Abstract
Delayed wound healing is one of the most challenging complications of diabetes mellitus (DM) in clinical medicine. This study has aimed to evaluate the effects of low-level laser therapy (LLLT) on human skin fibroblasts (HSFs) cultured in a high glucose concentration. HSFs were cultured either in a concentration of physiologic glucose (5.5 mM/l) or high glucose media (11.1 and15 mM/l) for either 1 or 2 weeks after which they were subsequently cultured in either the physiologic glucose or high concentration glucose media during laser irradiation. LLLT was carried out with a helium–neon (He–Ne) laser unit at energy densities of 0.5, 1, and 2 J/cm2, and power density of 0.66 mW/cm2 on 3 consecutive days. HSFs’ viability and proliferation rate were evaluated with the dimethylthiazol-diphenyltetrazolium bromide (MTT) assay. The LLLT at densities of 0.5 and 1 J/cm2 had stimulatory effects on the viability and proliferation rate of HSFs cultured in physiologic glucose (5.5 mM/l) medium compared to their control cultures (p = 0.002 and p = 0.046, respectively). All three doses of 0.5, 1, and 2 J/cm2 had stimulatory effects on the proliferation rate of HSFs cultured in high glucose concentrations when compared to their control cultures (p = 0.042, p = 0.000, and p = 0.000, respectively). This study showed that HSFs originally cultured for 2 weeks in high glucose concentration followed by culture in physiologic glucose during laser irradiation showed enhanced cell viability and proliferation. Thus, LLLT had a stimulatory effect on these HSFs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diabetes mellitus (DM), a metabolic disorder that gives rise to hyperglycemia and dysregulation of carbohydrates, fatty acids, and protein metabolism, is caused by a reduction in the production of insulin or tissue resistance to the effects of insulin [1]. In recent decades, there has been an unexpected rise in the worldwide prevalence of DM and its complications have become an important public health issue [2–4]. An estimated 285 million people were afflicted with DM worldwide in 2010 and estimates project that in 2030, 439 million people will suffer from its complications [5].
Delayed wound healing is one of the most challenging complications of DM in clinical medicine [6, 7]. According to research, collagen biosynthesis deficiency in un-wounded skin [8], decrease in the proliferation rate of fibroblasts [9], delay in connective tissue healing and ultra structural changes in connective tissue contraction as seen in a mesenteric diabetic rat model, delay in the release of insulin growth factors 1 and 2 (IGF I and II), and microangiopathies in diabetic mice are causes for delayed wound healing [10–12]. The effects of DM complications on skin cells have been evaluated by culturing skin cells in media that contain high concentrations of glucose to assess their responses [13–15].
The stimulatory effects of low-level laser therapy (LLLT) on the proliferation rate of keratinocytes and fibroblasts [16–19], and increase in collagen biosynthesis [17] have been investigated. Studies have also investigated the effects of light and LLLT on fibroblasts cultured in media with high glucose concentrations. Vinck et al. [20] irradiated chicken embryonic fibroblasts with green light emitting diodes at an energy density of 0.1 J/cm2 for 3 consecutive days and cultured them in media that contained a high concentration of glucose. In their research, the MTT assay showed that the proliferation rate of the irradiated cells was significantly higher than that of the control group. Houreld and Abrahamse, in three different studies, researched helium–neon (He–Ne) laser irradiation (632.8 nm) at an energy density of 16 J/cm compared with an energy density of 5 J/cm2 on wounded diabetic fibroblast cells and noted a reduction in cell migration, increased apoptosis and DNA damage [21], and decreased IL-6 release [22] and bFGF expression [23]. The power density in these experiments was 3.03 mW/cm2 while the total energy density (dose) were 5 and 16 J/cm2 and the cells were cultured for 24 h in complete media that consisted of an additional 17 mM/l d-glucose [21–23].
