Abstract
Central nervous system solitary fibrous tumors are a new pathological entity. To our knowledge, only 60 meningeal solitary fibrous tumors both in the spinal cord and in the brain have been described in the literature. The 56 previously reported cases of meningeal solitary fibrous tumors are critically reviewed. In addition, we report four new cases of solitary fibrous tumors of the meninges. There is a slight male prepoderance. Meningeal solitary fibrous tumors show a tendency to arise in the posterior fossa (26%) and spine (25%). The treatment was mainly total surgical excision. Radiotherapy was given only to four patients with tumors involving the cerebral parenchyma. Sporadic cases of recurrence and distant metastasis have been reported. The prognosis of meningeal solitary fibrous tumors is still unknown because the follow-up of the reported cases is short. It is probable that cases of solitary fibrous tumors of the meninges have been misdiagnosed as other tumors in the past. The best management of these tumors seems to be total surgical excision whenever possible. It is important that every new case of meningeal SFT be reported to throw light on this particular tumor and to affirm its status as a clinicopathological entity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Solitary fibrous tumor (SFT) is an uncommon spindle cell tumor that typically arises in the visceral pleura [1–4]. Extrapleural sites have been reported, including the pericardium, peritoneum, lung, liver, upper respiratory tract, tunica vaginalis testis, mediastinum, nasal cavities, thyroid and parotid glands, orbit and meninges [3, 5–13]. Histologically, SFTs consist of monomorphic spindle cells organized both into straight, curving or undulating fascicles and unstructured arrangement. Focally prominent bands of hyalinized collagen are characteristic, and the spindle cells are embedded in a conspicuous fibrous matrix [14]. The hystopathogenesis of these tumors has been a matter of debate, and both mesothelial and mesenchymal origin have been proposed [1, 15–20]. Recent studies confirm the mesenchymal origin of SFT [14]. To our knowledge, only 60 cases of SFT of the meninges have been described in the pertinent literature [14, 19, 21–48]. In this report, we describe four new cases of meningeal SFT that fulfill the diagnostic criteria for SFT and critically review the pertinent literature.
Material and methods
Between 1995 and 2000, four cases of SFT of the meninges were surgically treated at our institutes. All tumors were histologically reviewed. The diagnostic criteria for SFT were identical to those used for soft tissues and required the presence of strong CD34 immunoreactivity. The present cases had similar histology and immunohistochemistry. Macroscopically, the tumors were well-circumscribed, tan, and their surfaces were smooth. Upon sectioning, they had a thick, fibrous capsule. Microscopically, the tumor in all cases consisted of spindle cells in a collagenous background. The cellularity and the amount of collagen deposition were variable. Areas of relative hypocellularity with abundant collagen deposition were alternated with more cellular areas. Spindle cells were arranged in fascicles in some areas and in an unstructured pattern in others. There were many vessels. There was no evidence of necrosis, hemorrhage or microcystic degeneration. In all cases, MIB-1 labeling was <1%. Immunohistochemical analyses showed strong diffuse reactivity for CD34 and vimentin, but negative reaction for epithelial membrane antigen, S-100 protein and desmin.
Case 1
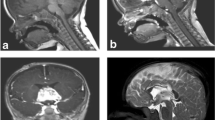
A 38-year-old man was admitted to our clinic with a 2-year history of right complete trigeminal neuralgia. Magnetic resonance imaging (MRI) demonstrated a right gasserian ganglion lesion with homogeneous enhancement after injection of gadolinium-DTPA (Fig. 1a).
a Case 1: axial contrast-enhanced T1-weighted images showing homogeneous enhancement of a left gasserian tumor. b Case 1: photomicrograph showing varying numbers of spindle cells arranged randomly in a collagenous background (H&E). c Case 2: sagittal contrast-enhanced T1-weighted images showing dyshomogeneous enhancement of the lesion similar to a neurinoma. d Case 2: photomicrograph showing a highly cellular tumor composed of spindle cells arranged in fascicles between bands of collagen (H&E)
A subtotal removal of the tumor was performed. The patient had a good recovery. The histopathologic diagnosis of the lesion was SFT (Fig. 1b). The postoperative course was uneventful. Fifty months after surgery, the illness was stable.
