Abstract
Background
Endoscopic submucosal dissection (ESD) has been used to treat patients with early gastric cancers (EGCs), including large and ulcerative lesions. Few published data exist on the long-term outcomes of this treatment with median follow-up periods of over 5 years; we therefore aimed to assess the long-term outcomes of EGC patients undergoing ESD.
Methods
A total of 1,956 consecutive patients with 2,210 EGC lesions at initial onset underwent ESD with curative intent at our hospital from 1999 to 2006. We performed a retrospective analysis of the 5-year survival of EGC patients undergoing curative ESD for absolute indications or for expanded indications.
Results
For the pathological curability, curative ESD for absolute indications, curative ESD for expanded indications of differentiated-type EGC, and curative ESD for undifferentiated-type EGC were achieved in 781, 713, and 43 patients, respectively. The median follow-up period was 83.3 months. Among the 1,537 patients, there were only two patients with recurrence, including one who developed a regional lymph node (LN) metastasis and one who developed a distant LN metastasis with local recurrence resulting in gastric cancer-related death. Seven died from metachronous gastric cancers. The 5-year rates of overall survival, disease-specific survival, and relative survival were 92.6, 99.9, and 105.0 %, respectively.
Conclusions
Based on the high rate of 5-year survival among EGC patients undergoing curative ESD for absolute indications or for expanded indications in the largest patient series with a median follow-up period of over 5 years, ESD could be employed as a standard treatment for EGC lesions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Endoscopic submucosal dissection (ESD) is now widely accepted as an initial treatment for early gastric cancers (EGCs), with a negligible risk of lymph node (LN) metastasis [1–4]. According to the Japanese gastric cancer treatment guideline 2010 (ver. 3), ESD is performed based on two independent sets of indications: absolute indications for standard treatment and expanded indications for investigational treatment [4]. A tumor meeting the absolute indications is defined as an “EGC lesion clinically diagnosed as an intramucosal (T1a) differentiated-type cancer ≤2 cm in diameter without ulceration [UL(−)].” On the other hand, the expanded indications include “tumors clinically diagnosed as T1a and of the (a) differentiated-type, UL(−) and >2 cm in diameter, (b) differentiated-type, UL(+) and ≤3 cm in diameter, or (c) undifferentiated-type, UL(−) and ≤2 cm in diameter.” Recently, the number of EGC patients undergoing ESD has been increasing in Japan. Although a number of reports have indicated favorable short- and long-term outcomes of ESD for EGC, there are few published data on the long-term outcomes of gastric ESD with a median follow-up period of over 5 years [5–13]. Therefore, we attempted to assess the long-term outcomes of ESD for EGC based on the data from our large consecutive patient series.
Methods
Patients
We included a total of 2,204 patients with 2,482 EGC lesions who underwent ESD after being clinically diagnosed as fulfilling the absolute or expanded indications for ESD at our hospital from January 1999 to December 2006 in order to assess 5-year long-term outcomes [4]. After excluding 248 patients with 272 EGC lesions (73 patients with 90 EGC lesions who had a history of endoscopic resection for other EGC lesions before 1999, 87 patients with 87 locally recurrent EGC lesions after endoscopic resection, 64 patients with 66 EGC lesions in the remnant stomach after gastrectomy, and 24 patients with 29 EGC lesions in the gastric tube after esophagectomy), 1,956 consecutive patients [male:female ratio 3.5:1; median age 67 years (range 29–93 years)] with 2,210 initial-onset EGC lesions in the normal stomach (upper/middle/lower thirds of the stomach 349/954/907) were assessed in this study.
The risks and benefits of the ESD procedure were thoroughly explained to every patient, and written informed consent was obtained from each patient prior to the treatment with ESD.
We retrospectively assessed the pathological curability of these EGC patients and the long-term outcomes of the EGC patients who underwent curative ESD for absolute indications or for expanded indications. The long-term outcomes included the 5-year rates of overall survival, disease-specific survival, and relative survival [14–17]. We also analyzed the incidences of local recurrence, LN and distant metastasis, and metachronous gastric cancers (MGCs) in these patients. The study protocol was approved by the Institutional Review Board of National Cancer Center (protocol no. 2013-125).
