Abstract
Spinal dural arteriovenous fistulas (SDAVFs) are rare pathologies with a yearly incidence of 5–10 new cases/million, constituting 60–80 % of spinal arteriovenous malformations. Clinical symptoms include progressive paraparesis, paresthesias, bladder, and bowel disturbances. The pathophysiology of SDAVFs is not well elucidated. Microneurosurgery and endovascular techniques are established treatment modalities for permanent fistula occlusion, which are oftentimes accompanied by an amelioration of neurological deficits in the long run. Here, we report our interdisciplinary neurosurgical/neuroradiological management strategy of SDAVFs in 32 patients who were evaluated retrospectively. We focused on clinical presentation, microneurosurgical and interventional technique, early, and late neurological results. Quality of life (QoL) was additionally assessed in 12 patients at last follow-up. We discuss the results against the background of the current literature. Our series and the literature indicate that clinical outcome after treatment of SDAVF is favorable in general. Both neurosurgical and neurointerventional therapies appear to be safe and effective, but short-term neurological deterioration after the intervention constitutes an as-of-yet unsolved problem. Beyond age and preoperative neurological state, presence of comorbidities had a significant influence on neurological outcome in our study sample. Self-assessed physical and mental QoL at long-term follow-up was reduced in quite a number of patients and was associated with a poorer neurological result as well as presence of comorbidities. The patients’ perspective in terms of QoL was first investigated in this study, but further research on QoL and psychosocial impairment of SDAVF patients is needed to enable individualized counseling and rehabilitation strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spinal dural arteriovenous fistulas (SDAVF), are rare pathologies with an incidence of only 5–10 new cases per million inhabitants per year [43]. Yet, these lesions constitute 60–80 % of spinal arteriovenous malformations. Most of SDAVF are located in the thoracolumbar region of the spine and usually become symptomatic in the sixth decade of life [9, 34, 47]. Diagnosis of SDAVF is generally difficult in clinical practice because of the rarity of the disease and the unspecific initial clinical presentation. First symptoms include gait disturbance and paraparesis, whereas in the later course bladder and bowel dysfunction can also be observed [12, 33, 34]. In contrast to the well-established traumatic and thrombotic pathogenesis of cranial dural arteriovenous fistulas [26, 28, 31], the pathomechanisms responsible for the development of SDAVFs are still not well elucidated. However, diagnosis and treatment of SDAVF are well known and described in the current literature [1, 9, 18, 34].
Microneurosurgical occlusion of the fistula was for many years the method of choice, but more recently, endovascular techniques have augmented the therapeutic armamentarium. It is, therefore, not surprising that studies on SDAVFs focus on the comparison of surgical and endovascular therapies [13–15, 19, 23, 35, 39] whereas the influence of clinical parameters (such as age, length of duration of symptoms and initial neurological presentation) on outcome and the patient’s perspective in terms of quality of life (QoL) have not been so comprehensively investigated.
The aim of our retrospective study was to identify potential clinical variables influencing outcome after treatment in our own series of 32 patients with spinal dural fistulas, treated interdisciplinary in cooperation with the Department of Neuroradiology and to fit them into the current literature. Moreover, we assessed the impact on functional impairment at late follow-up on health-related quality of life (QoL) in order to gain an insight on the patients’ perspective of the sequelae of this life-changing disease.
Patients and methods
A chart review was undertaken of all patients treated for spinal vascular diseases between January 2001 and December 2013 at our institution. All patients presenting with SDAVFs and treated at our institution were included in this retrospective survey, whereas patients with perimedullary fistulas and intramedullary AVMs as well as patients who refused treatment were excluded from this study.
Demographic and clinical data including age, gender, comorbidities, pre- and postoperative neurological state, radiological findings (i.e., level of SDAVF, extent of edema), surgical or endovascular technique, intraoperative findings (if applicable), and complications were obtained. All patients received a clinical follow-up examination at the neurosurgical outpatient department 3 months after therapy and had a second long-term follow-up. Motor impairment immediately before fistula occlusion, at 3-month follow-up and at last follow-up was classified according to the modified Aminoff-Logue myelopathy scale (Table 1) [36].
All SDAVFs except for one were initially detected by magnetic resonance imagining (MRI). In one patient with a cardiac pacemaker, the diagnosis was made by means of myelography. Diagnosis of SDAVF was verified in all cases using conventional spinal digital subtraction angiography (DSA).
