Abstract
Group A Streptococcus (GAS) is one of the most important agents of oropharyngeal infection. To avoid unnecessary antibiotic prescription, it is recommended the confirmation of GAS infection in pharyngeal swabs using culture or rapid antigen detection test (RADT). This study aimed to retrospectively analyse the incidence of GAS oropharyngeal infection, detected by RADT, in a paediatric population in the Centre of Portugal. Data was collected from the database of the Paediatric Hospital Emergency Department (ED) regarding patients admitted with symptoms suggesting acute pharyngitis, from January 2013 to December 2018, in a total of 18,304 cases. Among these, 130 clinical files were searched for symptoms, complications and additional visits to the ED. The results showed an average GAS infection prevalence of 33%, with seasonal variation. In preschool children, especially in patients less than 3 years old, where the guidelines do not routinely encourage RADT, GAS tonsillitis assumed an unexpected importance, with 731 positive tests in a total of 3128 cases. Scarlatiniform rash and oral cavity petechiae had significant correlation with streptococcal aetiology (p < 0.05). The statistical analysis also showed that different signs and symptoms assume different weights depending on the age group of the patient. The main conclusion is that the incidence of GAS infection in the studied population is higher than generally described in preschool children, suggesting the need for a more cautious approach to children under 3 years presenting acute pharyngitis, and that RADT in this age group would contribute to a decrease in the number of unnoticed cases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The term “tonsillitis” is used to indicate the presence of inflammation in the lymphoid tissue that lies between the palatoglossal and palatopharyngeal arches of the oral cavity (palatine tonsils). Since inflammation generally progresses to and associates with Waldeyer’s ring pathology, the terms “pharyngitis” and “adenotonsillitis” are often considered equivalent [1]. Being one of the most common causes of fever in children, it is one of the main reasons for seeking medical care [2, 3] and for school or work absenteeism (with an estimated cost ranging from US $224 million to US $539 million per year in the United States of America) [4].
Acute pharyngeal infections can be caused by a wide variety of microbial agents, mainly with viral aetiology (40%), with a benign and self-limited evolution, with bacteria accounting for about 30% of these infections. The remaining cases have no identified causal agents associated [2, 5]. Among bacteria, group A Streptococcus (GAS) is the most frequent [5,6,7] with an estimated 616 million new cases of GAS tonsillitis per year worldwide [8]; GAS is responsible for 15–30% of acute tonsillitis in children and 5 to 10% in adults [6], with the exception of very rare bacterial agents, including Corynebacterium diphtheriae, Neisseria gonorrhoeae and Arcanobacterium haemolyticum [37]. In the case of GAS infection, antimicrobial treatment has proven benefits: faster symptomatic relief, decreased risk of transmission and, most importantly, prevention of suppurative (cervical lymphadenitis, retropharyngeal or peritonsillar abscess, sinusitis, otitis media and mastoiditis) and non-suppurative complications (particularly acute rheumatic fever, if treatment is started within 9 days after the onset of symptoms) [6, 7, 9].
There is no international consensus on the most appropriate method to address acute tonsillitis, and standard clinical practice varies across countries. Most guidelines that recommend antibiotic treatment also encourage confirming GAS presence through the collection of pharyngeal exudate [10], avoiding unnecessary prescriptions and preventing possible disease complications. However, in countries such as the UK, Belgium or Netherlands, oropharyngeal swabs are not recommended. This maybe justified by different epidemiological issues (e.g. the prevalence of rheumatic fever), what Cochrane named as “clinical traditions”, different health systems and policies or the lack of reliable data on the incidence of complications and on the effectiveness of antibiotics in their prevention [1, 10].
In Portugal, the clinical guidelines (CG) 020/2012 issued by the health authorities recommend, with some exceptions, the aetiological screening for tonsillitis in paediatric patients aged 3 or over. Since there is a clinical similarity between streptococcal and non-streptococcal tonsillitis, and since the use of clinical scores is not recommended in children [11, 12], the recommendation is to confirm the presence of GAS by rapid antigen diagnostic test (RADT), and/or bacteriological culture. Cases in which clinical and epidemiological characteristics strongly suggest a viral aetiology—cough, rhinorrhea, hoarseness, diarrhoea, conjunctivitis and oral ulcers [7, 13]—are dismissed from these procedures.