The present study sought to evaluate the viability and proliferation rate of human skin fibroblasts (HSFs) initially cultured in media that had a high glucose concentration for either 1 or 2 weeks, followed by culture in media that contained either a physiologic concentration of glucose or high concentrations of glucose during exposure to LLLT. We used a He–Ne laser with a power density of 0.66 mW/cm2 at energy densities of 0.5, 1, and 2 J/cm2 for durations of 757, 1,512, and 3,024 s, respectively.
Materials and methods
Cell cultures
Normal HSFs cell line was purchased from the National Cell Bank of Iran (NCBI) affiliated with Pasteur Institute of Iran. HSFs were derived from a 30-year-old Caucasian female (Designation: HSF-PI 18, NCBI code: C194). The cell line was cultured in Dulbecco’s modified Eagle’s medium (DMEM; Gibco, Big Cabin, OK, USA) supplemented with 10 % fetal bovine serum (FBS; Gibco, USA), 1 % antibiotic–antimycotic solution (Sigma-Aldrich, St. Louis, MO, USA), and l-glutamine (1.16 g/l). The culture was incubated at 37°C in an atmosphere of 5 % CO2 and 95 % air, in 95 % humidity. HSFs were grown in media at glucose concentrations of 5.5 (physiologic concentration), 11.1 and 15 mM/l to determine the effects of hyperglycemia on fibroblasts. The cultured cells were passaged using 0.25 % trypsin in EDTA (Gibco, USA). Additionally, 0.5 ml of a cell solution with a concentration of approximately 105 cell/ml was added to each well of the 24-well plates. Both case and control groups contained six wells. There were four experimental groups (Table 1).
Non-irradiated HSFs were used as control groups for each of the experimental groups 1–4.
In order to study the effects of hyperglycemia on the changes in cell morphology and to determine the most effective hyperglycemic medium on cell proliferation, cells were cultured in media that consisted of three different glucose concentrations (5.5, 11.1, and 15 mM/l) for 3, 7, and 14 days (Table 2).
HSFs between the third and tenth passages were used in the experiments.
Low-level laser therapy
We used an He–Ne laser unit (IR-2000; Iranian Atomic Energy Agency, Tehran, Iran), to apply laser irradation to the total surface of each well at a15-cm irradiation distance, using a 632.8-nm wavelength and 10-mW power output. The beam was expanded by a biconvex lens (f = 6.5 mm, D = 7.5 mm, glass: BKD) and collimated by another biconvex lens (f = 20 mm, D = 22.3 mm). The irradiation was performed at energy densities of 0.5, 1, and 2 J/cm2, output power of 1.5 mW, power density of 0.66 mW/cm2, and 1.7-cm-diameter spot size of the laser which was used on the surface of the culture medium for 3 consecutive days. The irradiation durations were as follows: 757 s for an energy density of 0.5 J/cm2, 1,512 s for 1 J/cm2, and 3,024 s for 2 J/cm2. The laser output was controlled by a calibrated power meter made by the Iranian Atomic Energy Agency, Tehran, Iran [24]. LLLT was conducted 24 h after HSFs were transferred to the plates, once a day for 3 consecutive days.
Proliferation rate and cell viability assay
The proliferation rate and cell viability of the cultured fibroblasts was determined by the dimethylthiazol-diphenyltetrazolium bromide (MTT) assay 24 h after the last laser treatment. This assay detects surviving cultured cells and the degree to which they are activated [20]. In the MTT assay, tetrazolium salt is cleaved by microsomal enzymes (dehydrogenases) of the respiratory chain to form a purple crystallization product during incubation. The resultant product yields an estimate of cellular viability and metabolic activity. The culture supernatant was removed before adding 40 μl of the 5 mg ml−1 MTT solution plus 360 μl of culture medium to yield a final concentration of 0.5 mg/ml. After incubation for 4 h at 37 °C, the reaction was stopped by the addition of 200 μl acidic isopropanol to dissolve the dye crystals. Subsequently, 200 μl each of the control and laser-treated wells were transferred to two wells of a 96-well plate (100 μl/well) after 10 min of incubation in the dark. This resulted in duplication of the samples to enable a more accurate analysis of the results. There were a total of 12 samples in each group for the MTT assay. For quantification, OD was read on an ELISA reader (Anthos 2020 Microplate Readers, Biochrom Ltd., UK) that had a 492-nm filter. We used a 620-nm filter for background reading.
