Abstract
The effects of low-level laser on the wound healing and burn injuries have been previously examined to demonstrate some satisfactory results. Despite there are a few articles available to study photobiomodulation (PBM) effects on the pain relief of cesarean sectioned wound, however no systematic examination has been carried out so far regarding its healing. Here, the aim of this clinical study was to evaluate PBM effect on the cesarean-sectioned wound healing. PBM effects of semiconductor lasers are investigated at 658 and 660 nm with 100, 150 and 350 mW output powers on 40 patients. Due to the global increasing number of cesarean sections, we have decided to investigate the effect of laser as a reliable technique to recover the wounds fast. We considered women as the target group who had their first delivery giving the birth of their children by cesarean section. We selected patients are who treated by laser therapy using indium gallium aluminum phosphide (InGaAlP) semiconductor linear scanning type with beam cross section of 12 cm2 and the output power of 100 mW at 658 nm exposing a therapeutic dose of 2 J/cm2. The purpose is to accelerate the healing process of the wounds after delivery as an intervention group against the people who chose the conventional methods (using ointments, pills, etc.) to heal their cesarean sectioned wounds as the control group. Regarding the wounds of these two groups, the questionnaires were filled by patients to assess the severity of pain from visual analogue scale (VAS) based on the healing of wounds from redness, edema, ecchymosis, discharge, and distance between the two edges of the wound (REEDA) scale in the early hours after surgery and the post-treatment follow-up on the third, seventh, and the tenth days. The data collected by these questionnaires were analyzed using statistical package for social science)SPSS( as a statistical software to give out the comparative histograms. This study reports a clinical examination of PBM under intervention group of 40 patients ranging 18–40 years old with body mass index (BMI) of 29–36, during post-cesarean surgery to elucidate successful healing of the wounds and scars against conventional methods which considered as control group. Comparison of mean REEDA scores on the third day (p = 0.035), seventh day (p = 0.03), and tenth day (p = 0.02) after delivery exhibits that the two groups benefit a statistically significant difference with each other. For instance, the mean wound healing score in the intervention group was almost half of the mean wound healing score on the tenth day in the control group (1.09 ± 0.586 vs. 2.25 ± 0.422). The post-cesarean follow-up indicates that the patients treated by the laser therapy (intervention group) encounter better recovery than the control group.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The effect of low-level laser therapy (LLLT), also as known as photobiomodulation (PBM) therapy, on the wound healing and burn injuries has been previously examined to demonstrate some satisfactory results. Despite there are a few articles available to investigate low-level laser effects on the pain relief of cesarean sectioned wound, however no extensive study has been carried out so far regarding its healing [1,2,3]. PBM is one of the non-invasive treatment methods using light sources, which is performed by biological stimulation effects in photochemical processes. It basically does not increase the temperature at the macroscopic level. In medicine, light absorption reactions by receptors that can absorb light at specific wavelengths are widely used. The absorbing molecule in a light-activated form can cause chemical reactions that result in the treatment of a specific disease. This molecule should be a part of a main structure that can regulate the metabolic pathway. PBM or photobiomodulation effects do not increase the macroscopic temperature of the tissue, because the power density used in this method is low (the cooling rate of the irradiation tissue is faster than the rate of receiving energy from the radiation source). But it can cause a change in temperature at the microscopic level, which causes the start of cellular and molecular processes. The primary mechanism of PBM with intracellular receptors occurs as follows: Absorption of photon by intracellular compounds, creation of temperature difference in nearby points due to absorbed energy, release of Ca+2 from intracellular sources, initiation of processes dependent on Ca+2, increases in biological factors, formation of tissue metabolic glands, and regulation of nerve hormones. Radiation parameters, such as intensity, wavelength, and pulse mode, have important role in PBM. Figure 1 illustrates a simple model of PBM for enhancement of wound/scar recovery. The wound healing includes three phases: inflammation, proliferation, and restoration. During laser exposure, the movement of immune cells towards the wound becomes faster (inflammation phase), the production of fibroblasts and macrophages increases (proliferation phase), and collagen production will also enhance (restoration phase). The main effect of laser in wound healing is the release of cytokines and biological responses that reduce healing time. In both open and closed wounds and soft tissue injuries, PBM can be effective according to the following enhancement processes: increasing the activity of leukocytes and the activity of macrophages and stimulating collagen synthesis and angiogenesis and as a result oxygen delivery improvement, the proliferation of fibroblasts due to speeding up the collagen production, and the generation rate of epithelial cells alongside the reduction and the wound and infection, growth factors, and cell proliferation by ATP synthesis as well as keratinocytes proliferation. In these cases, lasers or LEDs are used with spectral range of 630–910 nm.
