Abstract
Patients with bacteraemia constitute an useful population for an audit of antibiotic treatments. Empirical antibiotic therapy (EAT) and its reassessment must take into account clinical data and microbiological results. Our aim was to determine the impact of these sequential steps of the therapy on survival. This was a retrospective multicentre study which included patients admitted to emergency departments (ED) for whom blood cultures were positive over a 4-month period. Microbial results were compiled from the database of the laboratories. The relevant information was extracted from the computerized patient’s chart. An efficient EAT was based on antibiotic susceptibility of the bacteria. An effective antibiotic reassessment (AR) was defined as any modification of the EAT. Unfavorable outcome was defined as death of the patient during in-hospital care. Three hospitals and two clinics took part in this study, 169 patients with bacteraemia being included. The diagnosis in ED was undetermined in 21 cases (12%), 35 patients (21%) required intensive care, and 23 died (14%). One hundred and thirty-six patients (80%) received an EAT, the latter being efficient in 107 cases (63%). An effective AR was performed in 116 cases (69%). In multivariate analysis, risks factors for death were: ongoing cancer AOR (adjusted odds ratio) 3.34, undetermined diagnosis in ED: AOR 9.34 and severe sepsis or shock: AOR 6.98. Effective AR was a protective factor: AOR 0.28 [0.09–0.81]. One third of bacteraemic patients in ED did not benefit from AR. Improvement of antimicrobial stewardship should be associated with a higher rate of survival.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Infectious diseases (ID) are unpredictable illnesses with substantial clinical and microbiological diversity and without any specific element for recognition. Furthermore, access to specialized ID departments, which are rare in the French healthcare system, is limited. In light of this, emergency departments (ED) constitute the main resource for patients with severe ID. Bacteraemia is a severe ID, which requires the administration of an efficient empirical antibiotic therapy (EAT) as soon as possible [1,2,3]. Empirical antibiotic therapies are based on both national and international guidelines, value-added by the awareness on local bacterial resistance [4,5,6,7]. However, an antibiotic reassessment (AR), which must lead to a documented treatment in case of a positive blood culture, is often not possible for the initial prescribers in the ED, since the results of bacteriological samples are generally not available until at least 12 to 72 h after the sampling.
With regard to EAT, when it comes to making a decision concerning the therapeutic options, the physician in charge must take into account the prevalence of multidrug-resistant (MDR) bacteria observed in the local area [3, 6,7,8]. In our own region, 10% of E. coli and more than 30% of K. pneumoniae isolates are now resistant to third-generation cephalosporins (Ceph-3). These endemic strains of MDR bacteria are one of the main reasons for launching an efficient antimicrobial stewardship programme (ASM) [7, 9,10,11].
Previous studies on bacteraemic patients underlined the need for a multidisciplinary approach in which clinicians, microbiologists, and ID specialists work in a coordinated and joint manner [12,13,14,15,16,17,18]. Also, other studies on ASM in the ED have revealed a significant impact in terms of lowering antibiotic use [18, 19], but very few studies have detailed the impact of ASM recommendations on the outcome of bacteraemic patients from ED [4]. Our aim was to evaluate the EAT prescribed in the ED and its reassessment for patients who had positive blood cultures, with an emphasis on the final outcome at the end of the hospitalization period.
Methods
This was a retrospective study performed in three public hospitals and two private medical clinics. The participating institutions worked in a professionnel multidisciplinary network for antibiotic stewardship, the objectives being practice audits, homogenization, and clinical research [20,21,22].
Each of these institutions had an ED, although as the two clinics had a close working relationship along complementary paths for different medical and surgical specialities, they were treated as a single institution for the purposes of this study.
All of the patients with positive blood cultures performed in the ED over the course of 4 consecutive months from January 2015 to April 2015 were enrolled. The full patient list was obtained from the digital databases of the laboratories. The number of blood samples obtained per patient in the ED was also reported, as was the antibiotic susceptibility of the strains.
