Abstract
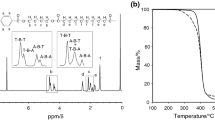
Hydroxyapatite-graft-poly(D-lactide) (HA-g-PDLA) was synthesized by ring-opening polymerization with HA as initiator and stannous octanoate (Sn(Oct)2) as catalyst. Thermogravimetric analysis (TGA) and Fourier transform infrared spectroscopy (FTIR) results indicate that PDLA chains are successfully grafted onto HA particles by covalent bond. Under two different processing temperatures (190 and 230 °C), the effect of the grafted PDLA chains on the crystallization behavior of poly(L-lactide)/HA-g-PDLA (PLLA/HA-g-PDLA) composite was investigated in the current study, comparing to neat PLLA and its four composites (PLLA/HA, PLLA/HA-g-PLLA, PLLA/PDLA, and PLLA/HA/PDLA). The crystallization rate of PLLA/HA-g-PDLA composite is highly enhanced comparing to PLLA, PLLA/HA and PLLA/HA-g-PLLA composites in which there are no stereocomplex (SC) crystallites. In addition, when the processing temperature rises from 190 °C to 230 °C, the acceleration of PLLA crystallization in PLLA/HA-g-PDLA composite is not influenced so much as other composites containing SC crystallites, such as PLLA/HA/PDLA or PLLA/PDLA. The differential scanning calorimetry (DSC) results demonstrate that even without SC crystallites, the crystallization of PLLA can still be accelerated a lot in this composite. This may be related to the interaction between the grafted PDLA chains and the amorphous PLLA chains in PLLA/HA-g-PDLA composite. The isothermal crystallization kinetics studies indicate that the nature of nucleation and crystal growth of PLLA/HA-g-PDLA composite are more likely 3D crystalline growth with heterogeneous nucleation mode, which are different from PLLA or other composites. This investigation could shed new light on the application of PLLA/HA composites.
Article PDF
Similar content being viewed by others

Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Change history
09 October 2020
In the version of this Article originally published, Fig. 7(f) was confused with Fig. 4(f). The corrected version of Fig. 7 is provided below. This graphical amendment does not affect the analyses, and additional text corrections are not needed.
References
Jain, A.; Kunduru, K. R.; Basu, A.; Mizrahi, B.; Domb, A. J.; Khan, W. Injectable formulations of poly(lactic acid) and its copolymers in clinical use. Adv. Drug Deliver. Rev.2016, 107, 213–227.
Du, N.; Guo, W. X.; Yu, Q. S.; Guan, S. L.; Guo, L. Y.; Shen, T.; Tang, H.; Gan, Z. H. Poly(D,L-lactic acid)-block-poly(N-(2-hydroxypropyl) methacrylamide) nanoparticles for overcoming accelerated blood clearance and achieving efficient anti-tumor therapy. Polym. Chem.2016, 7, 5719–5729.
Phetwarotai, W.; Tanrattanakul, V.; Phusunti, N. Synergistic effect of nucleation and compatibilization on the polylactide and poly(butylene adipate-co-terephthalate) blend films. Chinese J. Polym. Sci.2016, 34, 1129–1140.
Ma, C. L.; Pan, P. J.; Shan, G. R.; Bao, Y. Z.; Fujita, M.; Maeda, M. Core-shell structure, biodegradation, and drug release behavior of poly(lactic acid)/poly(ethylene glycol) block copolymer micelles tuned by macromolecular stereostructure. Langmuir2015, 31, 1527–1536.
Binan, L.; Tendey, C.; de, Crescenzo G.; El, Ayoubi R.; Ajji, A.; Jolicoeur, M. Differentiation of neuronal stem cells into motor neurons using electrospun poly-L-lactic acid/gelatin scaffold. Biomaterials2014, 35, 664–674.
Wang, Z. L.; Wang, Y.; Ito, Y.; Zhang, P. B.; Chen, X. S. A comparative study on the in vivo degradation of poly(L-lactide) based composite implants for bone fracture fixation. Sci. Rep.2016, 6, 20770.
