Abstract
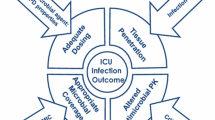
The World Health Organization (WHO) proposed a global priority pathogen list (PPL) of multidrug-resistant (MDR) bacteria. Our current objective was to provide global expert ranking of the most serious MDR bacteria present at intensive care units (ICU) that have become a threat in clinical practice. A proposal addressing a PPL for ICU, arising from the WHO Global PPL, was developed. Based on the supporting data, the pathogens were grouped in three priority tiers: critical, high, and medium. A multi-criteria decision analysis (MCDA) was used to identify the priority tiers. After MCDA, mortality, treatability, and cost of therapy were of highest concern (scores of 19/20, 19/20, and 15/20, respectively) while dealing with PPL, followed by healthcare burden and resistance prevalence. Carbapenem-resistant (CR) Acinetobacter baumannii, carbapenemase-expressing Klebsiella pneumoniae (KPC), and MDR Pseudomonas aeruginosa were identified as critical organisms. High-risk organisms were represented by CR Pseudomonas aeruginosa, methicillin-resistant Staphylococcus aureus, and extended-spectrum beta-lactamase (ESBL) Enterobacteriaceae. Finally, ESBL Serratia marcescens, vancomycin-resistant Enterococci, and TMP-SMX-resistant Stenotrophomonas maltophilia were identified as medium priority. We conclude that education, investigation, funding, and development of new antimicrobials for ICU organisms should focus on carbapenem-resistant Gram-negative organisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Multidrug-resistant (MDR) bacteria have become a health priority [1] and efforts have been made to prevent colonization, infection, and decrease mortality [2,3,4,5,6,7]. The World Health Organization (WHO) proposed a global priority pathogen list (PPL) of MDR bacteria to guide research, discovery, and development of new antibiotics [3, 8]. However, critically ill patients are particularly susceptible to infections arising from MDR bacteria [9, 10]. To develop a more solid understanding of the issues facing critically ill patients, we established the TOp TEn resistant Microorganisms (TOTEM) in critical care study group (Appendix 1). The scope was to identify the most important resistant bacteria for intensive care units (ICU) for which there is an urgent need for new therapies. The primary objective of the TOTEM study was to describe, as assessed by expert opinion and current evidence, a global list of the top ten most clinically relevant MDR bacteria affecting critically ill patients. The secondary objective was to prioritize the list to focus efforts proportionately according to perceived clinical need.
Methods
The study consisted of score prioritization by a panel of ten experts invited to prioritize organisms using MCDA. A steering committee (Appendix 2a) with experience of identification, prevention, and treatment of MDR bacteria in critically ill patients were invited to participate. They contributed in the revision of first drafts of the study protocol and selection of pathogens. Mycobacteria, Rickettsia, viruses, and parasites were excluded. Panel experts were suggested by the TOTEM project leader (JR) based on their prior experience or their expertise in clinical practice, clinical trials, and publications, seeking to provide global geographic coverage and membership from the range of professionals whose roles are impacted by MDR bacteria. MDR bacteria were defined as reported elsewhere [6]. The coordinating group represented intensivists, anesthesiologists, clinical microbiologists, and infectious disease (ID) consultants with experience in ICU settings (Appendix 2b). Pediatric and neonatal intensive care units (ICUs) were excluded. The list was ranked using the following (WHO) prioritization factors: all-cause mortality, healthcare and community burden, prevalence of resistance, 5-year trend of resistance, transmissibility and preventability, treatability, and current drug pipeline, with the addition of estimated cost of therapy. Definitions for the variables used in the prioritization list were reported elsewhere [8]. For each variable, scores were assigned from 1 (least) to 10 (most) according to importance and the average value was multiplied by 2 providing a maximal potential score of 20. The study used no patient-specific data and thus the need for ethical research committee approval or informed consent was waived.
Statistical and MCDA analysis
All responses were categorical variables presented as summary statistics, reporting proportions (percentages). The prioritization exercise was performed through the following steps: (1) selection of antibiotic-resistant organisms to be prioritized, (2) selection for criteria of prioritization, (3) data extraction and synthesis, (4) scoring of the alternatives and weighting of criteria by experts, and (5) finalization of the pathogens’ ranking. As a summary of sources of data on the different variables, participants were referred to the evidence-based information released by the WHO final report [8]. Data sources were PubMed and Ovid databases and did not have time restriction, last update in September 2016. The multiple-criteria decision analysis (MCDA) methodology has been detailed in Online Resource 1.