Contaminated blood cultures were defined by the isolation of common skin commensals (e.g., coagulase-negative staphylococci, “diphtheroids”, micrococcus, Bacillus spp.) When these were identified, they were not in keeping with the clinical features of the case.
Multidrug-resistant bacteria were ESBL-secreting Enterobacteriaeae (ESBL-E) or those secreting high-level cephalosporinases, Staphylococcus sp. resistant to methicillin, and vancomycin-resistant Enterococci.
All of the clinical data, therapeutic means, and outcomes were collected from the digital medical files of the patients. Co-morbidities were defined by the prescription of a specific treatment prior to the hospital care, or if the diagnosis was newly established during the hospital stay. More specifically, the diagnosis in the emergency room, the disease severity, and the final diagnosis available in the written records by the end of the hospitalizations were also reported. With regard to severity, we used criteria which were consensual at the time that the care was provided [8].
Healthcare-associated infections were defined as: 1) a diagnosis established ≥ 48 h after hospital admission, 2) when observed less than 1 month after surgery, and less than 1 year in case of insertion of a surgical device, and 3) when observed in association with an underlying urinary or venous catheterization.
An efficient EAT was defined by the susceptibility of the bacterial strain to at least one of the prescribed compounds. An effective AR was defined as any modification (including the first introduction) of the initial antibiotic treatment, irrespective of the time to change.
An unfavorable outcome was defined as death of the patient during the hospital stay.
Statistical analysis
The data were analyzed with Statview software version 4.5 and statistical significance was established at α = 0.05. Continuous variables were compared with the Mann–Whitney non-parametric test, and qualitative variables were compared with the χ2 or Fisher’s exact test, when appropriate. Logistic regression was used for multivariate analysis of the impact of EAT and effective antibiotic reassessment on all causes of in-hospital mortality, and results are presented as adjusted odds ratios (AORs) with their 95% confidence intervals (CIs). Variables were selected as candidates for the multivariate analysis on the basis of the level of significance of the univariate association with in-hospital mortality (p ≤ 0.1). Models were built sequentially, starting with the variable most strongly associated with the outcome and continuing until no other variable reached significance or altered the odds ratios of variables already in the model. When the final model was reached, each variable was dropped in turn to assess its effect.
Results
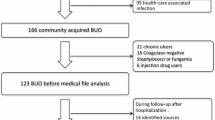
Two hundred and fourteen patients with positive blood cultures were included. The total number of blood cultures performed was 306, the patients having between one and four positive samples each.
Blood-culture contamination was observed in 45 cases (21%), coagulase negative Staphylococcus being the predominant bacterium (34 cases, 77%) followed by P. acnes in four cases (9%).
Thus, true bacteraemia was diagnosed in 169 cases, due to Enterobacteriaceae in 57% of the cases, Staphylococcus sp. in 16%. Streptococcus sp. and Enterococcus sp. accounted for 14% and 4% of the cases respectively (Table 1). Multidrug-resistant bacteria were isolated in 15 cases (9%), including 12 E-ESBL, 2 Enterobacteriaceae expressing high levels of cephalosporinases, and one methicillin-resistant Staphylococcus.
Demographic data are indicated in Table 2. Patients with urinary, respiratory, and digestive infections were the three main disease-related patient groups in the emergency room, accounting for 57% of the patients. It should be noted that in 38 cases (18%), no diagnosis was recorded in the patient’s medical file. The severity of the diseases was significant, as 40 patients (19%) required admission to intensive care.
After exclusion of the patients with contaminated blood cultures and those without infectious diseases (n = 13, 6%), 169 patients were analyzed. Healthcare-associated infections were observed in 35 cases (21%), including 24 in a medical context (main subgroup: 12 patients with cancer and central catheter and/or neutropenia) and 11 post-operative infections. These HCAI were associated with multidrug-resistant bacteria: 6/35 (17%) versus 9/134 (7%) respectively, p = 0.053.