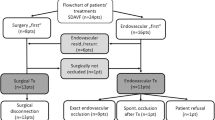
In all patients, the choice of treatment modality was discussed interdisciplinarily with neurosurgeons and interventional neuroradiologists experienced in treatment of cerebral and spinal vascular pathologies. Microneurosurgical obliteration was performed if the radiculomedullary arteries (anterior or posterior spinal artery) were part of or in close proximity to the feeding artery/fistula point or if, according to the neuroradiologic expertise the point of arteriovenous fistula could not be reached by embolization material, e.g., due to strong vessel elongation of the feeding artery or if branches of more than one segmental artery were involved in feeding the fistula. If neuroradiological intervention was deemed to be feasible with a comparable risk profile to the microneurosurgical procedure, the patient was offered this possible alternative treatment method. All patients were treated according to the same endovascular or neurosurgical protocol. All patients received post-therapeutic spinal DSA to ascertain successful obliteration of the fistula.
Endovascular technique
All embolizations were performed under general anesthesia. An arterial approach was obtained and a guiding catheter placed in the segmental artery; under fluoroscopy, a microcatheter was introduced coaxially through the feeding pedicle and advanced into the distal aspect of a feeding artery close to the fistula in the ideal case in wedge-position so that a liquid embolic agent could be pushed up to the proximal venous side. In seven patients, a mixture of Glubran® (cyanoacrylate glue, GEM s.r.l., Italy) and iodized oil (Lipiodol®, Guerbet, Aulnay-sous-Bois, France) in case-dependently variable concentrations (ranging from 1:3 up to 1:5) for appropriate flow characteristics and in one patient Onyx® (Covidien, Mansfield, MA, USA) was injected. In the ideal case the feeding pedicle, including the terminal feeders up to the fistulous point, as well as the beginning of the early draining vein was embolized in a continuous injection. A final spinal angiography of the initially feeding segmental artery and of the adjacent and contralateral segmental arteries was performed. An illustrative case is demonstrated in Fig. 1.
A 71-year-old female with thoracic SDAVF. Superselective angiogram of the right Th5 segmental artery (a) and plain film of glue cast (b) following embolization with 0.2 cm3 Glubran/Lipiodol (1:3). The right-sided Th5 segmental artery is occluded (c). Angiogram of the right-sided Th4 segmental artery showed residual filling of the SDAVF and simultaneous filling of the artery of Adamkiewicz (black arrow), no further embolization was performed
Surgical technique
All surgical procedures were performed under permanent control of somatosensory evoked potentials (SSEP) and motor evoked potentials (MSEP).
Targeted hemilaminectomy, laminoplasty, or laminectomy was performed according to spinal anatomy and the location of the fistula point and extended to the side of the spinal AV fistula. Then, the feeding radicular artery was identified first visually, followed by opening of the dura and isolation of the draining vein. A temporary aneurysm clip was placed on the fistulous point under electrophysiological monitoring to isolate the right feeder. Doppler ultrasonography [11] and indocyanine green (ICG) angiography [16] were also additional tools for identifying the feeder. Permanent occlusion of the fistula by means of coagulation and division was performed if potentials remained stable and the draining vein was no longer arterialized. The epidural feeding artery was also coagulated and disconnected if necessary. Intraoperative fluoroscopy was additionally performed since the year 2005 after acquisition of a PENTERO® microscope (Zeiss®, Oberkoden, Germany). The technique is illustrated in Fig. 2.
A 50-year-old male who suffered of hypaesthesia of both legs and gait disturbance. a T2-weighted MRI showing spinal cord edema b superselective angiography demonstrating right-sided dural AV fistula at the level of Th8, c after hemilaminectomy the fistula was exposed showing two feeding arteries (arrows), d fistula was then coagulated intradurally, e transsection with micro-scissor, f coagulation and transection of the feeding arteries epidurally
Investigation of quality of life
For investigation of patients’ QoL at late follow-up, the short form (SF)-36 was used. This instrument is a widely used and well-validated generic questionnaire on health- QoL. It differentiates between a physical and a mental component of QoL: The Physical Component Summary (SF-36 PCS) and Mental Component Summary (SF-36 MCS). Here, higher scores indicate a better QoL. The items of the questionnaire can be further subdivided into eight subscales. For the evaluation of the summary scores, the reference values for the German population were used [7].