Pathological curability
According to the Japanese gastric cancer treatment guideline 2010 (ver. 3), curative ESD for the absolute indications included: “en-bloc resection, tumor size ≤2 cm, differentiated-type histology, pT1a, horizontal margin (HM) 0, vertical margin (VM) 0, lymphatic invasion (ly) (−), venous invasion (v) (−)” [4, 18]. In addition, the resection is considered to be curative ESD for the expanded indications when all of the following conditions are fulfilled: “en-bloc resection, HM0, VM0, ly(−), v(−), and: (a) tumor size >2 cm, differentiated-type cancer, pT1a, UL(−); (b) tumor size ≤3 cm, differentiated-type cancer, pT1a, UL(+); (c) tumor size ≤2 cm, undifferentiated-type cancer, pT1a, UL(−); (d) tumor size ≤3 cm, differentiated-type cancer, pT1b (SM1).” Any resection that did not satisfy any of the above criteria was considered non-curative ESD. In the present study, based on the above mentioned confirmation of pathological curability, the patients were divided into (1.1) curative ESD for absolute indications; (1.2) curative ESD for expanded indications of differentiated-type EGC; (1.3) curative ESD for expanded indications of undifferentiated-type EGC; and (2) non-curative ESD. For patients with multiple EGCs, the curability was determined based on the highest of the above four categories fulfilled by the EGC (category 2; non-curative ESD >category 1.3; curative ESD for expanded indications of undifferentiated-type EGC >category 1.2; curative ESD for expanded indications of differentiated-type EGC >category 1.1; curative ESD for absolute indications).
The histological type was defined according to the major histological features of the lesion. According to the Japanese classification of gastric carcinoma and Japanese gastric cancer treatment guidelines, differentiated-type adenocarcinoma includes tubular adenocarcinoma and papillary adenocarcinoma, while undifferentiated-type adenocarcinoma includes poorly differentiated adenocarcinoma, signet-ring cell adenocarcinoma and mucinous adenocarcinoma [4, 18]. Infiltration of the submucosa (SM) is subclassified as SM1 (<500 μm from the muscularis mucosae) or SM2 (≥500 μm from the muscularis mucosae) [4, 18].
Follow-up care after ESD
According to the Japanese gastric cancer treatment guidelines, EGC patients undergoing curative ESD for absolute indications were followed up with esophagogastroduodenoscopy examinations every 6–12 months [4]. Patients undergoing curative ESD for expanded indications additionally underwent abdominal computed tomography, abdominal ultrasonography or endoscopic ultrasonography examinations every 6–12 months. In general, patients with non-curative ESD were referred for additional gastrectomy with LN dissection.
Collection of the long-term outcome data
The long-term outcome data on the EGC patients who underwent curative ESD for absolute indications or for expanded indications were collected retrospectively from the medical records from August 2013 to June 2014. For patients who had not visited our hospital regularly, the long-term outcome data were retrieved by requesting the referring physicians, contacting the patient’s home, or checking the statistical data maintained by the local government registry.
Statistical analysis
Survival time was calculated as the interval between the date of the first treatment and the date of death or the last date on which the patient was confirmed to be alive. Survival curves were drawn using the Kaplan–Meier method with the statistical analysis software SPSS, ver. 20 (SPSS Japan Inc., Tokyo, Japan). The relative survival rate was calculated as the ratio of the observed overall survival of the cancer patients divided by the expected survival of persons from the general population matched for age and sex, as obtained from the cohort survival table based on life tables for the Japanese population [14–17]. The relative survival rate was calculated according to the Ederer II method using Stata, Version 9.2 (Stata Corporation, College Station, TX, USA).