Among those 169 bacteraemic patients, 107 (63%) received an efficient EAT (see Table 3). The univariate analysis showed that disease severity as well as urinary, respiratory, or cutaneous infections were associated with the prescription of an EAT. By contrast, the absence of a presumptive diagnosis was associated with the lack of an EAT (p < 0.001).
Taking into account that 33/169 patients (19.5%) did not receive an EAT, the main treatment included a single compound in 31% of the cases, a combination in 42%, and more than two compounds in 5%. Also, Ceph-3 was the most commonly prescribed compound, with 32% of the patients being given these antibiotics.
Antibiotic reassessment was effective in 116/169 cases (69%), and it was not significantly more frequent in the case of an inefficient EAT, keeping in mind that out of 33 bacteraemia patients without EAT, 26 (79%) received an antibiotic reassessment, meaning the introduction of an efficient therapy.
Risk factors for death
Twenty-three patients (14%) died over the course of their in-hospital care. Table 4 indicates the risk factors for death using a multivariate analysis by logistic regression, including all variables associated with a trend for antibiotic use (p ≤ 0.1). Death was associated with cancer (p = 0.019); the absence of a diagnosis in the emergency room (p < 0.001); a severe sepsis or a septic shock (p < 0.001). Reassessment of the antibiotic therapy was a protective factor with an AOR 0.28 [0.09–0.81], p = 0.017.
Discussion
Our retrospective audit of antibiotic therapy for bacteraemic patients in the emergency departments yielded three important results: (1) an unexpected epidemiological observation that a high proportion of bacteraemia patients in the emergency rooms presented with HCAI, (2) a significant proportion of patients did not have any established diagnosis, and this had a negative impact on the prognosis, and (3) antibiotic therapy reassessment was associated with a better outcome. Lastly, as reported in multicentre antibiotic audits, differences in therapeutic practices led to different prognosis between institutions [21, 22].
A few previous studies have reported a high proportion (between 15 and 30%) of healthcare-associated infections in patients with positive blood cultures at hospital admission [4, 14, 17]. We found 12 bacteraemia (5.6%) due to E-ESBL. A recent study indicated that only 38% of E-ESBL infections were truly community-acquired [23]. Accordingly, we found a trend towards HCAI and MDR bacteria (p = 0.053). We believe that the determination of factors contributing to this high proportion of HCAI in patients with bacteraemia in emergency departments warrants further investigation.
The lack of diagnosis is usually reported in the literature under the term “sepsis” or “sepsis of unknown origin”, and may amount to between 15 and 30% of cases [24, 25]. There are difficulties in defining sepsis, which is a general term applied to a partially undetermined process, with no clinical criteria or decisive laboratory characteristics [8, 24]. It was, however, not possible to use new definitions of sepsis in our study which were unknown to the practitioners in 2015 [26]. Considering bacteria involved in community-acquired infections (see Table 1), in which Streptococcus species as well as Enterococcus and anaerobes are commonly isolated, our results and others suggest the use of a penicillin-derivative compound for a sepsis of unknown origin [25].
To the best of our knowledge, our study is the first to establish a relationship between the absence of even a putative diagnosis in the emergency department and an unfavorable outcome. This realistic approach should be systematically used for the evaluation of an empirical antibiotic therapy. Ultimately, 33 of our patients (19%) did not receive an EAT in the emergency room, and among patients receiving an EAT, the treatment was efficient against the bacteria involved in 107 cases (66%). Lastly, antibiotic reassessment was relatively frequent, being effective in 128 cases (60%), compared to what has been reported in previous studies (from 17 to 36%) [20, 27, 28]. Importantly, the multivariate analysis revealed that antibiotic reassessment was associated with increased survival during the in-hospital care, compared to patients without modification of the empirical antibiotic therapy. This result has been reported previously [29, 30]. Accordingly, in a prospective study in a university hospital including 3413 bacteraemia patients, the rate of mortality was 20% in cases of efficient EAT, compared to 34% in cases of inefficient EAT [29].
