Abstract
Background
Paclitaxel scheduled every 3 weeks has shown a response rate of ∼20% for gastric cancer, with modest hematological toxicity. Weekly administration of paclitaxel in patients with breast or ovarian cancer has shown equivalent efficacy and milder toxicity compared with an every-3 week schedule. We investigated, retrospectively, the antitumor effects and toxicity profiles of weekly paclitaxel for patients with metastatic or recurrent gastric cancer in clinical practice.
Methods
In 38 patients who had metastatic or recurrent histologically confirmed gastric cancer and a history of one prior chemotherapy regimen, other than paclitaxel or docetaxel, paclitaxel (8mg/m2) was administered weekly, three times every 4 weeks, with short-term premedication.
Results
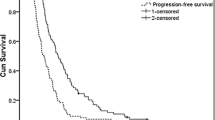
All 38 patients had had prior chemotherapy that included 5-fluorouracil, the fluoropyrimidine anticancer drug S-1, or cisplatin. The median number of courses in the present regimen was 6 (range, 1–44+). Dose intensity was 5mg/m2 per week, corresponding to 92% of the planned dose (6mg/m2 per week). The overall response rate was 24% (6/25) in measurable lesions, and pleural effusion and ascites disappeared in 2 of 7 patients (29%) and in 3 of 21 patients (14%), respectively. Median survival time was 151 days from the commencement of this treatment, with a median follow-up period of 260 days. Grade 3 or 4 leukopenia and neutropenia were observed in 11 (29%) and 12 (32%) patients, respectively. Seven patients (18%) died within 30 days of the last administration of paclitaxel.
Conclusion
Weekly paclitaxel seems to be active as second-line chemotherapy against metastatic and recurrent gastric cancer. Further study is needed to confirm the efficacy and safety of weekly paclitaxel.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
InstitutionalAuthorNameHealth and Welfare Statistics Association (1999) ArticleTitleThe movement of population J Health Welfare Stat 46 41–72
EK Rowinsky RC Donehower RJ Jones RW Tucker (1998) ArticleTitleMicrotubule changes and cytotoxicity in leukemic cell lines treated with taxol Cancer Res 48 4093–100
NM Lopes EG Adams TW Pitts BK Bhuyan (1993) ArticleTitleCell kill kinetics and cell cycle effects of taxol on human and hamster ovarian cell lines Cancer Chemother Pharmacol 32 235–42 Occurrence Handle10.1007/BF00685842 Occurrence Handle1:CAS:528:DyaK3sXlslektb8%3D Occurrence Handle8098996
D Fennelly C Aghajanian F Shapiro C O'Flaherty M McKenzie C O'Connor et al. (1997) ArticleTitlePhase I and pharmacologic study of paclitaxel administered weekly in patients with relapsed ovarian cancer J Clin Oncol 15 187–92 Occurrence Handle1:CAS:528:DyaK2sXnsV2ntA%3D%3D Occurrence Handle8996141
EA Eisenhauer WW ten Bokkel Huinink KD Swenerton L Gianni J Myles ME van der Burg et al. (1994) ArticleTitleEuropean–Canadian randomized trial of paclitaxel in relapsed ovarian cancer: high-dose versus low-dose and long versus short infusion J Clin Oncol 12 2654–66 Occurrence Handle1:STN:280:ByqD1c7otVM%3D Occurrence Handle7989941
JA Ajani J Fairweather P Dumas YZ Patt R Pazdur PF Mansfield (1998) ArticleTitlePhase II study of Taxol in patients with advanced gastric carcinoma Cancer J Sci Am 4 269–74 Occurrence Handle1:STN:280:DyaK1czlsVCnsg%3D%3D Occurrence Handle9689986
Y Yamada K Shirao A Ohtsu N Boku I Hyodo H Saitoh et al. (2001) ArticleTitlePhase II trial of paclitaxel by 3-h infusion for advanced gastric cancer with short premedication for prophylaxis against paclitaxel-associated hypersensitivity reactions Ann Oncol 12 1133–7 Occurrence Handle10.1023/A:1011680507956 Occurrence Handle1:STN:280:DC%2BD3MrjsFehtQ%3D%3D Occurrence Handle11583196
