Abstract
The unique marine and terrestrial ecosystems of the Galapagos Islands are highly vulnerable to human-based drivers of change, including the introduction of invasive species, unsustainable tourism, illegal fishing, overexploitation of ecosystem services, and climate change. These drivers can interact with climate-based drivers such as El Niño-Southern Oscillation (ENSO) at multiple temporal and spatial scales, exacerbating their negative impacts on already fragile ecosystems and the socioeconomic system of the Archipelago. In this review, we performed a literature review based on published literature from 1945 to 2020 and local and global climate databases to analyze drivers of change in the Galapagos. We developed and applied a spatial impact assessment model to identify high-ecological value areas with high sensitivity and exposure scores to environmental change drivers. We identified 13 priority HEVA that encompass ca. 23% (14,715 km2) of the Galapagos Archipelago, distributed in nearly 3% of the Galapagos Marine Reserve and 20% Galapagos National Park. Current and future impacts are likely to concentrate on the inhabited islands’ highlands, whereas marine impacts concentrate along most of the Galapagos Islands’ shorelines. These results are important for guiding the design and implementation of adaptation measures aimed at increasing ecosystem resilience and human adaptive capacity in the face of global environmental change. Overall, these results will be valuable in their application for preserving Galapagos biota, securing the provision of vital ecosystem services for resident human populations, and sustaining the nature-based tourism industry.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Climate change represents one of the main threats to the conservation and sustainable use of marine and terrestrial biodiversity worldwide (Mantyka-Pringle et al. 2012). Oceanic islands are especially vulnerable to this global climatic driver due to the fragility of their ecosystems, which are the result of complex evolutionary, geological, and environmental processes (Harter et al. 2015). The geographic isolation of oceanic islands, in combination with the long-term stability of the environmental conditions and natural selection, has promoted high levels of endemic and native species (Jansson 2003; Fordham and Brook 2010). Thus, evolutionary processes shaping island communities have originated insular species with unique behavioral and life-history traits, and ecological relationships suited to stable conditions. Insular species exhibit intrinsic characteristics that make them susceptible to habitat disturbance, including narrow ecological niches, natural restricted distributions, reduced competitive ability and predator awareness, and behavioral or habitat specializations (Cronk 1997; Fordham and Brook 2010; Sodhi et al. 2004). These ecological features make island ecosystems highly vulnerable to invasive species, whose colonization after natural or human-induced disturbances is facilitated by the absence of predators and low levels of interspecific competition (Vilà et al. 2011; Harter et al. 2015).
Climate change in combination with invasive species will exacerbate the degradation of island ecosystems (Keener et al. 2012; Hernández-Delgado 2015; Braje et al. 2017). Physical (e.g., rising air temperature, sea-level rise) and chemical changes (e.g., ocean acidification, O2 concentration declines) can affect both the composition and biodiversity of insular communities and the various functions of the ecosystem, transforming their structure (Keener et al. 2012; Ferreira et al. 2016; Harter et al. 2015). For example, rising sea surface temperature (SST) will result in increased rainfall that affects both low- and highland ecosystems, which likewise will alter plant growth and community structure, promote erosion, and provide better conditions for invasive species (Trueman and D’Ozouville 2010; Larrea Oña and Di Carlo 2011).
Climate change is occurring faster than expected by the scientific community (IPCC 2014; Smith et al. 2015), potentially exceeding the adaptive capacity and resilience of island ecosystems. This is happening in a context, in which most of these unique ecosystems are already degraded by a growing number of drivers of change, increasing the vulnerability of native and endemic species to climate change (Fordham and Brook 2010; Smale et al. 2019; Castrejón and Charles 2020).
The Galapagos Islands are located in the Eastern Tropical Pacific (ETP), 960 km west of mainland Ecuador (Fig. 1). This volcanic archipelago is located in the confluence of three major seasonally varying warm and cool water oceanic current systems, and it is strongly affected by El Niño-Southern Oscillation (ENSO), whose main influence area is the Equatorial Pacific Ocean (Liu et al. 2013; Glynn et al. 2018). Hence, the singular location of Galapagos makes it a unique place to assess the potential impacts of climate variability on the demography and life-history traits of Galapagos biota.
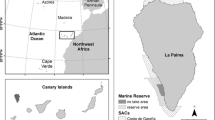
Map of the Galapagos Islands, with inset showing location of the archipelago relative to continental Ecuador. Surrounding lines denote the Galapagos Marine Reserve with its five bioregions as described by Edgar et al. (2004a): Far-Northern, Northern, Central-Southeastern, Western, and Elizabeth
The strong differences in oceanographic conditions across the archipelago have produced broad-scale and marine biogeographical patterns not observed in other parts of the world (Edgar et al. 2004a; Riegl et al. 2019a; Schiller et al. 2014). These unique features have made the Galapagos a nature-based tourism destination upon which the local economy depends on (Mathis and Rose. 2016). It generates annual revenues of USD 450 million, representing ca. 20% of Ecuador’s tourism gross domestic product, and ca. 80% of the local economy (Pizzitutti et al. 2017).
However, tourism has produced negative effects on the Galapagos natural environment, including the introduction of new invasive species, an increasing amount of waste, and growing use of local limited resources, mainly drinking water (Toral-Granda et al. 2017; Epler 2007; Larrea Oña and Di Carlo 2011; Pizzitutti et al. 2017). Besides the increasing number of tourists and invasive species, the Galapagos Islands face several other drivers of change, such as marine pollution, overfishing, and illegal fishing (Schiller et al. 2014; Alava et al. 2014; Salinas-De-León et al. 2020). All of these drivers of change can interact at multiple temporal and spatial scales with ENSO and climate change (Crain et al. 2008; Mantyka-Pringle et al. 2012; Harvey et al. 2013; Graham et al. 2011; Genner et al. 2010; Mouillot et al. 2013), exacerbating their impacts and threatening even more an already fragile system.
The global importance of the Galapagos Islands for biodiversity conservation and the high sensitivity of tropical island systems to drivers of change highlights the need to identify priority areas where conservation and management actions can be implemented to mitigate human impacts while increasing ecosystem resilience (Moreira et al. 2018; Cuesta et al. 2017; Fajardo et al. 2014; Kareiva et al. 2011). In this review, we discuss the main drivers of change that threaten the unique marine and terrestrial ecosystems of Galapagos. Then, we perform a spatial impact assessment model of the Galapagos Archipelago to identify high-ecological value areas (HEVA), which we define as areas of intrinsic biotic importance (singularity), highly exposed to climate change impacts and other human drivers, using available climate databases and a literature review of published literature. This spatial analysis is based on observed and expected changes in the terrestrial and marine ecosystems of the Galapagos Islands. Finally, within the obtained HEVA, and with the overarching goal of supporting policymaking and informing about conservation actions, we select specific HEVA that should be prioritized to develop ecosystem-based adaptation (EBA) measuresFootnote 1.
The Galapagos Islands
The archipelago is divided into five marine bioregions, referred to as Far-Northern, Northern, Central-Southeastern, Western, and Elizabeth (Fig. 1) (Edgar et al. 2004a). Each bioregion has distinctive reef fish and macro-invertebrate assemblages, which are unique combinations of species derived from Indo-Pacific, Panamanian, Peruvian, and endemic source areas (Edgar et al. 2004a). The abundance and distribution of these communities are strongly affected by the confluence of warm currents from the north and cool waters from the southwest (Riegl et al. 2019a). The western and central-south bioregions are characterized by colder upwelling conditions (Edgar et al. 2004a), while the northern bioregions exhibit higher SST than the central archipelago. However, the northern bioregions also exhibit persistent intrusions of colder and more nutrient-rich waters (Kislik et al. 2017; Riegl et al. 2019b).
Galapagos comprises approximately 234 islands, islets, and rocks with a total land area and coastline of ca. 7985 km and 1667 km (DPNG, Dirección del Parque Nacional Galápagos, 2014), which are enclosed in a multiple use area (MPA) of nearly 138,000 km2, the Galapagos Marine Reserve (GMR) (Fig. 1) (Heylings et al. 2002). The GMR encompasses a variety of ecosystems, ranging from coral reefs, coral communities, and mangroves along the shorelines (Glynn et al. 2018; Moity et al. 2019; Tanner et al. 2019) to rocky reefs and newly discovered kelp-forests on seabeds throughout the archipelago (Buglass 2018; Buglass et al. 2017; Eddy et al. 2019; Graham et al. 2007; Okey et al. 2004; Tompkins and Wolff 2016).
The GMR provides habitat for over 2900 fish species, aquatic invertebrates, and marine mammals, 20% of which are endemic (Schiller et al. 2014). The marine diversity in the GMR ranges from emblematic pelagic megafauna species such as whale sharks and mantas to endemic corals, groupers, and coral reef fish (Acuña-Marrero et al. 2014, 2018; Edgar et al. 2004a; Glynn et al. 2018; Hearn et al. 2014). Ecosystems within the GMR are important in the lifecycle of top predators that support shark diversity, shark nurseries, and other demersal ray-finned fishes (Hearn et al. 2010; Llerena et al. 2015; Salinas-De-León et al. 2015; Peñaherrera-Palma et al. 2017). The marine ecosystems of the GMR also provide important services to humans. This occurs mainly through fish productivity, where species such as red spiny lobster, sea cucumber, and demersal serranids are particularly exploited by artisanal fisheries (Hearn and Toral-Granda 2007, Hearn et al. 2005; Castrejón 2011).
Among the terrestrial environment, islands and islets exhibit a deserted landscape rather than a tropical forest typical of equatorial latitudes. Plants depend mostly on sporadic rain from December to June. However, islands higher than ~ 200 m can permanently have a dense fog (Porter 1979). The spatial variation of rainfall with altitude creates a vegetation zonation pattern of three main regions in the Galapagos Islands: (1) the dry lowlands, also referred as the arid zone, which occupies the majority of the archipelago (83% of total land area); (2) the transition zones; and the (3) humid zone or the highlands (Larrea Oña and Di Carlo 2011).
Regarding plant community assemblages, up to seven vegetation zones can be recognized (from lower to higher altitudes): (1) litoral, (2) arid, (3) transition, (4) Scalesia, (5) Brown, (6) Miconia, and (7) fern sedge zone. The plant biodiversity in each vegetation zone is adapted to the existing micro-climate conditions (Hamann 2001; Porter 1979). Smaller and lower islands typically have only littoral and arid/dry zones; seven of the islands are high enough to support humid zone ecosystems (Tye and Francisco-Ortega 2011).
The Galapagos Islands harbor over 600 plant species, of which 30% are endemic (Galapagos-Conservancy 2021) and mostly in the arid zone (Porter 1979). The humid zone has higher productivity due to its higher rainfall, which provides habitat for many native and endemic species (Larrea Oña and Di Carlo 2011). However, the humid zone is mostly degraded on inhabited islands due to land use and invasive plant species impacts (Laso et al. 2020; Watson et al. 2009). Protected land areas are managed by the Galapagos National Park (GNP), which covers 97% of the land area in the archipelago (GNP, 2021)
Drivers of change
The Galapagos Archipelago, like many tropical islands, is a system highly sensitive to human impacts (Fordham and Brook 2010) and is affected by climate dynamics (Grant and Grant 2006). The intrinsic sensitivity of the Galapagos has increased in recent decades due to the effects of the following drivers of change: (1) climate change; (2) unsustainable tourism and local population growth; (3) overfishing and illegal, undeclared, and unregulated (IUU) fishing; and (4) invasive species (Defeo et al. 2013; Castrejón and Charles 2020; Salinas-De-León et al. 2020). Throughout this article, we refer to drivers of change as any natural- or human-induced stressor that causes a change in ecosystems, as defined in Nelson et al. (2006) and Carpenter et al. (2006). The combined impacts of these drivers pose an unprecedented threat to the Galapagos system (Salinas-De-León et al. 2020).
Climate: Galapagos climate, El Niño-Southern Oscillation, and current and future trends
Galapagos climate
The Galapagos climate is a product of the interaction of oceanic currents surrounding the islands and the winds from the southeast (Trueman and D’Ozouville 2010). The influence of currents and winds is governed by interactions of the Intertropical Convergence Zone (ITCZ) and the El Niño-Southern Oscillation (ENSO) (Houvenaghel 1974; Sachs and Ladd 2010). Specifically, the ITCZ migration influences the main bi-seasonal characteristics of currents and winds of the Galapagos, whereas ENSO regulates yearly decadal fluctuations (Hamann, 1979, 1985; Hartten and Gage 2000). For most of the year, the ITCZ is located north of the archipelago and the southeast trade winds blow across the Galapagos, bringing cooled air from over the cold upwelled waters of the south pole. When the ITCZ migrates southwards closer to the Galapagos, the trade winds are reduced and warmer ocean currents from the north arrive at the archipelago (Alpert 1946).
The seasonality of the ITCZ combined with the topography of the archipelago results in two seasons: a warm, rainy season (January to May) and a cool, dry season (June to December) (Colinvaux 1972; Hamann 1979; Itow 2003). During the warm, rainy season, evaporation due to high SST leads to orographic rainfall that increases with altitude; thus, the lowlands only receive a marginal amount of rainfall and stay dry while the highlands become significantly humid (Hamann 1979; Snell and Rea 1999; Trueman and D’Ozouville 2010). Each island’s size, altitude, and exposure to wind determines the amount and seasonality of rainfall received. Furthermore, during the cool, dry season, the air is lowered in temperature by the ocean surface and is trapped below masses of warmer air, creating condensation. Condensation occurs above 250-m altitude and creates heavy mists and drizzle that are blown inland from the ocean, shifted upwards by the mountains, and consequently cooled, resulting in more intense rainfall in the highlands (Hamann 1979; Sachs and Ladd 2010; Trueman and D’Ozouville 2010).