The vast majority of RADTs, besides providing almost immediate results (minute-result period contrasting with the 18–48 h required for culture), have high specificity (> 95%) when compared to culture (reference method), promoting the confidence of the clinician to trust in a positive result. However, the sensitivity and specificity of these tests vary from 66 to 95% between studies [1, 7, 14, 15], meaning that a negative test result might prompt the clinician to ask for a verification culture [7].
According to the described, this study aimed, first and foremost, to characterise the oropharyngeal streptococcal infection, diagnosed by RADT, in children admitted to the Emergency Department of the paediatric hospital of Centro Hospitalar e Universitário de Coimbra (CHUC), in the central region of Portugal, during a 6-year period and to assess the adequacy of the local clinical practice with the clinical guidelines and worldwide epidemiology.
Materials and methods
Following ethical approval by the Ethics Committees, we analysed the data available in the paediatric hospital at the Centro Hospitalar e Universitário de Coimbra (CHUC) database of all RADTs performed from throat swabs in patients admitted to the Emergency Department (ED) of this hospital from 1 January, 2013, to 31 December, 2018 in a total of 18,336 cases. From these, only 18,304 cases were considered for this study because patients older than 18 years were excluded (n = 6), and tests with an inconclusive result were also excluded (n = 26). During the period in study, the RADT used for the qualitative detection of streptococcal antigens in samples from oropharynx swabs was the rapid immunochromatographic test BIOSYNEX® Strep A (Fumouze Diagnostics, France), with sensitivity and specificity (both related to culture) of, respectively, 94.4% (95% CI: 88.7–97.7%) and 97.3% (95% CI: 94.6–98.0%).
The tests carried out on adults (18 years or older), and the tests whose results were considered inconclusive were excluded. In total, the study was based on the information of 18,304 patients who performed rapid antigenic tests; parameters such as sex, age, harvest date and result of the RADT were taken into account in the descriptive statistical analysis.
Additionally, a group of 130 clinical files were consulted and thoroughly analysed to detect symptomatic patterns and additional visits to the ED; this group included patients between 2 and 10 years old (the age group with the largest number of streptococcal tonsillitis cases), and the analysis focused on both the signs and the symptoms presented as well as on their possible predictive value for detecting tonsillitis aetiology. Among these, 3 were excluded due to insufficient records, 79 had RADT-positive result and 48 were RADT-negative. A subgroup of 40 clinical files of patients under the age of 3 was also analysed—a group where the search for GAS is not routinely recommended—in order to verify the criteria for excluding the use of RADT.
The data obtained was treated using Microsoft® Excel® (Microsoft Office 365 ProPlus, version 16.0.11328.20390) and, when possible, compared with the existing literature. Linear regression tests (least squares method) were used to assess the possibility of associating symptoms and aetiology, with a p value < 0.05 being considered statistically significant.
Results
Characterisation of RADT results
This study included the RADT results of 18,304 patients, with a mean age of 6.29 ± 4.20 years (0–17 years), with 52% (n = 9445) of males and 48% (n = 8859) females. The annual percentage of bacterial aetiology cases ranged from a minimum of 27% in 2018 to a maximum of 39% in 2017 (Fig. 1), with an average GAS prevalence of 33% (6038 positive tests), with no differences between gender. In streptococcal tonsillitis cases, the mean age was 6.24 ± 3.74 years. At the hospital under study and during the period considered, the registration of RADT increased constantly every year, with the exception of 2015.
Rapid antigenic diagnostic tests (RADT) performed per year (2013–2018) at the paediatric hospital of CHUC, at the central region of Portugal. The results, deposited at the hospital database, represent the number of patients attended during the considered period, with a suspicion of streptococcal pharyngitis and that made RADT, in a total of 18,304 clinical cases. The bars represent the absolute number of RADT performed and the positive results per year; the line represents the percentage (%) of positive RADT results
Among the tested children, 15,176 (83%) were between 3 (inclusive) and 18 years old (exclusive); 3128 (17%) were younger (Fig. 2), corresponding to the age group for which the Portuguese clinical guidelines (CG) do not advise GAS test by default. The peak incidence of both acute tonsillitis (2502 RADT performed) and the highest number of streptococcal infection cases (863 positive tests) were registered in children aged 3 years old. The minimum number of tests performed was associated with 15-year-old patients (n = 379), and the minimum number of streptococcal tonsillitis (n = 86) was registered in patients aged 16 (Fig. 2). All age groups had a higher frequency of non-streptococcal aetiology, but in children with 4 to 10 and with 12 years, the GAS prevalence exceeded 35%, reaching its maximum in 9 years old patients (40%).