Case 2
A 54-year-old man presented with a 3-month history of left arm paresthesia and brachialgia. MRI revealed an intraspinal mass at the level of C7-D1 (Fig. 1c). The tumor resembled a spinal neurinoma and showed inhomogeneous contrast enhancement. Total excision was carried out, and the patient had good recovery. The histologic diagnosis of the lesion was SFT (Fig. 1d). There was no clinical or MRI evidence of recurrence after 15 months.
Case 3
A 29-year-old woman presented with a 3-month history of increasing headache, nausea and vomiting. MRI demonstrated a rounded lesion in the right cerebellar hemisphere with tentorial attachment and a small supratentorial extension. The adjacent ventricular recess appeared to be compressed. At operation, a well-demarcated lesion arising from the dura was subtotally excised. A radiotherapeutic course of radiosurgery with LINAC was performed (20 Gy) on the residual disease. The histopathological diagnosis was SFT. The postoperative course was uneventful. The patient was lost to follow-up, but 3 years after surgery was disease-free.
Case 4
A 34-year-old man presented with a 1-year history of headache and left limb weakness. Neurologic examination showed a slight left hemiparesis (4/5). MRI revealed a mass located in the right frontal lobe. The lesion did not enhance after injection of gadolinium-DTPA. The tumor was totally excised. The postoperative course was uneventful, and hemiparesis resolved completely. A diagnosis of SFT was made. Five years after surgery, the patient remains well.
Discussion
Most cases of SFT have been reported to arise in the visceral pleura [1–4]. However, these tumors have now been documented in many other sites outside the thoracic cavity [3, 5–13]. SFT of the meninges was first reported as a lesion distinct from fibrous meningioma by Carneiro et al. in 1996 [24]. Since then, a further 53 cases of SFT have been reported directly involving the neuraxis, both in the spinal cord and in the brain [14, 19, 21–48]. The reports of meningeal SFT accessible to us include 45 cases in the brain and 15 in the spine. Among 18 cases reported by Tihan et al. [44], four non-meningeal cases are included, and three cases reported by Perry [39] are partially described, so that, including the present series, we have found that meningeal SFT occurred in 20 women and 23 men with a mean age of 47.6 years (range: 11 to 73 years; Tables 1, 2). Meningeal SFTs show a tendency to arise in the posterior fossa (26%) and spine (25%). SFTs of the central nervous system typically show a clear dural origin so that they may mimic meningiomas radiographically and surgically [34]. However, most spinal SFTs have no dural attachment (Table 2).