Photobiomodulation (PBM) addresses the non-thermal laser-tissue interactions when low-level laser exposure is applied on the tissue of interest. The mechanism of PBM and low level laser therapy has been previously reviewed [4, 5]. In fact, PBM is a form of medicine that applies low-level coherent sources to the surface of the skin. On the other hand, Monte Carlo simulation of photon densities inside the dermis in PBM was carried out. It was shown that the backscattered photons in dermis layers, as highly scattering media, notably contribute to change the photon density along propagation axis due to the multiple scattering [6]. The interaction of photons with highly scattering media includes backscattering and the multiple scattering which deviate from Lambert law. In fact, the multiple scattering causes the photons accumulate in subcutaneous layers. Increasing the proliferation of different tissue cells after laser treatment accounts as the major mechanism of laser effect on the tissue. To evaluate the cell proliferation resulting from treatment, the number of cells is examined using cell counting method. Collagen which is mainly made from fibroblast leads to tissue/tensile strength. There have been many studies on the effect of laser on the proliferation of fibroblast, the amount of collagen released in the tissue, and the organization of collagen fibers. The low-power laser stimulates fibroblasts and accelerates the onset of cell proliferation following by alleviating inflammatory reactions and consequently the increase in collagen fibers [7].
Many laboratory and clinical studies have emphasized the acceleration of repair takes into account as the main reason for the effectiveness of the laser in the cell proliferation. The presence of fibroblasts and collagen fibers quantitatively/qualitatively are the evidences of the wound mechanical strength and tissue tensile strength. However, there is more consensus on the role of low-power lasers in the synthesis phenomenon. Not only the presence of fibroblasts is effective in the phenomenon of collagen production and mechanical strength of the wound, but also causes the formation of myofibroblasts to contract and accelerate its closure. Despite one believes that wound closure is not a good measure of wound healing, many studies have examined the effect of laser treatment on wound surface size. Rocha et al. [8] also mentioned the acceleration of wound surface closure in the animal model after 4 consecutive days using He–Ne laser treatment. Hopkins et al. [9], Simunovic et al. [10], and Al Watban et al.[11] and Bisht and Pereira et al. [12, 13] reported separate studies on human specimens giving rise to the accelerated surface closure of the wound after He–Ne laser therapy. In separate studies, Demir et al. [14] reported an increase in the number of fibroblasts in the cell proliferation phase against the control group after laser treatment. Bayat et al. [15] also pointed to the increase in the number of fibroblasts and the improvement of maximum tissue tension after laser treatment using low-power laser at 780 nm.