Results
Pathological curability
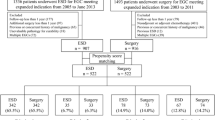
Investigating the pathological curability using a patient-based analysis, curative ESD for absolute indications, curative ESD for expanded indications of differentiated-type EGC, and curative ESD for undifferentiated-type EGC were reportedly achieved in 781, 713 and 43 patients, respectively (out of a total of 1,537 patients undergoing curative ESD for absolute indications or for expanded indications), while non-curative ESD was reported in 419 patients (Fig. 1). In addition, the 713 patients with curative ESD for expanded indications of differentiated-type EGC included 373 with differentiated intramucosal EGC >2 cm without ulceration, 212 with differentiated intramucosal EGC ≤3 cm with ulceration, and 128 with differentiated SM1 EGC ≤3 cm.
Long-term outcomes
The long-term outcomes in the 1,537 EGC patients who underwent curative ESD for absolute indications or for expanded indications are shown in Fig. 1. The median follow-up period was 83.3 months (range 0–180 months). For 92 % of the patients undergoing curative ESD, information on whether the patient was alive for at least 5 years or had died could be confirmed by investigating the individual patient data thoroughly. A patient who was followed up for 0 months died suddenly of acute aortic dissection (non-gastric cancer-related death) after the gastric ESD. For the remaining 8 % of patients undergoing curative ESD, information on whether the patients were alive for at least 5 years or they died could not be confirmed, as the median follow-up period of these patients was 46.5 months (range 3.3–56.9 months).
Among the 781 patients with curative ESD for absolute indications and 43 with curative ESD for expanded indications of undifferentiated-type EGC, there were no cases of local recurrence or regional or distant LN metastasis. In contrast, among the 713 with curative ESD for expanded indications of differentiated-type EGC, regional LN metastasis developed in one patient 4.1 years after the initial ESD, and distant LN metastasis with local recurrence was noted in one patient 7.2 years after the initial ESD (Fig. 1). The primary lesions in the two patients were SM1 invasive cancers. The patient who developed regional LN metastasis underwent salvage surgery, and no recurrence was detected during the follow-up period of 4.7 years after the surgery [19]. The other patient who developed distant LN metastasis with local recurrence received systemic chemotherapy but died of gastric cancer about 2 years after recurrence [20]. In addition, among the EGC patients with curative ESD for absolute indications, there were three cases of gastric cancer-related death from MGCs, while four patients among those with curative ESD for expanded indications of differentiated-type EGC died of MGCs (Fig. 1).
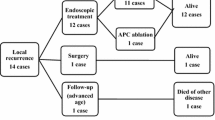
A total of 348 MGC lesions were detected in 244 patients (15.9 %). Further management and follow-up results for these patients with MGC lesions are shown in Fig. 2. Among the seven cases of gastric cancer-related death from MGCs, the MGCs were of a high malignancy grade at the time of their detection in three cases (cases 1, 2, and 3), even though they had received periodic follow-up examinations at our hospital; case 1 was a case of SM1-invasive gastric cancer with lymphatic invasion that was detected 1.0 years after the initial ESD and resulted in death 2.3 years after the initial ESD; case 2 was a case of gastric adenosquamous carcinoma with SM2 invasion that was detected 2.3 years after the initial ESD and resulted in death 4.6 years after the initial ESD; and case 3 was a case of gastric choriocarcinoma with subserosal invasion that was detected 5.1 years after the initial ESD and resulted in death 6.3 years after the initial ESD. In cases 4, 5, 6, and 7, fatal advanced MGCs were detected 5.1, 7.3, 8.7, and 7.3 years after the initial ESD at other hospitals because periodic surveillance examinations were not performed over the 5 years after the initial ESD. These patients died 7.3, 7.8, 9.0, and 8.4 years after the initial ESD. In addition, a case of advanced MGC was detected 6.3 years after the initial ESD at another hospital; in this case, the patient received only careful observation without active treatment due to her older age, and she died of abdominal aortic aneurysm rupture.