Statistical analysis
Statistical analyses were conducted using IBM SPSS Statistics 22 (Statistical Package of the Social Sciences, SPSS Inc., Armonk/USA). Descriptive statistics of interval-scaled data were expressed as mean ± standard deviations (SD) unless otherwise mentioned. Data was tested for normal distribution by conducting a Shapiro-Wilk test, in addition to histograms and Q-Q-Plots for every parameter. Because data did not meet the assumption of normality Wilcoxon signed-rank test for paired variables were used.
Categorical data was analyzed by Pearson Chi-Square test or, if expected frequencies were below 5, Fisher’s exact test. For bivariate correlation analyses Spearman’s rho was applied. Partial correlation analyses were used for controlling for the effect of age and the number of comorbidities. A p value of ≤ 0.05 was considered as statistically significant.
Results
Clinical characteristic of the study group
Thirty-two patients with SDAVF (6 females = 18.8 %; 26 males = 81.3 %) with a mean age of 64 ± 11.1 years (range 41–81 years) were included in this study.
Table 2 shows the neurological symptoms of the patients at the time of admission (multiple symptoms in one patient possible). Gait disturbances and sensory deficits were most frequent and observed in 81 and 94 %, respectively. The time from first symptoms to diagnosis of SDAVF as reported by the patients ranged from 1 month to 20 years (18.06 ± 43.1 months). At the time of treatment, patients had numerous comorbidities, most frequently hypertension, coronary heart disease and diabetes mellitus as well as knee and hip replacement surgery. Mean number of comorbidities was 2.2 ± 1.6. Four patients reported a trauma to the spine such as car accident with polytrauma and vertebral body fracture, injections to the lumbar spine region, heavy lifting with subsequent onset of symptoms, osteoporotic fracture and surgery for lumbar disc or spinal canal stenosis (Table 3). Figure 3 gives an overview of the locations of the fistulas. All but one patient had one fistula point each. The predominant fistula location was at the thoracic (T) 6 level (n = 8). According to the decision algorithm described above, 25 (79 %) patients underwent neurosurgical fistula obliteration, while 5 (16 %) were treated primarily interventionally by endovascular means. Two of these patients had to be operated upon after unsuccessful endovascular intervention. In another two patients, postoperative DSA showed persistent fistulas. One of them was re-operated whereas the other underwent successful neuroradiological embolization. By means of these therapies, complete obliteration of the fistula was achieved and documented by DSA in all patients.
Outcome
The interventions described above resulted in complete angiographically proven occlusion of the fistula in all patients. Three patients deteriorated neurologically after surgery. An epidural hematoma was diagnosed (Fig. 4) and evacuated surgically in one of them, leading to postoperative neurological improvement. In the other two patients (Table 4), the operative course was uneventful but MRI revealed persistent intramedullary edema. All other patients treated neurosurgically or interventionally improved or remained neurologically unchanged.
AL-scores as the primary outcome were available for all 32 patients at every follow-up performed. AL-score at 3-month follow-up (1.78 ± 1.50) did not improve significantly compared to AL-Score preoperatively (2.00 ± 1.41; p = 0.129). A long-term neurological follow-up was also available in all patients with a mean follow-up latency of 34.81 ± 49.41 months. At this follow-up, a significant improvement of AL-score to 1.69 ± 1.51 compared to the preoperative AL-Score was observed (p = 0.027) (Fig. 5).
A significant positive correlation between AL-Score before and 3 months after treatment follow-up (r s = 0.90; p < 0.01), but not duration of symptoms and the AL-Score 3 months after treatment (r s = 0.05; p = 0.769) was observed, with more than 81 % of the variance being explained by the AL-score before treatment. Patient age was also significantly correlated with AL-Score before (r s = 0.37; p < 0.05) and 3 month after treatment (r s = 0.41; p < 0.05). In addition, we correlated the number of comorbidities with the preoperative, postoperative and 3-month follow-up AL-score and there was a statistically significant correlation between those variables (p < 0.05), which remained statistically different (r s = 0.497; p = 0.006) for the last follow-up. When controlling for the effect of age (partial correlation), the correlation of patients AL-Score before and after surgery and at 3-months of follow-up was still significant (p < 0.01). No correlation between AL-Score at 3-month follow-up and localization of the SDAVF was observed (p = 0.422). All other clinical parameters investigated had no impact on patient outcome as assessed by the AL-score.