Reddy [16] examined the effects of two low-power lasers gallium arsenide (Ga-As) and helium neon (He–Ne) lasers at 21 J/cm2, 5 days a week for 3 weeks on a diabetic rat wound. It was concluded that both types of lasers improve the biomechanical properties, scaling up the amount of collagen. This attests that the effect of He–Ne lasers on biomechanical parameters is significantly greater than that of Ga-As ones. Stadler et al. [17] reported an increase in tissue tensile strength after laser treatment at 830 nm in diabetic female rats. Abergel et al. [18] investigated the effect of Ga-As and He–Ne lasers on fibroblast cultures with different intensities. An increase in collagen production by fibroblasts in both laser therapy groups is observed against the control one. Akio Yasukawa et al. [19] also studied the effect of He–Ne laser on rat’s surgical wound and reported a significant increase in tissue tensile strength and collagen fiber formation as well as tissue cohesion using 17 mW laser on the treated group. Hosseini et al. [20] showed that Ga-As and He–Ne lasers improve biomechanical properties following a 10-day treatment period on a daily basis and concluded that this event arises from the collagen production. The combination of two lasers would speed up the closure of the wound surface in full-thickness. Carvalho et al. [21] also reported the effect of He–Ne laser on rat surgical wounds and found an increase in collagen levels in laser-treated samples compared to the control wounds. Ikeuchi [22] examined the effect of lasers on surgical wounds elucidating that the tissue was more mature in the treatment group than in the control one on day 14th. In addition to the collagen secretion and growth factors, the myofibroblasts exhibit a direct contractile effect leading to the wound contraction and closure. However, Moulin et al. [23] concluded that the effect of the contractile process on the wound closure in human skin was less than that of other mammals.
Although the wound healing is reported to be enhanced by PBM in normal rodents [24, 25], PBM significantly improves wound healing in both diabetic rats [26, 27] and diabetic mice too [28, 29]. Eventually a case–control clinical trial and in vivo study of cesarean wounds reveal that PBM appears to increase proclatin secretion in those with low prolactin levels treated three times under 15–25-min laser exposure for 10 days [1].
The aim of this work is to treat systematically the scars of cesarean sectioned wound by making use of PBM under LLLT leading to the wound healing, following up day by day observations, relying on the regular assessment of the patients’ questionnaires. In fact, the conventional post-cesarean surgery wound management strategy includes the treatment of surgical site infection and the reduction of the number of complications clinically and physiologically using regular laser exposure on the wound area as well as the optimization of the laser parameters. Here, the biomodulation effect of semiconductor lasers at 658 and 660 nm with 100, 150, and 350 mW output powers is investigated on 40 patients who have been operated by cesarean. The clinical follow-up emphasizes that the laser therapy leads to the reduction of the recovery time and removal the scars as well as the notable pain alleviation.
The observed groups were selected among women with their first delivery, healthy and without underlying diseases, without high excess weight or severe underweight, and being in the appropriate age range. Surgical conditions, including the type of suture thread, were considered the same for all subjects under observation. Conventional assessments and treatments were started just after cesarean section for both groups. The intervention group received their first laser therapy on the first hours after surgery. Post-cesarean wound infection includes wound cellulitis or wound abdominal abscess causing redness, swellings, pain, and discharge from the wound. Cesarean may notably increase risk of asthma, systematic connective tissue disorders, and immune deficiencies.
Materials and methods
The patients’ consent is received before laser treatment according to typical testimonial given in the Appendix 1. In addition, we have no access to confidential personal information of the patients except their age, weight, height, and BMI. The 3 × 4 m2 laser treatment room has a patient bed that is disinfected with a portable UV lamp for each use. All sheets are disposable and have been disinfected before. The therapist was one of the expertized nurses of the hospital and was familiar with all laser safety and infection prevention protocols. She was not aware of the research objectives and performed therapy and completed the questionnaire blindly and presented it to the research team. In each therapy session, she changed his mask, gloves, and gown. The patient was required to use a disposable sterile patient’s gown for each therapy session. The bodies of linear and pen laser devices were placed under UV lamp radiation (Kyutee portable UV-C sterilizer, 35 µW/cm2 intensity at 6", 254 nm) before being turned on for therapy. The lighting of the room was provided by using 6 ceiling LED lamps, each with a power of 20 watts. This was adequate to create the normal lighting of the therapy room. The clinical observations alongside the filled questionnaires are assessed accordingly. In the course of assessment, as much as possible, an attempt has been made to prevent allocation bias by hiring a person who is unaware of the research objectives for therapy and data collection. The patients were not informed about the expected result of the laser therapy and had a vague idea of the outcome of the research, which prevented the placebo or cohort study in a large scale. The selectable conditions were set so that indicators such as age, weight, and conditions of cesarean sections for all patients undergo an almost reasonable range. The effect of placebo is neglected in this research. The assessor does not know which patient is within the control or intervention group. The questionnaires are arranged based on the number encoding to be blind for the assessor.