Limitations of our study are its retrospective nature, which prevents the reasons for the antibiotic reassessments from being known, while the impact of the transfer of the patients in clinical departments was not determined at all. Lastly, our definition for antibiotic reassessment based on treatment modifications may lead to an underestimation of the quality of the antibiotic treatment, but optimal EAT, according to previously published analysis [20], has not been observed.
Conclusion
Optimal antibiotic therapy given to patients with bacteraemia in the emergency room needs to distinguish healthcare-associated infections from community-acquired infections. This allows the EAT to be better defined, as well as its reassessment, which has a favorable impact on survival.
References
GML B, Wenzel RP (2005) Bacteremias: a leading cause of death. Arch Med Res 36:646–659
Padersen G, Schonheyder HC, Sorensen HT (2003) Source of infection and other factors associated with case fatality in community-acquired bacteremia — a Danish population-based cohort study from 1992 to 1997. Clin Microbiol Infect 9:793–802
Marchaim D, Zaidenstein R, Lazarovitch T, Karpuch Y, Ziv T, Weinberg M (2008) Epidemiology of bacteremia episodes in a single center: increase in gram-negative isolates, antibiotics resistance and patient age. Eur J Clin Microbiol Infect Dis Eur 27:1045–1051
Vallès J, Calbo E, Anoro E et al (2008) Bloodstream infections in adults: importance of healthcare-associated infections. J Inf Secur 56:27–34
Fätkenheuer G, Preuss M, Salzberger B et al (2004) Long-term outcomes and quality of care of patients with Staphylococcus Aureus bacteremia. Eur J Clin Microbiol Infect Dis 23:157–162
Société de Pathologie Infectieuse de Langue Francaise (2002) Comment améliorer la qualité de l’antibiothérapie dans les établissements de soins ? Méd Mal Infect 32:320–328
Pollack LA, Srinivasan A (2014) Core elements of hospital antibiotic stewardship programs from the centers for disease control and prevention. Clin Infect Dis 59(S3):97–100
Dellinger RP, Levy MM, Rhodes A et al (2013) Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med 41:580–637
Miliani K, L’Hériteau F, Alfandari S et al (2008) Specific control measures for antibiotic prescription are related to lower consumption in hospitals: results from a French multicentre pilot study. J Antimicrob Chemother 62:823–829
Haute Autorité de Santé (2008) Stratégie d’antibiothérapies et prévention des résistances bactériennes en établissement de santé. Available on : www.has-sante.fr
Carlet J, Le Coz P (2015) Tous ensemble, sauvons les antibiotiques. Propositions du groupe de travail spécial pour la préservation des antibiotiques: 150 pp. [http://www.social-sante.gouv.fr/IMG/pdf/rapport · antibiotiques.pdf]
Chen SY, Wu GHM, Chang SC et al (2008) Bacteremia in previously hospitalized patients: prolonged effect from previous hospitalization and risk factors for antimicrobial-resistant bacterial infections. Ann Emerg Med 51:639–646
Corona A, Wilson APR, Grassi M, Singer M (2004) Prospective audit of bacteraemia management in a university hospital ICU using a general strategy of short-course monotherapy. J Antimicrob Chemother 54:809–817
Vallès J, Rello J, Ochagavia A, Garnacho J, Alcalà MA (2003) Community-acquired bloodstream infection in critically ill adult patients: impact of shock and inappropriate antibiotic therapy on survival. Chest 123:1615–1624
Anatoliotaki M, Valatas V, Mantadakis E et al (2004) Bloodstream infections in patients with solid tumors: associated factors, microbial spectrum and outcome. Infection 32:65–71