Further management and follow-up results for the MGCs. ca cancer, ESD endoscopic submucosal dissection, MGC metachronous gastric cancer, M mucosa, SM submucosa. Hash curative ESD for absolute indications or for expanded indications. Asterisk excluding 1 case of gastric cancer-related death from primary EGC with distant LN metastasis and local recurrence and 7 cases of gastric cancer-related death from MGCs
The 5-year overall survival rate of a total of 1,537 EGC patients undergoing curative ESD for absolute indications or for expanded indications was 92.6 %. The 5-year overall survival rates of the EGC patients with curative ESD for absolute indications, with curative ESD for expanded indications of differentiated-type EGC, and with curative ESD for expanded indications of undifferentiated-type EGC were 93.0, 92.2, and 92.9 %, respectively (Fig. 3). The 5-year disease-specific survival rate of a total of 1,537 EGC patients with curative ESD for absolute indications or for expanded indications was 99.9 %. The 5-year disease-specific survival rates of the EGC patients with curative ESD for absolute indications, with curative ESD for expanded indications of differentiated-type EGC, and with curative ESD for expanded indications of undifferentiated-type EGC were 99.9, 99.9, and 100 %, respectively (Fig. 4). In addition, the 5-year relative survival rate of a total of 1,537 EGC patients with curative ESD for absolute indications or for expanded indications was 105.0 %, and the 5-year relative survival rates of the EGC patients with curative ESD for absolute indications, with curative ESD for expanded indications of differentiated-type EGC, and with curative ESD for expanded indications of undifferentiated-type EGC were 104.7, 105.6, and 100 %, respectively.
Discussion
Recently, ESD has been performed to treat EGCs, including large and ulcerative lesions, and there have been a number of published reports on the short- and long-term outcomes of ESD for EGC [1–13]. However, there are few published reports on the long-term outcomes with median follow-up periods of over 5 years (Table 1) [6–13]. Kosaka et al., Tanabe et al., and Ohnita et al. [10, 11, 13] reported favorable long-term outcomes of gastric ESD with median follow-up periods of over 5 years; however, the number of cases included in those studies was fewer than the number included in our study. Oda et al. [12] also reported the favorable long-term outcomes of curative gastric ESD over follow-up periods of at least 5 years; however, they reported preliminary results of a multicenter questionnaire survey without collecting individual patient data, meaning that they could not estimate the long-term outcomes using the Kaplan–Meier method. In addition, a previous study from our institution reported by Gotoda et al. [8] enrolled a larger number of patients (n = 1485) and revealed favorable long-term outcomes of curative gastric ESD. However, the median follow-up period was less than 4 years. Another previous study from our institution reported by Abe et al. [9] showed excellent long-term outcomes of curative ESD only for undifferentiated-type EGC lesions. To the best of our knowledge, this is the largest study demonstrating the high rate of 5-year survival of EGC patients undergoing curative ESD for absolute indications or for expanded indications with a median follow-up duration of over 5 years; the 5-year overall survival rate was comparable not only to that following surgical treatment, but also to that of persons from the general population with the same age and sex distribution. The present study indicated a 5-year overall survival rate after curative ESD for absolute indications or for expanded indications of around 92.6 % after a median follow-up duration of 5 years (83.3 months), which was as high as the reported rate of 93.5 % in patients with intramucosal gastric cancer after gastrectomy with LN dissection according to the current Japan Gastric Cancer Association nationwide registry [21]. In addition, more importantly, the 5-year relative survival rate of EGC patients undergoing curative ESD for absolute indications or for expanded indications in the present study was almost 100 %, which demonstrated that curative ESD can enable 5-year survival rates comparable to those of persons of matched sex and age from the general population to be achieved [14–17]. Furthermore, the 5-year disease-specific survival rate of 99.9 % noted in the present study was also comparable to the previously reported rate of 99.3 % for mucosal EGC patients treated by gastrectomy with LN dissection [22]. Therefore, the present study is very significant in that it allows precise clarification of the effectiveness of ESD for EGC from the perspective of long-term survival. ESD could be a standard treatment for the EGC lesions that meet not only the absolute but also expanded indications for ESD.