The histopathogenesis of SFTs has been debated, but now the mesenchymal origin of these tumors is generally accepted on the base of immunohistochemical, ultrastructural and cell culture evidence [6, 14, 49]. Immunohistochemical studies of SFT, showing strong CD34 reactivity of the cells, suggest that this tumor is mesenchymal in nature. CD34 is a transmembrane glycoprotein that has been found in hematopoietic stem cells, endothelial cells and an ubiquitous sub-population of fibroblasts, from which SFT may originate [50–54]. Furthermore, negative immunoreactivity to epithelial markers and, ultrastructurally, the absence of mesothelial structures confirm a mesenchymal origin of these tumors. Therefore, SFT should be classified into the group of rare mesenchymal, non-meningothelial tumors of the central nervous system. SFTs are usually negative also for vascular, neural crest and muscle markers [15, 34, 54] and are positive for vimentin. It should be highlighted that CD34 reactivity is not an exclusive marker of SFTs, but it can be identified also in meningiomas, neurofibromas and hemangiopericytoma as a result of the inclusion of dural fibroblasts within tumors. However, the pattern of CD34 reactivity in this latter tumor is weak and patchy, [34, 39, 51]. Immunoreactivity for bcl-2 has often been identified in SFTs and may be useful in the differential diagnosis [9, 22, 29, 34, 39, 51]. SFTs show distinctive cytological features that allow diagnosis in cytological specimens [55]. Clayton et al. [55], in a recent study, reported as characteristic features of SFT: the presence of oval to polygonal tumor cells, cellularity ranging from scant to moderate, a background containing irregular ropy elements of collagen and a few inflammatory cells, loose aggregates of cells enmeshed in a collagen matrix and nuclei uniformly bland, with evenly distributed, fine granules of chromatin. SFTs can mimic other tumors, rendering diagnosis difficult [4]. This is especially true when SFTs arise in uncommon sites such as the neuraxis. It is fundamental to be aware of this tumor and know that it must be included in the differential diagnosis with hemangiopericytoma and fibrous meningioma, schwannoma, neurofibroma and fibrosarcoma [24, 38, 39, 51]. This distinction is imperative, especially for those tumors that require postoperative radiotherapy. The fact that SFT has been diagnosed with increasing frequency in recent years as the result of improved methods of pathologic examination is reassuring.
The natural history of SFT is not completely defined because the period of follow-up of the reported cases of SFT is quite short. Histologically, the majority of SFT appear to be benign, but an analysis of 223 pleural SFT identified 82 cases with atypical features [4]. Clinically, 13–23% of pleural SFTs manifest with local invasion, recurrence, intrathoracic spread or distant metastasis [51]. SFT of the meninges generally pursues a slow, indolent and non-aggressive course, with only rare recurrence or metastasis [2, 28, 29, 37]. The malignant histological features described for SFTs of the pleura could be applied for meningeal SFTs. These malignant features include high cellularity, a high number of mitotic figures, necrosis and nuclear pleomorphism [4]. Three examples of clinically and histologically malignant meningeal SFTs have been reported [4, 29, 44]. All these cases showed an increased number of mitotic figures, hypercellularity, high proliferation index and nuclear pleomorphism. However, it has been noted that if the tumor is amenable to total removal, the above-mentioned histological findings are not predictive of malignant behavior [1, 3, 50]. Usually, the glucose metabolic rate of tumors is a good predictor of their malignancy [26]. Unfortunately, only one case of meningeal SFT studied with FDG-PET has been described [56]. In this case, FDG-PET demonstrated faint uptake of glucose in the tumor, which was of malignant nature. Also, expression of CD34 could be a useful predictor for SFTs because it may be lost focally in some high-grade tumors [44, 57]. Complete removal, rather than histological appearance, seems to be the most important prognostic marker of SFT [1, 21, 34]. This is mainly suggested by the experience with SFT at other sites, because the follow-up period is quite short in most cases of meningeal SFTs reported until now. SFTs seem to be successfully managed by surgery alone. A total excision has been possible for most meningeal SFTs, and evidence of recurrence or metastasis were present in rare cases [23, 24]. The role of postoperative radiotherapy in meningeal SFTs with atypical features is unknown because very few cases have been described. It is interesting to note that two spinal cases recurred and three intracranial cases recurred, of which one metastasized to the cervical lymph nodes and lung parenchyma. Unfortunately, it is difficult to establish if the recurrence rate of these meningeal types is a lot lower than that of the pleural (13–23%) or if this differences results from the shorter follow-up. In conclusion, prognosis of meningeal SFT remains unclear; consequently, careful and long-term follow-up remain mandatory for all meningeal SFTs.
