Abstract
Our previous in vitro study has shown that toluidine blue (TB)-mediated lethal photosensitization of periodontal pathogens (PPs) from periodontal patients is possible. The purpose of this study was to investigate whether TB-mediated photosensitization exerted damaging effects on periodontal tissues in mice. Twenty-four mice were randomly divided into four groups; the experimental photodynamic therapy (PDT) group was treated with 1 mg/ml TB and light irradiation (60 J/cm2, 635 nm, 337 s). Those in control groups were subjected to 140 J/cm2 laser irradiation alone or to 2.5 mg/ml TB alone or received neither TB nor light exposure. All the mice were killed 72 h after they had been subjected to PDT, and periodontal tissue samples were taken for histological examination. During the 72 h observation period, no mice showed any distress. No necrotic or inflammatory changes were found in the gingiva, dentin, dental pulp or alveolar bone of the mice in any of the groups in this study. The results suggest that TB-mediated PDT is a safe antimicrobial approach for the treatment of periodontopathy without damaging effects to adjacent normal tissues.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Periodontopathy is a common disease caused by bacterial infection in the adult population. It is often accompanied by the destruction of periodontal connective tissue and resorption of alveolar bone. Dental plaque is the major site of pathogens bringing about periodontopathy. If dental plaque increases and piles up, it will lead to gingivitis. Severe gingivitis can even develop into periodontitis.
Conventional mechanical debridement (i.e., scaling and root planing) can achieve a temporary decrease of microorganisms in the dental plaques [1]. However, microorganisms cannot be completely removed from the majority of periodontal pockets by mechanical therapy alone. Antimicrobial chemotherapy may further suppress the periodontal pathogens. Numerous systemic and local antimicrobial chemotherapeutic agents have been evaluated for the treatment of periodontopathy, with various degrees of success [2–5]. A lack of effectiveness of some of the antibiotics used may be due to the development of drug-resistant strains [6, 7]. To overcome the problems caused by the emergence of resistance, alternative antimicrobial approaches need to be developed.
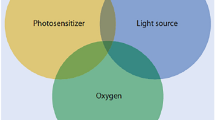
One potential alternative method is photodynamic therapy (PDT), showing great potential for the treatment of neoplastic and non-neoplastic diseases [8]. PDT involves a photosensitizer, light, and molecular oxygen [9]. After excitation with visible light, highly cytotoxic singlet oxygen and other reactive oxygen species (ROS) are generated [9–12].
The antimicrobial effect of PDT has been proven to be effective in killing a wide range of oral pathogens and drug-resistant bacteria. Many organisms that might cause infection in the oral gingiva have been shown to be susceptible to TB-mediated photodynamic action [13–15]. Owing to the localized and non-invasive nature of PDT, the side effects associated with many antibiotics are unlikely to occur. In our previous test, our findings suggested that TB-mediated photoinactivation of periodontal pathogens (PPs) from supragingival plaques is possible. However, for ideal bactericidal effects to be obtained, higher doses of light and photosensitizer would be required for treatment in vivo than their in planktonic counterparts. The best therapeutic effect was observed in treatment with 1 mg/ml TB combined with 12 J/cm2 at 159 mW/cm2 light irradiation [16]. Moreover, it is important for one to verify whether PDT exerts detrimental effects on the host tissues during the photoinactivation of pathogenic oral bacteria. The aim of this study was to determine whether any unfavorable effect was produced during TB-PDT treatment on normal periodontal tissues in mice, including gingiva, dentin, dental pulp and alveolar bone.
Materials and methods
Photosensitizer and laser
We prepared a TB solution by dissolving TB powder (Sinopharm Chemical Reagent, Shanghai, China) in sterile saline solution to give final concentrations of 1 mg/ml and 2.5 mg/ml, sterilized by the use of membrane filters of pore size 0.22 μm, and subsequently kept in the dark. A diode laser (HPD1202, HPD Inc., USA), with a continuous output of 0.26 W and a wavelength of 635 nm, was used to illuminate the mouse gingiva. The light was guided via a fiber-optic applicator with a 0.8 mm cylindrical diffusing tip. Before laser irradiation, the output power from the fiber tip was adjusted to 61 mW, and the irradiation area was adjusted, respectively, to a diameter of 7 mm and 6 mm to obtain 159 mW/cm2 and 212 mW/cm2 light doses. The laser output energy was carefully calibrated with a power meter (PS10, Coherent Inc., USA), and the size of the irradiation area was adjusted by our changing the distance between the fiber tip and the irradiation surface.