The analysis of the GAS prevalence in preschool children (≤ 5 years; Table 1) showed that although a rare pathology in children under 6 months (only 3 positive RADT in the 6-year period considered for this study), streptococcal tonsillitis assumes unexpected importance in patients younger than 3 years old; in this age group, where the CG does not routinely encourage the performance of RADT, 731 positive tests were registered in a total of 3128 clinical cases (23%).
Since the incidence of this infection is considered to be influenced by seasonal cycles, we looked at the variation according to the month. It was found that June and September were the months with, respectively, the highest (n = 1887) and lowest (n = 965) number of RADT performed. December was the month where more tonsillitis cases had GAS aetiology (n = 661) and August the month with the lower number of positive tests (n = 269). In May, June, October, November and December, the GAS prevalence rates were above 35%, with the maximum value occurring in November (39%). August was the month with the lowest percentage of streptococcal tonsillitis (25%) (Fig. 3).
The higher incidence of GAS infection was registered during spring and autumn, when, in Portugal, traditionally milder temperatures are registered. This was registered in all the years included in the study (Fig. 4a and b). Interestingly, the percentage of positive RADT peaked in May 2017 (49%, maximum GAS prevalence recorded in this study) and sharply declined in August every year. Illustrative of this tendency is the fact that the lowest percentage of positive RADT documented in this 6-year period (17%) was obtained in August 2013 (Fig. 4a). The monthly prevalence in each year (Fig. 4b) clearly shows the season cycle of this pathology.
Clinical data analysis
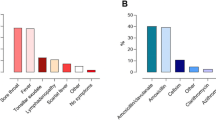
Among the 18,304 total RADT performed from 2013 until 2018, we randomly selected (using https://www.random.org) 130 patients whose clinical case files were carefully analysed concerning the symptoms registered by the paediatrician at hospital admission and additional visits to the Emergency Department. This group included patients of the age group with the largest number of streptococcal tonsillitis cases (2 to 10 years; a total of 13,842 cases). Three of these cases were excluded due to insufficient records. In the remaining 127 cases included in this analysis, 79 GAS-positive RADT and 48 GAS-negative, the most frequently described signs and symptoms were fever (83%), tonsillar hyperaemia (73%), odynophagia (73%) and tonsillar hypertrophy (43%). These 4 symptoms were also the most common, although in different proportions, both in streptococcal and in non-streptococcal tonsillitis (Supplementary information) (Fig. 5).
Frequency of signs and symptoms in patients with RADT-negative (GAS (−): group A Streptococcus–negative) or with RADT-positive (GAS (+): group A Streptococcus–positive) in the clinical files of a selected group of 127 patients, belonging to the age group with the highest incidence of GAS tonsillitis (2–10 years)
The statistical analysis of all the results showed that GAS tonsillitis is statistically correlated with the presence of scarlatiniform rash (p < 0.05) and palatal petechiae (p < 0.05). These signs increase the probability of a streptococcal cause by 33 and 23 percentage points, respectively. None of the analysed remaining variables showed similar statistical behaviour (Table 2).
However, when considering the subgroup of children under 3 years old (n = 38), scarlatiniform rash is the only sign that shows a positive statistical correlation (p < 0.05) with the bacterial aetiology (Supplemental information). Using the same linear regression analysis with results gathered from children between 3 and 10 years old (n = 89), we found a significant statistical correlation between bacterial tonsillitis and both cough and the presence of palatal petechiae. We also found p values < 0.05 for anterior rhinorrhoea and abdominal pain (Supplemental information), suggesting a higher probability of a non-streptococcal cause.