References
Briselli U, Mark EJ, Dickerson GR (1981) Solitary fibrous tumor of the pleura: eight new cases and review of 360 cases in the literature. Cancer 47:2678–2689
Dalton WT, Zolliker AS, Macaughey WTE, Jaques J, Kannerstein M (1979) Localized primary tumors of the pleura. Cancer 44:1465–1475
Dorfman DM, To K, Dickersin GR, Rosenberg AE, Pilch BZ (1994) Solitary fibrous tumor of the orbit. Am J Surg Pathol 18:281–287
England DM, Hochhlolzer L, Mc Carhy MJ (1989) Localized benign and malignant fibrous tumors of the pleura. Am J Surg Pathol 13:640–658
Caneselle-Teijeiro J, Varala-Duran, Fonseca E, Villanueva JP, Sobrinho-Simoes M (1994) Solitary fibrous tumor of the thyroid. Am J Clin Pathol 101:535–538
El-Naggar AK, Ro JY, Ayala AG, Ward R, Ordonez NG (1989) Localized fibrous tumor of the serosal cavities. Am J Clin Pathol 92:561–565
Kottke-Marchant K, Hart WR, Broughton T (1989) Localised fibrous tumor (localised fibrous mesothelioma) of the liver. Cancer 64:1096–1102
Parveen T, Fleischman J, Petrelli M (1992) Benign fibrous tumor of the tunica vaginalis testes. Report of a case with light, electron microscopi, and review of the literature. Arch Pathol Lab Med 116:277–861
Suster S, Nascimento AG, Miettinen M, Nickel JZ, Moran CA (1995) Solitary fibrous tumors of soft tissue. Am J Surg Pathol 19:1257–1266
Witkin GB, Rosai J (1989) Solitary fibrous tumor of the mediastinum: a report of 14 cases. Am J Surg Pathol 13:547–557
Witkin GB, Rosai J (1991) Solitary fibrous tumor of the upper respiratory tract: a report of six cases. Am J Surg Pathol 15:842–848
Young RH, Clement PB, McCaughei WTE (1990) Solitary fibrous tumors (“fibrous mesotheliomas”) of the peritoneum. Arch Pathol Lab Med 114:493–495
Yousem SA, Flynn SD (1988) Intrapulmonary localized fibrous tumor. Am J Clin Pathol 89:365–369
Brunori A, Cerasoli S, Donati R, Gingaspero F, Chiappetta F (1999) Solitary fibrous tumor of the meninges: two new cases and review of the literature. Surg Neurol 51:636–640
Flint A, Weiss SW (1995) CD34 and keratin expression distinguishes solitary fibrous tumor (fibrous mesothelioma) of pleura from desmoplastic mesothelioma. Hum Pathol 26:428–431
Foster EA, Ackerman LV (1960) Localized mesotheliomas of the pleura. The pathologic evaluation of 18 cases. Am J Clin Pathol 34:349–364
Hernandez FJ, Fernandez BB (1974) Localized fibrous tumor of the pleura: a light and electron microscopic study. Cancer 34:1667–1674
Kawai T, Yakumaru K, Mitaka A, Kageyama K, Torikata C, Shimosato Y (1978) Solitary (localized) pleural mesothelioma. A light and microscopic study. Am J Surg Pathol 2:365–375
Osamura RY (1977) Ultrastructure of localized fibrous mesothelioma of the pleura. Report of a case with histogenic considerations. Cancer 39:139–142
Stout AP, Murray MR (1942) Localized pleural mesothelioma. Investigations of its characteristics and histogenesis by the method of tissue culture. Arch Pathol 32:951–964
Ahn JY, Shim JY, Yang WIY, Kim TS (2001) Meningeal solitary fibrous tumor as an unusual cause of esophthalmos: case report and review of the literature. Neurosurgery 48:1362–1366
Alston SR, Francel PC, Jane JA (1997) Solitary fibrous tumor of the spinal cord. Am J Surg Pathol 21:477–483
