Abstract
Versatile predatory tactics might favor success during intra-guild predation. In the case of spiders, the preditory spitting spiders (Scytodidae) invade webs and feed on certain weaver spiders. Based on preliminary observations of Scytodes globula, we tested if this spider species as predator could feed on the sympatric hacklemesh weaver Metaltella simoni (Amphinectidae) as host, or if the host spider could prevent the attack. We exposed adult females of M. simoni on 2-week-old webs to adult female spitting spiders. The spitting spider preyed on the host spider in 28 % of the 72 trials in which the two spider species interacted. Leaving the retreat, approaching the predator, and touching the invader apparently made the host spider vulnerable to predation in 55 % of those interactions; therefore, those were considered risky behaviors. To the contrary, the host spider had a success rate of 67 % of surviving predation by performing defensive behaviors (moving hind legs, performing defensive displays). In four trials, the invader performed a defensive spit to deter the attack of the host spider, which points to the versatility of this trait. We found no effect of body size ratio of the spiders on the outcome of the interactions. The host spider left the retreat after the spitting spider entered its web, suggesting that the spitting spider could be performing aggressive mimicry, but notion is supported by only indirect evidence and needs further exploration. In summary, behavioral interplay mediated the outcome of the interactions between the two coexisting predators assessed in our study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Two or more coexisting species of predators can impose a selective pressure on each other, although the pressure imposed in such intraguild predation (IGP) interaction may be asymmetrical (Polis et al. 1989; Arim and Marquet 2004). Spiders are a specific predatory group in which IGP is expected to occur in close sympatry. Predatory strategies in spiders are diverse, versatile and plastic (Nelson and Jackson 2011, 2012). Araneophagy (attacking and feeding on/eating spiders) is a widespread and common tactic in many spider groups (Jackson 1992; Wise 1993; Jackson and Pollard 1996). Spiders predate on other spiders by actively hunting and aggressively attacking them in their own webs or by chasing and/or ambushing them on a solid substrate (Nelson and Jackson 2012). Spiders that invade webs and attack and successfully prey on the hosts are members of many spider families, including Gnaphosidae (Jarmar and Jackson 1986), Pholcidae (Jackson and Brassington 1987), Mimetidae (Jackson and Whitehouse 1986), Oxyopidae (Gonzaga et al. 1998), Salticidae (Nelson and Jackson 2012), and Scytodidae (Li 1996), as well as in kleptoparastic Theridiidae species (Whitehouse 1987; Whitehouse et al. 2002). Araneophagic spiders can on occasion vibrate the web of the host species (aggressive mimicry; Herberstein and Wignall 2011) in imitation of the vibrations made by conspecific males (Jackson and Wilcox 1990) or by potential prey entangled in the web (Li 1996).
The spitting spiders (Scytodidae) are notable among arachnids because they bear an enlarged cephalothorax with secondary modified venom glands that produce adhesive fluids and contractive silk threads which the spider spits at their prey to restrain them and allow a more controlled access before biting and injecting them with venom (Li et al. 1999; Ramires 1999; Ades and Ramires 2002; Clements and Li 2005; Suter and Stratton 2009; Ades et al. 2010). This spitting behavior is also used as a defensive mechanism (Gilbert and Rayor 1985; Jackson et al. 1998, 2002). The ‘saliva’ itself does not contain venom, as evidenced by the survival of attacked animals when biting has been prevented and after they have extracted themselves from the spit silk threads by grooming (Clements and Li 2005). After spitting, Scytodes spiders can perform an alternating movement with the pair of hind legs to wrap up their prey, while holding it with the second and third pair of legs (Gilbert and Rayor 1985; Barrantes and Eberhard 2007). The spitting spiders eat various spider species, and cannibalism has also been registered in S. longipes (Nentwig 1985; Li et al. 1999). The predatory behavior of the spitting spiders is plastic: when S. pallida attack bigger and more vibrating prey, they spit more times before the bite than when they attack smaller prey (Clements and Li 2005).
Scytodes globula Nicolet 1849 inhabits urban, semi-urban, and natural areas in Brazil, Argentina, and Uruguay (Aisenberg et al. 2011; Carvalho et al. 2012). In Uruguay, this species occurs in houses, buildings, gardens, parks, meadows, and agricultural fields, as well as natural forests and rocky areas. Prey-capture behaviors of S. globula have recently been described (Carvalho et al. 2012). This spider moves slowly when it encounters a prey and raises its first and second pair of legs, sometimes touching the potential prey (Carvalho et al. 2012), and then it spits and bites the prey. Scavenging has also been reported in this species (Vetter 2013). S. globula has been occasionally observed to consume harvestmen and spiders in natural conditions in Brazil and Uruguay (Carvalho et al. 2012; personal observations of the authors). In Uruguay, S. globula naturally inhabits the same tree bark and leaf litter as cribellate web-builder spiders, such as Badumna longiqua Koch 1867 (Desidae), Metaltella simoni Keyserling 1878 (Amphinectidae), and Kukulkania hibernalis Hentz 1842 (Filistatidae) (Simó et al. 2011).
We have occasionally seen S. globula approach a web of the hacklemesh weaver spider M. simoni and vibrate the web threads (F.G. Costa, unpublished data). This stimulus elicited the response of the host spider which came out of its retreat and subsequently was attacked by S. globula. We have also seen M. simoni responding to the presence of S. globula in their webs. Hence, both spider species may be predator or prey in the same interaction, a concept that has received only little attention from researchers (see Li 1996).
