai 2011), while the planktonic foods available for them are often limited by the size distribution of zooplankton (Okamura 2008). Larvae with small body sizes have insufficient swimming abilities (Tsukamoto et al. 1975), and their small mouths make it difficult to take in relatively large food items (Fukuhara and Fushimi 1986), both of which lead to inferior foraging efficiency compared to that displayed by larger larvae. Poor growth yields low fitness and thus weakly competitive larvae. An effective body size that is not below the survival threshold is expected to assure early success amongst conspecifics competing for foods. In conclusion, environments with varying incubation temperatures may have caused the landlocked form of Ayu to adaptively adjust its egg size to ensure that sufficient food is accessible to larvae. For a comprehensive understanding of its phenotypic plasticity, a common garden experiment should be carried out to indentify the role of genetic components in determining the egg size of landlocked Ayu in Lake Biwa.
References
Atkinson D (1994) Temperature and organism size—a biological law for ectotherms? Adv Ecol Res 25:1–18
Azuma M (1964) The life history of the landlocked Ayu-fish (Plecoglossus altivelis) in the Lake Biwa, with special reference to their developmental stages. Physiol Ecol Jpn 12:55–71
Azuma M (1970) Studies on the variability of the landlocked Ayu-fish, Plecoglossus altivelis T. et S., in Lake Biwa I. On the mode of distribution and some body form variation at early phases of development. Jpn J Ecol 20:63–76
Beacham TD, Murray CB (1990) Temperature, egg size, and development of embryos and alevins of five species of Pacific salmon: a comparative analysis. Trans Am Fish Soc 119:927–945
Chapman DW (1962) Aggressive behavior in juvenile coho salmon as a cause of emigration. J Fish Res Board Can 19:1047–1080
Coleman RM, Galvani AP (1998) Egg size determines offspring size in neotropical cichlid fishes. Copeia 1998:209–213
Einum S, Fleming IA (2002) Does within-population variation in fish egg size reflect maternal influences on optimum values. Am Nat 160:756–765
Folkvord A, Hunter JH (1986) Size-specific vulnerability of northern anchovy, Engraulis mordax, larvae to predation by fish. Fish Bull US 84:859–870
Fox CW, Czesak ME (2000) Evolutionary ecology of progeny size in arthropods. Ann Rev Entomol 45:341–369
Fukuhara O, Fushimi T (1986) Development and early life history of Ayu reared in the laboratory. Bull Jpn Soc Sci Fis 52:75–80
Iguchi K (1993) Latitudinal variation in ayu egg size. Nippon Suisan Gakkaishi 59:2087
Iguchi K, Kuwahara M (1999) Egg size variation in landlocked ayu from Lake Biwa system. Fish Sci 65:790–791
Iguchi K, Takeshima H (2006) Progress in analyzing fish population structure in ayu with today’s significance. Bull Fish Res Agen Suppl 5:187–195
Iguchi K, Yamaguchi M (1994) Adaptive significance of inter- and intrapopulational egg size variation in ayu Plecoglossus altivelis (Osmeridae). Copeia 1994:184–190
Iguchi K, Ohkawa T, Nishida M (2002) Genetic structure of land-locked ayu within the Biwa Lake system. Fish Sci 68:138–143
Kitano F, Nakano S, Maekawa K, Ono Y (1995) Effect of stream temperatures on longitudinal distribution of fluvial Dolly Varden and potential habitat loss due to global warming. Wildl Conserv Japan 1:1–11
Knutsen GM, Tilseth S (1985) Growth, development, and feeding success of Atlantic cod larvae Gadus morhua related to egg size. Trans Am Fish Soc 114:507–511
Okamura T (2008) Long-term changes in the water quality of Lake Biwa. Bull Shiga Pref Fish Exp St 52:33–41
Quinn TP, Hendry AP, Wetzel LA (1995) The influence of life history trade-offs and the size of incubation gravels on egg size variation in sockeye salmon (Oncorhynchus nerka). Oikos 74:425–438
Roff DA (1992) The evolution of life histories. Chapman and Hall, New York
Sakai A (2011) Factors affecting size fluctuation of landlocked Ayu Plecoglossus altivelis. Aquacult Sci 59:299–306
Sargent RC, Taylor PD, Gross MR (1987) Parental care and the evolution of egg size in fishes. Am Nat 129:32–46
Smith SS, Fretwell SD (1974) The optimal balance between size and number of offspring. Am Nat 108:499–506
Stearn SC (1992) The evolution of life histories. Oxford University Press, New York
Tsukamoto K, Kajihara T, Nishiwaki M (1975) Swimming ability of fish. Bull Jpn Soc Sci Fish 41:167–174
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Iguchi, K. Larger eggs at lower water temperature as a measure to assure effective hatchling size in the landlocked form of Ayu, Plecoglossus altivelis . Ichthyol Res 59, 20–25 (2012). https://doi.org/10.1007/s10228-011-0251-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10228-011-0251-1