Brunnemann RB, Ro JY, Ordonez NG, Mooney J, El-Naggar AK, Ayala AG (1999) Extrapleural solitary fibrous tumour: a clinico-pathological study of 24 cases. Mod Pathol 12:1034–1042
Carneiro SS, Scheithauer BW, Nascimento AG, Hirose TN, Davis TH (1996) Solitary fibrous tumor of the meninges: a lesion distinct from fibrous meningioma. A clinicopathologic and immunohistochemical study. Am J Clin Pathol 106:217–224
Centeno RS, Gorgulho Pedroso AA, Pereira EM, Neta AR (2002) Tumor fibroso solitario da meninge. Relato de Caso Arq Neuro-Psyquiatr 60:1–8
Challa VR, Kilpatrick SE, Ricci P, Wilson JA, Kelly DL (1998) Solitary fibrous tumors of the meninges. Clin Neuropathol 17:73–78
Donnelan RB, Govender D, Chite SH, Landers AT (2002) An unusual presentation of solitary fibrous tumour. Spine 25:749–751
Gentil Perret A, Mosnier JF, Duthel R, Brunon J, Barral F, Boucheron S (1999) Solitary fibrous tumor of the meninges. Ann Pathol 19:532–535
Hasegawa T, Matsuno Y, Shimoda T, Hasegawa F, Sano T, Hirohashi S (1999) Extrathoracic solitary fibrous tumors: their histological variability and potentially aggressive behaviour. Hum Pathol 30:1464–1473
Kanahara T, Hirokawa M, Shimizu M, Terayama K, Nakamura E, Hino Y, Mikawa Y, Manabe T (1999) Solitary fibrous tumor of the spinal cord. Report of a case with scrape cytology. Acta Cytol 43:425–428
Kataoka H, Akijama Y, Kubo S, Itoh H, Hamasuna R, Tjima N, Koono M (1999) Solitary fibrous tumor of the spinal rootlet: case report and literature survey. Pathol Int 49:826–830
Kuchelmeister K, Scheuerle A, Bohle RM, Kretshmer T, Richter HP, Schachenmayr W (1997) Solitary fibrous tumor—a new differential diagnosis in surgical neuropathology. Report of two cases. Clin Neuropathol 16:270–274
Malek AM, Waller SJ, Price DL, Madsen JR (1997) Solitary fibrous tumor presenting as a symptomatic intraspinal mass. Case report. Neurosurgery 40:844–847
Martin AJ, Fisher C, Igbaseimokumo U, Jarosz JM, Dean AF (2001) Solitary fibrous tumors of the meninges: case series and literature review. J Neurooncol 54:57–69
Mordani JP, Haq IU, Singh J (2000) Solitary fibrous humour of the spinal cord. Neuroradiology 42:679–681
Nawashiro H, Nagakawa S, Osada H, Katoh H, Ohnuki A, Tsuzuki N, Miyazawa T, Shima K, Ogata S, Aida S (2000) Solitary fibrous tumor of the meninges in the posterior cranial fossa: magnetic resonance imaging and histological correlation. Case report. Neurol Med Chir (Tokyo) 40:432–434
Ng HK, Choi PCL, Wong CW, To KF, Poon WS (2000) Metastatic solitary fibrous tumor of the meninges. Case report. J Neurosurg 93:490–493
Nikas DC, De Girolami U, Folkerth RD, Bello L, Zamani AA, Black PMcL (1999) Parasagittal solitary fibrous tumor of the meninges. Case report and review of the literature. Acta Neurochir (Wien) 141:307–313
Perry A, Scheitauer BW, Nascimento AG (1997) The immunophenotypic spectrum of meningeal hemangiopericytoma: a comparison with fibrous meningioma and solitary fibrous tumor of the meninges. Am J Surg Pathol 21:1354–1360
Prayson RA, McMahon JT, Barnett GH (1997) Solitary fibrous tumor of the meninges. Case report and review of the literature. J Neurosurg 86:1049–1052
Rodriguez L, Lopez J, Marin A, Cardozo D, Molina O, Cardozo J (2000) Solitary fibrous tumor of the meninges. Clin Neuropathol 19:45–48