Only 7 (18%) of the 38 clinical cases of patients with less than 3 years contained information of significant epidemiological context (close contact with confirmed GAS infection).
All patients with a positive RADT were treated with amoxicillin 50 mg/kg/day 2 ID, during 6 to 10 days. Seven patients with negative tests underwent antibiotic due to several reasons: (i) empiric therapy prescribed by the general practitioner (n = 2) without undergoing RADT (antibiotic was suspended during observation in the Emergency Department); (ii) justified by a concomitant clinic of acute otitis media (n = 2); (iii) due to worsening condition despite symptomatic relief measures (n = 1); (iv) in a prophylactic context after rat bite (n = 1) and, (v) due to difficulties in obtaining the oropharyngeal swab in an uncooperative patient with a significant epidemiological context (n = 1).
Tonsillitis complications were not detected in the selected clinical files. There were, however, four cases of additional visits to the Emergency Department in the 30 days following the first visit. Among these, only one patient, who returned 2 days later with a suspected encephalitis, had a negative RADT. In the remaining three cases, one child returned 2 days later (on the fifth day of illness) due to fever aggravation, with a suspected viral tonsillitis, and confirmed as asymptomatic GAS carrier; another patient returned after 10 days with similar complaints and still tested positive for GAS (inadequate duration of treatment - patient abandoned antibiotic after 5 days). Finally, a patient returned after 9 days, 2 days after completing antibiotic treatment, due to the appearance of a generalised maculopapular rash—suspicion of a late allergic reaction to amoxicillin.
Discussion
Streptococcus pyogenes, also known as group A Streptococcus (GAS), is an important human pathogen responsible for a wide spectrum of diseases. It causes disease especially in children, where superficial infections of the throat due to streptococcal infection are common [16]. This study constitutes a retrospective analysis of GAS pharyngitis in children who attended a paediatric hospital in the central region of Portugal and were subjected to RADT. We analysed 18,304 cases, registered from 1 January 2013 to 31 December 2018, and we believe that, when compared with other published data, the 6-year period of study allied to the particularly large sample size enabled a more robust investigation. Our results showed an average GAS prevalence of 33% (27–39%), similar to those reported by several studies held in Spain [17,18,19], who presented streptococcal prevalence of, respectively, 28.2%, 34.1% and 38.7%. These values show similar microbiological findings between the two countries, probably sustained not only by the close historical coexistence between Portuguese and Spanish populations but also due to similar geography and climate. In a broader context, a review [1] that analysed 98 studies from multiple countries, with a total of 101,121 participants, reached an average prevalence of 29.5% for streptococcal infection. In what regards the Portuguese regional data variability, the percentage of streptococcal tonsillitis we registered differs from the results presented before [2, 15]. The differences may be explained by the size of the population studied and the period of the year(s) considered, including cyclical fluctuations in the rate of infection only detectable in longer studies [20].
Traditionally, a higher incidence of infectious oropharynx pathology is described in early spring and winter [6, 21]. However, our results show that a higher incidence of GAS is registered during spring and autumn months, more precisely, in May and June, a result that corroborates the findings of Contessotto Spadetto and co-workers [17], and during October, November and December, seen as an autumn peak. Despite the slight differences, probably explained by climatic variations throughout different countries and regions, it can be concluded that this is a seasonal pathology. This has been reported before [22], but the novel conclusion that can be drawn from the data we gathered is that there is that streptococcal tonsillitis cases significantly drop in February and August. Especially in February, a very cold and humid month in Portugal, it would be expected a higher number of cases, has reported in other studies [23].