Of the 32 patients, 12 patients agreed to fill-in the SF-36 QoL questionnaires at last follow-up. The results of the Qol-assessment are displayed in Table 5. In regard to the physical component score a seriously decreased QoL, which was 1 to 2 standard deviations below average, compared to a German standard population, was observed in three patients [8]. For the mental component score, a standard deviation of 2 below average was identified in one patient.
Significant negative correlations between AL-score at the last follow-up and SF-36 domains were observed for the mental component summary score of the SF-36 (p = 0.023) and the subscales of the physical function scale (p = 0.002), general health perception (p = 0.024), social functioning (p = 0.005), role limitations due to emotional problems (p = 0.026) and general mental health (p = 0.003). When analyzing the impact of comorbidities, the correlation between AL-score and the SF-36 component score of mental health and the subscales of general health perception and role limitations due to emotional problems were no longer significant (p > 0.05, respectively).
Discussion
This retrospective study was performed to demonstrate the clinical course of SDAVF, identify variables influencing outcome, to assess the impact of functional impairment on QoL and to discuss the results in the light of the current literature.
Since the first anatomical description of SDAVF dates from the late nineteenth century Hebold and Gaupp [3] and the first presentation of a large patient series by Aminoff and Logue almost 100 years later, several studies on patients with SDAVF have been published, focusing mainly on the comparison of different treatment methods and the description of neurological outcome. Merland et al. published one of the first comparison study between surgical and interventional treatment of SDAVFs in 1985 [27]. Table 6 gives an overview of the recent comparative published studies in this field. As in the majority of these and other descriptive studies, our patient sample also exhibited the well-known clinical characteristics such as the lesion’s prevalence in middle-aged males, predominance of thoracolumbar location, progressive course of symptoms, and nearly uniform occurrence of lower-extremity weakness and sensory disturbance, as well as bladder dysfunction [3, 9, 19, 24, 26, 36].
In our patient series, two patients with prior severe trauma to the spine (one osteoporotic fracture; one polytrauma with vertebral body fracture and later fistula at the same level) and two patients with prior spine surgery were identified. This anamnestic information entices to speculate on a possible traumatic pathomechanism in spinal fistula development, especially since vertebral body fractures, spinal stenosis or disc prolapse and spinal surgery have been implicated in case reports as to be associated with SDAVF occurrence [6, 21, 37, 48]. Yet, the early neurological symptoms of SDAVF are unspecific and may be erroneously attributed to coexisting common abnormalities in older patients, such as spinal stenosis. In this context, Saladino et al. [36] reported, that 10 % of their patients underwent surgery other than occlusion of the fistula for symptoms that, in retrospect, were related to the SDAVF.
The authors of two cases of SDAVF in association with spondylolisthesis speculate on a pathomechanism in which local inflammation due to the spondylolisthesis could have led to subsequent development of an arteriovenous shunt [26, 31]. However, it seems equally possible that in those cases fistula development was the consequence of venous stasis with consecutive thrombosis. In analogy to the well-researched pathomechanisms of intracranial AV fistulas [6, 30, 31], we propose inflammation, infection, trauma, or thrombosis [25, 42, 45] to be possibly common but so far underreported causes of SDAVFs. Thorough clinical history in patients with SDAVF could reveal more anamnestic evidence for such pathophysiological correlations.
In accordance with the literature [9, 24, 36], the predominant fistula location in our series was in the middle and lower thoracic spine (25 patients, 78 %). Sacral locations of SDAVF constitute a rarity. Saladino et al. [36] reported that in only 6.4 % on sacral locations, we observed sacral location of fistula points in 9.3 % (3 patients). Higher thoracic fistula location has been described as a predictor for a poorer neurological outcome by Centano et al. [9], who postulated that the lower thoracic myelon has a better vascular collateral supply, making it less prone for fistula-related ischemia. Since significant relations between location of the SDAVF and clinical outcome could neither be observed in the large series by Saladino et al. [36] nor in our study, we cannot support this theory [2, 9, 10, 22, 35].