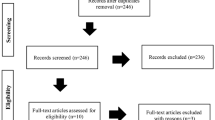
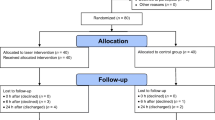
This research is a clinical trial that is non-randomized and one-sided on 80 people (40 individuals are in the intervention group, and 40 cases lie in the control group). The primiparous women are selected in Sarem Gynecology, Obstetrics, and Infertility Hospital for cesarean section. The parameters of the report are tabulated in Table 1.
The sample size is calculated using the mean difference method by the following formula.
The sample size is calculated using the mean difference method by the following formula.
REEDA scale is a tool for assessing perineal healing [23]. It includes five items related to the healing process such as hyperemia, edema, ecchymosis, discharge, and the apposition of the wound edges [24]. The research variables include the wound healing rate, quantitative variable of discrete scale type (total REEDA score for each person), pain intensity, and quantitative variable of discrete scale type (according to VAS questionnaire). REEDA questionnaire is an indicator as the quantitative variable of discrete scale type, quantitative variable of continuous scale type, weight, and quantitative variable of continuous scale type. Note that the typical questionnaire form is presented in the Appendix 2.
This trial is one-sided blind, because people decide according to their own opinion or their doctor’s opinion to do laser therapy or to be treated in the usual way to heal their wound, so it is not possible for a person not to know which group (control or test) she is placed in assessor blind. However, the laser treatment is performed by someone other than the researcher who is not aware of the objectives of the study.
The inclusion criteria for the study are primiparous women aged between 18 and 40 years with a normal body mass index; not having diseases that interfere with wound healing such as chronic systemic diseases, heart, kidney, liver, respiratory, coagulation, connective tissue, diabetes, anemia, cancer, and mental illnesses; no use of drugs effective on wound healing such as glucocorticoids, anticoagulants, immunosuppressants, broad-spectrum antibiotics, and chemotherapy; no reconstructive surgery in the abdomen; and no use of drugs and alcohol.
The exclusion criteria include in the following: not attending laser treatment on time, occurrence of allergy due to laser treatment, new trauma during the study for any reason, getting diseases that require antibiotic prescription during the study, failure to go to the clinic for follow-up, mother’s refusal to continue participating in the research, or expressing dissatisfaction for personal reasons.
In the intervention group, patients are required to visit the laser center of Sarem Hospital for 10 days, with 1 day in between, and undergo laser therapy by two types of low-power semiconductor lasers InGaAlP, from the Heltschl company, made in Austria, with a radiation cross section of 12 cm2 and the output power of 100 mW at 660 and 658 nm with a therapeutic dose of 2 J/cm2. The therapeutic window is located at 600–1200 nm [30]. In this spectral range, light penetrates into the biological tissues with less loss, and therapeutic work is carried out in deeper layers. In favor of an 8-cm long wound, the laser scans the first 4-cm section for 4 min, and then, it repeats for the adjacent section similarly. The laser exposure is carried out on one side of the wound in 4 min, and then, the head of the device is move to provoke the other side of the wound for another 4 min. If the length of the wound is longer, the duration of radiation will also increase. Since the doctor considers a specific cut length for the cesarean section based on the conditions of the patient and the baby, the duration of the laser radiation varies from person to person. The output power of the laser in linear mode is 100 mW, and the optimal irradiation time is 10–12 min at 658 nm achieving therapeutic laser dose. According to the size of the wound and the healing status of the wound, each person is exposed to laser radiation for 15–25 min. The method of laser radiation to the target area relies on: first, a rectangular cross section of 3 cm × 4 cm is covered by linear scanning by an InGaAlP laser. Figure 2 depicts a schematic of the irradiation set-up. The area of the cesarean section is exposed by laser radiation for a period of time determined by the doctor. Then, the areas where the depth of the wound is greater or the areas with more inflammation, such as the corners of the wound that have protrusion, are again exposed to radiation by the pen diode laser.