El Niño-Southern Oscillation
The Eastern Tropical Pacific (ETP) exhibits interannual SST variability that is dominated by the ENSO cycles (Wang and Fiedler 2006). El Niño (warm phase) events are characterized by high SST, a lack of west-to-east thermal gradient across the surface of the Pacific, and a weakening of the easterly trade winds (Snell and Rea 1999). In the Galapagos, El Niño produces high air temperatures, sustained high SST, increased rainfall, and a longer than usual warm season, whereas La Niña (cold phase) events result in abnormally cold conditions and drought (Sachs and Ladd 2010). Past strong El Niño events (1975–1976, 1982–1983, 1993–1984, and 1997–1998) triggered dramatic effects on both marine and terrestrial ecosystems (Snell and Rea 1999; Trueman and D’Ozouville 2010; Defeo et al. 2013; Martin et al. 2017). For example, the El Niño 1982–1983 decimated populations of endemic species, such as the Galapagos penguins (Spheniscus mendiculus), which are still recovering (Laurie 1985; Robinson and Del Pino 1985; Trillmich and Limberger 1985). Coral reefs suffered intensely during this period, with 98% of corals being wiped out by coral bleaching (Glynn, 1994; Lessios et al., 1983; Robinson, 1985) followed by a significant decrease in marine species diversity (Edgar et al. 2010; Stein Grove 1985). During El Niño events, the bottom of the food chain is also impacted by ENSO, as phytoplankton concentrations can decrease substantially (33–46%) as a result of high temperatures in the archipelago, leading to community-level reductions in biomass (Wolff et al. 2012).
The impact of ENSO events also extends to terrestrial ecosystems and communities. Heavy rainfall characteristic of El Niño can trigger massive increases in herbaceous plants, which can then stimulate increased abundances of exotic invasive species and vines (Larrea Oña and Di Carlo 2011). Over-flooding can also result in increased mortality for resident species, such as for arboreal plants (Aldaz and Tye 1999; Tye and Aldaz 1999) that have trunks smothered by vines (Hamann 1985; Tye and Aldaz 1999) and giant tortoises that die due to injury or drowning in flooded ravines (Marquez et al. 2008). Land birds (e.g., finches) are also negatively affected by El Niño events due to the intensity of perturbations and because high rainfall triggers more intense parasitism (Dudaniec et al. 2007; Fessl and Tebbich 2002; Grant et al. 2000). Despite the occurrence of ENSO events in the Galapagos for thousands of years, strong El Niño events are unusual (see Fig. 2—Riegl et al. 2019a). However, evidence suggests that El Niño events have increased in intensity and frequency over the last two decades due to warmer SSTs (Conroy et al., 2008, 2010; Rustic et al. 2015; Thompson et al. 2017).
Observed climatic trends
Mean air temperature has increased by ~ 0.5 °C since the late 1980s, in both lowland and highland regions (Fig. S1), as suggested by data from the National Meteorological and Hydrological Institute (INAMHI) climatological stations on the islands of Santa Cruz and San Cristobal, Ecuador. This increase in mean air temperature is higher during the warm/wet season on the coast in the cool/dry season (1.3 vs. 0.1 °C, respectively) (Fig. S1). In contrast to this increasing trend in mean air temperature, precipitation records from 1981 to 2017 suggest a decreasing trend across the archipelago, particularly in arid coastal areas (Fig. S2). Critically, the first two decades of this century are on average ~ 40% drier than those during the decade of 1981–1990 (Fig. S3). Despite this overall decreasing trend in precipitation in the archipelago, records from 2002 to 2017 suggest the precipitation pattern has not changed significantly in the coastal region of Santa Cruz and San Cristobal islands (Fig. S3A-B). This supports the hypothesis that ENSO events, particularly those from 1982 to 1983 and 1997 to 1998, have influenced the time series and prevented a clear interpretation of climatic trends. Although records from the islands of Santa Cruz and San Cristobal are essential in understanding climatic patterns, their variation due to island topology and exposure to oceanographic and climatic variables highlights the need to establish several more climatic stations in this region in order to understand climate variability throughout the entire archipelago.
In contrast to the data provided by the Santa Cruz and San Cristobal islands’ climate stations, a time series analysis using monthly datasets of CHELSA (Karger et al. 2017) from the last 34 years (1979 to 2013) covering the entire extent of the Galapagos Islands showed a small decrease of 0.06 °C in mean annual air temperature (Fig. S4A). These patterns of precipitation and air temperature demonstrate spatial variability, particularly with elevation (Fig. S4B-C). Data from this time series shows that annual precipitation across all of the Galapagos Islands ranges from 557 to 1324 mm and follows a clear positive trend along the elevation gradient. The upper areas (above 368 m asl) of the islands receive a mean annual rainfall of 909 mm, whereas lower areas (below 51 m asl) are exposed to an annual rainfall that can get up to 749 mm (Fig. S4B). In contrast to the positive trend in precipitation with elevation, the air temperature has a negative trend with elevation in the Galapagos Islands, with an adiabatic lapse rate of 0.55 °C per 100 m. The thermal amplitude spans from a mean air condition of 24 °C at sea level to as cold as 15 °C at 1600 m asl at the mountain summits of Santa Cruz, San Cristobal, or Isabela (Fig. S4C).
Within the GMR, SST for the period 2002–2018 shows a clear warming trend. Data from MODIS interannual variability shows an increase in diurnal and nocturnal SST at a rate of 0.06 °C year (Fig. S5A). This finding is in agreement with other reports suggesting that the equatorial Pacific has warmed 0.4–0.8° over the last 40 years (IPCC 2007) and that greater increases in SST are expected in this region due to greenhouse warming (Cai et al., 2015, 2018). However, due to the prevailing oceanic currents having differences depending on the particular region of the Galapagos Islands being examined, SST anomalies for the GMR have contrasting patterns (Fig. S5B). For the period 2002 to 2018, the Far-Northern and Northern bioregions have received the highest warming (up to 2.3 °C increase), whereas the Western bioregion has received the highest cooling (− 5.7 °C decrease) (Fig. S5B). Coastal areas around Floreana, Española, and San Cristóbal have also shown increased SST anomalies.
Projected changes in climate
The Climate Model Intercomparison Project (CMIP5) simulates a historical and future broad warming in the Eastern Pacific over the past century (Coats and Karnauskas 2017). However, small-scale temporal and spatial variability may be dominated by natural fluctuations in the climate system or with phenomena such as El Niño. Nonetheless, for the Galapagos Islands, global circulation models (GCMs) generally project warmer and wetter future conditions, consistent with current observations (Liu et al. 2013; Rial et al. 2017; Sachs and Ladd 2010).
Climatology projections, based on CHELSA grids (Karger et al. 2017) of mean annual air temperature and precipitation derived from 5 GCMs (CSIRO-MK3-6-0, HADGEM2-CC, HADGEM2-ES, MIROC-ESM-CHEM, and MRI-CGCM3) for two RCP scenarios (RCP 4.5, RCP 8.5), suggest that there will be significant anomalies in both temperature and precipitation for the year 2050 (period 2020–2060), with considerable differences between the RCP scenarios (Fig. S6). Temperature is expected to increase 6.2% up to 14.5%, in the RCP 4.5 and 8.5, respectively. This increase will be heterogeneous across the islands, with humid zones in the western islands showing the greatest increase (Fig. S6A). Precipitation projections also suggest a relative increase on all the islands (30.3% up to 50.2%), with greater increases in the highlands of Santa Cruz, Fernandina, and central/southern Isabela (Fig. S6B-C). Projected increases in annual rainfall are accentuated along the elevation gradient as major deviations from current conditions and are located at the upper elevation range, with a mean annual increase of 1.2% (i.e., 10.6 mm) (Fig. S7A). Precipitation is also predicted to increase in different transitions and arid zones throughout all islands. Relatively high precipitation increments are projected for the arid lowlands of the southern slopes of Floreana, Southern Isabela, and Santa Cruz. The arid ecosystems of Española, Marchena, Genovesa, Pinta, Santa Fe, and Pinzón will also be highly exposed to increased precipitation (Fig. S6B-C). These results are concordant with other Santa Cruz–based projections, which suggest air temperature will increase throughout the twenty-first century (1.8–5 °C and 3–5 °C for annual max. and min. air temperatures, respectively) while precipitation will accentuate its seasonal variation (2.5–4.5-mm per day increase in the rainy season and up to 3-mm per day reduction in the dry season) (CAF 2019).
The ETP is expected to have increased SSTs due to greenhouse warming, suggesting an increase in ENSO frequency and intensity (Cai et al., 2015, 2018). The dynamic downscaling of the impact of climate change on the ocean circulation dynamics in the Galapagos Islands projects an increase of a near 2 °C rising trend in SST anomaly in the El Niño 3.4 region for the period 2001–2050 (Liu et al. 2013). The observed warming trends in the dynamic model show that the entire Galapagos region is significantly affected by global climate change, yet the degree of exposition is not homogeneous across the archipelago. The upwelling region to the west of the Isabela Island shows relatively slower warming trends compared to the eastern Galapagos region (Liu et al. 2013).
The observed negative effects of El Niño indicate that an interaction between climate change and ENSO could pose a grave threat to the Galapagos Islands. The coupled impacts of both stressors could profoundly impact previously affected ecosystems and species (Boersma and Rebstock 2014; Salazar and Denkinger 2010), augment colonization dynamics of invasive exotic species (Ellis-Soto et al. 2017), disrupt ecological processes such as ocean productivity (Sachs and Ladd 2010) and fishing resources (Castrejón and Charles 2020), and change water regulation capacity through the altering of soil organic carbon stocks (Rial et al. 2017). Lastly, upward trends in sea levels are projected to continue throughout the twenty-first century (Nerem et al. 2018) and the sea level in the Galapagos has been slowly rising (~ 10 cm since 1985) (Fig. S7B). Sea level rise in the Galapagos Islands could increase the risk of coastal flooding and impact tourism and infrastructure, along with the reduction of marine and terrestrial habitats such as shallow reefs, mangroves, and nesting sites for marine iguanas and turtles (Larrea Oña and Di Carlo 2011).
The observed trends and future projections discussed above indicate a progressive divergence of current climate conditions in the Galapagos Islands from past confidence intervals characterized by climatic variables in this region. Continued increases in sea surface and air temperature coupled with more intense and erratic ENSO events may lead to a climate system in the Galapagos Islands with increased seasonality and stronger spatial heterogeneity (Wolff 2010).
Overfishing and illegal undeclared and unregulated fishing
Marine ecosystems provide a diverse array of services utilized by humans, including the support of fisheries (Barbier 2017). Fisheries are of paramount importance due to their roles in food security and sustaining livelihoods (Bell et al. 2018). However, the ecosystems that provide these services are threatened by climate change and human activities, reducing the benefits they can provide (Smale et al. 2019). Human activities that threaten these ecosystem services include overfishing of target species, which continues to be a persistent and growing problem, and poor water quality as a product of harmful algal blooms, offshore pollution, and oxygen depletion from land-based runoff and infrastructure (Barbier 2017).
The marine life of the Galapagos Islands has been commercially exploited since the late eighteenth century, marked with the hunting of Galapagos fur seals (Arctocephalus galapagoensis) (Townsend 1934), and local sperm whales (Physeter macrocephalus), which have never recovered from whaling activities (Cantor et al. 2017; Whitehead et al. 1997). Finfish fisheries in the Galapagos date back to the early nineteenth century (Castrejón 2011) and commercial fisheries were permanently established in 1945, where the main target species was the Galapagos grouper (Mycteroperca olfax), or locally referred to as bacalao (Schiller et al. 2014). Ecuadorian industrial fisheries are prevented from fishing within the borders of the GMR and are only allowed to operate within the exclusive economic zone (EEZ), an area that extends from outside of the GMR border to 320 km. Most of the legal and illegal fishing that occurs within and around the GMR comprises tunas and sharks (Carr et al. 2013; Schiller et al. 2014). The most important target species caught by the Ecuadorian industrial and artisanal fishing fleet are the skipjack, yellowfin, and bigeye tuna (Katsuwonus pelamis, Thunnus albacares, and Thunnus obesus, respectively) and mahi-mahi (Coryphaena hippurus) (Schiller et al. 2014; Castrejón 2020a). Sharks are caught incidentally in the tuna and mahi-mahi fishery and, together with IUU fishing, represent one of the main threats for shark conservation (Castrejón 2020b). The legal framework of Ecuador prohibits shark finning and commercial exploitation of sharks nationwide. In mainland Ecuador, the landing and trading of sharks are permitted only in those cases when these species are caught incidentally and as long as they are landed whole (fins and body). In contrast, the capture, landing, and trading of sharks are prohibited in the GMR, even if they were caught incidentally. Despite these measures, thousands of sharks are landed annually on the main fishing ports of mainland Ecuador, suggesting the existence of a fishery within the Ecuadorian EEZ that targets sharks illegally, including the GMR (Carr et al. 2013; Alava et al. 2017; Alava and Paladines 2017). Hence, the estimated landings of sharks very likely represent only a fraction of the total landings for this region (Schiller et al. 2014).
Galapagos artisanal fisheries target at least 68 fish species from 27 families (Schiller et al. 2014; Zimmerhackel et al. 2015). Exploited fishes are both demersal and pelagic and largely consist of serranids, tuna, wahoo, labrids, and mullets (Castrejón 2011). Galapagos fisheries also target invertebrates, mostly spiny and slipper lobsters (Panulirus penicillatus, Panulirus gracilis, and Scyllarides astori) (Bustamante et al. 2000; Hearn and Toral-Granda 2007). The brown sea cucumber (Isostichopus fuscus) is also harvested, but this fishery has remained closed since 2015. However, at least three other species (Stichopus horrens, Holothuria kefersteini, and H. atra) are illegally caught (Toral-Granda 2008).
The Galapagos Marine Reserve is a sanctuary for heavily exploited fish like tuna and sharks, which migrate consistently to and from the reserve (Hearn et al. 2016; Acuña-Marrero et al. 2017; Boerder et al. 2017). The maintenance of the GMR is beneficial for both industrial and artisanal fisheries, as it increases fish productivity both outside and inside the reserve (Boerder et al. 2017; Bucaram et al. 2018). However, the overexploitation, incidental catch, and illegal fishing, produced by Ecuadorian and foreign industrial and artisanal fisheries established along GMR’s boundaries (Boerder et al. 2017), reduce the effectiveness of the GMR to ensure the recovery of these commercial and protected species (Alava et al. 2017; Alava and Paladines 2017; Castrejón 2020b).