Animals
Twenty-four healthy male Kunming mice weighing 18–25 g were used in this study. The mice were kept under regular laboratory feeding and housing conditions.
Photodynamic therapy assay
The mice were anesthetized by inhalation of Fluothane (FCI Pharmaceuticals, Cheshire, UK). To gain easy access, we retracted their mouths with mouth retractors. Their heads were positioned to the left side so that topically applied TB solution would stay in place. The twenty-four mice were randomly divided into four groups, six mice in each. In the experimental PDT group, the mice were given 1 mg/ml TB, topically applied to the left side of the oral gingiva, and were left in the dark for 5 min. Then, they were irradiated (60 J/cm2 , 635 nm, 23 J). The micro-lens optical fiber was fixed to illuminate the gingiva in an area 6 mm in diameter. The control groups were treated as follows: (1) the mice were subjected to laser irradiation of 140 J/cm2; (2) the mice were given TB, or (3) the mice were given neither TB nor light exposure, The test parameters (photosensitizer and light doses used in this study) in each group are shown in Table 1.
Histological examination
Following PDT treatment, the treated area was visually inspected daily during the first 3 days. After the last observation, the mice were killed by cervical dislocation. Specimens of the whole periodontal tissue were fixed immediately in 10% formalin, and, 24 h later, the specimens were rinsed in running water and decalcified in nitric acid for 36–48 h. The sections were embedded in paraffin and stained with hematoxylin and eosin (H&E) for histological evaluation.
Results
During the 72 h observation period, no mice showed any distress. On macroscopic examination, none of the mice in any groups showed discernible changes in the gingiva.
Figure 1 shows an H&E-stained histological section of periodontal tissue treated by PDT (PDT group, 1 mg/ml TB and 60 J/cm2 light exposure), and Figs. 2, 3 and 4 show tissues from the controls: control group 1 (the mice were given laser irradiation alone); control group 2 (the mice received TB); control 3 (the mice were given neither TB nor light exposure). No necrotic or inflammatory changes were observed in the gingiva, dentin, dental pulp or alveolar bone of any of the mice in the four groups. The epithelium, connective tissue and bone structures were intact and remained microscopically unchanged. No distinct histological differences were detected between those four groups.
In brief, in all groups, no any histological changes, such as inflammatory cell infiltration, necrosis or damage to blood vessels, were observed in the periodontal soft tissues. What is more, no tissue damage was found in the dentin, dental pulp or alveolar bone.
Discussion
Periodontopathy is a common oral disease caused by dental plaque and involves the major pathogens. Conventional mechanical debridement can temporarily remove dental plaques, and antimicrobial chemotherapy may further suppress the periodontal pathogens [1]. However, the efficiency of those conventional treatments may not be ideal in some cases. Scaling and root planing are difficult to carry out in some sites such as furcations, deep invaginations and concavities. Administration of antibiotics, especially the systemic administration of antibiotics, may engender considerable side effects. Moreover, the growing bacterial resistance to antibiotics may further influence the efficacy of conventional therapy. To overcome these problems, alternative therapeutic modalities are greatly required.
PDT is a newly developed therapeutic technique in dentistry. It involves a photosensitizer, light of a specific wavelength and oxygen. During the PDT process, the photosensitizer is excited by the light in the presence of oxygen. Cytotoxic singlet oxygen, and other reactive oxygen species, are generated, that actively destroy unwanted target cells. PDT may provide a new modality in the management of various malignant and benign diseases, among others, periodontal disease. PDT has several unique properties, such as a non-invasive nature, high selectivity, ease of reaching deeply situated sites, repeatability and no resistance to drugs.
It has been demonstrated that PDT is effective in killing periodontal pathogens such as Porphyromonas gingivalis, Actinobacillus actinomycetemcomitans, Fusobacterium nucleatum, Prevotella intermedia and Streptococcus sanguis [17–20]. Among the many photosensitizers, TB, a cationic phenothiazine [1, 21], has been shown to be effective against many microorganisms, as well as their virulence factors, in vitro [2, 22–25]. Many periodontal microorganisms have been shown to be susceptible to TB-mediated PDT [12–14]. The effect of this therapeutic modality in treating PPs greatly depends on photosensitizer concentration, light intensity and light energy dose. It was reported that 100% of P. gingivalis can be killed by 25 μg/ml of TB and 4.4 J/cm2 of red light irradiation [26]. However, those studies were carried out on pure cultures of bacteria strains. In fact, the causative agents of periodontopathy in vivo are presented as an ecosystem that includes hundreds of species of bacteria. Our previous study in vitro has shown that it is possible to achieve photoinactivation of periodontal bacterial flora taken from patients, but greater photosensitizer and light doses were needed [16].