Another relevant aspect related to GAS is that it’s usually described low prevalence in children under 5 years of age. In fact, there are several studies showing this, in different countries and regions, such as Australia [24], Spain [25] and Israel [26]. In this last study, the authors claimed that only 33 (8%) would be true streptococcal infections, while the remaining 28 (6.7%) would be asymptomatic GAS carriers with tonsillitis due to viral superinfection. In literature, carriers (9 to 14% of healthy children) [27] are generally defined as patients with positive oropharyngeal cultures but without a specific immune response against GAS—such as increased anti-streptolysin O and anti-DNase B antibodies [1, 13, 26]. Nevertheless, the results obtained by us in this age group are similar to those described in Israel [28], reporting 26.4% positive oropharynx cultures in a group of symptomatic children; GAS was also detected in 6.7% of children in the control group (without symptoms), classified as asymptomatic carriers. Another study [29] described a GAS prevalence of 29% in children under 3 years old with complaints suggesting acute tonsillitis. In this context, and even though the Portuguese CG 020/2012 does not recommend testing for GAS in children with acute tonsillitis under the age of 3 years (unless there is a close contact with confirmed GAS infection), we believe that our results, which reflect the clinical practice of a paediatric hospital, indicate the importance of a revision of this directive. This would decrease the number of unnoticed cases and, consequently, a better therapeutic course would not be denied to an important section of the paediatric population. Although children under 3 years of age rarely develop non-suppurative complications, this age group can represent a source of streptococcal infection for older children, constituting a potential epidemiological risk factor for rheumatic fever [26].
In respect of the data concerning signs and symptoms, our study was limited to 127 clinical files randomly selected among the age group with the highest number of number of streptococcal tonsillitis cases, what can be considered a limitation in our data analysis. However, we believe that it can give important information about issues related to the clinics of this infectious pathology. In this context, fever, odynophagia and tonsillar hyperaemia and hypertrophy were, as expected, the most frequent findings, as corroborated by others [21, 22, 30]. Neither the symptoms nor the GAS results were influenced by the day from onset (data not shown). The linear regression analysis showed that the signs and symptoms present at the time of diagnosis assume different weights depending on the patient’s age group. Thus, although there are no pathognomonic signs, there are clinical characteristics that increase the pre-test probability of a streptococcal cause: (i) in all 127 clinical cases analysed (ages between 2 and 10 years), the presence of a scarlatiniform rash and the detection of palatal petechiae were the only features that correlated positively with a positive RADT; (ii) for children under 3 years of age, it was found that scarlatiniform rash increased the likelihood of GAS tonsillitis by 67 percentage points (Supplemental Results), but the presence of palatal petechiae lost the statistical weight it had when the whole sample was considered; iii) in patients in the age group between 3 and 10 years, we found not only a statistically positive association between streptococcal tonsillitis and the presence of cough and palatine petechiae but also a correlation between anterior rhinorrhoea and abdominal pain with the non-streptococcal cause.
These results are different from those presented by others who indicate that there is a significant streptococcal aetiology association with odynophagia, inflammatory adenopathy and rash [17], or with cough, scarlatiniform rash, tonsillar exudate, palatine petechiae and tonsillar hypertrophy [2] or, with the absence of cough, adenopathies and the use of a modified Centor score [30]. For sure, the relevance of cough is not consensual. The results reported in our study show some discrepancies with the consulted guidelines, the Portuguese CG 020/2012 and the Clinical Practice Guideline for the Diagnosis and Management of Group A Streptococcal Pharyngitis by the Infectious Diseases Society of America [7] describing the signs and symptoms more suggestive of a bacterial aetiology (fever, headache, nausea and vomiting, abdominal pain, petechiae, exudates, adenopathies, scarlatiniform rash or significant epidemiological context) or a viral cause (conjunctivitis, cough, hoarseness, runny nose, oral ulcers, diarrhoea, viral rash or absence of fever). These discrepancies may be due to different sample sizes, statistical methods with different robustness or inter-individual particularities in response to environmental aggressions. Nevertheless, they seem to indicate that the overlap in acute tonsillitis presentation is significant, even with aetiologies so different in nature that it does not allow the purely clinical distinction of the causal agent [1, 7, 13, 14].