Fambri, L.; Pegoretti, A.; Fenner, R.; Incardona, S.; Migliaresi, C. Biodegradable fibres of poly(L-lactic acid) produced by melt spinning. Polymer1997, 38, 79–85.
Tsuji, H.; Takai, H.; Saha, S. K. Isothermal and non-isothermal crystallization behavior of poly(L-lactic acid): effects of stereocomplex as nucleating agent. Polymer2006, 47, 3826–3837.
Schmidt, S. C.; Hillmyer, M. A. Polylactide stereocomplex crystallites as nucleating agents for isotactic polylactide. J. Polym. Sci., Part B: Polym. Phys.2001, 39, 300–313.
Xu, J. Z.; Zhang, Z. J.; Xu, H.; Chen, J. B.; Ran, R.; Li, Z. M. Highly enhanced crystallization kinetics of poly(L-lactic acid) by poly(ethylene glycol) grafted graphene oxide simultaneously as heterogeneous nucleation agent and chain mobility promoter. Macromolecules2015, 48, 4891–4900.
Song, L. Y.; Sun, L.; Jiang, N.; Gan, Z. H. Structural control and hemostatic properties of porous microspheres fabricated by hydroxyapatite-graft-poly(D,L-lactide) nanocomposites. Compos. Sci. Technol.2016, 134, 234–241.
Zhou, S. B.; Zheng, X. T.; Yu, X. J.; Wang, J. X.; Weng, J.; Li, X. H.; Feng, B.; Yin, M. Hydrogen bonding interaction of poly(D,L-lactide)/hydroxyapatite nanocomposites. Chem. Mater.2007, 19, 247–253.
Cui, W.; Li, X.; Zhou, S.; Weng, J. In situ growth of hydroxyapatite within electrospun poly(D,L-lactide) fibers. J. Biomed. Mater. Res., Part A2007, 82A, 831–841.
Wan, Y. Z.; Wu, C. Q.; Xiong, G. Y.; Zuo, G. F.; Jin, J.; Ren, K. J.; Zhu, Y.; Wang, Z. R.; Luo, H. L. Mechanical properties and cytotoxicity of nanoplate-like hydroxyapatite/polylactide nanocomposites prepared by intercalation technique. J. Mech. Behav. Biomed.2015, 47, 29–37.
Wei, J. C.; Sun, J. R.; Wang, H. J.; Chen, X. S.; Jing, X. B. Isothermal crystallization behavior and unique banded spherulites of hydroxyapatite/poly(L-lactide) nanocomposites. Chinese J. Polym. Sci.2010, 28, 499–507.
Liu, Z.; Chen, Y. H.; Ding, W. W.; Zhang, C. H. Filling behavior, morphology evolution and crystallization behavior of microinjection molded poly(lactic acid)/hydroxyapatite nanocomposites. Compos. Part A-Appl. S.2015, 72, 85–95.
Li, J.; Zheng, W.; Zheng, Y. F.; Lou, X. Cell responses and hemocompatibility of g -HA/PLA composites. Sci. China-Life Sci.2011, 54, 366–371.
Li, J.; Lu., X. L.; Zheng Y. F. Effect of surface modified hydroxyapatite on the tensile property improvement of HA/PLA composite. Appl. Surf. Sci.2008, 255, 494–497.
Wang, T. X.; Chow, L. C.; Frukhtbeyn, S. A.; Ting, A. H.; Dong, Q. X.; Yang, M. S.; Mitchell, J. W. Improve the strength of PLA/HA composite through the use of surface initiated polymerization and phosphonic acid coupling agent. J. Res. Natl. Inst. Stand. Technol.2011, 116, 785–796.
Qiu, X. Y.; Hong, Z. K.; Hu, J. L.; Chen, L.; Chen, X. S.; Jing, X. B. Hydroxyapatite surface modified by L-lactic acid and its subsequent grafting polymerization of L-lactide. Biomacromolecules2005, 6, 1193–1199.