Prior to clinical application, it is important for one to determine whether the increased photosensitizer concentrations and light doses would cause damage to host oral tissues. In our previous test, the best therapeutic effect was observed in treatment with 1 mg/ml TB combined with 12 J/cm2 at 159 mW/cm2 light irradiation. In this study, in order to review the safety of TB -mediated photosensitization to periodontal tissues in mice, the we increased the density of TB to 2.5 mg/ml and doses of light to 60 J/cm2;, those are much higher than in the optimal protocol in our previous experiment.
There have been several reports that PDT might kill pathogenic bacteria without damaging normal oral cells and tissues. In an in vitro study, using up to 2 μg/ml or 5 μg/ml TB in conjunction with 67.4 J/cm2 of light, there was no reduction in cell viability of human keratinocytes or fibroblasts, respectively, whereas 42.1 J/cm2 of light in the presence of 2.5 μg/ml TB was sufficient to kill Streptococcus sanguis [27]. The buccal mucosa of rats showed no necrotic or inflammatory changes after treatment with up to 200 μg/ml TB and 340 J/cm2 red light irradiation [28]. The present study demonstrated that, in comparison with all the mice in the three control groups (mice without any treatment, mice treated with 140 J/cm2 light irradiation alone, and mice treated with 2.5 mg/ml TB alone), the TB-PDT treated mice (1 mg/ml TB and 60 J/cm2 light irradiation from a 635 nm diode laser, much higher than the bacteria-killing doses) did not show any histological damages to the gingival anatomy, dentin, dental pulp or alveolar bone. The results of this study are encouraging, as even much higher doses of TB (to 2.5 mg/ml) and light (to 140 J/cm2) than those required for the eradication of microorganisms in vitro did not lead to any detectable adverse effects on the normal mice periodontal tissue. These results also further confirmed the safety of topically applied TB-mediated PDT and supported its potential application in the treatment of periodontopathy.
There are some reports in the literature that PDT may cause tissue damage in the oral cavity. Gingival ulceration in rabbits, following the systemic administration of 5 mg/kg disulfonated phthalocyanine and 20 J/cm2 of light exposure (675 nm), was observed by Meyer et al. [29]. Vesicles and edema were caused by treatment with a hematoporphyrin derivative at 20 mg/kg in combination with light illumination at 90 J/cm2 or 180 J/cm2 [30]. However, it should be pointed out that those two studies were carried out under very different conditions, in comparison with our study. Their photosensitizers were administered systematically, the light doses were much higher than in our study, and the light exposure was given at many hours after drug administration. Therefore, those two reports should not argue with the results of our study, lead to a confusing conclusion, or deny the safety of topically applied TB-mediated PDT in the treatment of periodontopathy.
Conclusion
The results of our study indicated that TB-mediated PDT (within the concentrations and light doses used in this study) could be an encouraging antimicrobial approach in the clinical treatment of periodontopathy, and they deserves further investigation.
References
Sbordone L, Ramaglia L, Gulletta E, Iacono V (1990) Recolonization of the subgingival microflora after scaling and root planing in human periodontitis. J Periodontol 61:579–584
Chaves ES, Jeffcoat MK, Ryerson CC, Snyder B (2000) Persistent bacterial colonization of Porphyromonas gingivalis, Prevotella intermedia, and Actinobacillus actinomycetemcomitans in periodontitis and its association with alveolar bone loss after 6 months of therapy. J Clin Periodontol 27:897–903
Loesche WJ, Syed SA, Morrison EC, Kerry GA, Higgins T, Stoll J (1984) Metronidazole in periodontitis. I. Clinical and bacteriological results after 15 to 30 weeks. J Periodontol 55:325–335
Renvert S, Dahlen G, Wikstrom M (1996) Treatment of periodontal disease based on microbiological diagnosis. Relation between microbiological and clinical parameters during 5 years. J Periodontol 67:562–571
Stabholz A, Nicholas AA, Zimmerman GJ, Wikesjo UM (1998) Clinical and antimicrobial effects of a single episode of subgingival irrigation with tetracycline HCl or chlorhexidine in deep periodontal pockets. J Clin Periodontol 25:794–800