Statistical analysis
Proliferation rates and cell viability assays for each group were performed on 12 samples (6 wells × 2 wells); each assay was performed once. All calculations were done with the Statistical Package for Social Science software (version 16, SPSS Inc.). The significance of the categorical findings was compared with that of the normally distributed variables by the Student t-test. The Mann–Whitney U-test was used for nonparametric data. One-way ANOVA followed by the post hoc Dunnett test compared the experimental groups cultured in different glucose concentrations with parametric data. A p value of less than 0.05 was considered statistically significant. Data were expressed as mean ± standard error of mean (SEM).
Results
Cell morphology
HSFs were well spread and showed typical elongation in the culture medium that had the normal glucose concentration (5.5 mM/l), whilst the HSFs cultured in the hyperglycemic medium (15 mM/l glucose) did not reach confluency during the same period of time (Fig. 1). Biological changes in cell morphology such as elongation and typical spindle shape were clearly visible in laser-treated HSFs at energy densities of 0.5, 1, and 2 J/cm2 (Fig. 2).
Morphology of cultured human skin fibroblasts (HSFs). Original magnification: ×100. a HSFs cultured in medium at physiologic glucose concentration (5.5 mM/l) after laser irradiation for 3 consecutive days at energy density of 0.5 J/cm2. b Non-irradiated HSFs cultured in medium at physiologic glucose concentration (5.5 mM/l). c HSFs cultured in medium at high glucose concentration (15 mM/l) after laser irradiation for 3 consecutive days at an energy density of 1 J/cm2. d Non-irradiated HSFs cultured in medium at a high glucose concentration (15 mM/l)
Proliferation rate and cell viability
The OD values of each studied group as well as the proliferation rate and cellular viability of the case and control groups are shown in Figs. 3, 4 and 5. In group one, the OD values were significantly higher at energy densities of 0.5 (Mann–Whitney U, p = 0.002) and 1 J/cm2 (Mann–Whitney U, p = 0.046) and significantly lower at 2 J/cm2 (Mann–Whitney U, p = 0.011) when compared with their respective control groups (Fig. 3).
Mean ± SEM of normal human skin fibroblast (HSFs) viability and proliferation rate in non-irradiated and laser-treated groups that were cultured in medium at physiologic glucose concentration (5.5 mM/l) as determined by MTT assay (group 1). There were 12 samples in each group. *Statistically significant data (p < 0.05)
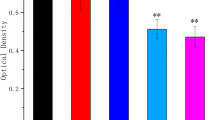
Cellular viability of cultured human skin fibroblasts (HSFs) in three media at different glucose concentrations (5.5, 11.1 and 15 mM/l) for 3, 7 and 14 days. There were 12 samples in each group. *One-way ANOVA test (post-hoc Dunnett test) showed that maintaining HSFs in media at glucose concentrations of 11.1 and 15 mm/l for 3 days significantly decreased the proliferation rate and viability compared to HSFs maintained at 5.5 mM/l (p < 0.05). Cell viability and proliferation rate were significantly lower in medium at the glucose concentration of 11.1 mM/l compared to 5.5 mM/l in the 14-day test. **Viability and proliferation rate of HSFs cultured in medium at the glucose concentration of 15 mm/l for 14 days was significantly lower than the two other media (p < 0.01)
a Mean ± SEM of normal skin fibroblasts (HSFs) viability in non-irradiated and laser-treated groups cultured in medium that had a high glucose concentration (15 mM/l) for 1 week before the LLLT (group 2) as determined by MTT assay. There were 12 samples in each group. Asterisk (*) indicates statistically significant data (p < 0.05). b Mean ± SEM of HSFs viability and proliferation rate in non-irradiated and laser-treated groups initially cultured in medium that had a high glucose concentration (15 mM/l) for both 1 week and during laser irradiation (group 3) as determined by MTT assay. There were 12 samples in each group. *Statistically significant data (p < 0.05). c Mean ± SEM of HSFs viability and proliferation rate in non-irradiated and laser-treated groups initially cultured in medium that had a high glucose concentration (15 mM/l) for 2 weeks, followed by culture in the physiologic concentration during laser irradiation (group 4) as determined by MTT assay. There were 12 samples in each group. *Statistically significant data (p < 0.05)