Huang, G. M.; Du, Z. Y.; Yuan, Z. Y.; Gu, L. H.; Cai, Q.; Yang, X. P. Poly(L-lactide) nanocomposites containing poly(D-lactide) grafted nanohydroxyapatite with improved interfacial adhesion via stereocomplexation. J. Mech. Behav. Biomed.2018, 78, 10–19.
Narita, J.; Katagiri, M.; Tsuji, H. Highly enhanced nucleating effect of melt-recrystallized stereocomplex crystallites on poly(L-lactic acid) crystallization. Macromol. Mater. Eng.2011, 296, 887–893.
Yamane, H.; Sasai, K. Effect of the addition of poly(D-lactic acid) on the thermal property of poly(L-lactic acid). Polymer2003, 44, 2569–2575.
Chang, M. C.; Tanaka, J. FTIR study for hydroxyapatite/collagen nanocomposite cross-linked by glutaraldehyde. Biomaterials2002, 23, 4811–4818.
Hong, Z. K.; Qiu, X. Y.; Sun, J. R.; Deng, M. X.; Chen, X. S.; Jing, X. B. Grafting polymerization of L-lactide on the surface of hydroxyapatite nano-crystals. Polymer2004, 45, 6699–6706.
Bahrololoom, M. E.; Javidi, M.; Javadpour, S.; Ma, J. Characterisation of natural hydroxyapatite extracted from bovine cortical bone ash. J. Ceram. Process. Res.2009, 10, 129–138.
Borum-Nicholas, L.; Wilson, O. Surface modification of hydroxyapatite. Part I. Dodecyl alcohol. Biomaterials2003, 24, 3671–3679.
Sun, Y.; He, C. B. Synthesis and stereocomplex crystallization of poly(lactide)-graphene oxide nanocomposites. ACS Macro. Lett.2012, 1, 709–713.
Narita, J.; Katagiri, M.; Tsuji, H. Highly enhanced accelerating effect of melt-recrystallized stereocomplex crystallites on poly(L-lactic acid) crystallization: effects of molecular weight of poly(D-lactic acid). Polym. Int.2013, 62, 936–948.
Nouri, S.; Dubois, C.; Lafleur, P. G. Homocrystal and stereocomplex formation behavior of polylactides with different branched structures. Polymer2015, 67, 227–239.
Jiang, L. Y.; Xiong, C. D.; Jiang, L. X.; Xu, L. J. Effect of hydroxyapatite with different morphology on the crystallization behavior, mechanical property and in vitro degradation of hydroxyapatite/poly(lactic-co-glycolic) composite. Compos. Sci. Technol.2014, 93, 61–67.
Yang, C. F.; Huang, Y. F.; Ruan, J.; Su, A. C. Extensive development of precursory helical pairs prior to formation of stereocomplex crystals in racemic polylactide melt mixture. Macromolecules2012, 45, 872–878.
Li, X. Y.; Liu, Y. P.; Tian, X. Y.; Cui, K. P. Molecular mechanism leading to memory effect of mesomorphic isotactic polypropylene. J. Polym. Sci., Part B: Polym. Phys.2016, 54, 1573–1580.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Nos. 51603005 and 51973010) and Beijing Natural Science Foundation (No. 2182052).
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Information
10118_2020_2374_MOESM1_ESM.pdf
Enhanced Crystallization Rate of Poly(L-lactide)/Hydroxyapatite-graft-poly(D-lactide) Composite with Different Processing Temperatures
Rights and permissions
About this article
Cite this article
Wang, M., You, LC., Guo, YQ. et al. Enhanced Crystallization Rate of Poly(L-lactide)/Hydroxyapatite-graft-poly(D-lactide) Composite with Different Processing Temperatures. Chin J Polym Sci 38, 599–610 (2020). https://doi.org/10.1007/s10118-020-2374-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-020-2374-1