When considering tonsillitis, although the disease is mostly self-limited, it is important to consider the post-infection complications, suppurative or non-suppurative. In our study, no complications of tonsillitis were detected in the consulted clinical files (n = 130); however, there were cases of children with positive RADT who returned to the Emergency Department within 1 month after the first visit. According to Piñeiro Perez and collaborators [25], suppurative complications occur in 1–2% of GAS acute tonsillitis not treated with antibiotics or after inadequate/incomplete treatment. Although the role of antibiotics is not perfectly defined in the remaining non-suppurative complications, it is known that, if effective in eliminating GAS, it reduces the risk of rheumatic fever by about 90% [32]. Nowadays, despite being a rare disease in developed countries, with an average annual incidence of 0.5 cases per 100,000 inhabitants, it remains as the leading cause of acquired cardiac pathology in children from poorer regions such as sub-Saharan Africa, India and parts of Australasia [1, 25, 31, 33]. The risk of developing rheumatic fever due to untreated tonsillitis is about 1%; its prevalence is closely related to the infectious GAS serotype but also to a genetic predisposition of the host, especially when there is cardiac involvement [16, 32]. The treatment, like the testing, for GAS has been described as having advantages and disadvantages [23, 34]. A prospective study in a cohort of 200 children showed that poor compliance with antibiotic treatment does not affect the incidence of complications [35]. Other prospective studies are currently progressing towards a more accurate knowledge about aspects related to GAS infection [36].
As a conclusion, our study shows that the prevalence of streptococcal tonsillitis in the centre of Portugal is in line with numerous studies conducted in several other countries. However, the percentage of GAS in preschool children, and especially under 3 years of age, proved to be higher than that reported by other authors and, most importantly, higher than the value accepted by clinical guidelines. We believe that this study will help to adapt the approach of both hospital teams and primary care units to paediatric patients with symptoms suggestive of acute tonsillitis. It indicates that it would be advisable to revise the currently available guidelines or to carry out further studies to assess the need for this revision. Like other authors, we found that the patient’s symptomatic condition is not enough to clearly indicate the aetiology of acute tonsillitis. This, together with the advantages of rapid sensitive tests, indicates that RADT is an important diagnosis resource in the prompt definition for the need of therapy, preventing post-infection complications or avoiding unnecessary antibiotic prescription.
Data availability
Not applicable.
References
Cohen JF, Bertille N, Cohen R, Chalumeau M (2016) Rapid antigen detection test for group a streptococcus in children with pharyngitis. Cochrane Database Syst Rev 7:CD010502. https://doi.org/10.1002/14651858.CD010502.pub2
Morais S, Teles A, Ramalheira E, Roseta J (2009) Streptococcal pharyngitis: clinical suspicion versus diagnosis. Acta Medica Port 22(6):773–778
Cohen JF, Cohen R, Levy C, Thollot F, Benani M, Bidet P, Chalumeau M (2015) Selective testing strategies for diagnosing group A streptococcal infection in children with pharyngitis: a systematic review and prospective multicentre external validation study. Cmaj 187(1):23–32. https://doi.org/10.1503/cmaj.140772
Pfoh E, Wessels MR, Goldmann D, Lee GM (2008) Burden and economic cost of group a streptococcal pharyngitis. Pediatrics 121(2):229–234. https://doi.org/10.1542/peds.2007-0484
Bisno AL (1996) Acute pharyngitis: etiology and diagnosis. Pediatrics 97(6 Pt 2):949–954
Bisno AL (2001) Acute pharyngitis. N Engl J Med 344(3):205–211. https://doi.org/10.1056/NEJM200101183440308