In the second step, proliferation rate and cell viability of the experimental groups cultured in different glucose concentrations was assessed by the MTT assay (Fig. 4). This assessment showed that the proliferation rate and cell viability of HSFs significantly decreased after 3 days in media with glucose concentrations of 11.1 and 15 mM/l compared to the medium with the normal glucose concentration of 5.5 mM/l (ANOVA test, post hoc Dunnett test, p < 0.05 for both). The lowest cell viability and proliferation rate was seen at the glucose concentration of 15 mM/l after 14 days compared to cells grown in glucose concentrations of 5.5 and 11.1 mM/l (ANOVA test, post hoc Dunnett test, p < 0.01 for both). Cell viability and proliferation rate of the HSFs cultured in the medium with the glucose concentration of 11.1 mM/l was significantly lower than those cultured in the medium with a physiologic glucose concentration (ANOVA test, post hoc Dunnett test, p = 0.003).
In the third part of the current study, HSFs were irradiated after a period of maintenance in hyperglycemic media. The OD values of each group were statistically analyzed (Fig. 5). LLLT at all energy densities of 0.5, 1, and 2 J/cm2 had stimulatory effects on the HSF proliferation rate and viability of group 2 in comparison with its control groups (Student t-test, p = 0.042, p < 0.01, and p < 0.01, respectively; Fig. 5a). On the other hand, only energy densities of 1 and 2 J/cm2 significantly increased the proliferation rate and cellular viability of the HSFs from group 3 (Mann–Whitney U-test, p < 0.01 and p = 0.03, respectively; Fig. 5b). In group 4, the HSFs were significantly activated (proliferated) at energy densities of 0.5 and 1 J/cm2 (Student t-test, p = 0.004 and p = 0.037, respectively; Fig. 5c).
Discussion
In the current study, HSFs grown in glucose concentrations of 11.1 and 15 mM/l for 3 and 15 days to stimulate a diabetic in vitro model showed significant inhibition of cellular viability and proliferation compared to HSFs grown in normal physiologic concentration medium. However there was no significant difference in cellular viability and proliferation rates between HSFs cultured in additional glucose medium and those grown in normal physiologic concentration medium for 7 days. Our results confirmed that concentrations of 11.1 and 15 mM/l glucose were necessary to cause a decline in HSF proliferation. These concentrations resulted in a remarkable reduction in the proliferation rate and cell viability compared to cells grown in 5.5 mM/l glucose. It seemed that the inhibitory effects of elevated glucose concentrations on the proliferation rate and cell viability of HSFs differed based on the different time periods that the cells were grown. This reflected the growth characteristics of HSFs in the current study.
In the present study, HSFs were cultured in a medium that contained a physiologic glucose concentration (5.5 mM/l) and irradiated with a He–Ne laser for 3 consecutive days. Morphological evaluation showed some changes in the laser-treated HSF population. There were more elongated, typically spindle-shaped cells, which suggested that the cells were more activated than the control group. These findings have proven that an appropriate energy density of LLLT can cause morphological changes in HSFs and turn them into activated cells, which is in agreement with the findings from other investigations [24–26]. These findings also indicate an important role for LLLT in wound healing. Activated, spindle-shaped HSFs produce more collagen during wound healing, which can accelerate wound closure and repair [25, 26].