The main aim of our study was to test whether the spitting spider (S. globula) is able to predate on the hacklemesh spider (M. simoni) by capturing it on the latter’s own web. Additional aims were to investigate the capture success of the spitting spider and to elucidate if behavioral and morphological (size ratio between the spiders) features affect that outcome. Also by exposing the host spiders to different vibratory cues in their respective web, we tested if the spitting spider performs aggressive mimicry. Finally, we examined the patterns of both prey capture and sexual behavior of this hacklemesh spider, which are unknown, to further our understanding of the components of the interactions between both spider species.
Materials and methods
Populations and study site
We collected spiders of both species (S. globula and M. simoni) from August to November 2012 in the provinces of Canelones (Parque Roosevelt, Marindia, and Piedras de Afilar), Lavalleja (Sierra de Minas and Monte de Ombúes de Arequita), and Montevideo, Uruguay. Individual spiders of both species were searched for under logs, rocks, and bricks, leaf litter, as well as under tree bark, up to 2.5 m aboveground in eucalyptus, pine, and cypress trees. The host (M. simoni) and spitting spider (S. globula) widely coexist in these areas, and on five occasions during field surveys we saw the two species under/on the same substrate (<15 cm apart), mostly on eucalyptus trees and underneath rocks. Additionally, it was common to find both spiders in the same tree at all the sites. Interactions between these two species in the field were registered by one of the authors (FGC, unpublished data). In one case, the spitting spider was feeding on the host spider in its web. In another case a spitting spider was at the edge of the host’s web; after we threw a housefly in the web, both spiders reacted, the spitting spider spat at the host spider and subsequently captured the housefly, while the host spider moved backwards.
The web of M. simoni consists of a cylindrical retreat with a thin sheet of cribellated silk threads and many loose threads attached to the substrates (the leaf litter or gaps between rocks). The retreat consists of a very dense cylinder funnel with a tangled mesh of hackle silk threads, with one or two entrances at the base or under the bark of the tree (height from the ground 0.0–1.5 m). This general structure of the webs in the field approximates those spun by M. simoni under laboratory conditions (Fig. 1). In the two dozen of webs collected in the field we observed many legs, heads, and dorsal parts of ants (probably Acromyrmex sp. and Camponotus sp.), the remains of spiders (mostly legs and chelicerae) from at least two unidentified species, as well as Diptera wings, Isoptera heads and mouth parts, and Coleoptera elytra, indicating that M. simoni feeds on a wide variety of prey. Aisenberg et al. (2011) noted that M. simoni approaches prey entangled in the web, bites it, and takes it to the retreat without wrapping the prey.
Prior to the trials, the spitting spiders were individually housed in petri dishes, and the host spiders were placed in adapted plastic bottles (see Fig. 1 and section Experimental design), both with a cotton ball soaked in water twice a week to maintain humidity. Individuals were kept under laboratory conditions [mean ± standard deviation (SD): 22.6 °C ± 2.7 (range 15–29 °C); 68.1 ± 5.6 % relative humidity (RH) (range 62–81 %]. Host spiders were fed once a week with a worker termite (Nasutitermes fulviceps Snyder 1949) until 1 week before the trial period. The spitting spiders were fed with a housefly every 2 weeks until 2 weeks before the trials. The feeding regimes were different because of the size differences of the prey offered, but also because under laboratory conditions it was more convenient to offer these different prey types to each spider. Additionally, in the field, the remains of termites have often been observed in the webs of M. simoni. No other prey was offered to any spider after these imposed 1- and 2-weeks, respectively. Ultimately, by controlling the starvation levels we standardized the motivation of the spiders of both species to approach and attack a potential prey (Clements and Li 2005).
Experimental arena in which adult females of the host spider Metaltella simoni were placed to allow web and retreat construction. The round tube placed into the right side of the container is where the spitting spider Scytodes globula was positioned to enter the bottle, after removal of a small petri dish that covered the bottle mouth. Web indicates the place above the sand and rocks where the host spider built the web and where most silk threads occurred
The individual spiders tested in the experimental setup were mostly newly matured virgin adult females raised in the laboratory (87 %), but we also tested recently molted penultimate females raised in the laboratory and or adult females collected from the field (potentially already mated). We did not observe any clear pattern between outcome of the interaction and the category of spiders in the trial (mature when collected or raised in the laboratory).
Experimental design
A single host spider was placed into one container 2 weeks before the trial. These containers were modified transparent plastic bottles (volume 600 ml; dimensions 22 cm high × 7 cm wide) im a horizontal position (for more details, see Coelho et al. 2010). A 6 × 10-cm window was constructed in the upper horizontal surface of the container, which could be opened during the trials to provide a clear view for observations and video recording. The substrate on the floor of the bottle consisted of a 2-cm-deep layer of white sand with small stones to allow spiders to attach threads (Fig. 1). The bottle opening was covered with liquid petrolatum (Hronsky 2006) in its inner side (also used on the inner surface of the window) to prevent the spiders from attaching threads at these positions.