The results of the present study were based on the medical records of EGC patients undergoing curative ESD for absolute indications or for expanded indications at a single major referral cancer center in Tokyo, Japan. Therefore, the background of these patients may not represent that of the entire country, as this cohort of patients belonged to a relatively high socioeconomic status and was more aware of the importance of early detection and treatment of gastric cancer. In addition, most of these patients may also have been relatively healthy, with a relatively low incidence of co-morbidities, which could have influenced the long-term outcomes of ESD. This was probably why the overall survival was better than the expected survival of comparable groups of patients matched for sex and age from the general population, with the 5-year relative survival rate being over 100 %. As an example of similar cases, there are several reports indicating that the survival of patients with prostate cancer managed by radical prostatectomy is better than the survival in the background population, with 5-year relative survival rates being over 100 %. These reports also mention similar reasons to ours to explain this finding [23, 24].
ESD preserves the stomach, thereby improving the quality of life of the patients as compared to surgery; however, a generally higher incidence of MGCs (in the range 1.8–12.8 %) has previously been reported after endoscopic treatment for EGC [25–27]. In the present study, MGCs occurred in 15.9 % with a median follow up of 83.3 months, consistent with previous reports. Unfortunately, we encountered seven cases of gastric cancer-related death associated with MGC, although the 5-year disease-specific survival rate was 99.9 %. Among these cases, four fatal advanced MGCs were detected over 5 years after the initial ESD because periodic surveillance examinations were not performed over the 5 years following the initial ESD. Thus, it is necessary to bear in mind the possibility of the development of fatal MGCs even if >5 years have passed since the curative ESD, although radical resection can be performed in most cases with MGCs. In addition, local recurrence and regional or distant LN metastasis remain critical issues. In this study, two patients in whom curative ESD was achieved for SM1 invasive EGC lesions developed regional LN metastasis or distant LN metastasis with local recurrence 4.1 or 7.2 years after the initial ESD [19, 20]. Therefore, more careful attention must be paid, in particular, to patients with SM1 invasive EGC lesions for which curative ESD has been achieved.
It seems reasonable to conclude from our results that ESD represents a highly effective treatment for EGC. However, this study had several limitations. First, the results of the present study were obtained from a retrospective assessment based on the medical records of patients with gastric ESD. In the present study, although 92 % of a large number of patients undergoing curative ESD for absolute indications or for expanded indications were successfully followed up in terms of obtaining survival information by investigating individual patient data thoroughly, the remaining 8 % were lost to follow-up within 5 years. It can be quite difficult to follow-up all consecutive patients for a lengthy period. Second, the present study was conducted at a single major referral cancer center in a large metropolitan area of Japan with many highly experienced endoscopists with specific expertise in ESD. Thus, a prospective multicenter nationwide study with a high follow-up rate over a period of 5 years or more is required for more precise evaluation of the long-term outcomes of gastric ESD. At present, several multicenter prospective cohort studies are ongoing [28–30]. Thirdly, the Helicobacter pylori (HP) infection statuses of all of the patients eligible for this study were not investigated, even though the presence of HP infection could have influenced the development of metachronous EGC lesions after endoscopic resection of EGC [31].
In conclusion, this is the largest study demonstrating the high rate of 5-year survival of EGC patients following curative ESD for absolute indications or for expanded indications with a median follow-up period of over 5 years. ESD could be employed as a standard treatment for EGC lesions that meet not only the absolute but also expanded indications for ESD.
References
Gotoda T, Yanagisawa A, Sasako M, Ono H, Nakanishi Y, Shimoda T, et al. Incidence of lymph node metastasis from early gastric cancer: estimation with a large number of cases at two large centers. Gastric Cancer. 2000;3:219–25.