To mitigate the impacts of human activities on the GMR and to ensure the sustainability of Galapagos small-scale fisheries, marine zoning plan was implemented (between 2000 and 2006) in combination with a co-management regime, and the allocation of exclusive fishing rights to local small-scale fishers (Heylings et al. 2002; Castrejón and Charles 2013). Approximately, 18% of the Galapagos coastline were declared as no-take zones, whose individual size ranged from small offshore islets to a 70-km span of coast, with no offshore boundaries legally established. However, the biased location of no-take zones in areas of low abundance of the most lucrative fishery resources (i.e., sea cucumbers and spiny lobsters), combined with a lack of effective enforcement and a high rate of non-compliance, severely limited the effectiveness of Galapagos marine zoning to improve the governance and sustainability of small-scale fisheries and the conservation of Galapagos marine biodiversity (Bucaram et al. 2013; Bucaram and Hearn, 2014; Defeo et al. 2014; Edgar et al. 2004b; Moity 2018).
The sea cucumber fishery collapsed in 2006 due to overfishing (Hearn and Toral-Granda 2007; Hearn et al. 2005; Toral-Granda 2008), while large apex-level fish such as the Galapagos grouper (M. olfax), the white-spotted sand bass (Paralabrax albomaculatus), and the olive grouper (Epinephelus cifuentesi) show signs of overexploitation (Danulat and Edgar, 2002; Schiller et al. 2014; Usseglio et al. 2016). Groupers and sand basses exhibit declines in landings and catch size compared to previous estimates, even in no-take zones (Burbano et al. 2014; Zimmerhackel et al. 2015; Usseglio et al. 2016). As a result, the catch composition has changed over time. Fish species previously with no economic value now are commercially exploited, including mullets (Xenomugil thoburni and Mugil galapaguensis), wahoo (Acanthocybium solandri), and pomfret (Seriola rivoliana) (Castrejón 2011; Danulat and Edgar 2002). Furthermore, the rate at which sharks are being extracted illegally from Galapagos is among the highest of any EEZ in the world (Schiller et al. 2014). Fishery assessments and genetic studies suggest that sharks in the ETP show signs of overexploitation (Carr et al. 2013; Pazmiño et al. 2017), and thus, urgent attention to illegal and incidental catch of sharks within and around the GMR is required.
Intensive fishing coupled with the reduced distribution of several Galapagos marine species (e.g., Galapagos grouper) makes them very susceptible to extinction (Schiller et al. 2014). Overexploitation of top predators, such as groupers or sand basses, can trigger cascading effects in the trophic chain, declining Galapagos marine diversity (Ruttenberg 2001; Ruiz and Wolff 2011). Furthermore, given the ecological role of sea cucumbers as nutrient recyclers (Purcell et al. 2011), the depletion of this species probably degraded the function and structure of Galapagos marine ecosystems. The reduction of spiny lobster stocks could be linked to an increasing presence of sea urchins (e.g., Eucidaris galapagensis) in the subtidal zone, leading to bioerosion and detriment of coral communities (Banks 2007; Glynn et al. 2015). However, this hypothesis is uncertain considering that, after a period of overexploitation, spiny lobster stocks have shown clear signs of recovery (Defeo et al. 2014; Szuwalski et al. 2016).
Invasive species
Invasive species have been introduced into the Galapagos both deliberately and by accident, including the introduction of farm animals and plants and the accidental introduction of rats, fire ants, and the parasitic fly (Philornis downsi) (Toral-Granda et al. 2017; Gardener et al. 2013; Larrea Oña and Di Carlo 2011). Until 2017, there were 1575 alien species across the archipelago (Toral-Granda et al. 2017). Among these, there are ca. 870 introduced plant species, of which 16% are invasive species and 3.3% transformers species, leading to plant communities structure modification (Buddenhagen and Tye, 2015; Trueman and D’Ozouville 2010). Invasive plant species not only impact native and endemic species abundance through competition and by transforming plant communities but can also be incorporated into the diet of native animals, aiding expansions in their distribution (Blake et al. 2012, 2015; Ellis-Soto et al. 2017). Invasive insects and vertebrates also cause negative impacts on native and endemic species decimating their populations. The larvae of the parasitic fly (P. downsi) feeds on the blood of chicks from native and endemic birds, causing high mortality rates (Deem et al. 2008; Jiménez-Uzcátegui et al. 2007; Lawson et al. 2017). Invasive fire ants predate on a variety of Galapagos wildlife, including reptiles, birds, and invertebrates (Causton et al. 2006; Herrera and Causton 2008; Wauters et al., 2016, 2017, 2018). Introduced mammalian species, mainly goats, rats, cats, and dogs, have decimated the abundance of diverse plant and animal species through predation and competition for the same ecological niches (Wiedenfeld and Jiménez-Uzcátegui 2008; Heleno et al. 2012; Renteria et al. 2012b).
The ecological impacts produced by invasive species can be exacerbated by climate oscillations that result in favorable conditions for these species (e.g., longer rainfall periods). ENSO increases rainfall season, which triggers massive growth of herbs and vines, changing the community structure of arid ecosystems and making them more susceptible to colonization by invasive species (Hamann 1985). In consequence, invasive plants have transformed entirely the composition of plant communities in the farmlands and pastures, located in the highlands of Galapagos inhabited islands (Laso et al. 2020; Watson et al. 2009) (Table S1). The increasing prevalence of pathogens and parasites during the rainfall season increases the mortality rates of bird populations, particularly of Galapagos finches and mockingbirds, by reducing their breeding and fledging success (Cimadom et al. 2014). This problem is exacerbated by rats and mice, which prey on native and endemic birds and whose abundance increases during the rainfall season.
The eradication of invasive species is extremely challenging and expensive (Renteria et al. 2012a), and projects aiming to eradicate invasive species in the Galapagos often meet a series of challenges, mainly with a lack of economic support for institutions, the denial by landowners to conduct fieldwork, or overly ambitious projects (Gardener et al. 2010). Despite these obstacles, plant eradications are feasible, realistic, and justifiable if well-known criteria are met. Buddenhangen and Tye (2015) have reported an up to 38% success rate for eradication programs in the Galapagos. In addition, several invasive vertebrates like goats, pigs, pigeons, rats, dogs, tilapia, and donkeys have been successfully eliminated from some of the islands or even from the entire archipelago (Carrion et al., 2007, 2011; Cruz et al. 2005; Phillips et al., 2012a, b). The removal of these harmful species has immediate positive results on the recovery of endangered native species (Carrion et al. 2011; Donlan et al. 2007). However, the eradication of invasive species is just one of several steps in being able to restore the terrestrial ecosystems of the Galapagos Islands (see Atkinson et al. 2008, Carrion et al. 2011).
Finally, although the impacts of invasive species have been extensively studied in Galapagos terrestrial ecosystems, very little is known about marine invasions in Galapagos and the magnitude of their impacts on marine ecosystems. At least 53 introduced marine invertebrates and 33 cryptogenic invertebrates, algae and halophytes, have been reported for Galapagos, most of them were probably brought by ships (Carlton et al. 2019; Keith et al. 2015). Given that research on marine alien species in Galapagos is relatively recent and that only a subset of habitats has been assessed, this suggests that marine alien species and their impacts are substantially underestimated. Therefore, regulating institutions should implement measures to study the advancement of alien species, reduce invasion risk, and minimize their impacts.
Unsustainable tourism and local population growth
Tourism is the main driver of change behind increasing demands for natural resources and population growth in the Galapagos, leading to an unsustainable development model that is fundamentally incompatible with the long-term conservation interests. In less than 10 years, the number of tourists that visit Galapagos has grown 417%, from 65,000 to 271,238 between 2000 and 2019 (Fig. S8A). Nature-based tourism is the primary economic engine of the Galapagos and generates annual revenues of USD 450,000,000 (Pizzitutti et al. 2017). This represents close to 20% of Ecuador’s tourism gross domestic product (GDP) and almost 80% of the local economy (Pizzitutti et al. 2017). The international representation of the Galapagos has transformed the islands into a world-class nature-based tourist destination, receiving a staggering 271.238 visitors in 2019 (DPNG, Dirección del Parque Nacional Galápagos, 2019).
The tourism industry has promoted demographic and economic growth for the Galapagos, resulting in ca. 30,000 residents (Epler 2007; Walsh and Mena 2016) that depend both directly and indirectly on the tourism industry (Fig. S8B). The population growth rate in the islands is three times higher than on the Ecuadorian mainland (Pizzitutti et al. 2017), while the economy is one of the fastest growing economies in the world. In response, the Ecuadorian Government has implemented restrictive migratory measures to avoid immigration into Galapagos. However, the resolution of this problem is more difficult than expected due to a complex intersection of economic, cultural, social, and political realities associated with the human development of inhabited islands (Brewington 2013; Epler 2007). Exponential rates of tourism arrivals have also negative feedbacks to local population, especially indirect effects on public health, as flux of migrants put increase pressure to the weak health systems, potable water network, and pressure over food security (Walsh and Mena 2016; Thompson et al. 2019; Nicholas et al. 2019; Houck et al. 2020).
Human development has aroused several problems that threaten natural resources, such as with oil spills inside the marine reserve (Charles Darwin Foundation 2002), water contamination and wastewater mismanagement (Alava et al. 2014; Ragazzi et al. 2016; Wikelski et al. 1996), destruction of native ecosystems (Brewington 2013; Laso et al. 2020), touristic site and trail overuse (Brewington 2013; Self et al. 2010), and plant and animals disturbance (Denkinger et al. 2013; French et al. 2010; Wikelski et al. 1996). One of the most pervasive byproducts of tourism is the introduction of invasive species (Nash 2009; Pizzitutti et al. 2017) (Fig. S8C), which have increased over time positively correlating with the increasing number of tourists (Toral-Granda et al. 2017). The effects of tourism have been so severe that UNESCO (United Nations Educational, Scientific and cultural Organization) added the Galapagos Islands to the list of “World Heritage in Danger” in 2007, listing uncontrolled development and mismanagement of tourism and growth in the human population as main reasons (Nash 2009). Additionally, the Galapagos conservation assessment by the IUCN was evaluated as of “significant concern” in the 2017 World Heritage Outlook, with tourism, invasive species, and climate change being the significant current threats (IUCN, Conseervation Outlook Assessment 2017). Galapagos is a prime example of a protected area suffering an environmental crisis that has been generated by the overexploitation of natural resources (Pizzitutti et al. 2017).
Overall, while the appealing combination of unique flora-and-fauna and beautiful landscapes in the Galapagos has helped boost the local economy and allowed the GNP to gain funds for its management and conservation initiatives, it has also brought problems to the archipelago. Climate change and tourism are interrelated drivers of change, as tourism contributes to climate change through the emission of greenhouse gases (GHG) related to accommodation, activities, and transport (Scott et al. 2008), and climate change disrupts ecosystem processes and the abundance and distribution of endemic species, which impacts the tourism industry. Thus, climate change scenarios in the Galapagos should be aligned with the tourism industry to mitigate the impacts and identify adaptation measures to increase both ecosystem and tourism industry resilience.
Projected impacts of environmental change
To select priority areas for the implementation of EBAs in the Galapagos (Colls et al. 2009), we built a spatially explicit model for impact assessment (Fig. 2). We used the concept of vulnerability for the identification of areas that would be highly sensitive and exposed to multiple drivers of change. The interaction of multiple drivers can result in additive, synergistic, or antagonistic outcomes with varying degrees of negative impacts (Crain et al. 2008). However, the outcomes of multiple drivers’ interactions in Galapagos’ ecosystems remain unknown. Therefore, we used a simple additive model approach, where the impact of drivers’ interactions is the product of their cumulative effects (Crain et al. 2008), and the magnitudes of exposure are differentiated and ranked. Our model does not pursue the precise estimation of the magnitudes of interactions but poses an approximation to the spatial distribution of different drivers and their heterogeneous and overlapped occurrence among the Galapagos Islands. The combined magnitudes of sensitivity and exposure submodels were used to identify areas of potential impacts (i.e., areas of biotic and abiotic importance where multiple drivers of change co-occur) (Fig. 2, Eq. 1). To this end, we used methods of multi-criteria and algebraic spatial modeling (Chakhar and Mousseau 2007; Dunčková et al. 2019; Greene et al. 2011; Lin 1998).
The magnitude of sensitivity was obtained by a literature review derived from the Galapagos-related scientific literature about the impact of climate change on terrestrial and marine ecosystems. The magnitude of exposure was obtained by combining environmental online databases (i.e., CHELSA, NOAA, land cover maps) with anthropogenic variables, such as terrestrial and marine public tourist use areas, reported targeted fishing and bycatch areas, and land use management status. The resulting impact model represents a hypothetical trajectory of potential environmental change-related impacts on a sensitive ecological system, assuming the absence of adaptation measures (Füssel and Klein 2006). Finally, it is not within the scope of this study the quantitative validation of the model, but to illustrate the spatial occurrence of the multiple drivers of change described in our literature review. However, the results of our impact assessment model were qualitatively validated by local management authorities and stakeholders through workshops and work meetings.
Sensitivity analysis
Literature review
The magnitude of sensitivity was assigned using a literature review that examined 135 published peer-reviewed studies from 1945 to 2018. We searched for Galapagos publications using the Web of Knowledge and Google Scholar and the keywords are as follows: “Galapagos”, “climate change”, and/or “environmental change”. Each study was evaluated for the following criteria: (1) invasive species impacts, (2) interactions between species, ecosystems, and services; (3) documented climate variability/change impacts of the studied population or area; (4) importance of the study area for the provision of environmental services; (5) exposure of the studied area or species to other impacts (e.g., overfishing); and (6) relevance of the studied area for the conservation and survival of a species. These criteria were scored in a binary fashion (Table S2), with a score of 1 being assigned for the respective criteria whenever any of the criteria was specified as relevant for the studied area/species in a study. In contrast, if the study did not highlight the abovementioned criteria, a score of 0 was assigned accordingly. Studies that analyzed areas with the highest sensitivity got a maximum score of 6, only if all the criteria were met. Additionally, several publications reported quantitative attributes that were added to the score, while others lacked specific spatial references (see Supplementary Materials). The final literature review score was the result of the spatial overlay of the 99 studies included in the metanalysis and the computation of an algebraic sum of the total criteria score of each study layer (Eq. 2).
