Abstract
This study analyzed the prevalence and antibiotic susceptibility of urogenital Ureaplasma urealyticum and Mycoplasma hominis isolated in Xi’an, China. A total of 2161 individuals from 2011 to 2015 were included, and antibiotic susceptibility tests were performed by using the Mycoplasma IST kit. Of the individuals studied, 1018 (47.11 %) were identified to be positive for urogenital mycoplasmas. The single U. urealyticum, single M. hominis, and dual U. urealyticum and M. hominis infections accounted for 772 (75.83 %), 66 (6.48 %), and 180 (17.68 %), respectively. The total positive rate was higher in females than in males (58.76 % vs. 28.86 %, p < 0.001). The highest total positive rate (48.88 %) was observed in individuals aged 25 years to 30 years. In symptomatic and asymptomatic individuals, the positive rates were both higher in females than in males (67.36 % vs. 31.02 %, p < 0.001 and 42.58 % vs. 7.69 %, p < 0.001, respectively) and individuals aged 25 years to 30 years, and those aged 30 years to 35 years had the highest positive rates (54.35 and 57.14 %, respectively). The U. urealyticum and M. hominis identified from single or dual infections displayed low resistance rates to josamycin, doxycycline, and minocycline (<10 %) in both the symptomatic and asymptomatic groups. These results suggest that females and individuals with symptoms and younger age had higher mycoplasma infection rates and that josamycin, doxycycline, and minocycline may be recommended for the clinical treatment of patients infected with urogenital mycoplasmas, irrespective of the symptoms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mycoplasma belong to the class Mollicutes characterized by small size, lacking of cell wall, extremely fastidious in vitro environment requirement, and tendency to form centered colonies on solid medium. Ureaplasma urealyticum and Mycoplasma hominis are commonly found in the urogenital tract of patients experiencing symptoms including infertility, orchitis, epididymitis, prostatitis, and nongonococcal urethritis [1, 2], and in asymptomatic individuals [3]. In recent years, due to the abuse of antibiotics, the resistance of U. urealyticum and M. hominis to antibiotics has been shown to have an increasing trend, reaching an unacceptable level, especially in developing countries [4, 5]. Therefore, it is necessary to monitor the local drug resistance rate of mycoplasmas regularly to provide guidance for the rational use of antibiotics in the clinic. The aim of this study was to investigate the prevalence and antibiotic resistance of U. urealyticum and M. hominis isolated from individuals in Xi’an, China.
Materials and methods
Clinical specimens
Endocervical and vaginal swabs, urethral swabs, and expressed prostatic secretion were obtained from the urogenital tracts of individuals between 18 and 76 (median 30) years old for urogenital mycoplasmas analysis. A total of 2161 individuals (1319 females and 842 males) were analyzed from January 2011 to December 2015. All of the individuals were outpatients attending two teaching hospitals (the First Affiliated Hospital of Xi’an Jiaotong University and the First Affiliated Hospital of Xi’an Medical University) in Xi’an, China, for reasons of gynecologic healthcare screening or the presence of symptoms of urogenital infection.
The individuals were divided into two categories, according to the presence (symptomatic) or absence (asymptomatic) of clear symptoms of genital tract infections (urethral or vaginal discharge, dysuria, urethral irritation, itching, genital lesions, and abdominal pain). There were 1625 individuals (861 females and 764 males) with symptoms and 536 individuals (458 females and 78 males) without symptoms.
Urogenital mycoplasmas culture and antibiotic susceptibility test
A Mycoplasma IST kit (Upper Bio-Tech, Shanghai, China) was used to determine the Mycoplasma identification, semi-quantification, and susceptibility. The inoculation and incubation of the specimens were conducted according to the manufacturer’s guidelines. The Mycoplasma IST strips provided information about the presence or absence of U. urealyticum and M. hominis, an estimate of the density of each organism [>104 colony-forming units (CFU)/mL], and antibiotic susceptibility to josamycin, doxycycline, minocycline, erythromycin, roxithromycin, azithromycin, ofloxacin, norfloxacin, and lincomycin. Briefly, the Mycoplasma IST strips were observed after incubation at 37 °C for 48 h. The red identification wells showed the presence of mycoplasmas, while the red enumeration wells indicated the density of mycoplasmas >104 CFU/mL. The rest of the wells were used for susceptibility testing. There were two concentration assay wells for all antibiotics. No discoloration to red in both of the wells implied sensitivity of the mycoplasma and discoloration to red in both of the wells signified resistance. Mycoplasma was moderately sensitive to the antibiotic tested if the low concentration assay wells turned red. The development or absence of red color on the relevant part of the strips provided an index of resistance or susceptibility to each antibiotic.