Ono H, Kondo H, Gotoda T, Shirao K, Yamaguchi H, Saito D, et al. Endoscopic mucosal resection for treatment of early gastric cancer. Gut. 2001;48:225–9.
Hirasawa T, Gotoda T, Miyata S, Kato Y, Shimoda T, Taniguchi H, et al. Incidence of lymph node metastasis and the feasibility of endoscopic resection for undifferentiated-type early gastric cancer. Gastric Cancer. 2009;12:148–52.
Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2010 (ver. 3). Gastric Cancer. 2011;14:113–23.
Oda I, Gotoda T, Hamanaka H, Eguchi T, Saito Y, Matsuda T, et al. Endoscopic submucosal dissection for early gastric cancer: technical feasibility, operation time and complications from a large consecutive series. Dig Endosc. 2005;17:54–8.
Isomoto H, Shikuwa S, Yamaguchi N, Fukuda E, Ikeda K, Nishiyama H, et al. Endoscopic submucosal dissection for early gastric cancer: a large-scale feasibility study. Gut. 2009;58:331–6.
Goto O, Fujishiro M, Kodashima S, Ono S, Omata M. Outcomes of endoscopic submucosal dissection for early gastric cancer with special reference to validation for curability criteria. Endoscopy. 2009;41:118–22.
Gotoda T, Iwasaki M, Kusano C, Seewald S, Oda I. Endoscopic resection of early gastric cancer treated by guideline and expanded National Cancer Centre criteria. Br J Surg. 2010;97:868–71.
Abe S, Oda I, Suzuki H, Nonaka S, Yoshinaga S, Odagaki T, et al. Short- and long-term outcomes of endoscopic submucosal dissection for undifferentiated early gastric cancer. Endoscopy. 2013;45:703–7.
Kosaka T, Endo M, Toya Y, Abiko Y, Kudara N, Inomata M, et al. Long-term outcomes of endoscopic submucosal dissection for early gastric cancer: a single-center retrospective study. Dig Endosc. 2014;26:183–91.
Tanabe S, Ishido K, Higuchi K, Sasaki T, Katada C, Azuma M, et al. Long-term outcomes of endoscopic submucosal dissection for early gastric cancer: a retrospective comparison with conventional endoscopic resection in a single center. Gastric Cancer. 2014;17:130–6.
Oda I, Oyama T, Abe S, Ohnita K, Kosaka T, Hirasawa K, et al. Preliminary results of multicenter questionnaire study on long-term outcomes of curative endoscopic submucosal dissection for early gastric cancer. Dig Endosc. 2014;26:214–9.
Ohnita K, Isomoto H, Shikuwa S, Yajima H, Minami H, Matsushima K, et al. Early and long-term outcomes of endoscopic submucosal dissection for early gastric cancer in a large patient series. Exp Ther Med. 2014;7:594–8.
Ederer F, Axtell LM, Cutler SJ. The relative survival rate: a statistical methodology. Natl Cancer Inst Monogr. 1961;6:101–21.
Parkin DM, Hakulinen T. Analysis of survival. In: Jensen OM, Parkin DM, MacLennan R, Muir CS, Skeet RG, editors. Cancer registration: principles and methods. IARC scientific publications no. 95. Lyon: International Agency for Research on Cancer; 1991. p. 159–76.
Henson DE, Ries LA. The relative survival rate. Cancer. 1995;76:1687–8.
Matsuda T, Ajiki W, Marugame T, Ioka A, Tsukuma H, Sobue T. Research Group of Population-Based Cancer Registries of Japan. Population-based survival of cancer patients diagnosed between 1993 and 1999 in Japan: a chronological and international comparative study. Jpn J Clin Oncol. 2011;41:40–51.
Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer. 2011;14:101–12.
Oya H, Gotoda T, Kinjo T, Suzuki H, Yoshinaga S, Taniguchi H, et al. A case of lymph node metastasis following a curative endoscopic submucosal dissection of an early gastric cancer. Gastric Cancer. 2012;15:221–5.