For laser treatment of cesarean section wounds, first, we set the area laser to the linear mode (rectangular), and according to the length of the wound, we have selected the duration of the laser radiation.
After finishing the linear laser beam, we start illumination by the pen or point laser. The laser pen is situated over the wound in a contact manner and press it down a little. If the wound is fresh and the person spends the first few days of treatment, let avoid the pressure exertion. The laser moves at intervals of 1 min along 1 cm. The output power of the pen laser is selected ranging from 150 to 350 mW at 660 nm. The laser parameters are tabulated in Table 2. The optimal time of exposure of the pen laser on the wound lasts 5–9 min. In order to have a better idea of the clinical conditions governing this study, some photos of the healing stages of the patients of both groups were taken. All the photos were taken by a digital camera (Sony Cyber-shot DSC- RX10 IV).
The basic evaluation to determine the pain intensity and the condition of the wound of the research units was carried out immediately before the start of the intervention; i.e., early hours after the cesarean section and the intervention begins 10 h after the cesarean section. The evaluation of the pain level on the third, seventh, and tenth days using the VAS visual measurement criteria and the degree of recovery using the REEDA scale on the third, seventh, and tenth days are examined and recorded. VAS visual pain assessment is a scale using points ranging 0–10 and is completed by patients. The REEDA scale was used to measure the healing rate of the cesarean wound, which examines redness, edema, ecchymosis, discharge, and the distance between the two edges of the wound, and gives a score between zero and three, where zero means “absence of variable” and 3 addresses “the presence of the maximum variable value”. REEDA’s scoring criteria are tabulated in Table 3. After summing up the scores, their classification is as follows: a score of zero means “improvement”, a score of 1–5 indicates “moderate improvement”, score of 6–10 indicates “weak recovery”, and number 11–15 declares no recovery. After collecting the required data, the results are analyzed using SPSS software and independent T and χ square statistical tests.
Results and discussion
During this study, none of the subjects left the study due to exclusion criteria, and all 80 people (40 in the control group and 40 in the intervention group) helped us fulfill the study.
Cesarean section examination of patients referred to Sarem obstetrics and gynecology hospital in Tehran shows that on the first day after delivery, none of the variables were redness, edema, and bruising, and the distance between the two edges of the wound and discharge was not significantly different between the two groups. It means that the basal wound of both groups was approximately the same, and in the two groups of cesarean wounds, there was no difference in terms of redness, edema, ecchymosis (bruising), discharge, and the distance between the two edges of the wound. This makes it easier for us to continue, because we have tried as much as possible to keep the two groups in the same conditions.
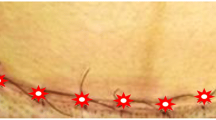
On the third day of the study, the two groups studied the variables of redness, edema, ecchymosis, and discharge; the distance between the two edges of the wound demonstrates a significant difference; and in all cases, the intervention group shows better conditions. Figure 3 depicts two pictures of a cesarean section of two patients of the two groups of interest on the third day.
On the seventh day after cesarean section, the situation was the same for the variables. The two groups have a significant difference with each other, and the wound healing process of the intervention group is clearly scaled up, indicating an enhancement of the healing process of cesarean section. Figure 4 illustrates two images of the cesarean section of two patients from the two groups on the seventh day.
On the tenth day (the last day) of follow-up on the control and intervention groups, the conditions evidenced again in favor of the intervention group. There was a significant difference between the intervention and control groups over all variables of redness, edema, ecchymosis, discharge, and wound edge distance. Thus, the intervention group lucidly healed faster than the control group. On the tenth day after delivery, their wounds reached the desired level of healing. Figure 5 displays a couple photos of a cesarean section of two patients from the two groups of interest on the tenth day.
