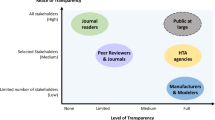
The introduction of health economic guidelines in various European countries allows partially different interpretations how to generate a model und which data, used within the model will be acdcepted by payers. With this survey we wanted to investigate in different European countries, to what extent models are considered in the decision making process concerning pricing and reimbursement of new drugs or devices, which role they might possibly play in the future and what the requirements are from a payer's and from a Health Economist's point of view.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Rights and permissions
About this article
Cite this article
, . Modeling – what is accepted by the authorities? . HEPAC 1, 44–46 (2000). https://doi.org/10.1007/s101980050009
Issue Date:
DOI: https://doi.org/10.1007/s101980050009