Preliminary observations showed that a 2-week period was sufficient time for M. simoni to build a web with a retreat and to be willing to response to vibrations on its web. Before each trial, we recorded the web structure and noted the number of threads attached to the substrate and bottle. For each trial, we placed a randomly chosen spitting spider in a plastic tube (5 long × 2 cm diameter) with both sides blocked with caps. After 30 min, we removed the caps and placed the tube in the opening of the bottle. We waited a maximum of 30 min for the spitting spider to exit the tub spontaneously and walk into the bottle and towards the host’s web (spitting spiders tend to remain still for long periods of time). If the spitting spider did not exit the tube within this 30-min period, we gently touched one hind leg with the tip of a forceps and waited an additional 30 min. If after the second 30-min period the spider had not left the tube, the trial was suspended. Since the spitting spider is mostly a nocturnal hunter, we performed the interactions in a dark room, illuminated with red light, after 17:00 h.
Each trial was run until either one of the spiders attacked the other, the spitting spider lost interest in approaching the host spider’s web, or neither spider reacted within a 30-min period. We assumed that the spitting spider was not willing to approach and or to attack the host spider if it left the web after touching the threads and put itself in the retracted leg position (Nentwig 1985), or if it left the bottle through the tube. Preliminary trials had shown that host spiders would not come out of their retreats when the spitting spider was walking in the web. Hence, if the spiders did not interact within 30 min of trial initiation we stopped the trial. In these cases and before the spitting spider was removed from the arena we threw a housefly into a midpoint of the web to observe if the spiders were willing to respond to vibrations in the web. We observed that in all cases both spiders responded by changing their body orientation and/or approaching to the vibration source, although they did not interact, suggesting that both spiders were willing to attack a prey in the web but not engage in a direct interaction.
To elucidate if body size of either spider was associated with the outcome of the interactions, 30 min after each trial we took three photographs of each animal with a Nikon Coolpix P100 digital camera (Nikon Group, Tokyo, Japan) through a dissecting microscope and measured the cephalothorax size in accordance with Clements and Li (2005). To calculate the cephalothorax area we used the software program ImageJ (Abràmoff et al. 2004) to measure (1) the maximum width and length of the cephalothorax and (2) its perimeter. We therefore obtained six values of cephalothorax area for each spider (since every spider was photographed three times). These six values were averaged to obtain an indicator of each spider’s body size.
Video recordings and analyses
We registered the details of the behavior of both species from video recordings made with a Sony Handycam DCR-SR87 digital camera (Sony Corp., Tokyo, Japan) and quantified the frequency and duration of the behavioral units (of both spiders, see below). We constructed one sequence of behaviors (ethogram) of both individuals similar to Soley and Taylor (2012), based on the behavioral units and situations noted (Tables 1, 2).
Response behavior of the host spider
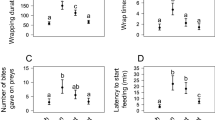
To investigate if the spitting spider performs aggressive mimicry in the web of the hacklemesh spider we compared the response of the host spider to three types of stimuli: (1) the spitting spider (described above, n = 72), (2) conspecific male (n = 8), and (3) potential prey (n = 5 with each of three types of prey). We released worker termites (N. fulviceps), houseflies (Musca domestica Linnaeus 1758), and leaf cutter ants (Acromyrmex sp.) in the web, near the opening of the retreat, subsequently video-recording the attack and recording the behaviors used by the spider to attack different types of prey. We also observed the approaching behavior of adult females of the host spider to conspecific males introduced in the bottle; on every occasion, the male walked towards the female in the web.
Statistical analyses
Chi-square tests were applied to determine if a transition from one behavior to another in the matrix (composed by all the behaviors of both species; Tables 1, 2) was more frequent than the rest of the transitions. To test if the size difference between individuals of both species is associated with the capture success of the spitting spider, we performed a logistic regression test using the size ratio of both individuals (mean cephalothorax area of S. globula/mean cephalothorax area of M. simoni) in the interaction as predictor variable and interaction outcome (predation/no predation) as response variable. The same analysis was performed using (1) spitting action versus no spitting action, (2) attack attempt by the spitting spider versus no attack attempt, and (3) host spider leaving its retreat versus not leaving as response variables. Data were analyzed with the statistical packages PAST 1.18 (Paleontological Statistics) software (Hammer et al. 2001) and NCSS 2001 (copyright 2000 Jerry Hintze).
Results
Observed behaviors during interactions between invader and host spiders
We recognized 12 behaviors performed by the spitting spider (Table 1) and 11 performed by the host spider (Table 2). The outcome of the interactions between the two spiders varied, and we identified seven types of interactions [three of which are shown in the video-recording in Electronic Supplementary Material (ESM) S1] (Table 3). In almost half (45.8 %) of the 72 interactions, the spitting spider entered the arena, touched the threads of the host spider, and walked on them—but did not interact with the host spider, or performed only a single, non-aggressive interaction (Fig. 2). In 13 cases the host spider did not come out of its retreat, and the spitting spider did not seem to notice the presence of the host spider (did not change its body orientation, neither walking towards the host nor searching for it). In 16.7 % of the total interactions the spitting spider walked directly towards the retreat and the host spider left the web and/or performed defensive displays. In the remaining 37.5 % of cases, the spitting spider spat at the host spider: in four cases apparently as a defensive tactic (video ESM S1; Table 1) and in 23 cases as an attempt to capture the host spider (Fig. 2). The spitting spider was successful in preying on the host spider in 11 of these latter 23 cases (representing 47.8 % of the direct attempts of feeding). The spitting spider was successful in preying on the host spider in 28.3 % of the 39 interactions in which the spiders oriented towards each other. The defensive displays of the host spider (legs and body movements; Table 2) seemed to discourage the attack by the spitting spider in 29.2 % of the cases, either in the first instance (“host spider’s resistance” followed by “defensive spit”, 12.5 %; Table 3; video ESM S1) and even after an aggressive spit (“spat but did not predate”, 16.7 %; Table 3; Fig. 2).