Biodiversity attributes
We complemented our literature review by separately assigning biodiversity attributes to terrestrial and marine ecosystems. These attributes considered different areas that accounted for the number of endangered species, species richness, the proportion of terrestrial endemic plant species to total plant species richness, and marine keystone speciesFootnote 2 distributions.
Maps of richness of endangered species accounted for the distribution of 28 threatened Galapagos species, as cataloged by IUCN’s red list. Polygons of each species’ distributions were downloaded from https://www.iucnredlist.org. This variable accounted for up to 16.5% and 7.6% of the sensitivity model variability in the terrestrial and marine models, respectively.
For terrestrial biodiversity attributes, we included the number of endemic plant (EP) species and mapped them on the seven vegetation zones recognized by Porter (1979), where we developed a ratio of the total 229 endemic species mapped for each vegetation zone. This variable contributed up to 9% of the sensitivity model variability (see Supplementary Materials).
Furthermore, for marine biodiversity attributes, we considered the distributions of important habitats and keystone species, including top predators’ density (Fig. S10). Habitats included areas of (1) sea cucumber and lobster catches (Buglass et al. 2017; Bustamante et al. 2000; Toral-Granda and Martínez 2005; Wolff et al. 2011), (2) shark nurseries (Llerena et al. 2015), (3) corals (Glynn et al. 2018), and (4) whale shark habitat (Hearn et al. 2016). For density and distributions of predators (PD), we included (1) endemism and richness of sharks (Acuña-Marrero et al. 2018; Hearn et al. 2014), (2) sperm whale densities (Cantor et al. 2017), (3) tiger shark densities (Acuña-Marrero et al. 2017), and (4) hammerhead shark densities (Hearn et al. 2010; Peñaherrera-Palma et al. 2017). Polygons for each habitat and predators’ density/distributions were scored with 1 whenever each was present. Marine habitats and predators’ density/distributions accounted for 2.2% and 3.3% of the sensitivity model variability, respectively (see Supplementary Materials).
Finally, we obtained the sensitivity score as the result of the literature review plus the biodiversity attributes (Eq. 3). Two models were obtained, one for terrestrial and the other for marine ecosystems. This output constitutes 50% of the potential impact model (Fig. 2, Eq. 1).
Sensitivity results
Half of the islands had high-frequency scores (within 20 and 40) and the highest sensitivity, including Santa Cruz, San Cristobal, Santiago, and Fernandina (Fig. S11A). Among terrestrial ecosystems, the Scalesia zone got the highest sensitivity scores, followed by the arid and transition zones. High-frequency scores were distributed differently among the terrestrial ecosystems (Fig. S11B). Furthermore, the Western and Elizabeth bioregions obtained the highest sensitivity, while the Central-Southeastern bioregion exhibited the highest frequency (Fig. S12A). Within marine ecosystems, corals and habitats of sea cucumbers and lobsters yielded the highest sensitivity, while corals and shark nurseries yielded the highest frequencies (Fig. S12B).
Our final spatial sensitivity model showed lowlands, arid zones, and mangroves as highly sensible areas (Fig. 3). The highland ecosystems of Santiago, Isabela, Santa Cruz, and San Cristobal also showed high sensitivity (Fig. 3a). Besides, the entire surface of Pinta island reported high sensitivity to environmental change stressors. Furthermore, we found sensitive areas within the GMR to be coastlines and islets from the Central-Southeastern bioregion (Fig. 3b). Other sensitive areas included the Bolivar Channel, Punta Abermarle, Caleta Iguana, Punta Essex, Elizabeth Bay, Punta Moreno and Alfaro in Isabela, Darwin and Wolf islands, Leon Dormido and Punta Pitt in San Cristobal, and the southeastern seabeds of Isabela and Cartago Bay.
The estimated sensitivity of terrestrial (a) and marine (b) ecosystems. Sensitivity was calculated based on the score of the literature review, status, and richness of IUCN red list species, endemism, and key species distributions. The sensitivity score is represented by hexagonal minimum mapping units of 3.46 km2. The scores of the terrestrial model are displayed individually for each island.
Exposure analysis
To estimate exposure, we combined direct drivers of change that included our previously obtained climatic trends and projections for air temperature, annual precipitation, and SST (Fig. S5-6), the distribution of targeted fishing and bycatch areas (Cerutti-Pereyra et al. 2020), and the magnitude of visits in tourist public use areas (PUA), with indirect drivers of change expressed in the distribution of land use and marine zones from the most recent Galapagos National Park Zoning Plan (GNP, 2021; DPNG, Dirección del Parque Nacional Galápagos, 2016; Fig. S13). This plan was designed in 2016, but it has yet to be agreed upon and enforced (see Supplementary Material).
Each of the exposure inputs had different weighted values within the spatial model, according to their level of impact (see Supplementary Material). The weighted values were assigned based on variables’ complexities and distributions. Global drivers of change (e.g., temperature, SST) have widespread effects on the ecosystems and are more complex to mitigate, whereas local drivers (e.g., tourism, fisheries) have sequential and localized effects, defined by zoning (defined by borders) or common pool resources on ecosystems, and are less complex to mitigate (Millennium Ecosystem Assessment 2005). In our marine exposure submodel, we assumed that SST and bycatch have a higher impact on marine ecosystems than regulated tourism or artisanal fisheries in sustainable use areas (SST > bycatch > sustainable use areas > PUA). Furthermore, for terrestrial ecosystems, we assume that precipitation and temperature have higher exposure values because the shifts in their temporal and spatial patterns may affect the productivity and distribution of native and invasive species. Followed by land use zones, the potential impacts in the transition areas are higher than the ones in sustainable use and touristic areas, given by the pressures on the boundaries of other zones and remaining native ecosystems (precipitation > temperature > transition areas > sustainable use area > PUA). The exposure areas of marine and terrestrial zones account for the pressures present in these areas that affect natural ecosystems, as land use change, admitted capacity of tourism, or overexploitation of natural resources (e.g., fisheries).
Spatial exposure model
Our terrestrial exposure model showed that most islands have a relatively high degree of exposure, with the highest being at the highlands of Isabela, Santa Cruz, San Cristobal, Fernandina, Floreana, Santiago, and Pinta (Fig. 4a). These results reflected the spatial co-occurrence of present and future drivers of change. For example, the four populated islands had concentrated areas with high exposure in the highlands, whereas the remnants of natural ecosystems on the Galapagos are threatened by agricultural expansion, increased prevalence of invasive species, high concentration of tourism, and high exposure to temperature and precipitation anomalies (Fig. S6).
Exposure of ecosystems in the Galapagos. Terrestrial exposure (a) was calculated by the admitted capacity of tourism sites (PUA), the presence of sustainable use and transition areas (Ministerio del Ambiente 2016), and estimated changes of precipitation and temperature for 2050 (RCP 4.5). Marine exposure (b) was calculated by the admitted capacity of marine tourism sites (PUA), the presence of sustainable use areas (outside of the no-take zones, ntkz), targeted fishing and bycatch areas, and sea surface temperature trends (2002–2017). The exposure is represented by hexagonal minimum mapping units of 3.46 km2
The marine exposure model revealed several exposed areas across bioregions (Fig. 4b). Marine exposure is a product of the interrelation between SST warming (Fig. S5), fishing activity susceptibility (in most of the GMR, as illustrated in the National Park Zoning of 2016), fishing bycatch (based on an experimental longline fishing study, Cerutti-Pereyra et al. 2020), and tourism. The majority of the exposed marine areas are concentrated in the Central-Southeastern, Western, and Elizabeth bioregions (Fig. 3b). The areas surrounding Fernandina, between Isabela and Floreana, and the seabeds between Santiago, and between Santa Cruz and Isabela, are particularly overexposed to overfishing and bycatch, despite the delimitation of no-take zones. The added exposure of all the abovementioned variables illustrates that most of the GMR is exposed to several drivers of environmental change.
Drivers of change in the Galapagos Islands: current and future impacts
Our impact assessment model identified current and future potential impacts of diverse drivers of change throughout the Galapagos Archipelago, based on the intrinsic sensitivity and degree of exposure of different bioregions, ecosystems, and islands (Figs. 5 and 6). The areas with higher impact scores were classified as high-ecological value areas (HEVA), which are defined as areas highly sensitive and exposed to drivers of environmental change. These areas are key for environmental service provision including freshwater, fisheries, and nature-based tourism activities. HEVA with the highest impacts were concentrated on the biggest and most inhabited islands, with a clear trend towards the highlands: the Miconia and Scalesia zones containing nearly 40% of all of the HEVA (Figs. 5 and 7). The island of Santa Cruz exhibited the highest impact, followed by San Cristobal, Floreana, and Isabela (Fig. 5a). Our impact assessment also identified a high concentration (ca. 20%) of HEVA in the transition and arid zones of different islands (Table S6, Figs. 5b and 7). The skewed spatial distribution of the HEVA towards the inhabited islands is related to the ecological importance of the humid forested ecosystems and the high endemism from the arid zone (Fig. S10). This is coupled with a projected variability in climate (Fig. S6) and the effects of the zoning in 2016 of the highlands on the inhabited islands (Fig. S13), which are primarily used for farmlands, pastures, and tourism, resulting in an increased concentration of invasive species and a constantly increasing demand for natural resources.
Although HEVA were widespread throughout marine ecosystems in our results, there were specific regions that concentrated uneven proportions of HEVA (Fig. 6). High impacts were clustered in the Far-Northern, Elizabeth, and the Central-Southeastern bioregions (Figs. 6a and 8). In the Western bioregion, HEVA were identified in the north and south boundaries of the Bolivar Channel and the central part of the archipelago (a marine corridor connecting Isabela, Santiago, Santa Cruz, Pinzon, and Rabida) (Fig. 8). The remaining HEVA were distributed along the islands’ shorelines, whose ecological importance relies on several ecosystem services, including nature-based tourism and fisheries (Table S7). Among marine macro-habitats, shark nurseries showed the highest impacts, followed by corals and the habitats of hammerhead and tiger sharks (Fig. 6b). The distribution of endemic species and macro-habitats (Fig. S10-10) in areas with high sensitivity, coupled with the rise of SST (Fig. S5, Table S5) throughout the GMR, might explain the high score impacts for sharks and corals.
Priority high-ecological value areas and stakeholder’s validation
To select HEVA that should be prioritized for implementing EBA measures aimed at increasing the resilience and adaptation capacity of the Galapagos Islands, we cross-validated our results with the assistance of technical staff and directors of the GNP during a 2-day workshop held in Santa Cruz, Galapagos, in February 2020. In this workshop, we used the results of our impact assessment models as inputs and chose 13 HEVA with terrestrial and marine ecosystems (Table 1, Fig. 9). Overall, the HEVA host endemic, vulnerable, and critically endangered species or ecosystems with limited distribution; comprise spawning zones, shark nurseries, and nesting sites for sea turtles and birds; harbor resilient coral reefs and communities; and are characterized by a high influx of tourists. Some HEVA report high diversity and biomass of marine species from different tropic guilds, are feeding grounds of multiple marine and terrestrial species, and could be considered potential climate change refugia. Moreover, some terrestrial ecosystems within the HEVA are buffering areas around the agricultural zone, register an increasing incidence of invasive species, but also include the last remnants of the Scalesia forest in the humid highlands. Finally, these areas are of prime importance for local livelihoods, especially for small-scale fisheries, but some of them are highly exposed to overfishing (for details of selected HEVA, see Table S8). Each HEVA is characterized by the following criteria: (1) expected climatic variability given by the spatial distribution of terrestrial future climate models; (2) representativeness, measured as HEVA distribution among bioregions; (3) habitat connectivity across the elevation gradient (i.e., number of terrestrial macro-habitats occurring on each HEVA); (4) marine habitat diversity (number of marine macro-habitats); and (5) HEVA relevance for environmental service provision (e.g., tourism, fishery, freshwater provision). The HEVA selected comprise 22.7% (14,715 km2) of the Galapagos Archipelago, distributed in 2.77% (3835 km2) of the GMR and 19.9% (1592 km2) of the GNP (the terrestrial protected area; Table 1, S8-9).
Priority high-ecological value areas (HEVA) for the development of adaptation measures against climate change. Priority HEVA are denoted as colored areas, where orange and red correspond to the fourth and fifth quintiles of the impact model score, respectively. The numbers next to the HEVA represent the estimated annual tourists at each HEVA. Stripped areas denote the admitted capacity of marine tourism sites (PUA) within HEVA. The estimated number of visitors was calculated by the ratio between the admitted capacity of visitors (CAV) and the average annual visits registered in 5 PUA (Puerto Ayora, Puerto Baquerizo Moreno, Puerto Chino, Puerto Villamil, and Sierra Negra). This may overestimate or underestimate the magnitude of visits in some areas but is an approximation of the average visits that PUA (with no data) may receive, given their actual CAV and the data in highly visited areas. Priority HEVA are as follows: (1) Corridor Wolf Volcano, Punta Albermarle, and Cape Marshall; (2) The Bolivar Channel and Elizabeth South; (3) Corridor Sierra Negra Volcano Isabela South; (4) Corridor Cartago Bay—San Luis seabed; (5) Santiago highland; (6) Conservation Area Santiago-Santa Cruz; (7) Floreana and Islets; (8) Marchena coral remnants; (9) Corridor la Galapaguera—Punta Pitt; (12) León Dormido (Kicker’s rock); (11) Corridor El Junco and Southern seabeds; (12) Española and Gardner islands; and (13) Darwin and Wolf islands
Based on the above-listed criteria, the HEVA were ranked for prioritizing the implementation of EBAs to confront climate change (Table S9-10). Four HEVA had the highest priorities: (1) Corridor Sierra Negra Volcano Isabela South; (2) Conservation area Santiago-Santa Cruz; (3) Corridor Wolf Volcano, Punta Albermarle, and Cape Marshall; and (4) The Bolivar Channel and Elizabeth South (Fig. 9). These four areas comprise more than half of the marine priority HEVA and one-third of the terrestrial priority HEVA (Table 1, S8). Overall, the selected priority HEVA constitute relevant areas for the distribution and life cycle of critically endangered and endemic species and relict ecosystems (e.g., Scalesia forest), which are interconnected by marine and terrestrial corridors. Furthermore, the prioritized areas are fundamental to sustain water, agriculture, and fishery provision for local inhabitants and the nature-based tourism industry.