Comparison of the total scores obtained from 5 variables of REEDA scale (redness, edema, ecchymosis, discharge, and distance between the two edges of the wound) showed that the comparison of mean REEDA score on the first day after delivery was not significantly different between the two groups (p = 0.126). Comparison of mean REEDA scores on the third day (p = 0.035), seventh day (p = 0.03), and tenth day (p = 0.02) after delivery exhibits that the two groups had a statistically significant difference with each other. The mean wound healing score in the intervention group was almost half of the mean wound healing score on the tenth day in the control group (1.09 vs. 2.25). Therefore, after 10 days, the intervention group shows an improvement as much as twice against the control group, and this is a satisfactory result. Tables 4 and 5 tabulate the comparison of the mean scores of the five variables of the REEDA scale on follow-up days, after delivery, by group, and compare the mean score of cesarean wound healing on the follow-up days by group, respectively. Figure 6 illustrates redness, edema, ecchymosis, discharge, and apposition of the wound edges during the follow-up of treatment days. The mean pain intensity in the first days after delivery is not much different between the control and intervention groups (up to the first three days), but in the following days, the pain intensity decreases more rapidly in the intervention group, which are presented in Table 6.
Figure 7 depicts the percentage of cesarean section basal wound healing in the intervention/control groups, which is obtained according to the average of REEDA. As can be seen, the moderate recovery in the intervention group is ~ 23%, poor recovery ~ 50%, and non-recovery ~ 27%. On the other hand, the percentage of cesarean section basal wound healing in the control group is based on REEDA’s average. In this group, the moderate recovery is ~ 40%, poor recovery is ~ 40%, and non-recovery is ~ 20%. The graphs emphasize that there is no significant difference in recovery status between intervention and control groups for the basal wound.
Figure 8 represents the percentage of cesarean section healing after 10 days in the intervention and control groups. These graphs are based on REEDA averages too. In the intervention group, the percentage of perfect recovery is ~ 43%, moderate recovery is ~ 53%, and poor recovery is ~ 4%. In the control group, these percentages are 28%, 60%, and 12%, respectively. These findings indicate that the rate of perfect recovery in the intervention group is lucidly higher than the control group, whereas the average recovery rate in the two groups is almost the same. Furthermore, the poor recovery in the control group demonstrates to be greater than that of the intervention group.
Conclusion
Patients always complain of permanent scars due to abdominal cesarean sections alongside the recovery elongation and the beauty failure. Hence, one makes attempt to find the ways to palliate the pain during the recovery as well as to efface the trace of suture at the end of treatment. Here, the aim is to use laser treatment based on PBM to biostimulate the tissues surrounding the section giving rise to the fading of surgical scars, reduction of pain, rapid healing of cesarean wound, and reduction of discharge. In fact, PBM enhances the activity of leukocytes and macrophages as well as stimulating collagen synthesis and angiogenesis. In addition to the short-term positive results, the aforementioned advantages reduce the use of drug uptake such as painkillers and antibiotics, to level down the side effects of medication in the long term. Not only the fading of the cesarean scar affects the patient’s consent, but also notably decreases the time and financial costs of cosmetic surgeries prescriptions aftermath. Here, in the course of the clinical study, according to REEDA and VAS criteria, PBM enhances the recovery success up to 15% for cesarean wounds 10 days after the operation against the patients who did not receive the laser treatment. Eventually, this clinical research gives us an insight to examine the larger statistical population to validate PBM as an effective post-cesarean treatment. Comparison of the total scores obtained from 5 variables of REEDA scale (redness, edema, ecchymosis, discharge, and distance between the two edges of the wound) showed that the mean REEDA score on the first day after delivery was not significantly different between the two groups (p = 0.126). Discrepancy of mean REEDA scores on the third day (p = 0.035), seventh day (p = 0.03), and tenth day (p = 0.02) after delivery elucidates that the two groups demonstrate a statistically significant difference with each other. The mean wound healing score in the intervention group was almost half of the mean wound healing score on the tenth day in the control group (1.09 vs. 2.25) which indicates the rapid recovery under laser therapy. Furthermore, regarding the statistical populations of intervention and control, around 17% of the patient population exhibits to be far of the average data. Thus, those are taken as out of range and to be improper for true statistical analysis. As a consequence, we come to conclusion that REEDA suffers from 17% systematic error. According to the statistical analysis given in this work, the competence of PBM process in hospitals and medical centers is well verified.