Kao CH, Kuo YC, Chen CC, Chang YT, Chen YS, Wann SR, Liu YC (2011) Isolated pathogens and clinical outcomes of adult bacteremia in the emergency department: a retrospective study in a tertiary referral Center. J Microbiol Immunol Infect 44:215–221
Chan J, Wong J, Saginur R, Forster AJ, van Walraven C (2015) Epidemiology and outcomes of bloodstream infections in patients discharged from the emergency department. CJEM 17:27–37
Trinh TD, Klinker KP (2015) Antimicrobial stewardship in the emergency department. Infect Dis Ther 4(Suppl 1):39–50
Borde JP, Kern WV, Hug M, Steib-Bauert M, de With K, Busch HJ, Kaier K (2015) Implementation of an intensified antibiotic stewardship programme targeting third-generation cephalosporin and fluoroquinolone use in an emergency medicine department. Emerg Med J 32:509–515
Étienne P, Roger PM, Brofferio P et al (2011) Antimicrobial stewardship program and quality of antibiotic prescriptions. Med Mal Inf 41:608–612
Roger PM, Tabutin J, Blanc V et al (2015) Prosthetic joint infection : a pluridisciplinary multi-center audit bridging quality of care and outcome. Med Mal Infect 45:229–236
Blanc V, Mothes A, Smets A et al (2015) Severe community-acquired pneumonia and positive urinary antigen test for S. Pneumoniae: amoxicillin is associated with a favourable outcome. Eur J Clin Microbiol Infect Dis 34:2455–2461
Zahar JR, Lesprit P, Ruckly S et al (2016) For the BacterCom Study Group. Predominance of healthcare-associated cases among episodes of community-onset bacteraemia due to extended β-lactamase-producing Enterobacteriaceae. Int J Antimicrob Agents 49:69–73
Gatewood MO, Wemple M, Greco S, Kritek PA, Durvasula R (2015) A quality improvement project to improve early sepsis care in the emergency department. BMJ Qual Saf 24:787–795
Courjon J, Demonchy E, Degand N, Risso K, Ruimy R, Roger PM (2017) Patients with community-acquired bacteremia of unknown origin: clinical characteristics and usefulness of microbiological results for therapeutic issues: a single-center cohort study. Ann Clin Microbiol Antimicrob 16:40. https://doi.org/10.1186/s12941-017-0214-0
Sterling SA, Puskarich MA, Glass AF, Guirgis F, Jones AE (2017) The impact of the Sepsis-3 septic shock definition on previously defined septic shock patients. Crit Care Med. https://doi.org/10.1097/CCM.0000000000002512
Leibovici L, Shraga I, Drucker M, Konigsberger H, Samra Z, Pitlik SD (1998) The benefit of appropriate empirical antibiotic treatment in patients with bloodstream infection. J Intern Med 244:379–386
Roger PM, Labate C, Serre S et al (2013) Factors associated with effective reassessment of antibiotic therapy on day 3. Med Mal Infect 43:123–127
Zaragoza R, Artero A, Camarena JJ, Sancho S, González R, Nogueira JM (2003) The influence of inadequate empirical antimicrobial treatment on patients with bloodstream infections in an intensive care unit. Clin Microbiol Infect 9:412–418
Enoch DA, Phillimore N, Mlangeni DA et al (2011) Outcome for gram-negative bacteraemia when following restrictive empirical antibiotic guidelines. Q J Med 104:411–419
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest.
Ethical approval
Not required, antibiotic audit being promoted by French National Health Agency.
Informed consent
Patients or relatives give their written consent for computerizing their personal data for hospitalization purpose and potential clinical researches.
Rights and permissions
About this article
Cite this article
Aillet, C., Jammes, D., Fribourg, A. et al. Bacteraemia in emergency departments: effective antibiotic reassessment is associated with a better outcome. Eur J Clin Microbiol Infect Dis 37, 325–331 (2018). https://doi.org/10.1007/s10096-017-3136-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-017-3136-z