In the current study, the MTT assay showed that LLLT at energy densities of 0.5 and 1 J/cm2 had stimulatory effects, whereas the 2 J/cm2 density showed inhibitory effects on the cellular viability of HSF cells cultured at a physiologic glucose concentration. These findings were similar to other investigations [16, 26, 27]. Grossman et al. studied the possible role of 780 nm low-power laser that emitted 6.5 mW with a focal spot of 0.32 cm2 on cultured normal human keratinocytes (NHK). Although the proliferation rate of NHK exposed to 0.45–0.95 J/cm2 laser was significantly enhanced relative to the sham-irradiated control, exposure to other energy densities was considerably less effective in enhancing the proliferation rate [16]. Medrado et al. [25] researched the effect of a Ga Al As diode emitting light with a wavelength of 670 nm and output of 9 mW on cutaneous wound healing in rats and found that treatment with an energy density of 4 J/cm2 was superior to 8 J/cm2. Hue et al. [27] investigated the underlying mechanisms involved in photostimulatory effects induced by He–Ne laser with energy densities of 0.5, 1, and 2 J/cm2 on A2058 cells in vitro. Their results showed that the effects of 1 J/cm2 were statistically better than those of 2 J/cm2 when using a He–Ne laser that had an output of 10 mW (p < 0.01 vs. p < 0.05) for most experiments.
Although the results of the current study and those of other authors concur that lower levels of laser energy density are more likely to enhance biostimulation [16, 26, 27], the existing literature also contains studies which report different results [24, 28, 29]. Mirzaei et al. [24] have investigated the effects of He–Ne laser irradiation at energy densities of 0.9–4 J/cm2 on cellular changes in cell and organ cultures of the skin of streptozotocin-induced diabetic rats. They showed that 4 J/cm2 irradiation with a power density of 0.66 mW/cm2 significantly increased fibroblast proliferation compared to the sham-exposed cultures [24]. Do Nascimento and Callera [28] investigated the effects of LLLT at different energy densities (0.1–2 J/cm2) on the capacity of long-term cryopreserved peripheral blood progenitor cells (PBPC) in vitro by using a 685-nm diode laser with 25-mW optical power as the irradiation source. These researchers concluded that LLLT at 1 J/cm2 elevated, whereas LLLT at 2 J/cm2 decreased the potential for long-term cryopreserved PBPC to support the growth of CFU in vitro [28]. Prabhu et al. [29] reported the positive effects of a new optical fiber probe-based He–Ne laser for tissue regeneration after exposing full-thickness excision wounds on mice to various laser doses of 1, 2, 3, 4, 6, 8, and 10 J/cm2 with the appropriate 2 J/cm2 control [29]. The wounds exposed to 2 J/cm2 immediately after wounding showed considerable contraction compared with the controls and other treatment schedules, which showed a significant decrease in healing time [29]. From these results [16, 26, 28] and those of the current study, it has been elucidated that energy density determines the effects of laser irradiation and both the stimulation and inhibition of cell properties can be obtained with the same laser.
The in vitro effects of LLLT on normal and wounded skin fibroblasts were examined by a number of investigators [30–35]. Abrahamse et al. investigated the cellular response to He–Ne laser irradiation. The findings of these experiments revealed that doses of 0.5, 2.5, 5, and 10 J/cm2 were sufficient to produce measurable changes in fibroblast cells and a dose of 10 J/cm2 appeared to produce a significant amount of cellular and molecular damage [31]. Thus, the significant stimulatory effect of LLLT on increasing proliferation and viability of fibroblasts as observed in the current study and other studies [30–35] can enhance the wound healing process in vivo [34].