Statistical analysis
The Chi-squared test or Fisher’s exact test was used to compare the occurrence of strains susceptible or resistant to different antibiotics. A p-value < 0.05 was regarded as statistically significant.
Results
Prevalence of U. urealyticum and M. hominis in the urogenital tract
Of the 2161 tested specimens, 1018 (47.11 %) were positive for urogenital mycoplasma. In these positive specimens, 772 (75.83 %) were single positive for U. urealyticum, 66 (6.48 %) were single positive for M. hominis, and 180 (17.68 %) were dually positive for U. urealyticum and M. hominis (Table 1). The incidence of single U. urealyticum infection was far higher than that of single M. hominis or dual U. urealyticum/M. hominis infection. The positive rates of all three patterns of infection showed no significant difference over the 5-year period studied (p > 0.05).
The positive rates of urogenital mycoplasma in the symptomatic and asymptomatic groups were 50.28 and 37.50 %, respectively, and showed a significant difference (p < 0.05). However, the positive rates of all three patterns of infection in the symptomatic and asymptomatic groups did not show a significant difference (p > 0.05).
Distributions of urogenital mycoplasmas in different genders
The positive rates of urogenital mycoplasmas in male and female individuals in each year are shown in Table 2. In females, the total positive rate in the 5 years was 58.76 %. The positive rates of U. urealyticum, M. hominis, and U. urealyticum/M. hominis mixed infection were 46.25, 3.49, and 9.02 %, respectively (data not shown). In males, the total positive rate in the 5 years was 28.86 %. The positive rates of U. urealyticum, M. hominis, and U. urealyticum/M. hominis mixed infection were 19.36, 2.37, and 7.13 %, respectively (data not shown). Neither females nor males showed any significant difference in the positive rates between different years (p > 0.05).
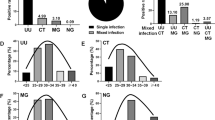
Subgroup analysis according to symptoms show that the positive rates in males and females were 31.02 and 67.36 %, respectively, in the symptomatic group and 7.69 and 42.58 %, respectively, in the asymptomatic group (Fig. 1). The positive rates of both males and females were significantly increased in the symptomatic group compared with the asymptomatic group (both p < 0.001, Fig. 1).
Distributions of urogenital mycoplasmas in different age groups
The total positive rates of urogenital mycoplasmas in the 20 to 25 years, 25 to 30 years, and 40 to 45 years age groups were relatively higher (>45 %) compared with the other age groups, and were drastically decreased in patients older than 50 years. The highest positive rate (48.88 %) was detected in the 25 to 30 years age group (Fig. S1).
The positive rates in both the symptomatic and asymptomatic groups were increased in individuals younger than 30 years and then decreased in individuals older than 35 years. The positive rates in the symptomatic group peaked in the 25 to 30 years age group (54.35 %) and decreased to the lowest level in the 35 to 40 years and older than 50 years age groups (35.19 and 33.33 %, respectively). Different from the symptomatic group, the highest positive rates in the asymptomatic group was observed in the 30 to 35 years age group (57.14 %) and the lowest positive rates were detected in the 18 to 20 years and 45 to 50 years age groups (0 %) (Fig. S2 and Fig. S3).
Antibiotic susceptibility of urogenital mycoplasmas in vitro
Over the 5 years, the resistance rates of U. urealyticum were less than 10 % (0–7.35 %) to josamycin, doxycycline, minocycline, erythromycin, roxithromycin, and azithromycin, and less than 15 % (7.02–13.97 %) to ofloxacin, while they were higher than 60 % (60.91–99.13 %) to norfloxacin and lincomycin (Table 3). The resistance rates of M. hominis were less than 10 % (0–9.90 %) to josamycin, doxycycline, and minocycline, while they were higher than 40 % to norfloxacin and higher than 80 % to erythromycin, roxithromycin, and azithromycin (Table 4). Besides, the resistance rates of M. hominis to ofloxacin and lincomycin showed an upward trend and were higher than 40 % in 2015. The resistance rates of U. urealyticum/M. hominis mixed infection were less than 10 % (0–9.52 %) to josamycin, doxycycline, and minocycline, but higher than 40 % to ofloxacin and higher than 85 % to erythromycin, roxithromycin, azithromycin, norfloxacin, and lincomycin (Table 5).