To show the impact of nature-based tourism on the islands, we overlaid the priority HEVA with the estimated potential visits of public use areas (PUA) Reck et al. (2010). We calculated the average ratio between the admitted capacity of visitors (CAV, for its Spanish acronym) and the average annual visits registered in five PUA (Puerto Ayora, Puerto Baquerizo Moreno, Puerto Chino, Puerto Villamil, and Sierra Negra) for the period 2016–2019. Then, we estimated the number of visits for not monitored PUA given their actual CAV multiplied by the calculated average ratio (actual PUA CAV * 0.05). This approximation to the potential visits that PUA with no data may receive (given their actual CAV and the available data from highly visited areas) adds to a maximum capacity of up to 526,080 annual visitors in the entire GNP. Specifically, the priority HEVA exhibited an estimated capacity of up to 383,200 annual tourists, equivalent to more than half of the potential total annual tourists the Galapagos Islands could receive (Fig. 9, Table S8). HEVA with the highest capacity were (6) Conservation Area Santiago-Santa Cruz (110,400), (11) Corridor El Junco and Southern seabeds (67,520), (3) Corridor Sierra Negra Volcano Isabela South (55,200), and (7) Floreana and Islets (44,160). This estimation outweighs the number of tourists registered in 2019 (Fig. S8) for the regulated tourist sites. Our estimations suggest that the high influx of tourists could be affected by drivers of change, especially in marine-related touristic activities. Besides, the estimated maximum capacity should be reevaluated concerning sustainable ecosystem capacity, as many visitors that arrive directly to the inhabited islands visit nearby tourism attractions that are not recorded in the PUA/CAV statistics (GNP, personal comment). There is a lack of records regarding tourist visits and only five PUA out of 66 keep visit records. According to our estimations, more than 200,000 visits may account for the non-monitored/regulated tourism in the islands, which may exceed the sustainable ecosystem capacity.
Conclusions
This research presents the first study evaluating the current and potential ecological impacts of major drivers of change that threaten terrestrial and marine ecosystems of the Galapagos Islands, including climate change, unsustainable tourism and local population growth, IUU fishing, and invasive species. Our literature review, coupled with the spatial impact assessment model, identified 13 areas of high-ecological value area (HEVA) distributed across the Archipelago, equivalent to ca. 23% (14,715 km2) of the marine and terrestrial habitats. These HEVA represent areas most vulnerable to climate-based and human drivers of change that threaten the conservation and sustainable use of Galapagos’ marine and terrestrial biodiversity. They also constitute important areas for the distribution and life cycle of critically endangered and endemic species and relict ecosystems (e.g., Scalesia forest).
Our impact assessment model demonstrated that current and potential impacts over HEVA are likely to concentrate on the four inhabited islands’ highlands due to their prolonged periods of transformation. Projected changes are expected to increase invasive species encroachment, potentially impacting endemic Galapagos biodiversity and freshwater availability. In contrast, areas of higher impact for marine ecosystems concentrate along shorelines of most Galapagos islands, which could profoundly affect food security and livelihoods for Galapagos artisanal fisheries and the nature-based tourism industry.
The four HEVA with the highest priority to focus ecosystem-based adaptation measures are (1) Conservation Area Santiago-Santa Cruz; (2) Corridor Sierra Negra Volcano Isabela South; (3) Corridor Wolf Volcano, Punta Albermarle, and Cape Marshall; and (4) The Bolivar Channel and Elizabeth South.
Based on this review results, we recommend to the Government of Ecuador, international cooperation, civil society organizations, and productive sectors, to create strategic alliances to design, agree upon, and implement a set of ecosystem-based adaptation (EBA) measures. These EBAs need to ensure the well-being of local livelihoods and the conservation of Galapagos’ unique marine and terrestrial ecosystems by increasing the resilience and adaptation capacity of the Archipelago against current and future threats. Specifically, it is urgent to implement the following EBA measures: (1) restoring the humid highland ecosystems of the four inhabited islands as a means to increase freshwater provision, secure agricultural production, and reduce exotic species invasions; (2) improving the design and effectiveness of Galapagos marine zoning, through an adaptive co-management of the Galapagos Marine Reserve to reduce IUU fishing and protect the most suitable areas to ensure commercial stocks recovery, based on climate change risk assessment; (3) strengthening marine biosecurity programs for invasive species; (4) restoring selected coral reef habitats through experimental coral breeding and exclusion areas; (5) reducing the impact of diving, anchoring, and pollution related to tourism operations in selected marine HEVAS; (6) strengthening ongoing ecological and socioeconomic monitoring programs to produce the scientific data required to understand how climate change will interact with other non-climatic drivers and how they will impact the Galapagos Islands. This will support the design of scientific-sound base adaptation measures and the evaluation of their effect on increasing ecosystem resilience and human adaptive capacity.
Notes
An EBA is a measure designed to simultaneously reduce poverty, protect or restore biodiversity and ecosystem services, and remove atmospheric greenhouse gases. Therefore, an EBA integrates the use of biodiversity and ecosystem services into an overall strategy to help people adapt to the adverse impacts of climate variability and change (Scarano, 2017). EBAs are particularly relevant for Galapagos to safeguard one of most the important biodiversity and climate change hotspots in the world.
A keystone species has a disproportionately large effect on its natural environment relative to its abundance (Paine 1995). Therefore, a keystone species plays a critical role in maintaining the structure of an ecological community, affecting many other organisms in an ecosystem, and helping to determine the types and numbers of various other species in the community.
References
Acuña-Marrero D, Jiménez J, Smith F, Doherty JrPF, Hearn A, Green JR et al. (2014) Whale shark (Rhincodon typus) seasonal presence, residence time and habitat use at Darwin Island, Galapagos Marine Reserve. PLoS One 9:e115946. https://doi.org/10.1371/journal.pone
Acuña-Marrero D, Smith ANH, Hammerschlag N, Hearn A, Anderson MJ et al (2017) Residency and movement patterns of an apex predatory shark (Galeocerdo cuvier) at the Galapagos Marine Reserve. PLoS One 12:e0183669. https://doi.org/10.1371/journal.pone.0183669
Acuña-Marrero D, Smith ANH, Salinas-De-León P, Harvey ES, Pawley MDM et al (2018) Spatial patterns of distribution and relative abundance of coastal shark species in the Galapagos Marine Reserve. Mar Ecol Prog Ser 593:73–95. https://doi.org/10.3354/meps12505
Alava JJ, Paladines F (2017) Illegal fishing on the Galapagos high seas. Sci 357:1362. https://doi.org/10.1126/science.aap7832
Alava JJ, Palomera C, Bendell L, Ross PS (2014) Pollution as an emerging threat for the conservation of the Galapagos Marine Reserve: environmental impacts and management perspectives. In: Denkinger J, Vinueza L (eds) The Galapagos Marine Reserve, social and ecological interactions in the Galapagos Islands. Springer Science+Business Media New, New York, pp 247–283. https://doi.org/10.1007/978-3-319-02769-2
Alava J, Barragán-Paladines M, Denkinger J, Muñoz-Abril L, Jiménez P et al (2017) Massive Chinese fleet jeopardizes threatened shark species around the Galápagos Marine Reserve and waters off Ecuador: implications for national and international fisheries policy. Int J Fish Sci Res 1:1001.
Aldaz I, Tye A (1999) Effects of the 1997-98 El Nino event on the vegetation of Alcedo Volcano. Isabela Island Not Galapagos 60:25–28.
Alpert ML (1946) Notes on the weather and climate of Seymour Island. Galapagos archipelago Bull Am Meterological Soc 27:200–209
Atkinson R, Renteria JL, Simbaña W (2008) The consequences of herbivore eradication on Santiago: are we in time to prevent ecosystem degradation again? Galapagos Rep 2007–2008:121–124
Banks S (2007) Subtidal ecological monitoring of the coastal management subzones: 2004 to 2006. Galapagos Rep 2006–2007:128–132
Barbier EB (2017) Marine ecosystem services. Curr Biol 27:R507–R510. https://doi.org/10.1016/j.cub.2017.03.020
Bell JD, Cisneros-Montemayor A, Hanich Q, Johnson JE, Lehodey P et al (2018) Adaptations to maintain the contributions of small-scale fisheries to food security in the Pacific Islands. Mar Policy 88:303–314. https://doi.org/10.1016/j.marpol.2017.05.019
Blake S, Wikelski M, Cabrera F, Guezou A, Silva M et al (2012) Seed dispersal by Galápagos tortoises. J Biogeogr 39:1961–1972. https://doi.org/10.1111/j.1365-2699.2011.02672.x
Blake S, Guézou A, Deem SL, Yackulic CB, Cabrera F (2015) The dominance of introduced plant species in the diets of migratory Galapagos tortoises increases with elevation on a human‐occupied island. Biotropica 47:246–258. https://doi.org/10.1111/btp.12195
Boerder K, Bryndum-buchholz A, Worm B (2017) Interactions of tuna fisheries with the Galápagos marine reserve. Mar Ecol Prog Ser 585:1–15. https://doi.org/10.3354/meps12399
Boersma PD, Rebstock GA (2014) Climate change increases reproductive failure in Magellanic Penguins. PLoS One 9:e85602. https://doi.org/10.1371/journal.pone.0085602
Braje TJ, Leppard TP, Fitzpatrick SM, Erlandson JM (2017) Archaeology, historical ecology and anthropogenic island ecosystems. Environ Conserv 44:286–297. https://doi.org/10.1017/S0376892917000261
Brewington L (2013) The double bind of tourism in Galapagos Society. In: Walsh, SJ, Mena CF. (Eds.) Science and conservation in the Galapagos Islands: frameworks & perspectives, social and ecological interactions in the Galapagos Islands 1. © Springer Science+Business Media, LLC, pp 105–125. https://doi.org/10.1007/978-1-4614-5794-7
Bucaram SJ, Hearn A (2014) Factors that in fluence the entry–exit decision and intensity of participation of fishing fleet for the Galapagos lobster fishery. Mar Policy 43:80–88. https://doi.org/10.1016/j.marpol.2013.05.005
Bucaram SJ, White JW, Sanchirico JN, Wilen JE (2013) Ocean & Coastal Management Behavior of the Galapagos fishing fleet and its consequences for the design of spatial management alternatives for the red spiny lobster fishery. Ocean Coast Manag 78:88–100. https://doi.org/10.1016/j.ocecoaman.2013.03.001
Bucaram SJ, Hearn A, Trujillo AM, Rentería W, Bustamante RH et al (2018) Assessing fishing effects inside and outside an MPA: the impact of the Galapagos Marine Reserve on the industrial pelagic tuna fisheries during the first decade of operation. Mar Policy 87:212–225. https://doi.org/10.1016/j.marpol.2017.10.002
Buddenhagen CE, Tye A (2015) Lessons from successful plant eradications in Galapagos: commitment is crucial. Biol Invasions 17:2893–2912. https://doi.org/10.1007/s10530-015-0919-y
Buglass S (2018) Exploring the “Bajos” or Shallow Seamount of the Galapagos Marine Reserve. Charles Darwin Foundation. https://www.darwinfoundation.org/en/blog-articles/438-exploring-the-bajos-or-shallow-seamounts-of-the-galapagos-marine-reserve. Accessed 3 Feb 2021
Buglass S, Reyes H, Ramirez-González J, Eddy TD, Salinas-De-León P et al (2017) Evaluating the effectiveness of coastal no-take zones of the Galapagos Marine Reserve for the red spiny lobster, Panulirus penicillatus. Mar Policy 88:204–212. https://doi.org/10.1016/j.marpol.2017.11.028
Burbano DV, Mena CF, Guarderas P, Vinueza L (2014) Shifting baselines in the Galapagos white fin fishery, using fisher’s anecdotes to reassess fisheries management: the case of the Galapagos Grouper. In: Denkinger J, Vinueza L (eds) The Galapagos Marine Reserve, social and ecological interactions in the Galapagos Islands. Springer Science+Business Media New, New York, pp 227–246. https://doi.org/10.1007/978-3-319-02769-2
Bustamante RH, Reck GK, Ruttenberg BI, Polovina J (2000) The Galápagos spiny lobster fishery. In: Phillips, B., Kittaka, J. (Eds.) Spiny lobsters: fisheries and culture. Fishing News Books, pp 210–220. https://doi.org/10.1002/9780470698808.ch12