In the third part of the present investigation, LLLT at 0.5, 1, and 2 J/cm2 energy densities was used to irradiate hyperglycemic cultured fibroblasts. This resulted in a significant increase in cellular viability and proliferation. Other studies have used an in vitro diabetic wound model and cultured cells in complete media that contained additional 17 mM/l d-glucose for 24 h [21–23, 36]. Some of the studies irradiated cells with a He–Ne laser at 5 and 16 J/cm2 once a day for 2 non-consecutive days [21], whereas others conducted a single laser irradiation [22]. Other researchers used LLLT once a day on days 1 and 4, allowing for a period of 72 h between irradiations [23], whereas other studies compared LLLT on days 1 and 2 (24 h between irradiations) to LLLT administered on day 1 and day 4 (72 h between irradiations) [35]. LLLT at the energy density of 16 J/cm2 when compared to the energy density of 5 J/cm2 at a power density of 3.03 mW/cm2 reduced cell migration, increased apoptosis and DNA damage [21], and decreased in IL-6 release [22] and bFGF expression [23]. Houreld et al. have concluded that the irradiation interval played an important role in wound healing in vitro [35].
The most striking results of the present study showed that LLLT at energy densities of 0.5, 1 and 2 J/cm2 had significant stimulatory effects on the proliferation rate and cell viability of HSFs initially cultured in medium that consisted of a high glucose concentration for 1 or 2 weeks, followed by culture in medium that had a physiologic glucose concentration during laser irradiation. The best significant stimulatory response was observed in HSFs cultured in high glucose concentration media for 2 weeks followed by growth in normal glucose concentration during laser irradiation at 0.5 J/cm 2. Our results, for the first time, have shown that glycemic control plays an important role in the proliferation and viability of laser–treated HSFs initially grown in medium that has a high concentration of glucose. Clearly, further molecular investigations are required to determine the precise mechanism of the action of LLLT with respect to increased fibroblast proliferation rate and cell viability in a highly concentrated glucose medium.
Conclusion
In light of our results, LLLT with a He–Ne laser at energy densities of 0.5, 1, and 2 J/cm2 increased the proliferation rate and cell viability of HSFs at both physiologic and high glucose concentrations. We found that HSFs initially cultured in a physiologic glucose concentration and maintained at that physiologic glucose concentration during irradiation responded to laser irradiation in a dose-dependent manner. HSFs that were initially cultured in high concentrations of glucose and then maintained at a physiologic glucose concentration during laser irradiation also responded to laser irradiation in a dose-dependent manner. These cells had the best response when irradiated at an energy density of 0.5 J/cm2.
References
Robertson RP (2004) Chronic oxidative stress as a central mechanism for glucose toxicity in pancreatic islet beta cells in diabetes. J Biol Chem 279:42351–42354. doi:10.1074/jbc.R400019200, Epub 2004 Oct 8
Alberti G, Zimmet P, Shaw J, Bloomgarden Z, Kaufman F, Silink M (2004) Type 2 diabetes in the young: the evolving epidemic: the International diabetes federation consensus workshop. Diabetes Care 27:1798–1811. doi:10.2337/diacare.27.7.1798
De Fronzo R, Bonadonna RC, Ferrannini E (1992) Pathogenesis of NIDDM: a balanced overview. Diabetes Care 15:318–368. doi:10.2337/diacare.15.4.508
