Abstract
Distinguishing primary ovarian cancer from metastatic colorectal cancer is often difficult by a conventional pathological examination alone. We assessed the usefulness of p53 gene mutation analysis for the differential diagnosis of ovarian adenocarcinoma. A 66-year-old woman suffered multiple organ metastases, including the liver, para-aortic lymph node, and right ovary, following an operation for advanced sigmoid colon cancer. She underwent ovarian resection after effective chemotherapy against the liver and para-aortic lymph node cancer. Histological analysis suggested primary ovarian cancer. Therefore, we applied p53 gene mutation analysis for the differential diagnosis of primary versus metastatic ovarian cancer from sigmoid colon cancer. The direct sequence of the p53 gene demonstrated the same gene mutation in codon 211 (ACT to ATT) in both the sigmoid colon and ovarian cancers. According to the International Agency for Research on Cancer TP53 mutation database, this type of p53 mutation in colorectal cancer and ovarian cancer is 0.13% (5/3,693) and 0% (0/1,494), respectively. Therefore, we determined that the ovarian tumor was metastatic. Although p53 gene mutation analysis has been applied in some cases, this modality is very useful for the differential diagnosis of primary and metastatic cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The incidence of metastatic ovarian tumors from extragenital primary sites accounts for 4.4–12.5% of malignant ovarian tumors [1–3], and 1–8% of cases with colorectal cancer metastasize to the ovary [4, 5]. Distinguishing primary ovarian cancer from metastatic colorectal cancer is often difficult by a conventional pathological examination alone. Although a past history of malignancies is very important, Abu-Rustum et al. reported that 23% of ovarian tumors were primary ovarian malignancies even after a history of past colorectal cancer [6]. Analysis of size and laterality, as well as immunohistochemical markers such as cytokeratins 7 (CK7) and 20 (CK20), β-catenin, CDX2, and P504S, are useful for differential diagnosis, but these modalities lack objectivity [7–11]. Therefore, the final diagnosis depends on a comparison of clinical history and histological results. Apart from these modalities, a comparison of specific gene mutations between the primary and metastatic sites is objective. Analysis of the p53 gene mutation pattern has been used in the diagnosis of primary versus metastatic pulmonary adenocarcinoma, hepatocellular carcinoma, and epithelial ovarian cancer [12–15]. We analyzed the p53 gene mutation pattern to distinguish between primary sigmoid colon and metachronous ovarian cancers.
Case report
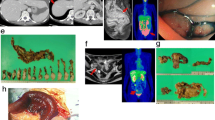
A 66-year-old woman with ileus induced by sigmoid colon cancer underwent a Hartmann procedure in July 2006. A histological study of the resected specimen showed that atypical cells formed a glandular structure with invasion to the surrounding stroma. The tumor cells were disseminated into the peritoneal cavity, including mesenteries of the small intestine and sigmoid colon. Histological diagnosis is as follows: moderately differentiated adenocarcinoma of colon (tub2), pSI, INFbeta, pPM0, pDM0, n0, ly 2, v 0, pT4N0P3H0, Stage IV. Although she had been undergoing systemic adjuvant chemotherapy (FOLFOX) since August 2006, computed tomography (CT) demonstrated tumors in the liver, para-aortic lymph node, and right ovary in September 2006 (Fig. 1). Although we consulted the gynecologist, distinguishing between the primary and metastatic ovarian tumor was difficult by CT and ultrasound examination. Although the tumors in the liver and para-aortic lymph node responded to chemotherapy, the right ovarian tumor was enlarged in November 2006. Therefore, a different chemotherapy (FOLFIRI) was started in December 2006 because the availability of 5-FU, irinotecan, and oxaliplatin in the course of treatment is associated with improvement of survival of patients with advanced colorectal cancer [7]. The tumors in the liver and para-aortic lymph node disappeared following the FOLFIRI therapy, but the ovarian tumor enlarged further, and by February 2007, the ovarian tumor had enlarged to 20 cm (Fig. 2). The patient suffered from renal dysfunction for reasons of obstruction of both ureters by the ovarian tumor. The serum tumor marker carcinoembryonic antigen was elevated to 28.2 ng/ml (normal, <5.0 ng/ml), but CA125 was normal (18.3 U/ml).