Since only the occlusion of the fistulous point will prevent progressive myelopathy caused by venous hypertension [4, 3], a causal therapy is required in all patients harboring SDAVF. Up to date, endovascular and surgical treatment have both been proven safe and effective [13, 19]. In the literature, many authors state a preference for surgical treatment since it is easy, safe, and effective to permanently exclude the fistula microneurosurgically [20, 24, 33, 36, 40, 41]. This is also our experience, especially in cases with more than one fistulous point (see Fig. 3). In our series, surgery generally yielded good results with only two persisting fistulas postoperatively which were located at the lumbar and sacral level of the spine. In one of those patients, the fistulous point at the L1/2 level was located and divided uneventfully at second surgery. In the other, the fistulous point was located very deep at the S2/3 level. Because of the anatomical and vascular characteristics of this region, especially these deeply located sacral SDAVF represent a special challenge for surgical treatment because of the laborious surgical approach. This second patient was treated without complications by endovascular embolization. An endovascular approach could have general advantages in such cases which are difficult to access surgically.
Clinical outcome was good in general in our series, with 30/32 patients improved and only 2 (6.3 %) patients worsened neurologically after surgery without further improvement. Both patients had already deteriorated in the early postoperative period despite regular results of postoperative MRI and spinal DSA. Such unexplainable neurological deterioration has also been described in 11 (7.1 %) patients in the series of Saladino et al. [36], who postulated this to be caused by an increase in abdominal pressure due to intraoperative positioning. We do not assume this to be the cause for deterioration in our patients. Knowing that Hassler et al. [17, 18] reported that the balance between normal vascularization/drainage and ischemia/edema could be negatively affected by any increase in abdominal pressure, all of our patients had put in a prone position for surgery with a freely hanging belly, carefully avoiding any pressure being put on the abdomen. Another possible explanation for the early neurological deterioration is occurrence of partial thrombosis of the venous component causing acute onset ischemia. Indirect support for this theory stems from endovascular series in patients who always receive effective anticoagulation with heparin after embolization and in those where such early neurological deterioration was not observed [23].
In contrast to other authors, we did not observe an influence of duration of symptomatology before treatment with late outcome [9, 14, 17, 23, 32, 35, 36, 38, 39, 41, 44]. In our patient population, only age, preoperative AL-score and number of comorbidities had an impact on late neurological result, while level of fistula point, pretreatment duration of neurological deficits, or other clinical variables did not. Nevertheless, these results should not preclude timely diagnosis and treatment of SDAVFs since clinical experience shows that even patients with severe neurological deficits of short duration can improve substantially. Preoperative AL-score was also a major factor determining outcome in other studies [9, 12, 29, 41, 46], while a high or moderate influence of age on neurological outcome has only been described in two further studies [9, 32]. We postulate that age may have an impact on neurological result because of the higher comorbidities in elderly patients possibly exerting a negative influence on rehabilitation measures.
QoL of patients with SDAVF has so far not been investigated before. In our exploratory series, QoL as assessed by the SF-36 was reduced, as expected. In the 12 patients of whom questionnaires were available, we observed significant negative correlations between several physical QoL domains of the SF-36 but also the mental component summary score with AL-scores at last follow-up, which indicates that physical impairment by fistula-related symptomatology influences psychological well-being negatively. However, some of the lost significance after taking the effect of comorbidities into account again shows that not only disease-specific impairment but also the general state of health and age need to be taken into consideration in counseling the individual SDAVF patient in terms of prognosis and rehabilitation.
Conclusion
Our series and the literature indicate that clinical outcome after treatment of SDAVF is good in general. Both neurosurgical and neurointerventional therapies appear to be safe and effective. However, since microneurosurgery provides direct access to the fistula point, in the interdisciplinary approach utilized in our setting, this was the treatment of choice in most of our cases and also in most other series. Deep-seated sacral fistulas may constitute an exemption from this approach. Neurological deterioration of some patients after adequate treatment is still a problem. Thrombosis of the draining vein is a possible pathophysiological explanation. Further studies should clarify whether postoperative heparinization could prevent progressive edema and postoperative neurological deterioration. In contrast to other studies, in this investigation, the length of the diagnostic process had no statistically significant impact on late outcome. However, age and presurgery AL-Score were relevant factors for the physical disability of patients with arteriovenous fistula after surgery. We assume that neurological recovery of patients is not only explained by fistula-related symptoms but also the higher comorbidity of older patients.
The patients’ perspective was first investigated in this study, but further research on QoL and psychosocial impairment of SDAVF patients is needed to enable individualized counseling and rehabilitation strategies.