Slavik T, Bentley RC, Gray L, Fuchs HE, McLendon RE (1998) Solitary fibrous tumor of the meninges occurring after irradiation of a mixed germ cell tumor of the pineal gland. Clin Neuropathol 17:55–60
Zamecnik M, Michal M (1998) Solitary fibrous tumor (fibrous mesothelioma). Report of 2 cases in an extraosseous location. Cesk Patol 34:58–62
Tihan T, Viglione M, Rosenblum MK, Olivi A, Burger PC (2003) Solitary fibrous tumors of the central nervous system. Clinicopathological review of 18 cases and comparison with meningeal hemangiopericytoma. Arch Pathol Lab Med 27:432–439
Barron J, Lownie SP, Lee DH, Hammond RR (2001) June 2001: 61 year old woman with confusion and obtundation. Brain Pathol 11:485–487
Suzuki SO, Fukui M, Nishio S, Iwaki T (2000) Clinicopathological features of solitary fibrous tumor of the meninges: an immunohistochemical reappraisal of cases previously diagnosed to be fibrous meningioma or hemangiopericytoma. Pathol Int 50:808–817
Morimitsu Y, Masayoshi N, Masanori H, Hiroshi H (2000) Extrapleural solitary fibrous tumor: clinicopathologic study of 17 cases and molecular analysis of the p53 pathway. Acta Pathol Microbiol Immunol Scand 108:617–625
Vorster SJ, Prayson RA, Lee JH (2000) Solitary fibrous tumor of the thoracic spine. Case report and review of the literature. J Neurosurg 92 [Suppl 2]:217–220
Alvarez-Fernandez E, Diez-Nau MD (1979) Malignant fibrosarcomatous mesothelioma and benign pleural fibroma (localized fibrous mesothelioma) in tissue culture. Cancer 43:1658–1663
Chan JKC (1997) Solitary fibrous tumor-everywhere, and a diagnosis in vogue. Histopathology 31:568–576
Cummings TJ, Burchette JL (2002) CD34 and dural fibroblasts: the relationship to solitary fibrous tumor and meningioma. Acta Neuropathol 102:349–354
Nickoloff BJ (1911) The human progenitor cell antigen (CD34) is localized on endothelial cells, dermal dendritic cells, and perifollicular cells in formalin-fixed normal skin, and on proliferating endothelial cells and stromal spindle-shaped cells in Kaposi’s sarcoma. Arch Dermatol 27:523–529
Traweek ST, Kandalaft PL, Metha P, Battifora H (1991) The human hematopoietic progenitor cell antigen (CD34) in vascular neoplasia. Am J Clin Pathol 96:25–31
Van de Rijm M, Rouse RV (1994) CD34 a review. Appl Immunohistochem 2:71–80
Clayton AC, Salomao DR, Keeney GL, Nascimento AG (2001) Solitary fibrous tumor: a study of cytologic features of six cases diagnosed by fine-needle aspiration. Diagn Neuropathol 25:172–176
Hayashi S, Kurihara H, Hirato J, Sasaki T (2001) Solitary fibrous tumor of the orbit with extraorbital extension: case report. Neurosurgery 49:1241–1245
Yokoi T, Tsuzuki T, Yatabe Y, Suzuki M, Kurumaya H, Koshikawa T, Kuhara H, Kuroda M, Nakamura N, Nakatani Y, Kakudo K (1998) Solitary fibrous tumor: significance of p53 and CD34 immunoreactivity in its malignant transformation. Histopathology 32:423–432
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Caroli, E., Salvati, M., Orlando, E.R. et al. Solitary fibrous tumors of the meninges. Neurosurg Rev 27, 246–251 (2004). https://doi.org/10.1007/s10143-004-0331-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-004-0331-z