A laparotomy was performed to relieve symptoms including abdominal distension and hydronephrosis after improvement of renal function by inserting stents into the ureters. The laparotomy showed a huge cystic ovarian tumor without ascites or dissemination (Fig. 3). The uterus and left ovary were resected by the gynecologist. The damaged bladder and left ureter were reconstructed by the urologist. The damaged small intestine was reconstructed and resected. An intraoperative histological examination of the resected ovarian tumor showed that the tumor cells formed a well-differentiated glandular structure rather than a moderately differentiated one. From this finding, the tentative diagnosis of the intraoperative specimen was as follows: suspected endometrioid adenocarcinoma, grade 1, suggesting primary ovarian cancer rather than metastatic ovarian cancer from the sigmoid colon cancer. Therefore, we closed the colostomy, and re-anastomosis was performed. The operation time was 9 h and 45 min with 6,360 ml blood loss. Even though the clinical history of advanced sigmoid colon cancer strongly indicated it as the primary site, the different responses of the ovary and other organs to chemotherapy, rapid growth of the ovarian tumor, laterality of the tumor, and conventional histological study suggested a primary ovarian cancer. To determine a diagnosis for the ovarian cancer, we analyzed the p53 gene mutation in both sigmoid colon and ovarian cancers. Written informed consent was obtained from the patient before the first operation. This procedure was approved by the institutional review board in our hospital.
Specimens were fixed with 10% formalin, embedded in paraffin, and cut. Tumor cells were microdissected with a laser capture microdissection system (Leica, AS-LMD; Leica Microsystems, Wetzler, Germany), and genomic DNA was obtained from the microdissected cells using the QIAamp DNA micro kit (Qiagen, Valencia, CA, USA). The fragments containing the p53 gene hotspot were amplified by polymerase chain reaction (PCR) using the following primer sets: 5′-CTCTTCCTACAGTACTCCCCTGC and 5′-GCCCCAGCTGCTCACCATCGCTA for exon 5, 5′-GATTGCTCTTAGGTGTGGCCCCTC and 5′-GGCCACTGACAACCACCCTTA ACC for exon 6, 5′-TGCCACAGGTCTCCCCAAGG and 5′-GCAACAGCAGGCCAGTGT GCA for exon 7, and 5′-CCTGAGTAGTGGTAATCTACTG and 5′-TGCTTGCTTACCTCGC TTA for exon 8. The amplified fragment was directly sequenced with an ABI3100 sequencer (Applied Biosystems, Foster City, CA, USA).
The direct sequence of the p53 gene demonstrated the same gene mutation in codon 211 (ACT to ATT) in both sigmoid colon and ovarian cancers. According to the International Agency for Research on Cancer (IARC) TP53 database, this type of p53 mutation in colorectal cancer and ovarian cancer is 0.13% (5/3,693) and 0% (0/1,494), respectively. We also analyzed the immunohistochemical expression of CK7 and CK20 in sigmoid colon and ovarian cancers, and both tumors were CK7 negative and CK20 positive (Fig. 4). Based on the results of p53 gene mutation analysis and CK7/CK20 expression pattern, we determined that the ovarian tumor was metastatic adenocarcinoma from the sigmoid colon with no metastasis to the uterus and lymph nodes. Cytological examination in the peritoneal cavity has not been done. Although the patient received chemotherapy after the operation, she suffered from liver metastasis, left axillary lymph node metastasis, and dissemination into the pelvic cavity, one after another. She underwent resection of the liver metastasis in November 2007 and a resection of the left axillary lymph node metastasis in 2008. She received radiation therapy for dissemination in the pelvic cavity in 2009. Immunohistochemical staining demonstrated CK7-negative/CK20-positive tumor both in the liver metastasis and the left axillary lymph node metastasis (data not shown). She survived 42 months after the first operation with evidence of recurrence in the pelvic cavity.