References
Afshar JK, Doppman JL, Oldfield EH (1995) Surgical interruption of intradural draining vein as curative treatment of spinal dural arteriovenous fistulas. J Neurosurg 82(2):196–200
Aminoff MJ, Logue V (1974) Clinical features of spinal vascular malformations. Brain J Neurol 97(1):197–210
Aminoff MJ, Logue V (1974) The prognosis of patients with spinal vascular malformations. Brain J Neurol 97(1):211–218
Aminoff MJ, Barnard RO, Logue V (1974) The pathophysiology of spinal vascular malformations. J Neurol Sci 23(2):255–263
Andres RH, Barth A, Guzman R, Remonda L, El-Koussy M, Seiler RW, Widmer HR, Schroth G (2008) Endovascular and surgical treatment of spinal dural arteriovenous fistulas. Neuroradiology 50(10):869–876
Asakuno K, Kim P, Kawamoto T, Ogino M (2002) Dural arteriovenous fistula and progressive conus medullaris syndrome as complications of lumbar discectomy. Case report. J Neurosurg 97(3 Suppl):375–379
Bullinger M (1996) Assessment of health related quality of life with the SF-36 health survey. Die Rehabil 35(3):XVII–XXVII, quiz XXVII–XXIX
Bullinger M, Morfeld M, Kohlmann T, Nantke J, van den Bussche H, Dodt B, Dunkelberg S, Kirchberger I, Kruger-Bodecker A, Lachmann A, Lang K, Mathis C, Mittag O, Peters A, Raspe HH, Schulz H (2003) SF-36 health survey in rehabilitation research. Findings from the North German network for rehabilitation research, NVRF, within the rehabilitation research funding program. Die Rehabil 42(4):218–225
Cenzato M, Debernardi A, Stefini R, D’Aliberti G, Piparo M, Talamonti G, Coppini M, Versari P (2012) Spinal dural arteriovenous fistulas: outcome and prognostic factors. Neurosurg Focus 32(5), E11
Dhandapani S, Gupta A, Singh J, Sharma BS, Mahapatra AK, Mehta VS (2013) Spinal dural arterio-venous fistula: clinico-radiological profile and outcome following surgical occlusion in an Indian neurosurgical center. Neurol India 61(4):406–410
Eide PK, Sorteberg AG, Meling TR, Sorteberg W (2013) Directional intraoperative Doppler ultrasonography during surgery on cranial dural arteriovenous fistulas. Neurosurgery 73(2 Suppl Operative):ons211–ons222, discussion ons222-213
Fugate JE, Lanzino G, Rabinstein AA (2012) Clinical presentation and prognostic factors of spinal dural arteriovenous fistulas: an overview. Neurosurg Focus 32(5), E17
Gemmete JJ, Chaudhary N, Elias AE, Toma AK, Pandey AS, Parker RA, Davagnanam I, Maher CO, Brew S, Robertson F (2013) Spinal dural arteriovenous fistulas: clinical experience with endovascular treatment as a primary therapy at 2 academic referral centers. AJNR Am J Neuroradiol 34(10):1974–1979
Gokhale S, Khan SA, McDonagh DL, Britz G (2014) Comparison of surgical and endovascular approach in management of spinal dural arteriovenous fistulas: a single center experience of 27 patients. Surg Neurol Int 5:7
Hacein-Bey L, Konstas AA, Pile-Spellman J (2014) Natural history, current concepts, classification, factors impacting endovascular therapy, and pathophysiology of cerebral and spinal dural arteriovenous fistulas. Clin Neurol Neurosurg 121:64–75
Hanel RA, Nakaji P, Spetzler RF (2010) Use of microscope-integrated near-infrared indocyanine green videoangiography in the surgical treatment of spinal dural arteriovenous fistulae. Neurosurgery 66(5):978–984, discussion 984–975
Hassler W, Thron A (1994) Flow velocity and pressure measurements in spinal dural arteriovenous fistulas. Neurosurg Rev 17(1):29–36
Hassler W, Thron A, Grote EH (1989) Hemodynamics of spinal dural arteriovenous fistulas. An intraoperative study. J Neurosurg 70(3):360–370
Inagawa S, Yamashita S, Hiramatsu H, Kamiya M, Tanaka T, Sakahara H, Aoyama H (2013) Clinical results after the multidisciplinary treatment of spinal arteriovenous fistulas. Jpn J Radiol 31(7):455–464
Jellema K, Tijssen CC, Sluzewski M, van Asbeck FW, Koudstaal PJ, van Gijn J (2006) Spinal dural arteriovenous fistulas—an underdiagnosed disease. A review of patients admitted to the spinal unit of a rehabilitation center. J Neurol 253(2):159–162