Outcomes of the 72 interactions between the spitting spider Scytodes globula and the hacklemesh weaver Metaltella simoni (host spider). See Table 3 for a more detailed description of the seven types of outcomes
Behavioral sequences and differences in body sizes
Overall, the outcome of the interactions seemed to be affected by the behaviors of both spiders (ESM video S1; Fig. 3). In many trials the movements of the spitting spider walking on the silk threads were followed by a movement from the host spider—moving inside the retreat, emerging from the retreat, and/or walking on the web. If the host spider left the retreat and met the spitting spider, the host spider performed plucks of the web (pluck behavior), waving movement of the forelegs (wave foreleg), and potentially defensive displays towards the spitting spider (leg wave defensive display) (ESM video S1; Table 2; Fig. 3). These behaviors caused the spitting spider to react differently, remaining motionless, walking away, waving forelegs, making a defensive spit, or making an aggressive spit (Table 1; Fig. 3). Whether or not the spitting spider attempted an attack immediately or remained near the host spider, the aim of the latter spider was most a return to its retreat and/or escaping the interaction by walking away from the retreat or the web and exiting the arena (Table 2; Fig. 3). A successful attack by the spitting spider was preceded by behaviors of the host spider, such as pluck, wave foreleg, and leg wave defensive display (Fig. 3). To the contrary, if the host spider remained in its retreat or went back to it having first exited it, attack or predation was unlikely. Nonetheless, certain strong movements by the host spider allowed it to escape after a spit. The 12 cases in which the spitting spider spat but did not predate the host was followed by active squirming of the host spider (Table 2), which may have allowed it to free itself from the spat threads (Fig. 3). In terms of location, in six of the 11 trials (55 %) in which the spitting spider successfully predated, the host spider was outside of the retreat and motionless in the web before the spitting spider entered the arena; whereas only in nine of the 61 trials (15 %) in which the spitting spider was unsuccessful was the host spider outside the retreat (proportion X 21 = 5.61, p = 0.02).
Ethogram of the interactions of the spitting spider S. globula (Scytodidae) and the hacklemesh weaver M. simoni (Amphinectidae). The behavioral units indicated in squares and ovals are those performed by S. globula (predator) and M. simoni (host), respectively. Thickness of arrows are proportional to the frequency of the transitions of one behavioral unit leading to the next one. Below The 10–15 % transitions (10 % was the cut-off point) and the 100 % transitions. Total = 72 interactions. See text and Tables 1–3 for description of the behavioral units
Our analysis of the size ratio of the spiders (mean S. globula cephalothorax area/mean M. simoni cephalothorax area) did not reveal any clear association with the outcome of the interactions. This measure apparently did not affect the probability of the spitting spiders to spit, predate, or cease the attack towards the host spider (Fig. 4). The size ratio of the spiders also did not affect the probability of the host spider to successfully defend itself or not defend itself (host spider’s resistance) (Table 3; Fig. 4), by inducing the spitting spider to make a defensive spit and/or to abandon the attack.
Outcome of the 72 interactions between the spitting spider S. globula (Scytodidae) and the hacklemesh weaver M. simoni (Amphinectidae) according to their size ratio (mean S. globula cephalothorax area/mean M. simoni cephalothorax area; see Materials and methods section for further description of the measures). Each figure part represents a different scenario resulting from the interactions. See Results section and Table 3 for a detailed description of the outcomes. Logistic regressions did not indicate an association of the size ratio on the outcome: a X 21 = 0.002, p = 0.98, odds ratio (OR) = −0.71, b X 21 = 0.36, p = 0.55, OR = −1.20, c X 21 = 1.87, p = 0.17, OR = −0.23, d X 21 = 1.09, p = 0.30, OR = −0.61
Behavioral responses of the host spider to conspecific mates and potential prey
Conspecific males
The females of the host spider seldom seemed willing to leave the retreat (or even change its body orientation) when a conspecific male spider was walking or standing still in the web close to the retreat or performing a series of quick taps on the threads in the web with its pedipalps. In three of the 12 trials with conspecific males, the female changed its body orientation in the retreat 30–40 s after the male contacted silk threads, possibly following by the waving of its forelegs alternately in the entrance of the retreat (Table 2). In two cases the female left the retreat quickly, but then stopped and remained still <2 cm away from the edge of the retreat. This behavior was followed by a series of short steps and quick touches by the female on the web with its pedipalps (those touches were followed by male’s touches to the web). The movement of the M. simoni male walking on the web seemed to cause a more conspicuous deflection of the web than the movement of the predator spitting spider. On two occasions the female left the retreat and after making quick contact with the male’s legs and/or dorsum, she turned abruptly and went back to the retreat. In two trials the female approached the male, and after he made various series of vibrations on the threads with its pedipalps, both spiders put together their legs and ventral side of their bodies, made three or four turns and then separated, with the female returning to the retreat. Three other series of similar vibration were followed by a mating, all inside the retreat.