Discussion
The 5-year survival rate after resection of metastatic ovarian tumors from nongynecologic organs was worse than those from gynecologic organs (19% vs. 47% [1]; 5.4% vs. 14.8% [3]). In colorectal cancers, 5-year survival for patients with synchronous ovarian metastases was 9%, versus 20% for metachronous ovarian metastases. Resection of metastatic disease was associated with an improved 5-year survival for metachronous ovarian metastasis if patients were disease free postoperatively (24% vs. 0%, P < 0.0001) [5].
Although the prognosis of metastatic ovarian tumors from colorectal cancer is poor, the patient was disease free postoperatively after resection of metachronous ovarian metastasis. Therefore, the exact diagnosis is very important for adjuvant chemotherapy because the regimen of chemotherapy differs between primary and metastatic ovarian cancer.
Distinguishing primary ovarian cancer from metastatic ovarian cancer is often difficult by conventional histological examination alone. Therefore, a clinical history of the malignancies and the size and laterality of the ovarian tumor are helpful for a final diagnosis [8]. It has been reported that metastatic ovarian tumors from colorectal cancer are misdiagnosed as primary ovarian tumors in 45% of patients [6]. In our case, a clinical history of advanced sigmoid colon cancer strongly suggested a metastatic ovarian tumor. However, the different response of the ovarian tumor to chemotherapy from other metastatic lesions and the rapidly growing and inconsistent morphology from the primary lesion suggested the possibility of a primary ovarian tumor. An immunohistochemical study is useful for the differential diagnosis of the ovarian tumor [9–12]. The most popular modality is a combination of CK7 and CK20. Generally, primary colon cancer shows a CK7-negative and CK20-positive status, which occurs in 68% of all the cases [10]. Especially, a CK7-negative and CK20-positive status in metastatic ovarian tumor from colorectal cancers is 70–91% [9, 10, 12]. On the other hand, a CK7-negative and CK20-positive status in primary mucinous ovary tumors is 5–14% [9, 12]. The novel immunohistochemical markers including CDX2, β-catenin, and P504S are useful modalities when they are used together [11, 12]. The problems with an immunohistochemical examination include exceptions and lack of quantity.
The p53 gene mutation pattern has been applied for the differential diagnosis of lung and liver tumors [13–15]. The lung is the target organ when squamous cell carcinomas or adenocarcinomas metastasize from the lung itself or from other organs. The liver often bears multiple hepatocellular carcinoma nodules. The distinction between multifocal disease and metastatic disease from one nodule is important for diagnosis of clinical stage. The p53 gene mutation analysis also demonstrates that advanced epithelial ovarian cancer is unifocal in origin when an ovarian tumor is compared to a resected omental tumor [16]. The frequency of the p53 gene mutation is approximately 30% in ovarian cancer, and no specific mutational hotspot has been found [15]. On the other hand, the frequency of p53 gene mutation is approximately 40–50% in colorectal cancer, and five specific hotspot codons have been found [17]. We applied p53 gene mutation analysis to confirm that the ovarian tumor was a metastatic tumor from sigmoid colon cancer. An identical p53 mutation is present in both primary and metastatic colorectal cancer with a very high frequency [18, 19]. According to the IARC TP53 database (http://www-p53.iarc.fr/index.html), the frequency of this type of p53 mutation in colorectal cancer and ovarian cancer is 0.13% (5/3,693) and 0% (0/1,494), respectively. Therefore, we finally diagnosed the ovarian tumor as metastatic. Although p53 gene mutation analysis has been applied in only some cases, it is very useful for the differential diagnosis of primary and metastatic cancer.