Kang JW, Koo JH, Kim DK, Joo YJ, Kim TH, Song SH (2011) Myelopathy caused by spinal dural arterio-venous fistula after first lumbar vertebral body fracture—a case report. Ann Rehabil Med 35(5):729–732
Kataoka H, Miyamoto S, Nagata I, Ueno Y, Hashimoto N (1999) Intraoperative microdoppler monitoring for spinal dural arteriovenous fistulae. Surg Neurol 52(5):466–472
Kirsch M, Berg-Dammer E, Musahl C, Bazner H, Kuhne D, Henkes H (2013) Endovascular management of spinal dural arteriovenous fistulas in 78 patients. Neuroradiology 55(3):337–343
Krings T, Geibprasert S (2009) Spinal dural arteriovenous fistulas. AJNR Am J Neuroradiol 30(4):639–648
Lawton MT, Jacobowitz R, Spetzler RF (1997) Redefined role of angiogenesis in the pathogenesis of dural arteriovenous malformations. J Neurosurg 87(2):267–274
Meisel K, Yee A, Stout C, Kim W, Cooke D, Halbach V (2013) Arteriovenous fistula after ventriculostomy in aneurysmal subarachnoid hemorrhage. Neurology 80(23):2168
Merland JJ, Assouline E, Rufenacht D. (1985/1986) Dural spinal arteriovenous fistulae draining into medullary veins: clinical and radiological results of treatment (embolization and surgery) in 56 cases. Neuroradiology :283–289
Miyachi E, Izumi T, Matsubara N, Naito T, Haraguchi K, Wakabayashi T (2011) Mechanism of the formation of dural arteriovenous fistula: the role of the emissary vein. Interv Neuroradiol J Perither Neurorad Surg Proced Relat Neurosci 17(2):195–202
Narvid J, Hetts SW, Larsen D, Neuhaus J, Singh TP, McSwain H, Lawton MT, Dowd CF, Higashida RT, Halbach VV (2008) Spinal dural arteriovenous fistulae: clinical features and long-term results. Neurosurgery 62(1):159–166, discussion 166–157
Nishimura Y, Natsume A, Ginsberg HJ (2014) Spinal dural arteriovenous fistula associated with L-4 isthmic spondylolisthesis. J Neurosurg Spine 20:670–674
Nishiyama Y, Kanemaru K, Yoshioka H, Hanihara M, Horikoshi T, Kniouchi H (2013) Intracranial pial arteriovenous fistula caused by dural tenting: case report. Neurologia Medico-Chirurgica
Ofran Y, Yovchev I, Hiller N, Cohen J, Rubin SA, Schwartz I, Meiner Z (2013) Correlation between time to diagnosis and rehabilitation outcomes in patients with spinal dural arteriovenous fistula. J Spinal Cord Med 36(3):200–206
Oldfield EH, Di Chiro G, Quindlen EA, Rieth KG, Doppman JL (1983) Successful treatment of a group of spinal cord arteriovenous malformations by interruption of dural fistula. J Neurosurg 59(6):1019–1030
Ropper AE, Gross BA, Du R (2012) Surgical treatment of type I spinal dural arteriovenous fistulas. Neurosurg Focus 32(5), E3
Ruiz-Juretschke F, Perez-Calvo JM, Castro E, Garcia-Leal R, Mateo-Sierra O, Fortea F, Iza B, Garbizu JM, Villoria F (2011) A single-center, long-term study of spinal dural arteriovenous fistulas with multidisciplinary treatment. J Clin Neurosci Off J Neurosurg Soc Australas 18(12):1662–1666
Saladino A, Atkinson JL, Rabinstein AA, Piepgras DG, Marsh WR, Krauss WE, Kaufmann TJ, Lanzino G (2010) Surgical treatment of spinal dural arteriovenous fistulae: a consecutive series of 154 patients. Neurosurgery 67(5):1350–1357, discussion 1357–1358
Santiago R, Guenther E, Carroll K, Junkins EP Jr (2006) The clinical presentation of pediatric thoracolumbar fractures. J Trauma 60(1):187–192
Sasaki O, Yajima N, Ichikawa A, Yamashita S, Nakamura K (2012) Deterioration after surgical treatment of spinal dural arteriovenous fistula associated with spinal perimedullary fistula. Neurol Med Chir 52(7):516–520
Sherif C, Gruber A, Bavinzski G, Standhardt H, Widhalm G, Gibson D, Richling B, Knosp E (2008) Long-term outcome of a multidisciplinary concept of spinal dural arteriovenous fistulae treatment. Neuroradiology 50(1):67–74
Symon L, Kuyama H, Kendall B (1984) Dural arteriovenous malformations of the spine. Clinical features and surgical results in 55 cases. J Neurosurg 60(2):238–247
Tacconi L, Lopez Izquierdo BC, Symon L (1997) Outcome and prognostic factors in the surgical treatment of spinal dural arteriovenous fistulas. A long-term study. Br J Neurosurg 11(4):298–305