Results
After MCDA, mortality and treatability were of highest concern (Scores of 19/20) while dealing with PPL, followed by cost of treatment, healthcare burden, and resistance prevalence. Carbapenem-resistant (CR) Acinetobacter baumannii, Klebsiella pneumoniae-expressing carbapenemase (KPC), and MDR Pseudomonas aeruginosa were classified as critical organisms. High-risk organisms were represented by CR P. aeruginosa, methicillin-resistant Staphylococcus aureus (MRSA), and extended-spectrum beta-lactamase (ESBL) Enterobacteriaceae. Finally, ESBL Serratia marcescens, vancomycin-resistant Enterococci, and TMP-SMX-resistant Stenothophomonas maltophilia were identified as medium priority. Distribution of scores is detailed in Table 1. In the PPL scoring, CR A. baumannii, KPC, and MDR P. aeruginosa scored high for mortality, treatability, and cost of treatment while MDR P. aeruginosa, KPC, and ESBL K.pneumoniae were prioritized for healthcare burden. Overall prevalence of resistance was high for ESBL Enterobacteriaceae. Along with other critical and high-priority pathogens, S. marcescens too scored high among difficult to treat pathogens. Preventability was worst with KPC followed by MRSA.
Discussion
CR Acinetobacter baumannii, CR Klebsiella pneumoniae, and MDR Pseudomonas aeruginosa were classified as critical organisms (priority 1), confirming the WHO PPL [8]. In contrast, priority 2 represented by high-risk organism is markedly different. However, this finding is not a surprise as the risk factors for the selection of resistant organisms in hospitals vary from the community. Our findings emphasize a global concern regarding Gram-negative bacteria.
Indeed, while dealing with PPL, mortality and treatability were considered the highest priority followed by cost of treatment, healthcare burden, and resistance prevalence in MCDA analysis. Carbapenem-resistant organisms were indisputably perceived as the highest threat for mortality, treatability, and cost. The results support the difficulty faced in managing MDR P.aeruginosa infections in ICUs [11]. Mortality by CR organisms is contributed particularly by the non-availability of effective drugs rather than increased virulence [12,13,14,15]. Currently, the biggest gap exists in the investigational pipeline for compounds active against CR A. baumannii, which is perceived as a critical organism for treatability. Our findings suggest that CR A. baumannii is of major concern, despite it being considered conventionally of low virulence [16]. Not surprisingly, given the focus on intensive care major concerns, the prioritization list came up with a different ranking of pathogens and resistance markers than the WHO PPL, which takes a more global view.
WHO reports estimate approximately 30% of ICU patients are affected by at healthcare-associated infections while incidence is 3-fold higher in low- and middle-income countries [17]. Several reports from these countries suggest the lack of surveillance data thus having a negative influence on the implementation of preventive measures [18,19,20,21,22]. Two EPIC studies in a span of 10 years have demonstrated a 20% increase in prevalence of ICU-acquired infections [23, 24].
There are a number of limitations to this study. The survey panel has not uniformly represented the regions of global hotspots of MDR infections, such as Asia, whereas Europe is over-represented. The study did not take into consideration the current evidence for infections in respect to the frequency and burden, discrepancies in CDC vs ECDC definitions, underlying immune status, sub-classification of infections based on underlying condition (medical, trauma, burns, cardiac surgery, special patient population etc), pediatric patients, and public health threats. Other bacterial pathogens causing severe infections that are potentially drug resistant and are acquired in the community were not covered. The strengths include the study methodology (MCDA) incorporating expert opinion and evidence-based data that showed a high stability of the final ranking and its future adaptability for regional updates of the priority pathogen lists.
Conclusions
Carbapenem-resistant Acinetobacter baumannii, carbapenemase-expressing Klebsiella pneumoniae, and MDR Pseudomonas aeruginosa were classified as critical organisms (priority 1) causing ICU infections. Education, investigation, funding, and development of new antimicrobials for ICU organisms should be focused on the identified priorities.