References
Yada-Hashimoto N, Yamamoto T, Kamiura S et al (2003) Metastatic ovarian tumors: a review of 64 cases. Gynecol Oncol 89:314–317
Turan T, Aykan B, Koc S et al (2006) Analysis of metastatic ovarian tumors from extragenital primary sites. Tumori 92:491–495
Salamalekis E, Bakas P, Sykiotis K et al (2004) Outcome of patients with ovarian metastatic tumors. Report of 83 cases and review. Eur J Gynaecol Oncol 25:713–715
Hanna NN, Cohen AM (2004) Ovarian neoplasms in patients with colorectal cancer: understanding the role of prophylactic oophorectomy. Clin Colorectal Cancer 3:215–222
Huang PP, Weber TK, Mendoza C et al (1998) Long-term survival in patients with ovarian metastases from colorectal carcinoma. Ann Surg Oncol 5:695–698
Abu-Rustum N, Barakat RR, Curtin JP (1997) Ovarian and uterine disease in women with colorectal cancer. Obstet Gynecol 89:85–87
Grothey A, Sargent D, Goldberg RM et al (2004) Survival of patients with advanced colorectal cancer improves with the availability of fluorouracil-leucovorin, irinotecan, and oxaliplatin in the course of treatment. J Clin Oncol 22:1209–1214
Yemelyanova AV, Vang R, Judson K et al (2008) Distinction of primary and metastatic mucinous tumors involving the ovary: analysis of size and laterality data by primary site with reevaluation of an algorithm for tumor classification. Am J Surg Pathol 32:128–138
Cathro HP, Stoler MH (2002) Expression of cytokeratins 7 and 20 in ovarian neoplasia. Am J Clin Pathol 117:944–951
Park SY, Kim HS, Hong EK et al (2002) Expression of cytokeratins 7 and 20 in primary carcinomas of the stomach and colorectum and their value in the differential diagnosis of metastatic carcinomas to the ovary. Hum Pathol 33:1078–1085
Logani S, Oliva E, Arnell PM et al (2005) Use of novel immunohistochemical markers expressed in colonic adenocarcinoma to distinguish primary ovarian tumors from metastatic colorectal carcinoma. Mod Pathol 18:19–25
Vang R, Gown AM, Wu LS et al (2006) Immunohistochemical expression of CDX2 in primary ovarian mucinous tumors and metastatic mucinous carcinomas involving the ovary: comparison with CK20 and correlation with coordinate expression of CK7. Mod Pathol 19:1421–1428
Oda T, Tsuda H, Scarpa A et al (1992) Mutation pattern of the p53 gene as a diagnostic marker for multiple hepatocellular carcinoma. Cancer Res 52:3674–3678
Noguchi M, Maezawa N, Nakanishi Y et al (1993) Application of the p53 gene mutation pattern for differential diagnosis of primary versus metastatic lung carcinomas. Mol Pathol 2:29–35
Nakazato Y, Tanaka R, Seki E et al (2008) Differential diagnosis of primary versus metastatic pulmonary adenocarcinomas using gene mutation analyses: a case report. J Thorac Oncol 3:931–934
Mok CH, Tsao SW, Knapp RC et al (1992) Unifocal origin of advanced human epithelial ovarian cancers. Cancer Res 52:5119–5122
Russo A, Bazan V, Iacopetta B et al (2005) The TP53 colorectal cancer international collaborative study on the prognostic and predictive significance of p53 mutation: influence of tumor site, type of mutation, and adjuvant treatment. J Clin Oncol 23:7518–7528
Khan ZA, Jonas SK, Le-Marer N et al (2000) P53 mutations in primary and metastatic tumors and circulating tumor cells from colorectal carcinoma patients. Clin Cancer Res 6:3499–3504
Albanese I, Scibetta AG, Migliavacca M et al (2004) Heterogeneity within and between primary colorectal carcinomas and matched metastases as revealed by analysis of Ki-ras and p53 mutations. Biochem Biophys Res Commun 325:784–791
Conflict of interest statement
No author has any conflict of interest.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Yamano, T., Morii, E., Arai, I. et al. Diagnosis of primary versus metastatic ovarian adenocarcinoma using p53 gene mutation analysis. Int J Clin Oncol 15, 621–625 (2010). https://doi.org/10.1007/s10147-010-0096-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-010-0096-z