Terada T, Higashida RT, Halbach VV, Dowd CF, Tsuura M, Komai N, Wilson CB, Hieshima GB (1994) Development of acquired arteriovenous fistulas in rats due to venous hypertension. J Neurosurg 80(5):884–889
Thron A (2001) Spinal dural arteriovenous fistulas. Radiologe 41(11):955–960
Ushikoshi S, Hida K, Kikuchi Y, Miyasaka K, Iwasaki T, Abe H (1999) Functional prognosis after treatment of spinal dural arteriovenous fistulas. Neurol Med Chir 39(3):206–212, discussion 212–203
Wachter D, Hans F, Psychogios MN, Knauth M, Rohde V (2011) Microsurgery can cure most intracranial dural arteriovenous fistulae of the sinus and non-sinus type. Neurosurg Rev 34(3):337–345, discussion 345
Wakao N, Imagama S, Ito Z, Ando K, Hirano K, Tauchi R, Muramoto A, Matsui H, Matsumoto T, Matsuyama Y, Ishiguro N (2012) Clinical outcome of treatments for spinal dural arteriovenous fistulas: results of multivariate analysis and review of the literature. Spine 37(6):482–488
Westphal M, Koch C (1999) Management of spinal dural arteriovenous fistulae using an interdisciplinary neuroradiological/neurosurgical approach: experience with 47 cases. Neurosurgery 45(3):451–457, discussion 457–458
Yoshino O, Matsui H, Hirano N, Tsuji H (1998) Acquired dural arteriovenous malformations of the lumbar spine: case report. Neurosurgery 42(6):1387–1389
Disclosure
The authors have no personal financial or institutional interest in any of the drugs, materials, or devices described in this article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
Torstein R. Meling, Oslo, Norway
In this issue of NSR, Özkan et al. present their interdisciplinary neurosurgical/neuroradiological management strategy of spinal dural arteriovenous fistulas (SDAVFs) in 32 patients. The article is a retrospective clinical report. Although somewhat difficult to decipher as the table with patient characteristics is incomplete, from the text it seems that the treatments were endovascular in 5/32 (two of whom were later operated due to residual SDAVFs), microsurgical in 25/32 and another 2/32 patients of whom we only learn about due to their retreatments (endovascular and microsurgical, respectively). Follow-up was 100 % at 3 months. Outcome parameters were obliteration rates, modified Aminoff-Logue (AL) myelopathy scale and quality of life (QoL). The obliteration rates after primary therapy is not stated, but a complete fistula obliteration was eventually achieved and documented by angiography in all patients. The AL-score at the 3-month follow-up did not improve significantly (p = 0.13). Three patients (9 %) deteriorated neurologically after surgery, caused by an epidural hematoma in one and persistent intramedullary edema in two patients. With respect to clinical outcome at 3 months, 30/32 patients improved and only two (6.3 %) patients worsened neurologically after surgery. Of the 32 patients treated, only 12 patients filled in the SF-36 QoL questionnaires, making firm conclusions impossible, although the authors use a German standard population as a reference. Not surprisingly, there is a negative correlation between the AL-scores and QoL as measured by the SF-36 questionnaire, but the statistics are not shown, only the p values. The authors conclude that clinical outcome after treatment of SDAVF is good and that microneurosurgery was the treatment of choice in most cases since it provides direct access to the fistula point. It could preferably have been backed up by their own data showing that retreatment was far more frequent after endovascular than microsurgical treatment in their hands.
Rights and permissions
About this article
Cite this article
Özkan, N., Kreitschmann-Andermahr, I., Goerike, S.L. et al. Single center experience with treatment of spinal dural arteriovenous fistulas. Neurosurg Rev 38, 683–692 (2015). https://doi.org/10.1007/s10143-015-0645-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-015-0645-z