Data Availability
The corresponding author is able to send the data to the reader according to his/her request.
References
Mokmeli S, Khazemikho N, Niromanesh S, Vatankhah Z (2009) The application of low-level laser therapy after cesarean section does not compromise blood prolactin levels and lactation status. Photomed Laser Surg 27(3):509–512
de Holanda AAMP, de Sena KRR, da Silva FEM, Pegado R, Micussi MTABC (2020) Low-level laser therapy improves pain in postcesarean section: a randomized clinical trial. Lasers Med Sci 35(5):1095–1102
Poursalehan S, Nesioonpour S, Akhondzadeh R, Mokmeli S (2018) The effect of low-level laser on postoperative pain after elective cesarean section. Anesth Pain Med 8(6):e84195
Hamblin M, Demidova T (2006) Mechanisms of low level light therapy. Proc of SPIE 6140:614001
Hamblin M (2016) Photobiomodulation or low-level laser therapy. J Biophotonics 9(11–12):1122–1124
Parvin P, Eftekharnoori S, Dehghanpour HR (2009) Monte Carlo simulation of photon densities inside the dermis in LLLT (Low Level Laser Therapy). Opt Spectrosc 107(3):489
Nakashima T, Ueda H, Misawa H, Suzuki T, Tominaga M, Ito A, Numata S, Kasai S, Asahi K, Vernon JA, Meikle MB (2002) Transmeatal low-power laser irradiation for tinnitus. Otol Neurotol 23(3):296–300
Rocha Júnior AM, Vieira BJ, Andrade LCFd, Aarestrup FM (2007) Effects of low-level laser therapy on the progress of wound healing in humans: the contribution of in vitro and in vivo experimentalstudies. J Vasc Bras 6(3):257–265
Hopkins JT, Mcloda TY, Seegmiller JG, Baxter GD (2004) Low level laser therapy facilitates super ficial wound healing in human: a tripl -blind, sham controlled study. J Athl Train 39(3):223–229
Simunovic Z, Ivankovich AD, Depolo A (2000) Wound healing of animal and human body sport and traffic accident injuries using low-level laser therapy treatment: a randomized clinical study of seventy-four patients with control group. J Clin Laser Med Surg 18(2):67–73
Al-Watban FAH, Zhang XY, Andres BL (2007) Low-level laser therapy enhances wound healing in diabetic rats: a comparison of different lasers. Photomed Laser Surg 25(2):72–77
Bisht D, Gupta SC, Misra V (1994) Effect of low intensity laser radiation on healing of open skin wounds in rats. Indian J Mec Res 100:43–46
Pereira AN, Eduardo CDP, Matsone E, Marques MM (2002) Effect of low _ power laserirradiation on cell growth & procollagen synthesis of cultured fibroblast. Laser Surg & Med 31:263–267
Demir H, Balay H, Kirnap M (2004) A comparative study of the effects of electrical stimulation & laser treatment on experimental wound healing in rats. J Rehabil Res Dev 41:147–154
Bayat M, Azari A, Golmohammadi MG (2010) Effects of 780-nm low-level laser therapy with a pulsed gallium aluminum arsenide laser on the healing of surgically induced open skin wound of rat. Photomed Laser Surg 28(4):465–470
Reddy G (2003) Comparison of the photostimulatory effects of visible He-Ne and infrared Ga-As lasers on healing impaired diabetic rat wounds. Lasers Surg Med 33(5):344–351
Stadler I, Lanzafame RJ, Evans R, Narayan V, Dailey B, Buehner N, Naim JO (2001) 830nm irradiation increases the wound tensile strength in a diabetic murine model. Lasers Surg Med 28(3):220–226
Abergel RP, Lyons RF, White RA, Dwyer RM, Castel JC, Uitto J (1987) Biostimulation of wound healing in vivo by a helium-neon laser. Ann Plast Surg 18:47–50