Groop LC, Widen E, Ferrannini E (1993) Insulin resistance and insulin deficiency in pathogenesis of type 2 (non-insulin dependent) diabetes mellitus: errors of metabolism or of methods ? Diabetologia 36:1326–1331. doi:10.1007/BF00400814
Shaw JE, Sicree RA, Zimmet PZ (2010) Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Care 87:4–14
Goodson WH, Hunt TK (1979) Wound healing and the diabetic patient. Surg Gynerol Obstet 19:600–608
Greenhalgh DG (2003) Wound healing and the diabetes mellitus. Clin Plast Surg 30:37–45. doi:10.1016/S0094-1298(02)00066-4
Kern P, Moczar M, Robert L (1979) Biosynthesis of skin collagens in normal and diabetic mice. Biochem J 102:337–345
Franzen LE, Roberg K (1995) Impaired connective tissue repair in streptozotocin induced diabetes shows ultrastructoral signs of impaired contraction. J Surg Res 58:407–414
Brown DL, Kane CD, Chernausek SD, Greenhalgh DG (1997) Differential expression and localization of insulin-like growth factors l and II in cutaneous wound. Am J Pathol 151:715–724
Algenstaedt P, Schaefer C, Biermann T, Hamann A, Schwarzloh B, Greten H, Rüther W, Hansen-Algenstaedt N (2003) Microvascular alterations in diabetic mice correlate with level of hyperglycemia. Diabetes 52:542–549. doi:10.1016/S0736-0266(03)00060-3
Loots MAM, Lamme EN, Mekkes JR, Bos JD, Middelkoop E (1999) Cultured fibroblasts from chronic diabetic wounds on the lower extremity (non-insulin-dependent diabetes mellitus) show disturbed proliferation. Arch Dermatol Res 291:93–99. doi:10.1007/s004030050389
Benazzoug Y, Borchiellini C, Labat-Robert J, Robert L, Kern P (1998) Effect of high- glucose concentration on the expression collagens and fibronectin by fibroblasts in culture. Exp Gerontol 33:445–455
Yevdokimova NY (2003) High glucose–induced alterations of extracellular matrix of human skin fibroblasts are not dependent on TSP-1–TGF β 1 pathway. J Diabetes Complications 17:355–364. doi:10.1016/S1056-8727(02)00225-8
Deveci M, Gilmont RR, Dunham WR, Mudge BP, Smith DJ, Marcelo CL (2005) Glutathione enhances fibroblast collagen concentration and protects keratinocytes from apoptosis in hyperglycaemic culture. Br J Dermatol 152:217–224. doi:10.1111/j.1365-2133.2004.06329.x
Grossman N, Schneid N, Reuveni H, Holery S, Lubart (1998) 780 nm low-power diode laser irradiation stimulates proliferation of keratinocyte culture, involvement of reactive oxygen species. Lasers Surg Med 22:212–218
Almeida-Lopez L, Rigau J, Zangaro RA, Guidugli-Neto J, Jaeger MM (2001) Comparison of low-level laser therapy effects on cultured human gingival fibroblasts proliferation using different irradiance and same fluence. Lasers Surg Med 29:179–184
Hawkin DH, Abrahamse H (2006) The role of laser fluence a in cell viability, proliferation, and membrane integrity of wounded human skin fibroblasts following helium–neon laser irradiation. Lasers Surg Med 36:74–83. doi:10.1117/12.641172
Evans DH, Abrahamse H (2008) Efficacy of three different laser wavelengths for in vitro wound healing. Photodermol Photoimmunol Photomed 24:199–210
Vinck EM, Gaginie BJ, Cornelissen MJ, Declerecq HA, Cambier DC (2005) Green light emitting diode irradiation enhances fibroblast growth impaired by high glucose level. Photomed Laser Surg 23:167–217. doi:10.1089/pho.2005.23.167
Houreld NN, Abrahamse H (2007) In vitro exposure of wounded diabetic fibroblast cells to a helium–neon laser at 5 and 16 J/cm2. Photomed Laser Surg 25:78–84. doi:10.1089/pho.2007.990
Houreld N, Abrahamse H (2007) Irradiation with a 632.8 nm helium–neon laser with 5 J/cm2 stimulates proliferation and expression of interleukin-6 in diabetic wounded fibroblast cells. Diabetes Technol Ther 9(5):451–459. doi:10.1089/dia.2007.0203