AA Garcia CG Leichman HJ Lenz J Baranda R Lujan Y Casagrande et al. (2001) ArticleTitlePhase II trial of outpatient schedule of paclitaxel in patients with previously untreated metastatic, measurable adenocarcinoma of the stomach Jpn J Clin Oncol 31 275–8 Occurrence Handle10.1093/jjco/hye060 Occurrence Handle1:STN:280:DC%2BD3MvhsVGhtA%3D%3D Occurrence Handle11463806
A Ohtsu N Boku F Tamura K Muro Y Shimada K Saigenji et al. (1998) ArticleTitleAn early phase II study of a 3-h infusion of paclitaxel for advanced gastric cancer Am J Clin Oncol 21 416–9 Occurrence Handle10.1097/00000421-199808000-00021 Occurrence Handle1:STN:280:DyaK1cznt1emsw%3D%3D Occurrence Handle9708646
T Arai T Hamaguchi K Shirao Y Shimada Y Yamada K Muro et al. (2003) ArticleTitleWeekly paclitaxel in patients with heavily treated advanced gastric cancer Proc Am Soc Clin Oncol 22 321
H Cho A Shotsu K Konishi M Kanari A Tsuburaya O Kobayashi et al. (2002) ArticleTitleWeekly paclitaxel for the second-line treatment of recurrent and advanced cancer (RAGC) Proc Am Soc Clin Oncol 21 357
EA Gehan MC Tefft (2000) ArticleTitleWill there be resistance to the RECIST (Response evaluation criteria in solid tumors)? J Natl Cancer Inst 92 179–81 Occurrence Handle10.1093/jnci/92.3.179 Occurrence Handle1:STN:280:DC%2BD3c7it1GjsA%3D%3D Occurrence Handle10655425
A Ohtsu Y Shimada K Shirao N Boku I Hyodo H Saito et al. (2003) ArticleTitleRandomized phase III trial of fluorouracil alone versus fluorouracil plus cisplatin versus uracil and tegafur plus mitomycin in patients with unresectable, advanced gastric cancer: The Japan Clinical Oncology Group Study (JCOG9205) J Clin Oncol 21 54–9 Occurrence Handle10.1200/JCO.2003.04.130 Occurrence Handle1:CAS:528:DC%2BD2cXpsVWru7k%3D Occurrence Handle12506170
N Boku A Ohtsu Y Shimada K Shirao S Seki H Saito et al. (1999) ArticleTitlePhase II study of a combination of irinotecan and cisplatin against metastatic gastric cancer J Clin Oncol 17 319–23 Occurrence Handle1:CAS:528:DyaK1MXltVyrtg%3D%3D Occurrence Handle10458249
Y Sakata A Ohtsu N Horikoshi K Sugimachi Y Mitachi T Taguchi (1998) ArticleTitleLate phase II study of novel oral fluoropyrimidine anticancer drug S-1 (M tegafur–0.M gimestat–M otastat potassium) in advanced gastric cancer patients Eur J Cancer 34 1715–20 Occurrence Handle10.1016/S0959-8049(98)00211-1 Occurrence Handle1:CAS:528:DyaK1cXntlShurw%3D Occurrence Handle9893658
W Koizumi M Kurihara S Nakano K Hasegawa (2000) ArticleTitlePhase II study of S-1, a novel oral derivative of 5-fluorouracil, in advanced gastric cancer. For the S-1 Cooperative Gastric Cancer Study Group Oncology 58 191–7 Occurrence Handle10.1159/000012099 Occurrence Handle1:CAS:528:DC%2BD3cXjvVeitr4%3D Occurrence Handle10765119
K Sugimachi Y Maehara N Horikoshi Y Shimada Y Sakata Y Mitachi et al. (1999) ArticleTitleAn early phase II study of oral S–1, a newly developed 5-fluorouracil derivative for advanced and recurrent gastrointestinal cancers. The S–1 Gastrointestinal Cancer Study Group Oncology 57 202–10 Occurrence Handle10.1159/000012032 Occurrence Handle1:CAS:528:DyaK1MXmvF2msr0%3D Occurrence Handle10545788
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hironaka, S., Zenda, S., Boku, N. et al. Weekly paclitaxel as second-line chemotherapy for advanced or recurrent gastric cancer. Gastric Cancer 9, 14–18 (2006). https://doi.org/10.1007/s10120-005-0351-6
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10120-005-0351-6