Potential prey
During the initial 2 s following the contact of a potential prey on the web threads, the host spider changed its body orientation in the retreat so that it was oriented toward the prey. The spider then left its retreat, met the prey, and attacked it, but the speed of leaving the retreat, of moving towards the web sheet, and of physical contact with the potential prey varied. For example, when the prey consisted of ants and termites, the spider left the retreat slowly (10 s) and walked into the sheet slowly. Its movements seemed cautious: it made short and slow steps (compared to the approach to flies; see below), alternating them with 2- to 3-s-long pauses. The spider touched the prey many times with both pairs of forelegs. To the contrary, when the prey was a housefly, the spider left the retreat quickly and made contact with the prey in <1 s after the prey touched a thread. The flies seemed to cause strong vibrations to the web with its wings.
While facing towards any type of prey the spider waved its forelegs very quickly (Table 2), after which it touched the prey with its forelegs and pedipalps. Following the touches, the spider made a strong bite, which commonly secured the prey to its mouth parts (especially when attacking flies) and quickly took the prey back to the retreat, entering with a backwards movement of its body. We never saw the spider performing the wrapping behavior in any of the 15 attacks to each prey type.
Discussion
We found that a behavioral interplay affected the outcome of the interaction between the predator spitting spider and the host hacklemesh spider, and not size ratio. Our data show that when the host engaged in risky behaviors—namely, when it did not perform aggressive behaviors and when it left the retreat—it became more vulnerable in 55 % of the interactions to an attack and, consequently, to predation. To the contrary, aggressive defensive behaviors by the host spider, such as leg waving displays and moving forelegs, favored its survival, with a 67 % success rate when defensive behaviors were performed. Additionally, such defensive behaviors caused the spitting spider to switch its predatory attempt into a predatory avoidance status, by walking away or even performing a defensive spit. The web’s retreat played a crucial factor in host spider survival because it provided a hiding place for the host spider to survive a web invasion. The predatory avoidance function of the retreats has been suggested for other sheet and cob weaver spiders (Konigswald et al. 1990; Riechert and Hedrick 1990; Henschel and Jocqué 1994). Hence, remaining still in the retreat after sensing the vibration of the spitting spider walking on the web seems to be a beneficial response of the host spider. Several times we observed that the host spider returned to the retreat after making contact with the invader; ultimately, it escaped the retreat and abandoned the web quickly. The defensive strategies (remaining in the retreat, performing aggressive displays, and escaping the web) did not seem to follow a particular sequence, and M. simoni may perform them depending on the invader’s behavior.
In terms of size, the interaction resulted in any outcome irregardless of whether the spitting spider was smaller, larger, or of similar size than the host spider. For example, we registered that one spitting spider preying on a host spider double its size. It is possible that spitting allows these spiders to pursue and attack larger and dangerous predators. The spitting spider may evaluate its chances of a successful attack on the basis of the behaviors of the host spider, as suggested by our results.
We acknowledge that our study was unable to provide substantial information from the field; nonetheless, the anecdotic observations, the fact that these spiders do coexist, and the laboratory trials we conducted suggest that the spitting spider S. globula has the potential to impose predation pressure on the host spider M. simoni. Our main argument is that the capture success of the spitting spider was close to 30 % in our trials. This predatory success appears to be high considering that both species are predators of a similar size. Our results meet the pattern reported for IGP in carnivores (either top or intermediate, the case of our spiders) of approximately 30 % (Polis et al. 1989; Arim and Marquet 2004). Yasuda et al. (2001) studied IGP in two predatory ladybird species (Coleoptera) and found an asymmetrical predatory success: very low (<20 %) for Coccinella septempunctata Linnaeus 1758 on Harmonia axyridi Pallas 1773, and high (~60 %) in the inverse case. What is certainly novel from our results is that the spitting spider was never predated. Therefore, our experimental scenario could represent a case of extreme asymmetric predatory success during IGP; however, further studies are required to test this hypothesis.
The host spider showed alternative defensive behavioral tactics to avoid interacting with and being predated by the spitting spider. The complex behavioral interaction we observed between these two spiders could be the result of a long coexistence during evolutionary time. The current coexistence of these two predator spider species suggests extended ecological and behavioral interactions (see Denno et al. 2004; Perkins et al. 2007). Ingram et al. (2012) studied IGP between fishes (the prickly sculpin Cottus asper Richardson 1836 capturing the three-spined stickleback Gasterosteus aculeatus Linnaeus 1758) under laboratory conditions and found less predatory success on the stickleback fishes from populations that coexist with sculpins than those that did not coexist, suggesting adaptive anti-predatory responses in the prey. To test the hypothesis of IGP predation affecting the ecology of both species, future research should focus on (1) the predation pressure of the spitting spider in field conditions, and (2) the abundance of the host spiders in conditions of coexistence with the spitting spider compared to sites in which they do not coexist.