Houreld NN, Abrahamse H (2007) Laser light influence cellular viability and proliferation in diabetic-wounded fibroblast cells in a dose- and wavelength dependent manner. Lasers Med Sci 23(1):11–18. doi:10.1007/s10103-007-0445-y
Mirzaei M, Bayat M, Mosafa N, Mohsenifar Z, Piryaei A, Farokhi B, Rezaei F, Sadeghi Y, Rakhshan M (2007) Effect of low-level laser therapy on skin fibroblasts of streptozotocine-diabetic rats. Photomed Laser Surg 25:517–523. doi:10.1089/pho.2007.2098
Pourreau-Schneider N, Ahmed A, Soudry M, Jacquemier J, Kopp F, Franquin JC, Martin PM (1990) Helium–neon laser treatment transforms fibroblasts into myofibroblasts. Am J Pathol 137:171–178
Medrado AR, Puyliese LS, Reis SR, Andrade ZA (2003) Influence of low level laser therapy on wound healing and its biological action upon myofibroblasts. Lasers Surg Med 32:239–244. doi:10.1002/lsm.10126
Hue WP, Wang JJ, Yu CL, Lan CC, Chen GS, Yu HS (2007) Helium–neon laser irradiation stimulates cell proliferation through photostimulatory effects in mitochondria. J Invest Dermatol 127(8):2048–2057
Do Nascimento RX, Callera F (2006) Low-Level laser therapy at difference energy densities (0.1–2.0 J/cm2) and its effects on the capacity of human long-term cryopreserved peripheral blood progenitor cells for the growth of colony-forming units. Photomed Laser Surg 24:601–604. doi:10.1089/pho.2006.24.601
Prabhu V, Rao SB, Rao NB, Aithal KB, Kumar P, Mahato KK (2010) Development and evaluation of fiber optic probe-based helium–neon low-level laser therapy system for tissue regeneration: an in vivo experimental study. Photochem Photobiol 86:1364–1372. doi:10.1111/j.1751-1097.2010.00791.x
Van Breugel HH, Bar PR (1992) Power density and exposure time of He–Ne laser irradiation are more important than total energy dose in photo-biomodulation of human fibroblasts in vitro. Lasers Surg Med 12:528–537. doi:10.1002/lsm.1900120512
Hawkins DH, Abrahamse H (2006) The role of laser fluence in cell viability, proliferation and membrane integrity of wounded human skin fibroblasts following helium–neon laser irradiation. Lasers Surg Med 38:78–83. doi:10.1002/lsm.20271
Hawkins D, Abrahamse H (2005) Biological effects of helium–neon laser irradiation on normal and wounded human skin fibroblasts. Photomed Laser Surg 23(3):251–259. doi:10.1089/pho.2005.23.251
Hawkins DH, Abrahamse H (2006) Effects of multiple exposures of low-level laser therapy on cellular responses of wounded human skin fibroblasts. Photomed Laser Surg 24:705–714. doi:10.1089/pho.2006.24.705
Karu T (1989) Photobiology of low-power laser effects. Heal Phys 56:691–704. doi:10.1097/00004032-198905000-00015
Dadpay M, Sharifian Z, Bayat M, Bayat M, Dabbagh A (2012) Effect of pulsed infra-red low level laser irradiation on open skin wound healing of healthy and strepzotocin-induced diabetic rats by a biomechanical evaluation. J Photochem Photobiol B 111:1–8, Epub 2012 Mar 16
Houreld NN, Abrahamse H (2007) Effectiveness of helium–neon laser irradiation on viability and cytotoxicity of diabetic-wounded fibroblast cells. Photomed Laser Surg 25(6):474–481. doi:10.1089/pho.2007.1095
Acknowledgments
We thank the late Mrs. Jamileh Rezaei. We also extend our thanks to the Vice Chancellor of Research at Shahid Beheshti University of Medical Sciences, Tehran, Iran, for financial support.
Conflict of interest
No competing financial interests exist.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Esmaeelinejad, M., Bayat, M., Darbandi, H. et al. The effects of low-level laser irradiation on cellular viability and proliferation of human skin fibroblasts cultured in high glucose mediums. Lasers Med Sci 29, 121–129 (2014). https://doi.org/10.1007/s10103-013-1289-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-013-1289-2