CAF 2019. Índice de vulnerabilidad al cambio climático en la isla de Santa Cruz. CAF-Banco de Desarrollo de América Latina, pp. 252
Cai W, Santoso A, Wang G, Yeh S, An SI et al (2015) ENSO and greenhouse warming. Nat Clim Chang 5:849–859. https://doi.org/10.1038/NCLIMATE2743
Cai W, Wang G, Dewitte B, Wu L, Santoso A et al (2018) Increased variability of eastern Pacific El Niño under greenhouse warming. Nature 564:201–206. https://doi.org/10.1038/s41586-018-0776-9
Cantor M, Eguiguren A, Merlen G, Whitehead H, Ayora P et al (2017) Galápagos sperm whales (Physeter macrocephalus): waxing and waning over three decades. Can J Zool 95:645–652. https://doi.org/10.1139/cjz-2016-0266
Carlton JT, Keith I, Ruiz GM (2019) Assessing marine bioinvasions in the Galápagos Islands: implications for conservation biology and marine protected areas. Aquat Invasions 14:1–20. https://doi.org/10.3391/ai.2019.14.1.01
Carpenter SR, Bennett EM, Peterson GD (2006) Editorial: special on scenarios for ecosystem services. Ecol Soc 11:32. https://doi.org/10.5751/es-01609-110232
Carr LA, Stier AC, Fietz K, Montero I, Gallagher AJ et al (2013) Illegal shark fishing in the Galápagos Marine Reserve. Mar Policy 39:317–321. https://doi.org/10.1016/j.marpol.2012.12.005
Carrion V, Donlan CJ, Campbell K (2007) Feral donkey (Equus asinus) eradications in the Galápagos. Biodivers Conserv 16:437–445. https://doi.org/10.1007/s10531-005-5825-7
Carrion V, Donlan CJ, Campbell KJ, Lavoie C, Cruz F (2011) Archipelago-wide island restoration in the Gala Islands: reducing costs of invasive mammal eradication programs and reinvasion risk. PLoS One 6:e18835. https://doi.org/10.1371/journal.pone.0018835
Castrejón M (2011) Co-Manejo Pesquero en la Reserva Marina de Galápagos: Tendencias, retos y prespectivas de cambio. Fundación Charles Darwin, Santa Cruz, Galápagos
Castrejón M (2020a) The Ecuadorian tuna and mahi-mahi fisheries: characterization, trends, and management strategies. Island Conservation and Pew Bertarelli Ocean Legacy Project, pp. 82
Castrejón M (2020b) La pesca ilegal, no declarada y no reglamentada (INDNR) en el Pacífico Este Tropical: caracterización, causas, soluciones y prioridades de inversión. World Wildlife Fund y Fundación PACÍFICO, pp. 201
Castrejón M, Charles A (2013) Improving fisheries co-management through ecosystem-based spatial management: the Galapagos Marine Reserve. Mar Policy 38:235–245. https://doi.org/10.1016/j.marpol.2012.05.040
Castrejón M, Charles A (2020) Human and climatic drivers affect spatial fishing patterns in a multiple-use marine protected area: the Galapagos Marine Reserve. PLoS One 15:1–32. https://doi.org/10.1371/journal.pone.0228094
Causton ACE, Peck SB, Sinclair BJ, Roque-Albelo L, Hogson CJ et al (2006) Alien insects: threats and implications for conservation of Galapagos Islands. Ann Entomol Soc Am 99:121–143. https://doi.org/10.1603/0013-8746(2006)099[0121:AITAIF]2.0.CO;2
Cerutti-Pereyra F, Moity N, Dureuil M, Ramírez-González J, Reyes H et al (2020) Artisanal longline fishing the Galapagos Islands–effects on vulnerable megafauna in a UNESCO World Heritage site. Ocean Coast Manag 183:104995. https://doi.org/10.1016/j.ocecoaman.2019.104995
Chakhar S, Mousseau V (2007) An algebra for multicriteria spatial modeling. Comput Environ Urban Syst 31:572–596. https://doi.org/10.1016/j.compenvurbsys.2007.08.007
Charles Darwin Foundation (2002) Biological Impacts of the Jessica Oil Spill on the Galápagos Environment. Charles Darwin Foundation for the Galapagos Islands, Puerto Ayora
Cimadom A, Ulloa A, Meidl P, Zöttl M, Zöttl E et al (2014) Invasive parasites, habitat change and heavy rainfall reduce breeding success in Darwin’s Finches. PLoS One 9:e107518. https://doi.org/10.1371/journal.pone.0107518
Coats S, Karnauskas KB (2017) Are simulated and observed twentieth century tropical pacific sea surface temperature trends significant relative to internal variability? Geophys Res Lett 44:9928–9937. https://doi.org/10.1002/2017GL074622
Colinvaux PA (1972) Climate and the Galapagos Islands. Nat 240:17–20. https://doi.org/10.1038/240017a0
Colls A, Ash N, Ikkala N (2009) Ecosystem-based adaptation: a natural response to climate change, Journal of Semitic Studies. IUCN, Gland, p 16. https://doi.org/10.1093/jss/XXXIII.1.1
Conroy JL, Restrepo A, Overpeck JT, Steinitz-kannan M, Cole JE et al (2008) Unprecedented recent warming of surface temperatures in the eastern tropical Pacific Ocean. Nat Geosci 2:46–50. https://doi.org/10.1038/ngeo390
Conroy JL, Overpeck JT, Cole JE (2010) El Niño/Southern Oscillation and changes in the zonal gradient of tropical Pacific sea surface temperature over the last 1.2 ka. Pages News 18:32–34. https://doi.org/10.22498/pages.18.1.32
Crain C, Kroeker K, Halpern BS (2008) Interactive and cumulative effects of multiple human stressors in marine systems. Ecol Lett 11:1304–1315. https://doi.org/10.1111/j.1461-0248.2008.01253.x
Cronk QCB (1997) Islands: stability, diversity, conservation. Biodivers Conserv 6:477–493. https://doi.org/10.1023/A:1018372910025
Cruz F, Donlan CJ, Campbell K, Carrion V (2005) Conservation action in the Galápagos: feral pig (Sus scrofa) eradication from Santiago Island. Biol Conserv 121:473–478. https://doi.org/10.1016/j.biocon.2004.05.018
Cuesta F, Peralvo M, Merino-Viteri A, Bustamante M, Baquero F et al (2017) Priority areas for biodiversity conservation in mainland Ecuador. Neotropical Biodivers 3:93–106. https://doi.org/10.1080/23766808.2017.1295705
Danulat E, Edgar GJ (2002) Reserva Marina de Galápagos. Línea base de la biodiversidad. Fundación Charles Darwin/Servicio Parque Nacional Galápagos, Santa Cruz, Galápagos
Deem SL, Cruz M, Jiménez-Uzcátegui G, Fessl B, Miller ER et al (2008) Pathogens and parasites: an increasing threat to the conservation of Galapagos avifauna. Galapagos Rep 2007-2008:125–130. http://www.ecologyandsociety.org/vol9/iss1/art5/
Defeo O, Castrejón M, Ortega L, Kuhn AM, Gutierrez NL et al (2013) Impacts of climate variability on Latin American small-scale fisheries. Ecol Soc 18:30. https://doi.org/10.5751/ES-05971-180430
Defeo O, Castrejón M, Perez-Castañeda R, Castilla JC, Gutierrez NL et al (2014) Co-management in Latin American small-scale shell fisheries: assessment from long-term case studies. Fish Fish 17:176–192. https://doi.org/10.1111/faf.12101
Denkinger J, Parra M, Muñoz JP, Carrasco C, Murillo JC et al (2013) Are boat strikes a threat to sea turtles in the Galapagos Marine Reserve? Ocean Coast. Manag 80:29–35. https://doi.org/10.1016/j.ocecoaman.2013.03.005
Donlan CJ, Campbell K, Cabrera W, Lavoie C, Carrion V et al (2007) Recovery of the Galápagos rail (Laterallus spilonotus) following the removal of invasive mammals. Biol Conserv 138:520–524. https://doi.org/10.1016/j.biocon.2007.05.013
DPNG, Dirección del Parque Nacional Galápagos (2014) Plan de Manejo de las Áreas Protegidas de Galápagos para el Buen Vivir. Ministerio del Ambiente – Parque Nacional Galápagos, Puerto Ayora, Galápagos http://awsassets.panda.org/downloads/_1_plan_de_manejo_del_png_2014__final_.pdf
DPNG, Dirección del Parque Nacional Galápagos (2016) Zonificación de las áreas protegidas de Galápagos, Sistema de zonificación de las áreas protegidas de Galápagos. Ministerio del Ambiente–Parque Nacional Galápagos. pp 105
DPNG, Dirección del Parque Nacional Galápagos (2019) Informe anual de visitantes a las áreas protegidas de Galápagos del año 2019. http://www.galapagos.gob.ec/estadistica-de-visitantes/
Dudaniec RY, Fessl B, Kleindorfer S (2007) Interannual and interspecific variation in intensity of the parasitic fly, Philornis downsi, in Darwin’s finches. Biol Conserv 139:325–332. https://doi.org/10.1016/j.biocon.2007.07.006
Dunčková L, Bednarik M, Krčmář D, Marschalko M, Tornyai R (2019) GIS-based multicriteria evaluation of foundation conditions. Bull Eng Geol Environ 78:2903–2917. https://doi.org/10.1007/s10064-018-1283-9
Eddy TD, Friedlander AM, Salinas de León P (2019) Ecosystem effects of fishing & El Niño at the Galápagos Marine Reserve. Peer J 7:e6878. https://doi.org/10.7717/peerj.6878
Edgar G, Banks S, Fariña JM, Calvopiña M, Martínez C (2004a) Regional biogeography of shallow reef fish and macro-invertebrate communities in the Galapagos Archipelago. J Biogeogr 31:1107–1124. https://doi.org/10.1111/j.1365-2699.2004.01055.x
Edgar G, Bustamante RH, Fariña JM, Calvopiña M, Martinez C et al (2004b) Bias in evaluating the effects of marine protected areas: the importance of baseline data for the Galapagos Marine Reserve. Environ Conserv 31:1–7. https://doi.org/10.1017/S0376892904001584
Edgar GJ, Banks S, Brandt M, Bustamante RH, Chiriboga A et al (2010) El Niño, grazers and fisheries interact to greatly elevate extinction risk for Galapagos marine species. Glob Chang Biol 16:2876–2890. https://doi.org/10.1111/j.1365-2486.2009.02117.x
Ellis-Soto D, Blake S, Soultan A, Guézou A, Guézou A et al (2017) Plant species dispersed by Galapagos tortoises surf the wave of habitat suitability under anthropogenic climate change. PLoS One 12:e0181333. https://doi.org/10.1371/journal.pone.0181333
Epler B (2007) Tourism, the economy, population growth, and conservation in Galapagos. Puerto Ayora, Santa Cruz Island, Galapagos Islands
Fajardo J, Lessmann J, Bonaccorso E, Devenish C. Muñoz J (2014) Combined use of systematic conservation planning, species distribution modelling, and connectivity analysis reveals severe conservation gaps in a megadiverse country (Peru). PLoS One 9:e114367. https://doi.org/10.1371/journal.pone.0114367
Ferreira MT, Cardoso P, Borges PAV, Gabriel R, Brito de Azevedo E et al (2016) Effects of climate change on the distribution of indigenous species in oceanic islands (Azores). Clim Chang 138:603–615. https://doi.org/10.1007/s10584-016-1754-6
Fessl B, Tebbich S (2002) Philornis downsi-a recently discovered parasite on the Galápagos archipelago-a threat for Darwin’s finches? Ibis 144:445–451. https://doi.org/10.1046/j.1474-919X.2002.00076.x
Fordham DA, Brook BW (2010) Why tropical island endemics are acutely susceptible to global change. Biodivers Conserv 19:329–342. https://doi.org/10.1007/s10531-008-9529-7
French SS, Denardo DF, Greives TJ, Strand CR, Demas GE (2010) Human disturbance alters endocrine and immune responses in the Galapagos marine iguana (Amblyrhynchus cristatus). Horm Behav 58:792–799. https://doi.org/10.1016/j.yhbeh.2010.08.001
Füssel HM, Klein RJT (2006) Climate change vulnerability assessments: an evolution of conceptual thinking. Clim Chang 75:301–329. https://doi.org/10.1007/s10584-006-0329-3
Galapagos-Conservancy (2021) Galapagos-Conservancy (2021) Galapagos Conservancy Web (Home > About Galapagos > Biodiversity > Plants). https://www.galapagos.org/about_galapagos/about-galapagos/biodiversity/plants/. Accessed 3 February 2021
Gardener MR, Atkinson R, Rentería JL (2010) Eradications and people: lessons from the plant eradication program in Galapagos. Restor Ecol 18:20–29. https://doi.org/10.1111/j.1526-100X.2009.00614.x
Gardener MR, Trueman M, Buddenhagen C, Heleno R, Jäger H (2013) Plant invasions in protected areas: patterns, problems and challenges. In: Foxcroft, L. et al (Ed.) Plant invasions in protected areas: patterns, problems and challenges. Springer Science+Business Media, pp 1–656. https://doi.org/10.1007/978-94-007-7750-7
Genner MJ, Sims DW, Southward AJ, Budd GC, Masterson P et al (2010) Body size-dependent responses of a marine fish assemblage to climate change and fishing over a century-long scale. Glob Chang Biol 16:517–527. https://doi.org/10.1111/j.1365-2486.2009.02027.x
Glynn PW (1994) State of coral reefs in the Galapagos Islands: natural vs anthropogenic impacts. Mar Pollut Bull 29:131–140. https://doi.org/10.1016/0025-326X(94)90437-5
Glynn PW, Riegl B, Purkis S, Kerr JM, Smith TB (2015) Coral reef recovery in the Galapagos Islands: the northernmost islands (Darwin and Wenman). Coral Reefs 34:421–436. https://doi.org/10.1007/s00338-015-1280-4
Glynn PW, Feingold JS, Baker A, Banks S, Baums ID et al (2018) State of corals and coral reefs of the Galapagos Islands (Ecuador): past, present and future. Mar Pollut Bull 133:717–733. https://doi.org/10.1016/j.marpolbul.2018.06.002
Graham MH, Kinlan BP, Druehl LD, Garske LE, Banks S (2007) Deep-water kelp refugia as potential hotspots of tropical marine diversity and productivity. Proc Natl Acad Sci USA 104:16576–16580. https://doi.org/10.1073/pnas.0704778104
GNP (2021) National Park, Park, Dirección del Parque Nacional Galápagos Web. https://www.galapagos.gob.ec/en/national-park/Accessed 3 February 2021