A potential mechanism involved in predatory avoidance by M. simoni could be the fine-scaled and precise discrimination of vibratory cues on its web (Masters et al. 1986; Landolfa and Barth 1996; Blamires et al. 2011). This was evident when we registered that the reaction of the host spider varied between, and even among, different vibrations in its web (spitting spider, conspecific males, and potential prey). Responding with the appropriate behavior to each cue seemed to be crucial for M. simoni, and such cautiousness may be a mechanism to prevent the occurrence of aggressive mimicry of the spitting spider reported in other web-building spiders (Jackson and Wilcox 1990; Li 1996).
We did not observe explicit behavioral evidence of aggressive mimicry in the behavior of the spitting spider or the host spider. However, we consider that our results do provide indirect evidence for aggressive mimicry, since the predatory success of the spitting spider was commonly preceded by the host spider leaving its retreat and meeting with the web invader, thus indicating that the presence of one type of vibration in the web favors the capture success of S. globula. This may be the mechanism that the spitting spider is taking advantage of, which supports the evolutionary maintenance of such a risky predatory tactic as invading a predator’s web (see below). During aggressive mimicry, predatory spiders often rely on specific vibratory stimuli performed in the outer extremes of the host’s web (see revision in Herberstein and Wignall 2011); however, there is always the risk of the invader becoming entangled in the host’s web. Cribellated hacklemesh webs, as those of M. simoni, may impose an additional constraint on the invader since hundreds of cribellar fibers mechanically interlock and catch setae and other stiff irregularities of the prey (Opell 2013), but it remains unclear why cribellate weaver spiders do not get entangled in their own web (Opell 2013). In our case, the spitting spider could be using the anti-adhesion traits known for orb weaver spiders to avoid getting entangled in their adhesive silk webs: branched setae, careful engagement movements of the legs, or legs with a chemical coating layer (Briceño and Eberhard 2012). However, it is preliminary to conclude that S. globula performs aggressive mimicry (by mimicking the vibration walking prey) on M. simoni’s web. To address this issue it is necessary a gain a more complete understanding of the biomechanical performance of spiders on cribellated silk, record and compare the vibrations of many sources on the web, and test this hypothesis experimentally.
The spitting spider seemed to engage in risky behavior when attacking the host spider. M. simoni is a well-defended potential prey item: (1) it inhabits a long and dense web with a protective retreat; (2) the cribellated silk threads accurately transmit vibrations; (3) it bears long and strong hind legs and has chelicerae, venom, and silk (Aisenberg et al. 2011; this study). Nonetheless, it is noticeable that we did not find any instance in which the host spider preyed upon the spitting spider. It would be expected that a wandering predator such as the spitting spider would aim at capturing an easier and apparently less harmful prey item than M. simoni. The distinctive spitting tactic of S. globula seems to function as both a predatory and defensive strategy, with spitting allowing the spider to control a predatory interaction from a distance (Li et al. 1999; Clements and Li 2005). Hence, this feature could favor the attack attempts on M. simoni by increasing the probability of subduing the prey, but also by using defensive spits to deter a counterattack. Additionally, we found that in the tree barks in which these two predators coexist there are few large invertebrates as potential prey; therefore, M. simoni (as well as other weaver spiders therein; Simó et al. 2011) may be attractive and nutritionally rewarding prey.
Our study is characterized by two possible confounding factors that we did not control for: (1) individual variation in the silk threads attached to the substrates and (2) the density of the silk threads in the web, which are known to vary in sheet weaver spiders (the agelenid Melpomeme sp: Rojas 2011; the pholcid Physocyclus globosus: Escalante 2013). Although we did not measure this latter parameter quantitatively, qualitative observations of the silk lines of the 15-day-old webs of the host spider (fed under the same regimen) suggested that this host spider tends to individually vary the number of threads attached to the substrate. Also, the length of the retreat and thread density seemed to vary between individuals. However, we did not detect any pattern between the apparent density of silk lines, the retreat, or the substrate and the outcome of the interaction. Hence, the web-building behavior and the web itself as a phenotype are apparently very plastic, although further examination and detailed descriptions of these features are required to sustain this suggestion.
In summary, our data illustrate the importance of studying the natural history and behavior of both predator and potential prey to unravel the factors mediating the interactions of two predators. We report novel observations on the ecology of the environment in which such interactions occur between the spitting and the host spider, as well as on their defensive and predatory behavior. The spitting spider can gain an indirect benefit from its interaction with M. simoni by using the host’s web to attack prey attached to it, either after the host spider has escaped or after S. globula has preyed on it. The high abundance of M. simoni adult females in pitfall traps (Costa et al. 2006; Costa and Simó 2014) suggests that these weaver spiders often engage in the expensive strategy of abandoning the web and wandering in the leaf litter. However, extended observations of the spitting spider after preying upon M. simoni and other weaver spiders and an experimental approach are needed to test the benefit of occupying an already built web. Finally, this is the first detailed description of the variation in the prey approaching and capture behavior of M. simoni. Future studies could benefit from our study to deeply address the sexual and predatory behavior of this relatively understudied cribellated weaver species, testing our hypothesis about the plasticity in their response in different contexts.