Shulman ST, Bisno AL, Clegg HW, Gerber MA, Kaplan EL, Lee G, Martin JM, Van Beneden C (2012) Clinical practice guideline for the diagnosis and management of group A streptococcal pharyngitis: 2012 update by the Infectious Diseases Society of America. Clin Infect Dis 55(10):e86–e102. https://doi.org/10.1093/cid/cis629
Carapetis JR, Steer AC, Mulholland EK, Weber M (2005) The global burden of group A streptococcal diseases. Lancet Infect Dis 5(11):685–694. https://doi.org/10.1016/S1473-3099(05)70267-X9
Noh HJ, Freitas CA, Souza Rde P, Simoes JC, Kosugi EM (2015) Lemierre syndrome: a rare complication of pharyngotonsillitis. Braz J Otorhinolaryngol 81(5):568–570. https://doi.org/10.1016/j.bjorl.2015.03.009
Matthys J, De Meyere M, van Driel ML, De Sutter A (2007) Differences among international pharyngitis guidelines: not just academic. Ann Fam Med 5(5):436–443. https://doi.org/10.1370/afm.741
Cohen R, Levy C, Ovetchkine P, Boucherat M, Weil-Olivier C, Gaudelus J, de la Rocque F, Bingen E (2004) Evaluation of streptococcal clinical scores, rapid antigen detection tests and cultures for childhood pharyngitis. Eur J Pediatr 163(4–5):281–282. https://doi.org/10.1007/s00431-004-1416-y
Shaikh N, Swaminathan N, Hooper EG (2012) Accuracy and precision of the signs and symptoms of streptococcal pharyngitis in children: a systematic review. J Pediatr 160(3):487–493 e3. https://doi.org/10.1016/j.jpeds.2011.09.011
Tanz RR, Shulman ST (2007) Chronic pharyngeal carriage of group A streptococci. Pediatr Infect Dis J 26(2):175–176. https://doi.org/10.1097/01.inf.0000255328.19808.be
Gerber MA, Shulman ST (2004) Rapid diagnosis of pharyngitis caused by group A streptococci. Clin Microbiol Rev 17(3):571–580. https://doi.org/10.1128/CMR.17.3.571-580.2004
Plainvert C, Duquesne I, Touak G, Dmytruk N, Poyart C (2015) In vitro evaluation and comparison of 5 rapid antigen detection tests for the diagnosis of beta-hemolytic group A streptococcal pharyngitis. Diagn Microbiol Infect Dis 83(2):105–111. https://doi.org/10.1016/j.diagmicrobio.2015.06.012
Barnett TC, Bowen AC, Carapetis JR (2019) The fall and rise of group A Streptococcus diseases. Epidemiol Infect 147(e4):1–6. https://doi.org/10.1017/S0950268818002285
Contessotto Spadetto C, Camara Simon M, Aviles Ingles MJ, Ojeda Escuriet JM, Cascales Barcelo I, Rodriguez Sanchez F (2000) Rational use of antibiotics in pediatrics: impact of a rapid test for detection of beta-haemolytic group A streptococci in acute pharyngotonsillitis. An Esp Pediatr 52(3):212–219
Flores Mateo G, Conejero J, Grenzner Martinel E, Baba Z, Dicono S, Echasabal M, Gonzalo Santos C, Aliaga A, Barredo M, Ruiz L, Carrau M (2010) Early diagnosis of streptococcal pharyngitis in paediatric practice: validity of a rapid antigen detection test. Aten Primaria 42(7):356–361. https://doi.org/10.1016/j.aprim.2010.01.011
Regueras De Lorenzo G, Santos Rodriguez PM, Villa Bajo L, Perez Guirado A, Arbesu Fernandez E, Barreiro Hurle L, Nicieza Garcia M (2012) Use of the rapid antigen technique in the diagnosis of Streptococcus pyogenes pharyngotonsillitis. An Pediatr (Barc) 77(3):193–199. https://doi.org/10.1016/j.anpedi.2012.01.012
Noah ND (1989) Cyclical patterns and predictability in infection. Epidemiol Infect 102(2):175–190. https://doi.org/10.1017/s0950268800029848
Choby BA (2009) Diagnosis and treatment of streptococcal pharyngitis. Am Fam Physician 79(5):383–390
Penalba Citores AC, Riano Mendez B, Maranon Pardillo R, Miguez Navarro C, Vazquez Lopez P, Guerrero Soler MM, Merello Godino C (2007) Incidence of streptococcal pharyngitis. An Pediatr (Barc) 67(3):220–224. https://doi.org/10.1016/s1695-4033(07)70610-0
Martin JM (2015) The mysteries of streptococcal pharyngitis. Curr Treat Options Pediatr 1(2):180–189. https://doi.org/10.1007/s40746-015-0013-9
Edmond KM, Grimwood K, Carlin JB, Chondros P, Hogg GG, Barnett PL (1996) Streptococcal pharyngitis in a paediatric emergency department. Med J Aust 165(8):420–423