Further analysis indicated that the resistance rates of U. urealyticum to the tested antibiotics did not show any difference between the symptomatic and asymptomatic groups (p > 0.05, Fig. 2). However, the resistance rates of M. hominis to erythromycin, roxithromycin, azithromycin, and norfloxacin in the symptomatic group (75–100 %) were significantly higher than those in the asymptomatic group (25–55 %, p < 0.001 or 0.05, Fig. 3). The resistance rates of U. urealyticum/M. hominis mixed infection to roxithromycin, azithromycin, and norfloxacin in the symptomatic group (95–99 %) were significantly higher than those in the asymptomatic group (50–75 %, p < 0.001 or 0.05, Fig. 4).
Discussion
In this study, we confirmed that U. urealyticum and M. hominis were commonly detected in urogenital tract specimens. The positive rates of mycoplasmas were in the range 48.40–46.66 % from 2011 to 2015, and no obvious difference was observed among different years. Ureaplasma urealyticum single infection is the most common infection (75.83 %), followed by U. urealyticum/M. hominis mixed infection (17.68 %) and M. hominis single infection (6.48 %). The positive rates of all three patterns of infection showed no significant difference over the 5 years studied. The overall trend of positive rates and distribution of three infection patterns were consistent with previous studies [6–9], but the total positive rates were obviously higher than the results of Serbia and Denmark [6, 7] and lower than the results of Africa and Hangzhou, China [8, 9].
Furthermore, our study confirmed that the positive rate in the symptomatic group (50.28 %) was higher than that in the asymptomatic group (37.50 %). Meanwhile, in these positive individuals, most urogenital mycoplasma infections (80.26 %) may have caused the symptoms and one-fifth of urogenital mycoplasma infection patients (19.74 %) had no symptoms. This finding confirmed that there are inapparent infections of urogenital mycoplasmas and indicates that some infected cases may be missed if the diagnosis of urogenital mycoplasma infections is based on symptoms alone without culture results.
Our study also confirmed that the positive rate in females was much higher than that in males, which is consistent with a recent report in China [8]. Furthermore, in symptomatic and asymptomatic individuals, the positive rates in females were both higher than in males. The ratio of symptom-free persons among positive females was 34.72 %, significantly higher than that in males (9.26 %). This is different from the results reported in Hungary [10]. The positive rate in females was much higher than that in males, with or without symptoms. This may be related to the special physiological structure of the urinary tract, vaginal blood, birth control type, and/or the lack of an estrogen effect of women, especially users of intrauterine devices which may have an increased risk of U. urealyticum and M. hominis colonization, as indicated by another study [3].
Our study also found that a certain relationship existed between mycoplasma infections and the ages of individuals. Previous studies have also reported that age and high-risk sexual behavior were risk factors of urogenital mycoplasma infection [3, 11]. The high positive rates of U. urealyticum and M. hominis are mainly distributed among young people, in particular those aged 18 to 20 years, 25 to 30 years, and 40 to 45 years, in both men and women. However, the infection rates in individuals older than 50 years are drastically decreased. These findings are similar to the results obtained from Serbia, Denmark, Hungary, Italy, Turkey, and Beijing, China [6, 7, 10, 12–14]. Furthermore, our study shows that the positive rates in the symptomatic and asymptomatic groups among the age groups peaked and decreased to the lowest levels in different age groups. The positive rates of the asymptomatic group among those aged 30 to 40 years was >50 %. This finding indicates that it is necessary to have routine screening for urogenital mycoplasma, especially for women aged 30 to 40 years old. All of these results suggest that a certain relationship existed between mycoplasma infections and individuals’ gender and age. However, there is still a lack of proof regarding the causality of urogenital mycoplasma infection and gender and age, and further study is needed.