References
Abràmoff MD, Magalhães PJ, Ram SJ (2004) Image processing with ImageJ. Biophot Int 11:36–42
Ades C, Ramires EN (2002) Asymmetry of leg use during prey handling in the spitting spider Scytodes globula (Scytodidae). J Insect Behav 15:563–570
Ades C, Ramires EN, Marcon MR (2010) Asymmetry and experience in the predatory probing behavior of spitting spiders Scytodes globula Nicolet, 1849 (Araneae, Scytodidae). Rev Bras Zoocien 12:91–94
Aisenberg A, Toscano-Gadea CA, Ghione S (2011) Guía de arácnidos del Uruguay. Ediciones de la Fuga, Montevideo
Arim M, Marquet PA (2004) Intraguild predation: a widespread interaction related to species biology. Ecol Lett 7:557–564
Barrantes G, Eberhard WG (2007) The evolution of prey-wrapping behaviour in spiders. J Nat Hist 41:1631–1658
Blamires SJ, Chao YC, Liao CP, Tso IM (2011) Multiple prey cues induce foraging flexibility in a trap-building predator. Anim Behav 81:955–961
Briceño RD, Eberhard WG (2012) Spiders avoid sticking to their webs: clever leg movements, branched drip-tip setae, and anti-adhesive surfaces. Naturwissenschaften 99:337–341
Carvalho LA, Souza ES, Willemart RH (2012) Behavioral analysis of the interaction between the spitting spider Scytodes globula (Araneae: Scytodidae) and the harvestmen Discocyrtus invalidus (Opiliones: Gonyleptidae). J Arachnol 40:332–337
Clements R, Li D (2005) Regulation and non-toxicity of the spit from the pale spitting spider Scytodes pallida (Araneae: Scytodidae). Ethology 111:311–321
Coelho L, Aisenberg A, Costa FG (2010) Testing female receptiveness to novel or previous mating partners in a wolf spider. Behavior 147:383–395
Costa FG, Simó M (2014) Fenología de las arañas epígeas de una zona costera del sur de Uruguay: un estudio bianual con trampas de caída. Bol Soc Zool Uruguay 23(1):1–15
Costa FG, Simó M, Aisenberg A (2006) Composición y ecología de la fauna epígea de Marindia (Canelones, Uruguay) con especial énfasis en las arañas: un estudio de dos años con trampas de intercepción. In: Menafra R, Rodríguez-Gallego L, Scarabino F, Conde D (eds) Bases para la conservación y el manejo de la costa uruguaya. Vida Silvestre Uruguay, Montevideo, pp 427–436
Denno RF, Mitter MS, Langellotto GA, Gratton C, Finke DL (2004) Interactions between a hunting spider and a web-builder: consequences of intraguild predation and cannibalism for prey suppression. Ecol Entomol 29:566–577
Escalante I (2013) Ontogenetic and sexual differences in exploration and web construction in the spider Physocyclus globosus (Araneae: Pholcidae). Arachnology 16:61–68
Gilbert C, Rayor RS (1985) Predatory behavior of spitting spiders (Araneae: Scytodidae) and the evolution of prey wrapping. J Arachnol 13:231–241
Gonzaga MO, Santos AJ, Dutra GF (1998) Web invasion and araneophagy by Peucetia tranquillini Mello-Leitão, 1992 (Araneae, Oxyopidae). J Arachnol 26:249–250
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: Paleontological statistics software package for education and data analysis. Palaeontol Electron 4(1). Available at: http://palaeo-electronica.org/2001_1/past/issue1_01.htm
Henschel JR, Jocqué R (1994) Bauble spiders: a new species of Achaearanea (Araneae: Theridiidae) with ingenious spiral retreats. J Nat Hist 28:1287–1295
Herberstein ME, Wignall A (2011) Deceptive signals in spiders. In: Herberstein ME (ed) Spider behavior flexibility and versatility. Cambridge University Press, Cambridge
Hronsky M (2006) Magnetic resonance study of the spider Nephila edulis and its liquid dragline silk. PhD and Habilitation thesis. Swiss Federal Institute of Technology, Zurich. doi:10.3929/ethz-a-005299740
Ingram T, Svanba R, Kraft NJB, Kratina P, Southcott L, Schluter D (2012) Intraguild predation drives evolutionary niche shift in threespine stickleback. Evolution 66:1819–1832
Jackson RR (1992) Eight-legged tricksters. Spiders that specialize in catching other spiders. Bioscience 42:590–598
Jackson RR, Brassington RJ (1987) The biology of Pholcus phalangioides (Araneae, Pholcidae): predatory versatility, araneophagy and aggressive mimicry. J Zool 211:227–238
Jackson RR, Pollard SD (1996) Predatory behavior of jumping spiders. Annu Rev Entomol 41:287–308
Jackson RR, Whitehouse MEA (1986) The biology of New Zealand and Queensland pirate spiders (Araneae: Mimetidae): aggressive mimicry, araneophagy and prey specialization. J Zool 210:279–303
Jackson RR, Wilcox RS (1990) Aggressive mimicry, prey-specific predatory behaviour and predator recognition in the predator- prey interactions of Portia fimbriata and Euryattus sp., jumping spiders from Queensland. Behav Ecol Sociobiol 26:111–119
Jackson RR, Li D, Fijn N, Barrion AT (1998) Predator-prey interaction between aggressive-mimic jumping spiders (Salticidae) and araneophagic spitting spiders (Scytodidae) from the Philippines. J Insect Behav 11:319–342