References
De Waale J, Akova M, Antonelli M et al (2018) Antimicrobial resistance and antibiotic stewardship programs in the ICU: a position statement from ESICM/ESCMID/WAAAR round table on multidrug resistance. Intensive Care Med 44:189–196
National efforts to combat antibiotic-resistant bacteria through science | NIH: National Institute of Allergy and Infectious Diseases n.d. https://www.niaid.nih.gov/news-events/national-efforts-combat-antibiotic-resistant-bacteria-through-science (Accessed Aug 2, 2018)
Tacconelli E, Carrara E, Savoldi A et al (2018) Discovery, research and development of new antibiotics: the WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect Dis 18:318–327
Lucet JC, Koulenti D, Zahar JR (2014) Persistence of colonization with MDRO following discharge from the ICU. Intensive Care Med 40:603–605
Sievert D, Ricks P, Edwards J, Schneider A, Patel J, Srinivasan A et al (2013) Antimicrobial-resistant pathogens associated with healthcare-associated infections: summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2009-2010. Infect Control Hosp Epidemiol 34:1–14
Magiorakos A-P, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, Giske CG et al (2012) Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect 18:268–281
Otter JA, Mutters NT, Tacconelli E, Gikas A, Holmes AH (2015) Controversies in guidelines for the control of multidrug-resistant Gram-negative bacteria in EU countries. Clin Microbiol Infect 21:1057–1066
WHO. Prioritization of pathogens to guide discovery, research and development of new antibiotics for drug resistant bacterial infections, including tuberculosis. Essent Med Heal Prod 2017:88. doi:WHO reference number: WHO/EMP/IAU/2017.12
Poulakou G, Matthaiou DK, Bassetti M, Erdem H, Dimopoulos G, Curcio DJ et al (2017) “Salvage treatment” for infections by extensively- and pan-drug-resistant pathogens is common and often sub-optimal. Intensive Care Med 43:1164–1166
Zaragoza R, Ramirez P, Lopez-Pueyo MJ (2014) Nosocomial infections in intensive care units. Enferm Infecc Mircobiol Clin 32:320–327
Borgatta B, Gattarello S, Mazo CA, Imbiscuso AT, Larrosa MN, Lujan M et al (2017) The clinical significance of pneumonia in patients with respiratory specimens harbouring multidrug-resistant Pseudomonas aeruginosa: a 5-year retrospective study following 5667 patients in four general ICUs. Eur J Clin Microbiol Infect Dis 36:2155–2163
Grundmann H, Glasner C, Albiger B, Aanensen DM, Tomlinson CT, Andrasević AT et al (2017) Occurrence of carbapenemase-producing Klebsiella pneumoniae and Escherichia coli in the European survey of carbapenemase-producing Enterobacteriaceae (EuSCAPE): a prospective, multinational study. Lancet Infect Dis 17:153–163
Kern WV (2018) Multidrug resistant bacteria: antibiotic prescription and antibiotics of last resort. DDtsch Med Wochenschr 143:643–650
Micek ST, Wunderinck RG, Kollef MH et al (2015) An international multicenter retrospective study of Pseudomonas aeruginosa nosocomial pneumonia: impact of multidrug resistance. Crit Care 19:219
Zilberberg MD, Shorr AF, Micek ST, Vazquez-Guillamet C, Kollef MH (2014) Multi-drug resistance, inappropriate initial antibiotic therapy and mortality in Gram-negative severe sepsis and septic shock: a retrospective cohort study. Crit Care 18:596
Paterson DL, Harris PNA (2015) Editorial Commentary: the new Acinetobacter equation: hypervirulence plus antibiotic resistance equals big trouble. Clin Infect Dis 61:155–156
Health care-associated infections FACT SHEET. http://www.who.int/gpsc/country_work/gpsc_ccisc_fact_sheet_en.pdf. Accessed Nov 5th, 2018
Talaat M, El-Shokry M, El-Kholy J, Ismail G, Kotb S, Hafez S et al (2016) National surveillance of health care–associated infections in Egypt: developing a sustainable program in a resource-limited country. Am J Infect Control 44:1296–1301
Lim C, Takahashi E, Hongsuwan M, Wuthiekanun V, Thamlikitkul V, Hinjoy S et al (2016) Epidemiology and burden of multidrug-resistant bacterial infection in a developing country. Elife 5. https://doi.org/10.7554/eLife.18082
Dondorp AM, Limmathurotsakul D, Ashley EA (2018) What’s wrong in the control of antimicrobial resistance in critically ill patients from low- and middle-income countries? Intensive Care Med 44:79–82
Alp E, Damani N (2015) Healthcare-associated infections in intensive care units: epidemiology and infection control in low-to-middle income countries. J Infect Dev Ctries 9:1040–1045
Iwuafor AA, Ogunsola FT, Oladele RO, Oduyebo OO, Desalu I, Egwuatu CC et al (2016) Incidence, clinical outcome and risk factors of intensive care unit infections in the Lagos University Teaching Hospital (LUTH), Lagos, Nigeria. PLoS ONE 11:e0165242. https://doi.org/10.1371/journal.pone.0165242
Vincent JL, Bihari DJ, Suter PM, Bruining HA, White J, Nicolas-Chanoin MH et al (1998) The prevalence of nosocomial infection in intensive care units in Europe. Results of the European Prevalence of Infection in Intensive Care (EPIC) study. EPIC international advisory committee. JAMA 274:639–644
Vincent JL, Rello J, Marshall J, Silva E, Anzueto A, Martin CD et al (2009) International study of the prevalence and outcomes of infection in intensive care units. JAMA 302:2323–2329. https://doi.org/10.1001/jama.2009.1754
Belton V, Stewart TJ (2002) Multiple criteria decision analysis: an integrated approach. Springer US
Acknowledgements
We appreciate comments in the design from Nieves Larrosa, Barcelona, Spain. The study was developed in part from the Observership Programme, ESCMID, Basel, Switzerland (Vandana KE).