Mycoplasma lacks cell wall structures, and is insensitive to penicillins, cephalosporins, and other beta-lactam antibiotics. It is not susceptible to sulfonamides or trimethoprim because of its inability to synthesize folic acid. Currently, three kinds of antibiotics, including fluoroquinolones, tetracyclines, and macrolides, are commonly used for treating mycoplasma infections. However, mycoplasma can be resistant to the antibiotics due to the emergence of different resistance mechanisms [1, 10, 15]. We demonstrate that the susceptibility profiles do not show significant changes over the study period. Ureaplasma urealyticum, M. hominis, and U. urealyticum/M. hominis mixed infection all displayed relatively lower resistance rates to josamycin, doxycycline, and minocycline (<10 %), but relatively higher resistance rates to the other tested antibiotics. Moreover, we found that the resistance rates of U. urealyticum/M. hominis mixed infection isolates to most of the tested antibiotics were higher than that of U. urealyticum or M. hominis single infection isolates, indicating that the U. urealyticum/M. hominis coinfection will be more difficult to treat than U. urealyticum or M. hominis single infection. The resistance rates of U. urealyticum to ofloxacin were obviously lower than the recently published result in Hangzhou and Changzhou, China [11, 16]. There are geographical differences among antibiotic resistance, and, therefore, local susceptibility testing in vitro is recommended to avoid failure of therapy [17].
Moreover, our study indicates that all the U. urealyticum, M. hominis, and U. urealyticum/M. hominis mixed isolates from symptomatic and asymptomatic patients with genital infections are highly sensitive to josamycin, doxycycline, and minocycline, while the resistant rates of M. hominis to the other tested antibiotics in the asymptomatic group were significantly lower than that in the symptomatic group. Especially, the resistant rates of M. hominis to ofloxacin and norfloxacin are less than 30 %. It is suggested that ofloxacin and norfloxacin may also be selected for the asymptomatic M. hominis infection.
In conclusion, this study demonstrated that females and individuals with symptoms and younger age had higher mycoplasma infection rates. Josamycin, doxycycline, and minocycline may be recommended for the clinical treatment of patients infected with U. urealyticum, M. hominis, and U. urealyticum/M. hominis with or without symptoms, and ofloxacin and norfloxacin can be used as candidate drugs for the clinical treatment of asymptomatic M. hominis infection in Xi’an, China.
References
Leli C, Mencacci A, Bombaci JC, D’Alò F, Farinelli S, Vitali M, Montagna P, Bietolini C, Meucci M, Perito S, Bistoni F (2012) Prevalence and antimicrobial susceptibility of Ureaplasma urealyticum and Mycoplasma hominis in a population of Italian and immigrant outpatients. Infez Med 20(2):82–87
Zhang N, Wang R, Li X, Liu X, Tang Z, Liu Y (2014) Are Ureaplasma spp. a cause of nongonococcal urethritis? A systematic review and meta-analysis. PLoS One 9(12):e113771. doi:10.1371/journal.pone.0113771
Tibaldi C, Cappello N, Latino MA, Masuelli G, Marini S, Benedetto C (2009) Vaginal and endocervical microorganisms in symptomatic and asymptomatic non-pregnant females: risk factors and rates of occurrence. Clin Microbiol Infect 15(7):670–679. doi:10.1111/j.1469-0691.2009.02842.x
Azizmohammadi S, Azizmohammadi S (2015) Antimicrobial susceptibility patterns of Ureaplasma urealyticum and Mycoplasma hominis isolated from pregnant women. Iran Red Crescent Med J 17(12):e17211. doi:10.5812/ircmj.17211
Okeke IN, Laxminarayan R, Bhutta ZA, Duse AG, Jenkins P, O’Brien TF, Pablos-Mendez A, Klugman KP (2005) Antimicrobial resistance in developing countries. Part I: recent trends and current status. Lancet Infect Dis 5(8):481–493. doi:10.1016/S1473-3099(05)70189-4