Jackson RR, Pollard SD, Li D, Fijn N (2002) Interpopulation variation in the risk related decisions of Portia labiata, an araneophagic jumping spider (Araneae, Salticidae), during predatory sequences with spitting spiders. Anim Cogn 5:215–223
Jarmar EAR, Jackson RR (1986) The biology of Taieria erebus (Araneae, Gnaphosidae), an araneophagic spider from New Zealand: Silk utilisation and predatory versatility. N Z J Zool 13:521–541
Konigswald A, Lubin Y, Ward D (1990) The effectiveness of the nest of a desert widow spider, Latrodectus revivensis, in predatory deterrence. Psyche 97:75–80
Landolfa MA, Barth FG (1996) Vibrations in the orb web of the spider Nephila clavipes: cues for discrimination and orientation. J Comp Physiol A 179:493–508
Li D (1996) Prey preference of specialized jumping spiders (Araneae: Salticidae). PhD thesis. University of Canterbury. Christchurch
Li D, Jackson RR, Barrion AT (1999) Parental and predatory behaviour of Scytodes sp., an araneophagic spitting spider (Araneae: Scytodidae) from the Philippines. J Zool 247:293–310
Masters WM, Markl HS, Moffat AJM (1986) Transmission of vibration in a spider’s web. In: Shear WA (ed) Spiders: webs, behavior and evolution. Stanford University Press, Stanford, pp 49–69
Nelson X, Jackson RR (2011) Flexibility in the foraging strategies of spiders. In: Herberstein ME (ed) Spider behaviour: flexibility and versatility. Cambridge University Press, Cambridge, pp 31–56
Nelson X, Jackson RR (2012) The role of numerical competence in a specialized predatory strategy of an araneophagic spider. Anim Cogn 15:699–710
Nentwig W (1985) Feeding ecology of the tropical spitting spider Scytodes longipes (Araneae, Scytodidae). Oecologia 65:284–288
Opell BD (2013) Cribellar thread. In: Nentwig W (ed) Spider ecophysiology. Springer, Berlin, pp 303–315
Perkins TA, Riechert SE, Jones TC (2007) Interactions between the social spider Anelosimus studiosus (Araneae, Theridiidae) and foreign spiders that frequent its nests. J Arachnol 35:143–152
Polis GA, Myers CA, Holt RD (1989) The ecology and evolution of intraguild predation: potential competitors that eat each other. Annu Rev Ecol Syst 20:297–330
Ramires EN (1999) Uma abordagem comparativa ao comportamento defensivo, agonístico e locomotor de três espécies de aranhas do gênero Loxosceles (Sicariidae). PhD thesis. Instituto de Psicología, Universidad de São Paulo, São Paulo
Riechert SE, Hedrick AV (1990) Levels of predation and genetically based anti-predator behaviour in the spider, Agelenopsis aperta. Anim Behav 40:679–687
Rojas A (2011) Sheet-web construction by Melpomene sp. (Araneae: Agelenidae). J Arachnol 39:189–193
Simó M, Laborda Á, Jorge C, Guerrero JC, Alves Dias M, Castro M (2011) Introduction, distribution and habitats of the invasive spider Badumna longinqua (L. Koch, 1867) (Araneae: Desidae) in Uruguay, with notes on its world dispersion. J Nat Hist 45:1637–1648
Soley F, Taylor PW (2012) Araneophagic assassin bugs choose routes that minimize risk of detection by web–building spider. Anim Behav 84:315–321
Suter RB, Stratton GE (2009) Spitting performance parameters and their biomechanical implications in the spitting spider, Scytodes thoracica. J Insect Sci 9:1–15
Vetter RS (2013) Scavenging behavior in spitting spiders, Scytodes (Araneae: Scytodidae). J Arachnol 41:392–394
Whitehouse MEA (1987) “Spider eat spider”: the predatory behavior of Rhomphaea sp. from New Zealand. J Arachnol 15:355–362
Whitehouse MEA, Agnarsson I, Miyashita T, Smith D, Cangialosi K, Masumoto T, Li D, Henaut Y (2002) Argyrodes: Phylogeny, sociality and interspecific interactions–a report on the Argyrodes symposium, Badplass 2001. J Arachnol 30:238–245
Wise DH (1993) Spiders in ecological webs. Cambridge University Press, Cambridge
Yasuda H, Kikuchi T, Kindlmann P, Sato S (2001) Relationships between attack and escape rates, cannibalism, and intraguild predation in larvae of two predatory ladybirds. J Insect Behav 14:373–384
Acknowledgments
We thank M. Masís for her extensive help in measuring the body size of the spiders. We also thank E. Stanley, L. Montes de Oca, S. Fierro, M. J. Albo, V. Mello, C. Toscano-Gadea, and all of the staff of the Laboratorio de Etología, Ecología y Evolución of the IIBCE for their assistance in the field and in laboratory during this project. D. Li, R. H. Willemart, R. R. Jackson, F. Pérez-Miles, M. Simó, and C. Viera provided important guidance and suggestions, as well as the literature to improve this project. Two anonymous reviewers contributed substantially to the improvement of of this manuscript. Financial support was provided by PEDECIBA (AA), ANII (AA and FGC), and the SEP-UCR (IE).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Escalante, I., Aisenberg, A. & Costa, F.G. Risky behaviors by the host could favor araneophagy of the spitting spider Scytodes globula on the hacklemesh weaver Metaltella simoni . J Ethol 33, 125–136 (2015). https://doi.org/10.1007/s10164-014-0420-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10164-014-0420-6