Abe S, Oda I, Nakajima T, Suzuki H, Nonaka S, Yoshinaga S, et al. A case of local recurrence and distant metastasis following curative endoscopic submucosal dissection of early gastric cancer. Gastric cancer. 2015;18:188–92.
Nashimoto A, Akazawa K, Isobe Y, Miyashiro I, Katai H, Kodera Y, et al. Gastric cancer treated in 2002 in Japan: 2009 annual report of the JGCA nationwide registry. Gastric Cancer. 2013;16:1–27.
Sasako M, Kinoshita T, Maruyama K. Prognosis of early gastric cancer. Stomach Intest. 1993;28:139–46 (abstract in English).
Mathers MJ, Roth S, Klinkhammer-Schalke M, Gerken M, Hofstaedter F, Wilm S, et al. Patients with localised prostate cancer (t1–t2) show improved overall long-term survival compared to the normal population. J Cancer. 2011;2:76–80.
Røder MA, Brasso K, Berg KD, Thomsen FB, Gruschy L, Rusch E, et al. Patients undergoing radical prostatectomy have a better survival than the background population. Dan Med J. 2013;60(4):A4612.
Nakajima T, Oda I, Gotoda T, Hamanaka H, Eguchi T, Yokoi C, et al. Metachronous gastric cancers after endoscopic resection: how effective is annual endoscopic surveillance? Gastric Cancer. 2006;9:93–8.
Kobayashi M, Narisawa R, Sato Y, Takeuchi M, Aoyagi Y. Self-limiting risk of metachronous gastric cancers after endoscopic resection. Dig Endosc. 2010;22:169–73.
Han JS, Jang JS, Choi SR, Kwon HC, Kim MC, Jeong JS, et al. A study of metachronous cancer after endoscopic resection of early gastric cancer. Scand J Gastroenterol. 2011;46:1099–104.
Oda I, Shimazu T, Ono H, Tanabe S, Iishi H, Kondo H, et al. Design of Japanese multicenter prospective cohort study of endoscopic resection for early gastric cancer using web registry (J-WEB/EGC). Gastric Cancer. 2012;15:451–4.
Kurokawa Y, Hasuike N, Ono H, Boku N, Fukuda H. Gastrointestinal Oncology Study Group of Japan Clinical Oncology Group. A phase II trial of endoscopic submucosal dissection for mucosal gastric cancer: Japan Clinical Oncology Group Study JCOG0607. Jpn J Clin Oncol. 2009;39:464–6.
Takizawa K, Takashima A, Kimura A, Mizusawa J, Hasuike N, Ono H. Gastrointestinal Endoscopy Study Group (GIESG) of Japan Clinical Oncology Group (JCOG); Stomach Cancer Study Group (SCSG) of Japan Clinical Oncology Group (JCOG). A phase II clinical trial of endoscopic submucosal dissection for early gastric cancer of undifferentiated type: Japan Clinical Oncology Group study JCOG1009/1010. Jpn J Clin Oncol. 2013;43:87–91.
Fukase K, Kato M, Kikuchi S, Inoue K, Uemura N, Okamoto S, et al. Japan Gast Study Group. Effect of eradication of Helicobacter pylori on incidence of metachronous gastric carcinoma after endoscopic resection of early gastric cancer: an open-label, randomised controlled trial. Lancet. 2008;372:350–2.
Acknowledgments
We thank Dr. Koichi Hosokawa, Dr. Hiroyuki Ono, and Dr. Takuji Gotoda (our mentors at the National Cancer Center Hospital in Tokyo) for their efforts in developing ESD.
Conflict of interest
None.
Ethical standard
The study was approved by the Institutional Review Board of National Cancer Center (protocol no. 2013-125).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Suzuki, H., Oda, I., Abe, S. et al. High rate of 5-year survival among patients with early gastric cancer undergoing curative endoscopic submucosal dissection. Gastric Cancer 19, 198–205 (2016). https://doi.org/10.1007/s10120-015-0469-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10120-015-0469-0