Funding
The study was funded in part by Centro de Investigacion Biomedica en Red de Enfermedades Respiratorias (CIBERES), Instituto de Salud Carlos III, Madrid, Spain, and Observership Programme from ESCMID, Basel, Switzerland.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr. Rello served in the speaker’s bureau or consultant for Pfizer, Anchoagen, ROCHE. The remaining authors have no conflicts of interest to declare.
Ethical approval
Not required.
Electronic supplementary material
Online Resource 1
(DOCX 13.9 kb)
Appendices
Appendix 1. TOTEM Study Investigators
Argentina: Luna CM, Reina R; Bulgaria: Dobrevska R, China: Deng H, Leiqing L, Liu L, 温, 沈延飞, LX, Wang D, Yuetian Y, Zhang G, Zh Zhang, Zheng C, YW, ZRYHC; Colombia: Del Rio G; Dominican Republic: Rojas R; Ethiopia: Amare D. France: Alfandri S, Argemi X, Dellamonica J. Kernies S, Lesprit P, Greece: Arvanitik K, Papanikolaou M, Tsigou E, Soultati I, Platsouka E, Katsiari M, Nikolaou C, Tsiodras S; Italy: Antonelli M, Cascio A, DiPascale G, Garofalo E, Girardis M, Leone D; India: Vandana KE, Kaniyarakkal V, Munim F, Nath S, Patil S, Suchitra U; Israel: Yahav D; Kenya: Misango DO; Kosovo:Gecaj-Gashi A; Kuwait: Rotimi V; Mexico: Aguilar D, Araujo-Melendez J, Franco-Zendejas R, Lagunes L, Lemus J, Perales Martinez DE, Rivera Chavez M; Netherlands: Schouten J; Oman: Khamis F; Pakistan: Nizamuddin S, Portugal: Santos L, Santos-Ribeiro E; Romania: Miftode E; South Africa: Alekar S, Baker D, Ballot D, Black V, Bhamjee S, Brannigan L, Hunt IA, Kotze J, Lowman W, Levy B, Mer M, Morar R,Michell W, Nana T, Pahad H, Tsai M, Schleicher GK, Shaddock E, Shoul E, Smith C, Richards GA, Van der Merwe L, Welkovics N; Spain: Borges M, Diaz E, J Garnacho-Montero, Maseda E, Mañez R, Rello J, Samso E, Serrano R, Solvio J, Vidaur L, Zaragoza R; Thailand: Wongsurakiat P; Turkey: Abravci N, Akbudak I, Akkoyunlu Y, Altındiş M, Aydin DÇelebi G, Emel A, Emine A,, Erdem H, Gulden E, Guner R, Kızmaz Y, Yalçin A, Kepenek E, Sener A, Tekin R, Tulek N, Ulu, Unuvar G; United Kingdom: Buckley J, Conway-Morris A, Dunn M, Hall A,Hobrok M, Felton T, Fletcher S, Marshall B, McConnell H, McKee R, McAuley D, McFie C, Morton B, Naisbitt J, Rooney K, Szakmany T, Yates B, Zochios V; Venezuela: Von der Osten J
Appendix 2a- Steering Committee members
Jordi Rello, Spain (Chair); Joana Alves, Portugal; Leonel Lagunes, Mexico; Jeroen Schouten, Netherlands; Celine Pulcini, France; Nieves Larrosa, Spain; Mervyn Mer, South Africa; Emine Alp, Turkey; Zhongheng Zhang, China.
Appendix 2b- Scoring Committee members
Emine Alp, Turkey; Andrew Conway-Morris, UK; Leonel Lagunes, Mexico; Davide Leoni, Italy; Jose Nicolas, Colombia; Jordi Rello, Spain, Vandana KE, India; Richard Wunderink, USA; Zhongheng Zhang, China.
Rights and permissions
About this article
Cite this article
Rello, J., Kalwaje Eshwara, V., Lagunes, L. et al. A global priority list of the TOp TEn resistant Microorganisms (TOTEM) study at intensive care: a prioritization exercise based on multi-criteria decision analysis. Eur J Clin Microbiol Infect Dis 38, 319–323 (2019). https://doi.org/10.1007/s10096-018-3428-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-018-3428-y