Graham NAJ, Chabanet P, Evans RD, Jennings S, Letourneur Y et al (2011) Extinction vulnerability of coral reef fishes. Ecol Lett 14:341–348. https://doi.org/10.1111/j.1461-0248.2011.01592.x
Grant PR, Grant RB (2006) Evolution of character displacement in Darwin’s finches. Sci 313:224–226. https://doi.org/10.1126/science.1128374
Grant PR, Grant BR, Keller LF, Petren K (2000) Effects of El Nino events on Darwin’s finch productivity. Ecol 81:2442–2457. https://doi.org/10.1890/0012-9658(2000)081[2442:EOENOE]2.0.CO;2
Greene R, Devillers R, Luther JE, Eddy BG (2011) GIS-based multiple-criteria decision analysis. Geogr Compass 5(6):412–432. https://doi.org/10.1111/j.1749-8198.2011.00431.x
Hamann O (1979) On climatic conditions, vegetation types, and leaf size in the Galapagos Islands. Biotropica 11:101–122. https://doi.org/10.2307/2387785
Hamann O (1985) The El Niño influence on the Galapagos vegetation. In: Robinson G, del Pino ME (eds) El Niño En Las Islas Galápagos: El Evento de 1982-1983. Fundación Charles Darwin para las Islas Galápagos, Quito, pp 299–330
Hamann O (2001) Demographic studies of three indigenous stand- 1593 forming plant taxa (Scalesia, Opuntia, and Bursera) in the 1594 Galapagos Islands, Ecuador. Biodivers Conserv 10:223–250. https://doi.org/10.1023/A:1008952319105
Harter DEV, Irl SDH, Seo B, Steinbauer MJ, Gillespie R et al (2015) Impacts of global climate change on the floras of oceanic islands–projections, implications and current knowledge. Perspect Plant Ecol Syst 17:160–183. https://doi.org/10.1016/j.ppees.2015.01.003
Hartten LM, Gage KS (2000) ENSO’s impact on the annual cycle: The view from Galápagos. Geophys Res Lett 27(3):385–388. https://doi.org/10.1029/1999GL010953
Harvey BP, Gwynn-Jones D, Moore PJ (2013) Meta-analysis reveals complex marine biological responses to the interactive effects of ocean acidification and warming. Ecol Evol 3:1016–1030. https://doi.org/10.1002/ece3.516
Hearn A, Toral-Granda MV (2007) Reproductive biology of the red spiny lobster, Panulirus penicillatus and the Galapagos slipper lobster, Scyllarides astori in the Galapagos Islands. Crustaceana 80:297–312
Hearn A, Marti P, Toral-Granda MV, Murillo C (2005) Population dynamics of the exploited sea cucumber Isostichopus fuscus in the western Galapagos Islands. Ecuador Fish Oceanogr 14:377–385. https://doi.org/10.1111/j.1365-2419.2005.00342.x
Hearn A, Ketchum J, Klimley AP, Espinoza E, Peñaherrera C (2010) Hotspots within hotspots? Hammerhead shark movements around Wolf Island, Galapagos Marine Reserve. Mar Biol 157:1899–1915. https://doi.org/10.1007/s00227-010-1460-2
Hearn AR, Acuña D, Ketchum JT, Peñaherrera C, Green J et al (2014) Elasmobranchs of the Galapagos Marine Reserve. In: Denkinger J, Vinueza L (eds) The Galapagos Marine Reserve, social and ecological interactions in the Galapagos Islands. Springer Science+Business Media New, New York, pp 23–59. https://doi.org/10.1007/978-3-319-02769-2
Hearn AR, Green J, Román MH, Acuña-Marrero D, Espinoza E et al (2016) Adult female whale sharks make long-distance movements past Darwin Island (Galapagos, Ecuador) in the Eastern Tropical Pacific. Mar Biol 163:1–12. https://doi.org/10.1007/s00227-016-2991-y
Heleno RH, Olesen JM, Nogales M, Vargas P, Traveset A (2012) Seed dispersal networks in the Galápagos and the consequences of alien plant invasions. Proc R Soc B 280:20122112. https://doi.org/10.1098/rspb.2012.2112
Hernández-Delgado EA (2015) The emerging threats of climate change on tropical coastal ecosystem services, public health, local economies and livelihood sustainability of small islands: cumulative impacts and synergies. Mar Pollut Bull 101:5–28. https://doi.org/10.1016/j.marpolbul.2015.09.018
Herrera HW, Causton CE (2008) Distribution of fire ants Solenopsis geminata and Wasmannia auropunctata (Hymenoptera: Formicidae) in the Galapagos Islands. Galapagos Res 65:11–14. http://aquaticcommons.org/id/eprint/21133
Heylings P, Bensted-Smith R, Altamirano M (2002) Zonificación e historia de la reserva marina de Galápagos. In: Danulat E, Edgar GJ (eds) Reserva Marina de Galápagos. Línea Base de La Biodiversidad. Fundación Charles Darwin/Servicio Parque Nacional Galápagos, Santa Cruz, Galápagos, pp 10–22
Houck KM, Terán E, Ochoa J, Zapata GN, Gomez AM et al (2020) Drinking water improvements and rates of urinary and gastrointestinal infections in Galápagos, Ecuador: assessing household and community factors. Am J Hum Biol 32:e23358. https://doi.org/10.1002/ajhb.23358
Houvenaghel GT (1974) Equatorial undercurrent and climate in the Galapagos Islands. Nat 250:565–566. https://doi.org/10.1038/250565a0
IPCC (2007) Climate Change 2007 Synthesis report. Contribution of Working Groups I, II and III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. IPCC, Geneva
IPCC (2014) Climate Change 2014: Synthesis report, in: Core-Writing-Team. In: Pachauri RK, Meyer LA (eds) Contribution of Working Group I, II, and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. IPCC, Geneva, p 151
Itow S (2003) Zonation pattern, succession process and invasion by aliens in species-poor insular vegetation of the Galapagos Islands. Glob Environ Res 7:39–58
IUCN, Conseervation Outlook Assessment (2017) Galapagos Islands. https://worldheritageoutlook.iucn.org/node/957/pdf/en?year=2017
Jansson R (2003) Global patterns in endemism explained by past climatic change. Proc R Soc B 270:583–590. https://doi.org/10.1098/rspb.2002.2283
Jiménez-Uzcátegui G, Milstead B, Márquez C, Zabala J, Buitrón P et al (2007) Galapagos vertebrates: endangered status and conservation actions, Galapagos Report 2006-2007. Ingala, Puerto Ayora, Galapagos
Kareiva P, Tallis H, Ricketts T, Daily G, Polasky S (2011) Natural capital: theory and practice of mapping ecosystem services. Press, Oxford University
Karger DN, Conrad O, Böhner J, Kawohl T, Kreft H (2017) Climatologies at high resolution for the earth’s land surface areas. Sci Data 4:1–20. https://doi.org/10.1038/sdata.2017.122
Keener V, Marra J, Finucane M, Spooner D, Smith MH (2012) Climate change and Pacific Islands: indicators and impacts. Report for the 2012 Pacific Islands Regional Climate Assessment (PIRCA). Island Press, Washington DC
Keith I, Dawson T, Collins K, Banks S (2015) Marine invasive species in the Galapagos Marine Reserve: a case for additional research, improved management, and policy review. In: Galapagos Report 2013-2014. GNPD, GCREG, CDF and GC, Puerto Ayora, Galapagos, pp 83–88
Kislik E, Saltos GM, Torres G, Borbor-Córdova M (2017) Biological hotspots in the Galapagos Islands: exploring seasonal trends of ocean climate drivers to monitor algal blooms. Int J Bioeng Life Sci 11:784–794. https://doi.org/10.5281/zenodo.1314831
Larrea Oña I, Di Carlo G (2011) Climate change vulnerability assessment of the Galapagos Islands. WWF and Conservation International, USA.
Laso FJ, Ben L, Rivas-Torres G, Sampedro C, Arce-Nazario J (2020) Land cover classification of complex agroecosystems in the non-protected highlands of the Galapagos Islands. Remote Sens 12:1–39. https://doi.org/10.3390/rs12010065
Laurie A (1985) The effects of the 1982-83 El Niño on marine iguanas. In: Robinson G, Del Pino ME (eds) El Niño En Las Islas Galápagos: El Evento de 1982-1983. Charles Darwin Foundation, Quito, pp 199–209
Lawson LP, Fessl B, Vargas HF, Farrington HL, Cunninghame FH et al (2017) Slow motion extinction: inbreeding, introgression, and loss in the critically endangered mangrove finch (Camarhynchus heliobates). Conserv Genet 18:159–170. https://doi.org/10.1007/s10592-016-0890-x
Lessios HA, Glynn PW, Robertson DR (1983) Mass mortalities of coral reef organisms. Sci 222:715. https://doi.org/10.1007/BF00288252
Lin FT (1998) Many sorted algebraic data models for GIS. Int J Geogr Inf Sci 12:765–788. https://doi.org/10.1080/136588198241491
Liu Y, Xie L, Morrison JM, Kamykowski D (2013) Dynamic downscaling of the impact of climate change on the ocean circulation in the Galápagos Archipelago. Adv Meteorol 2013:1–18. https://doi.org/10.1155/2013/837432
Llerena Y, Peñaherrera C, Espinoza E, Hirschfeld M, Wolff M et al (2015) Nursery grounds of blacktip sharks (Carcharhinus limbatus) in mangrove-fringed bays in the central part of the Galapagos archipelago. In: Galapagos Report 2013-2014. GNPD, GCREG, CDF and GC, Puerto Ayora, Galapagos, pp 103–110
Mantyka-Pringle CS, Martin TG, Rhodes JR (2012) Interactions between climate and habitat loss effects on biodiversity: a systematic review and literature review. Glob Chang Biol 18:1239–1252. https://doi.org/10.1111/j.1365-2486.2011.02593.x
Marquez C, Wiedenfeld D, Naranjo S, Llerena W (2008) 1997-8 el Niño and the Galapagos tortoises Geochelone vandenburghi on Alcedo Volcano, Galapagos. Galapagos Res 65:7–10
Martin NJ, Conroy JL, Noone D, Cobb KM, Konecky BL et al (2017) Seasonal and ENSO influences on the stable isotopic composition of Galápagos precipitation. J Geophys Res Atmos 123:261–275. https://doi.org/10.1002/2017JD027380
Mathis A, Rose J (2016) Balancing tourism, conservation, and development: a political ecology of ecotourism on the Galapagos Islands. J Ecotour 15:64–77. https://doi.org/10.1080/14724049.2015.1131283
Millennium Ecosystem Assessment (2005) Ecosystems and human well-being: wetlands and water synthesis. World Resources Institute, Washington, DC
Ministerio del Ambiente (2016) Ministerial agreement No. 093 (2016). Accessed at: http://www.galapagos.gob.ec/wpcontent/uploads/downloads/2016/10/Acuerdo_Ministerial_N093_Reforma_Santuario_Marino.pdf
Moity N (2018) Evaluation of no-take zones in the Galapagos Marine Reserve, zoning plan 2000. Front Mar Sci 5:1–5. https://doi.org/10.3389/fmars.2018.00244
Moity N, Delgado B, Salinas-De-Leon P (2019) Mangroves in the Galapagos Islands: distribution and dynamics. PLoS One 14:e0209313. https://doi.org/10.1371/journal.pone.0209313
Moreira M, Fonseca C, Vergílio M, Calado H, Gil A (2018) Spatial assessment of habitat conservation status in a Macaronesian island based on the InVEST model: a case study of Pico Island (Azores, Portugal). Land Use Policy 78:637–649. https://doi.org/10.1016/j.landusepol.2018.07.015
Mouillot D, Graham NAJ, Villéger S, Mason NWH, Bellwood DR (2013) A functional approach reveals community responses to disturbances. Trends Ecol Evol 28:167–177. https://doi.org/10.1016/j.tree.2012.10.004
Nash S (2009) Ecotourism and other invasions. Biosci 59:106–110. https://doi.org/10.1525/bio.2009.59.2.3
Nelson GC, Bennett E, Berhe AA, Cassman K, DeFries R (2006) Anthropogenic drivers of ecosystem change: an overview. Ecol Soc 11:29. https://doi.org/10.5751/ES-01826-110229
Nerem RS, Beckley BD, Fasullo JT, Hamlington BD, Masters D (2018) Climate-change–driven accelerated sea-level rise detected in the altimeter era. Proc Natl Acad Sci USA 115:2022–2025. https://doi.org/10.1073/pnas.1717312115
Nicholas K, Bentley M, Terán E, Thompson A (2019) Water Security in the Galápagos: socioecological determinants and health implications. EcoHealth. 117:111–124. https://doi.org/10.1007/s10393-019-01456-w
Okey TA, Banks S, Born AF, Bustamante RH, Calvopiña M et al (2004) A trophic model of a Galápagos subtidal rocky reef for evaluating fisheries and conservation strategies. Ecol Model 172:383–401. https://doi.org/10.1016/j.ecolmodel.2003.09.019
Paine R (1995) A conversation on refining the concept of keystone species. Conserv Biol 4:962–964. https://doi.org/10.1046/j.1523-1739.1995.09040962.x
Pazmiño DA, Maes GE, Simpfendorfer CA, Salinas-De-León P, Van Herwerden L (2017) Genome-wide SNPs reveal low effective population size within confined management units of the highly vagile Galapagos shark (Carcharhinus galapagensis). Conserv Genet 18:1151–1163. https://doi.org/10.1007/s10592-017-0967-1
Peñaherrera-Palma C, Espinosa E, Hearn AR, Ketchum J, Semmens JM et al (2017) Report of the population status of hammerhead sharks in the Galapagos Marine Reserve, Galapagos Report 2015-2016. GNPD, GCREG, CDF and GC, Puerto Ayora, Galapagos
Phillips RB, Cooke BD, Carrión V, Snell HL (2012a) Eradication of rock pigeons, Columba livia, from the Galapagos Islands. Biol Conserv 147:264–269. https://doi.org/10.1016/j.biocon.2012.01.013
Phillips RB, Wiedenfeld DA, Snell HL (2012b) Current status of alien vertebrates in the Galapagos Islands: invasion history, distribution, and potential impacts. Biol Invasions 14:461–480. https://doi.org/10.1007/s10530-011-0090-z
Pizzitutti F, Walsh SJ, Rindfuss RR, Gunter R, Quiroga D et al (2017) Scenario planning for tourism management: a participatory and system dynamics model applied to the Galapagos Islands of Ecuador. J Sustain Tour 25:1117–1137. https://doi.org/10.1080/09669582.2016.1257011