Yasukawa A, Hrui H, Koyama Y, Nagai M, Takakuda K (2007) The effect of low reactive-level laser therapy (LLLT) with helium-neon laser on operative wound healing in a rat model. J Vet Med Sci 69(8):799–806
Hosseini Sanati M, Torkaman G, Hedayati M, Mokhtari M (2010) Effect of Ga-As (904nm) and He- Ne (632 8nm) laser on the improvement of biomechanical characteristics recovery in full Thickness wound. Lasers Med 7(1):6–13
Carvalho PdTCd, Mazzer N, Reis FAd, Belchior ACG, Silva IS (2006) Analysis of the influence of low-power HeNe laser on the healing of skin wounds in diabetic and non-diabetic rats. Acta Cir Bras 21(3):177–183
Ilkeuchi S, Ohsaka F, Asanami A, Nomoto T (1989) Effects of low power He-Ne laser on the healing of full-thickness skin defects. Laser Dent 2:85–89
Moulin V, Auger FA, Garrel D, Germain L (2000) Role of wound healing myofibroblasts on reepithelialization of human skin. Burns 26:2–12
Pereira AN, Eduardo Cde P, Matson E, Marques MM (2002) Effect of low-power laser irradiation on cell growth and procollagen synthesis of cultured fibroblasts. Lasers Surg Med 31:263–267
Young S, Bolton P, Dyson M, Harvey W, Diamantopoulos C (1989) Macrophage responsiveness to light therapy. Lasers Surg Med 9:497–505
Fujimaki Y, Shimoyama T, Liu Q, Umeda T, Nakaji S, Sugawara K (2003) Low-level laser irradiation attenuates production of reactive oxygen species by human neutrophils. J Clin Laser Med Surg 21:165–170
Chen YS, Hsu SF, Chiu CW, Lin JG, Chen CT, Yao CH (2005) Effect of low-power pulsed laser on peripheral nerve regeneration in rats. Microsurgery 25:83–89
Miloro M, Halkias LE, Mallery S, Travers S, Rashid RG (2002) Low-level laser effect on neural regeneration in Gore-Tex tubes. Oral Surg. Oral Med. Oral Pathol Oral Radiol Endod 93:27–34
Balaban P, Esenaliev R, Karu T, Kutomkina E, Letokhov V, Oraevsky A, Ovcharenko N (1992) He-Ne laser irradiation of single identified neurons, Lasers Surg. Med 12:329–337
Neimz M (2007) Laser-tissue interactions. Springer, Berlin
Acknowledgements
We are very interested to express our thanks and appreciation for the help of Dr. Afshin Kazerouni and Dr. Elahe Mollasalehi, from the staff of Sarem obstetrics and gynecology hospital, for helpful technical consults. We are very grateful to the expertized nurse Masoumeh Mardani for performing laser therapy and blind assessment of patients.
Author information
Authors and Affiliations
Contributions
Conceptualization: Hamid Reza Deghanpour; Methodology: Hamid Reaz Dehghanpour, Aliakbar Golchini; Clinical affairs: Parvaneh Ganjali, Aliakbar Golchini; Software: Parvaneh Gnajali, Omid Heidari; Validation: Hamid Reza Dehghanpour; Formal analysis: Hamid Reza Dehghanpour, Aliakbar Golchini; Investigation: Hamid Reza Dehghanpour, Aliakbar Golchini; Data curation: Parvaneh Ganjali; writing—original draft preparation: Hamid Reza Dehghanpour; Writing—review and editing: Hamid Reza Dehghanpour, Parviz Parvin, Hossein Eshghifard; Visualization: Hamid Reza Dehghanpour, Parvaneh Ganjali; Supervision, Hamid Reza Dehghanpour; Project administration: Hamid Reza Dehghanpour and Aliakbar Golchini.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix 1

Appendix 2


Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dehghanpour, H.R., Parvin, P., Ganjali, P. et al. Evaluation of photobiomodulation effect on cesarean-sectioned wound healing: a clinical study. Lasers Med Sci 38, 171 (2023). https://doi.org/10.1007/s10103-023-03774-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10103-023-03774-6