Skiljevic D, Mirkov D, Vukicevic J (2016) Prevalence and antibiotic susceptibility of Mycoplasma hominis and Ureaplasma urealyticum in genital samples collected over 6 years at a Serbian university hospital. Indian J Dermatol Venereol Leprol 82(1):37–41. doi:10.4103/0378-6323.172903
Salado-Rasmussen K, Jensen JS (2014) Mycoplasma genitalium testing pattern and macrolide resistance: a Danish nationwide retrospective survey. Clin Infect Dis 59(1):24–30. doi:10.1093/cid/ciu217
Song T, Ye A, Xie X, Huang J, Ruan Z, Kong Y, Song J, Wang Y, Chen J, Zhang J (2014) Epidemiological investigation and antimicrobial susceptibility analysis of Ureaplasma species and Mycoplasma hominis in outpatients with genital manifestations. J Clin Pathol 67(9):817–20. doi:10.1136/jclinpath-2014-202248
Kouegnigan Rerambiah L, Ndong JC, Medzegue S, Elisee-Ndam M, Djoba Siawaya JF (2015) Genital Mycoplasma infections and their resistance phenotypes in an African setting. Eur J Clin Microbiol 34(6):1087–1090. doi:10.1007/s10096-015-2326-9
Pónyai K, Mihalik N, Ostorházi E, Farkas B, Párducz L, Marschalkó M, Kárpáti S, Rozgonyi F (2013) Incidence and antibiotic susceptibility of genital Mycoplasmas in sexually active individuals in Hungary. Eur J Clin Microbiol Infect Dis 32(11):1423–1426. doi:10.1007/s10096-013-1892-y
Ye G, Jiang Z, Wang M, Huang J, Jin G, Lu S (2014) The resistance analysis of Ureaplasma urealyticum and Mycoplasma hominis in female reproductive tract specimens. Cell Biochem Biophys 68(1):207–210. doi:10.1007/s12013-013-9691-8
De Francesco MA, Caracciolo S, Bonfanti C, Manca N (2013) Incidence and antibiotic susceptibility of Mycoplasma hominis and Ureaplasma urealyticum isolated in Brescia, Italy, over 7 years. J Infect Chemother 19(4):621–627. doi:10.1007/s10156-012-0527-z
Bayraktar MR, Ozerol IH, Gucluer N, Celik O (2010) Prevalence and antibiotic susceptibility of Mycoplasma hominis and Ureaplasma urealyticum in pregnant women. Int J Infect Dis 14(2):e90–e95. doi:10.1016/j.ijid.2009.03.020
Wang QY, Li RH, Zheng LQ, Shang XH (2014) Prevalence and antimicrobial susceptibility of Ureaplasma urealyticum and Mycoplasma hominis in female outpatients, 2009–2013. J Microbiol Immunol Infect 49(3):359–362. doi:10.1016/j.jmii.2014.06.007
Samra Z, Rosenberg S, Dan M (2011) Susceptibility of Ureaplasma urealyticum to tetracycline, doxycycline, erythromycin, roxithromycin, clarithromycin, azithromycin, levofloxacin and moxifloxacin. J Chemother 23(2):77–79. doi:10.1179/joc.2011.23.2.77
Zhu C, Liu J, Ling Y, Dong C, Wu T, Yu X, Hou Y, Dong L, Cheng X (2012) Prevalence and antimicrobial susceptibility of Ureaplasma urealyticum and Mycoplasma hominis in Chinese women with genital infectious diseases. Indian J Dermatol Venereol Leprol 78(3):406–407. doi:10.4103/0378-6323.95480
Okeke IN, Klugman KP, Bhutta ZA, Duse AG, Jenkins P, O’Brien TF, Pablos-Mendez A, Laxminarayan R (2005) Antimicrobial resistance in developing countries. Part II: strategies for containment. Lancet Infect Dis 5(9):568–580. doi:10.1016/S1473-3099(05)70217-6
Acknowledgments
The project was sponsored by the National Natural Science Foundation of China (no. 81201326).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics statements
This study was initiated after receiving approval from the Institutional Review Board of the First Affiliated Hospital of Xi’an Jiaotong University and carried out according to the Helsinki Declaration. Informed consent was obtained from all the participants.
Rights and permissions
About this article
Cite this article
Zeng, XY., Xin, N., Tong, XN. et al. Prevalence and antibiotic susceptibility of Ureaplasma urealyticum and Mycoplasma hominis in Xi’an, China. Eur J Clin Microbiol Infect Dis 35, 1941–1947 (2016). https://doi.org/10.1007/s10096-016-2745-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-016-2745-2