Porter DM (1979) Endemism and evolution in Galapagos Islands vascular plants. In: Bramwell D (ed) Plants and islands. Academic Press, London, pp 225–256
Purcell SW, Mercier A, Conand C, Vero M, Lovatelli A et al (2011) Sea cucumber fisheries: global analysis of stocks, management measures and drivers of overfishing. Fish Fish 14:34–59. https://doi.org/10.1111/j.1467-2979.2011.00443.x
Ragazzi M, Catellani R, Rada EC, Torretta V, Salazar-valenzuela X (2016) Management of urban wastewater on one of the Galapagos Islands. Sustain 8:2–19. https://doi.org/10.3390/su8030208
Reck G, Casafont M, Naula E, Oviedo M (2010) SIMAVIS–System of managing visitors of the Galapagos National Park. Galapagos Report 2009-2010:93–102
Renteria JL, Gardener MR, Panetta DF, Atkinson R, Crawley MJ (2012a) Possible impacts of the invasive plant Rubus niveus on the native vegetation of the Scalesia Forest in the Galapagos Islands. PLoS One 7:e48106. https://doi.org/10.1371/journal.pone.0048106
Renteria JL, Gardener MR, Panetta FD, Crawley MJ (2012b) Management of the invasive hill raspberry (Rubus niveus) on Santiago Island, Galapagos: eradication or indefinite control? Invasive Plant Sci Manag 5:37–46. https://doi.org/10.1614/IPSM-D-11-00043.1
Rial M, Martínez-Cortizas A, Taboada T, Rodríguez-Lado L (2017) Soil organic carbon stocks in Santa Cruz Island, Galapagos, under different climate change scenarios. Catena 156:74–81. https://doi.org/10.1016/j.catena.2017.03.020
Riegl B, Johnston M, Glynn PW, Keith I, Rivera F (2019a) Some environmental and biological determinants of coral richness, resilience and reef building in Galápagos (Ecuador). Sci Rep 9:10322. https://doi.org/10.1038/s41598-019-46607-9
Riegl B, Glynn PW, Banks S, Keith I, Rivera F et al (2019b) Heat attenuation and nutrient delivery by localized upwelling avoided coral bleaching mortality in northern Galapagos during 2015/2016 ENSO. Coral Reefs 38:773–785. https://doi.org/10.1007/s00338-019-01787-8
Robinson G (1985) Influence of the 1982-83 El Niño on Galapagos marine life. In: Robinson G, Del Pino ME (eds) El Niño En Las Islas Galápagos: El Evento de 1982-1983. Fundación Charles Darwin para las Islas Galápagos, Quito, pp 153–190
Robinson G, Del Pino ME (1985) El Niño en las Islas Galápagos: El evento de 1982-1983. Charles Darwin Foundation for the Galápagos Isles, Quito
Ruiz DJ, Wolff M (2011) The Bolivar Channel Ecosystem of the Galapagos Marine Reserve: energy flow structure and role of keystone groups. J Sea Res 66:123–134. https://doi.org/10.1016/j.seares.2011.05.006
Rustic GT, Koutavas A, Marchitto TM, Braddock LK (2015) Dynamical excitation of the tropical Pacific Ocean and ENSO variability by Little Ice Age cooling. Sci 350:1537–1541. https://doi.org/10.1126/science.aac9937
Ruttenberg BI (2001) Effects of artisanal fishing on marine communities in the Galapagos Islands. Conserv Biol 15:1691–1699. https://doi.org/10.1046/j.1523-1739.2001.99556.x
Sachs BJP, Ladd SN (2010) Climate and oceanography of the Galapagos in the 21st century: expected changes and research needs. Galapagos Res 67:50–54
Salazar S, Denkinger J (2010) Possible effects of climate change on the populations of Galapagos pinnipeds. Galapagos Res 67:45–49
Salinas-de-León P, Rastoin E, Acuña-Marrero D (2015) First record of a spawning aggregation for the tropical eastern Pacific endemic grouper Mycteroperca olfax in the Galapagos Marine Reserve. J Fish Biol 87:179–186. https://doi.org/10.1111/jfb.12703
Salinas-De-León P, Andrade S, Arnés-Urgellés C, Bermudez JR, Bucaram S et al (2020) Evolution of the Galapagos in the Anthropocene. Nat Clim Chang 10:380–382. https://doi.org/10.1038/s41558-020-0761-9
Scarano FR (2017) Ecosystem-based adaptation to climate change: concept, scalability and a role for conservation science. Perspect Ecol Conserv 15:65–73. https://doi.org/10.1016/j.pecon.2017.05.003
Schiller L, José J, Grove J, Reck G, Pauly D (2014) The demise of Darwin’s fishes: evidence of fishing down and illegal shark finning in the Galapagos Islands. Aquat Conserv Mar Freshw Ecosyst 25:431–446. https://doi.org/10.1002/aqc.2458
Scott D, Amelung B, Ceron J.-P, Dubois G, Gössling S et al (2008) Climate change and tourism: responding to global challenges, CTO/CRSTDP Regional Workshop. Caribbean Regional Sustainable Tourism Development Programme CTO Lot 3: Sustainable Tourism Policy Development. Davos, Switzerland.
Self RM, Self DR, Bell-Haynes J (2010) Marketing tourism in the Galapagos Islands: ecotourism or greenwashing? Int Bus Econ Res J 9:111–126. https://doi.org/10.19030/iber.v9i6.590
Smale DA, Wernberg T, Oliver ECJ, Thomsen M, Harvey BP (2019) Marine heatwaves threaten global biodiversity and the provision of ecosystem services. Nat Clim Chang 9:306–312. https://doi.org/10.1038/s41558-019-0412-1
Smith SJ, Edmonds J, Hartin CA, Mundra A, Calvin K (2015) Near-term acceleration in the rate of temperature change. Nat Clim Chang 5:333–336. https://doi.org/10.1038/NCLIMATE2552
Snell H, Rea S (1999) The 1997-98 El Niño in Galapagos: can 34 years of data estimate 120 years of pattern? Not Galápagos 60:11–20
Sodhi NS, Liow LH, Bazzaz FA (2004) Avian extinctions from tropical and subtropical forests. Annu Rev Ecol Syst 35:323–345. https://doi.org/10.1146/annurev.ecolsys.35.112202.130209
Stein Grove J (1985) Influence of the 1982-1983 El Niño event upon the ichthyofauna of the Galapagos archipelago. In: Robinson G, Del Pino ME (eds) El Niño En Las Islas Galápagos: El Evento de 1982-1983. Charles Darwin Foundation for the Galápagos Isles, Quito, pp 191–198
Szuwalski CS, Castrejon M, Ovando D, Chasco B (2016) An integrated stock assessment for red spiny lobster (Panulirus penicillatus) from the Galapagos Marine Reserve. Fish Res 177:82–94. https://doi.org/10.1016/j.fishres.2016.01.002
Tanner MK, Moity N, Costa MT, Jarrin JRM (2019) Mangroves in the Galapagos: ecosystem services and their valuation. Ecol Econ 160:12–24. https://doi.org/10.1016/j.ecolecon.2019.01.024
Thompson DM, Conroy JL, Collins A, Hlohowskyj SR, Overpeck JT et al (2017) Tropical Pacific climate variability over the last 6000 years as recorded in Bainbridge Crater Lake, Galápagos. Paleoceanography 32:903–922. https://doi.org/10.1002/2017PA003089
Thompson AL, Nicholas KM, Watson E, Terán E, Bentley ME (2019) Water, food, and the dual burden of disease in Galapagos. Am J Hum Biol 32:e23344. https://doi.org/10.1002/ajhb.23344
Tompkins P, Wolff M (2016) Galápagos macroalgae: a review of the state of ecological knowledge. Rev Biol Trop 65:375. https://doi.org/10.15517/rbt.v65i1.18139
Toral-Granda V (2008) Galapagos Islands: a hotspot of sea cucumber fisheries in Latin America and the Caribbean In: Toral-Granda V, Vasconcellos A, Lovatelli M (eds.) Sea Cucumbers. A Global Review of Fisheries and Trade. FAO Fisheries and Aquaculture Technical Paper, Rome, FAO, pp. 231–253.
Toral-Granda MV, Martínez PC (2005) Population density and fishery impacts on the sea cucumber (Isostichopus fuscus) in the Galapagos marine reserve. FAO Fish Tech Pap
Toral-Granda V, Causton CE, Jäger H, Trueman M, Izuriet JC et al (2017) Alien species pathways to the Galapagos Islands, Ecuador. PLoS One 12:e0184379. https://doi.org/10.1371/journal.pone.0184379
Townsend C (1934) The fur seals of the Galapagos Islands. Zool:43–56
Trillmich F, Limberger D (1985) Drastic effects of El Niño on Galapagos pinnipeds. Oecologia 67:19–22. https://doi.org/10.1007/BF00378445
Trueman M, D’Ozouville N (2010) Characterizing the Galapagos terrestrial climate in the face of global climate change. Galapagos Res 67:26–37
Tye A, Aldaz I (1999) Effects of the 1997-98 El Niño event on the vegetation of Galapagos. Not Galápagos 60:22–24
Tye A, Francisco-Ortega J (2011) Origins and evolution of Galapagos endemic vascular plants. In: Bramwell D, Caujapé-Castells J (eds) The biology of island floras. Cambridge University Press, Cambridge, pp 89–153
Usseglio P, Friedlander AM, Koike H, Zimmerhackel J, Schuhbauer A et al (2016) So long and thanks for all the fish: overexploitation of the regionally endemic Galapagos Grouper Mycteroperca olfax (Jenyns, 1840). PLoS One 11:e0165167. https://doi.org/10.1371/journal.pone.0165167
Vilà M, Espinar JL, Hejda M, Hulme PE, Jarošík V et al (2011) Ecological impacts of invasive alien plants: a literature review of their effects on species, communities and ecosystems. Ecol Lett 14:702–708. https://doi.org/10.1111/j.1461-0248.2011.01628.x
Walsh SJ, Mena CF (2016) Interactions of social, terrestrial, and marine sub-systems in the Galapagos Islands. Ecuador Proc Natl Acad Sci 113:14536–14543. https://doi.org/10.1073/pnas.1604990113
Wang C, Fiedler PC (2006) Progress in Oceanography ENSO variability and the eastern tropical Pacific: a review. Prog Oceanogr 69:239–266. https://doi.org/10.1016/j.pocean.2006.03.004
Watson J, Trueman M, Tufet M, Henderson S, Atkinson R (2009) Mapping terrestrial anthropogenic degradation on the inhabited islands of the Galapagos Archipelago. Oryx 44:79–82. https://doi.org/10.1017/S0030605309990226
Wauters N, Dekoninck W, Hendrickx F, Herrera HW, Fournier D (2016) Habitat association and coexistence of endemic and introduced ant species in the Galapagos Islands. Ecol Entomol 41:40–50. https://doi.org/10.1111/een.12256
Wauters N, Fournier D, Dekoninck W (2017) Do agricultural zones on Santa Cruz Island, Galápagos, host native and endemic arthropods? Pan-Pac Entomol 93:141–158. https://doi.org/10.3956/2017-93.2.141
Wauters N, Dekoninck W, Nagy ZT, Fournier D (2018) Impact of laying date and fire ants on hatchlings of Chelonoidis porteri on Santa Cruz island, Galápagos. Ecuador Herpetol Conserv Biol 13:479–487
Whitehead HAL, Christal J, Dufault S (1997) Past and distant whaling and the rapid decline of sperm whales off the Galapagos Islands. Conserv Biol 11:1387–1396. https://doi.org/10.1046/j.1523-1739.1997.96246.x
Wiedenfeld DA, Jiménez-Uzcátegui GA (2008) Critical problems for bird conservation in the Galapagos Islands. Cotinga 29:22–27
Wikelski M, Carbone C, Trillmich F (1996) Lekking in marine iguanas: female grouping and male reproductive strategies. Anim Behav 52:581–596. https://doi.org/10.1006/anbe.1996.0199
Wolff M (2010) Galapagos does not show recent warming but increased seasonality. Galapagos Res 67:38–44
Wolff M, Schuhbauer A, Castrejón M (2011) A revised strategy for the monitoring and management of the Galapagos sea cucumber Isostichopus fuscus (Aspidochirotida: Stichopodidae). Rev Biol Trop 60:539–551. https://doi.org/10.15517/rbt.v60i2.3912
Wolff M, Ruiz DJ, Taylor M (2012) El Niño induced changes to the Bolivar Channel ecosystem (Galapagos): comparing model simulations with historical biomass time series. Mar Ecol Prog Ser 448:7–22. https://doi.org/10.3354/meps09542
Zimmerhackel JS, Schuhbauer AC, Usseglio P, Heel LC, Salinas-De-León P (2015) Catch, bycatch and discards of the Galapagos Marine Reserve small-scale handline fishery. Peer J 3:e995. https://doi.org/10.7717/peerj.995
Acknowledgements
We thank the Directorate of the Galapagos National Park, WWF-Ecuador and San Francisco University for sharing thematic and base maps of Galapagos, particularly the zoning plan of 2016, PUA, seabed’s distribution, and statistics of visitors. We also thank INAMHI for their datasets from climatic stations. We are thankful for the Galapagos National Park staff and several stakeholders that participated in our validation exercise and provided valuable inputs that guided the selection of the priority HEVA. Special thanks to Gabriela Maldonado, Mariuxi Farias, Manuela González, and Tarsicio Granizo from WWF-Ecuador for providing assistance in designing and implementing the workshop in the Galapagos National Park. We are particularly grateful for Gabriela Maldonado for her valuable comments and support throughout this study. We also thank Nicole Barbour for comments on the manuscript.
Funding
All authors acknowledge WWF-Ecuador and Universidad de las Américas for the financial support provided FC acknowledges Universidad de las Américas for making this publication Open Access. MC acknowledges the financial support provided by the Inter-American Institute for Global Change Research (IAI).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by Tony Weir
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 3171 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Escobar-Camacho, D., Rosero, P., Castrejón, M. et al. Oceanic islands and climate: using a multi-criteria model of drivers of change to select key conservation areas in Galapagos. Reg Environ Change 21, 47 (2021). https://doi.org/10.1007/s10113-021-01768-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10113-021-01768-0