Feres M, Haffajee AD, Goncalves C et al (1999) Systemic doxycycline administration in the treatment of periodontal infections. II. Effect on antibiotic resistance of subgingival species. J Clin Periodontol 26:784–792
Olsvik B, Tenover FC (1993) Tetracycline resistance in periodontal pathogens. Clin Infect Dis 16[Suppl 4]:S310–S313
Dougherty TJ (2000) An update on photodynamic therapy applications. J Clin Laser Med Surg 20:3–7
Ochsner M (1997) Photophysical and photobiological processes in the photodynamic therapy of tumours. J Photochem Photobiol B 39:1–18
Oleinick NL, Evans HH (1998) The photobiology of photodynamic therapy: cellular targets and mechanisms. Radiat Res 150[5 Suppl]:S146–S156
Zhou CN (1989) Mechanisms of tumor necrosis induced by photodynamic therapy. J Photochem Photobiol B 3:299–318
Moan J, Berg K (1991) The photodegradation of porphyrins in cells can be used to estimate the lifetime of singlet oxygen. Photochem Photobiol 53:549–553
Millson CE, Wilson M, MacRobert AJ, Bown SG (1996) Ex-vivo treatment of gastric Helicobacter infection by photodynamic therapy. J Photochem Photobiol B Biol 32:59–65
Canete M, Villanueva A, Juarranz A (1993) Uptake and photo effectiveness of two thiazines in HeLa cells. Anticancer Drug Des 8:471–477
Darzynkiewicz Z, Carter SP (1988) Photosensitizing effects of the tricyclic heteroaromatic cationic dyes pyronin Y and toluidine blue O (tolonium chloride). Cancer Res 48:1295–1299
Qin Y, Luan X, Bi L et al (2007) Toluidine-blue-mediated photoinactivation of periodontal pathogens from supragingival plaques. Lasers Med Sci DOI 10.1007/s10103-007-0454-x
Kömerik N, Nakanishi H, MacRobert AJ (2003) In vivo killing of Porphyromonas gingivalis by toluidine blue-mediated photosensitization in an animal model. Antimicrob Agents Chemother 47(3):932–940
Chan Y, Lai CH (2003) Bactericidal effects of different laser wavelengths on periodontopathic germs in photodynamic therapy. Lasers Med Sci 18:51–55
Wilson M, Burns T, Pratten J, Pearson GJ (1995) Bacteria in supragingival plaque samples can be killed by low-power laser light in the presence of a photosensitizer. J Appl Bacteriol 78:569–574
Wilson M, Sarkar S, Bulman J (1994) Effect of blood on the lethal photosensitization of bacteria in subgingival plaque from patients with chronic periodontitis. Lasers Med Sci 8:297–303
Klausen B, Evans RT, Sfintescu C (1989) Two complementary methods of assessing periodontal bone level in rats. J Dent Res 97:494–499
Jackson Z, Meghji S, MacRobert A, Henderson B, Wilson M (1999) Killing of the yeast and hyphal forms of Candida albicans using a light-activated antimicrobial agent. Lasers Med Sci 14:150–157
Wilson M, Dobson J, Harvey W (1992) Sensitisation of oral bacteria to killing by low power laser radiation. Curr Microbiol 25:77–81
Wilson M, Yianni C (1995) Killing of methicillin-resistant Staphylococcus aureus by low-power laser light. J Med Microbiol 42:62–66
Komerik N, Wilson M, Poole S (2000) The effect of photodynamic action on two virulence factors of gram-negative bacteria. Photochem Photobiol 72:676–680
Bhatti M, MacRobert A, Meghji S, Henderson B, Wilson M (1997) Effect of dosimetric and physiological factors on the lethal photosensitization of Porphyromonas gingivalis in vitro. Photochem Photobiol 65:1026–1031
Soukos N, Wilson M, Burns T, Speight PM (1996) Photodynamic effects of toluidine blue on human oral keratinocytes and fibroblasts and Streptococcus sanguis evaluated in vitro. Lasers Surg Med 18:253–259
Kömerik N, Curnow A, MacRobert AJ, Hopper C, Speight PM, Wilson M (2002) Fluorescence biodistribution and photosensitising activity of toluidine blue O on rat buccal mucosa. Lasers Med Sci 176:86–92
Meyer M, Speight P, Bown SG (1991) A study of the effects of photodynamic therapy on the tissues of the rabbit jaw. Br J Cancer 64:1093–1097
Pe MB, Sano K, Inokuchi T (1993) Effects of photodynamic therapy in the normal mouse tongue. J Oral Maxillofac Surg 51:1129–1134
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luan, X.L., Qin, Y.L., Bi, L.J. et al. Histological evaluation of the safety of toluidine blue-mediated photosensitization to periodontal tissues in mice. Lasers Med Sci 24, 162–166 (2009). https://doi.org/10.1007/s10103-007-0513-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-007-0513-3