Pineiro Perez R, Hijano Bandera F, Alvez Gonzalez F, Fernandez Landaluce A, Silva Rico JC, Perez Canovas C, Calvo Rey C, Cilleruelo Ortega MJ (2011) Consensus document on the diagnosis and treatment of acute tonsillopharyngitis. An Pediatr (Barc) 75(5):342 e1–342 13. https://doi.org/10.1016/j.anpedi.2011.07.015
Nussinovitch M, Finkelstein Y, Amir J, Varsano I (1999) Group A beta-hemolytic streptococcal pharyngitis in preschool children aged 3 months to 5 years. Clin Pediatr (Phila) 38(6):357–360. https://doi.org/10.1177/000992289903800606
Shaikh N, Leonard E, Martin JM (2010) Prevalence of streptococcal pharyngitis and streptococcal carriage in children: a meta-analysis. Pediatrics 126(3):e557–e564. https://doi.org/10.1542/peds.2009-2648
Berkovitch M, Vaida A, Zhovtis D, Bar-Yohai A, Earon Y, Boldur I (1999) Group A streptococcal pharyngotonsillitis in children less than 2 years of age--more common than is thought. Clin Pediatr (Phila) 38(6):361–363. https://doi.org/10.1177/000992289903800607
Woods WA, Carter CT, Schlager TA (1999) Detection of group A streptococci in children under 3 years of age with pharyngitis. Pediatr Emerg Care 15(5):338–340. https://doi.org/10.1097/00006565-199910000-00011
Kose E, Sirin Kose S, Akca D, Yildiz K, Elmas C, Baris M, Anil M (2016) The effect of rapid antigen detection test on antibiotic prescription decision of clinicians and reducing antibiotic costs in children with acute pharyngitis. J Trop Pediatr 62(4):308–315. https://doi.org/10.1093/tropej/fmw014
Wessels MR (2011) Clinical practice. Streptococcal pharyngitis. N Engl J Med 364(7):648–655. https://doi.org/10.1056/NEJMcp1009126
Gerber MA, Baltimore RS, Eaton CB, Gewitz M, Rowley AH, Shulman ST, Taubert KA (2009) Prevention of rheumatic fever and diagnosis and treatment of acute Streptococcal pharyngitis: a scientific statement from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee of the Council on Cardiovascular Disease in the Young, the Interdisciplinary Council on Functional Genomics and Translational Biology, and the Interdisciplinary Council on Quality of Care and Outcomes Research: endorsed by the American Academy of Pediatrics. Circulation 119(11):1541–1551. https://doi.org/10.1161/CIRCULATIONAHA.109.191959
Olivier C (2000) Rheumatic fever-is it still a problem? J Antimicrob Chemother 45(Suppl):13–21. https://doi.org/10.1093/jac/45.suppl_1.13
ESCMID Sore Throat Guideline Group, Pelucchi C, Grigoryan L, Galeone C, Esposito S, Huovinen P, Little P, Verheij T (2012) Guideline for the management of acute sore throat. Clin Microbiol Infect 18(Suppl 1):1–28. https://doi.org/10.1111/j.1469-0691.2012.03766.x
Sarrell EM, Giveon SM (2012) Streptococcal pharyngitis: a prospective study of compliance and complications. ISRN Pediatr 2012:796389. https://doi.org/10.5402/2012/796389
Bennett J, Moreland NJ, Oliver J et al (2019) Understanding group A streptococcal pharyngitis and skin infections as causes of rheumatic fever: protocol for a prospective disease incidence study. BMC Infect Dis 19:633 doi.org/10.1186/s12879-019-4126-9
Vu MLD, Rajnik M (2020) Arcanobacterium Haemolyticum. In: StatPearls [Internet]. StatPearls Publishing, Treasure Island
Funding
TG and JL are funded through the COMPETE 2020 - Operational Programme for Competitiveness and Internationalisation and Portuguese national funds via FCT – Fundação para a Ciência e a Tecnologia, under project UIDB/04539/2020 (Strategic Plan CNC.IBILI).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Code availability
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 36 kb)
Rights and permissions
About this article
Cite this article
Mendes, N., Miguéis, C., Lindo, J. et al. Retrospective study of group A Streptococcus oropharyngeal infection diagnosis using a rapid antigenic detection test in a paediatric population from the central region of Portugal. Eur J Clin Microbiol Infect Dis 40, 1235–1243 (2021). https://doi.org/10.1007/s10096-021-04157-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-021-04157-x