Abstract
Staphylococcus aureus bacteremia (SAB) is a fatal disease. We aimed to describe risk factors for long-term mortality with SAB. We analyzed data from a retrospectively collected database including 1,692 patients with SAB. We considered variables of infection and background conditions for the analysis of long-term survival. The Kaplan–Meier procedure was used for analysis of long-term survival. Variables significantly associated with mortality were analyzed using a Cox regression model. We included 1,692 patients in the analysis. Patients were followed for up to 22 years. Within one year, 62% of patients died and within 5 years 72% died. A total of 82% of patients aged 65 years and older died within 5 years. Independent predictors of long-term mortality were older age (Hazard ratio 1.029, 95% confidence interval 1.022–1.036), female gender (HR 1.302, 95% CI 1.118–1.517), pneumonia or primary/ unknown source of infection (HR 1.441, 95% CI 1.230–1.689), dementia (HR 1.234, 95% CI 1.004–1.516), higher Charlson score (HR 1.155, 95% CI 1.115–1.196), shock at onset (HR 1.776, 95% CI 1.430–2.207) and arrival to hospitalization from an institution (HR 1.319, 95% CI 1.095–1.563). Long-term survival of patients older than 65 years and of women with SAB is severely curtailed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Staphylococcus aureus is a leading cause of bacteremia. The annual incidence of S. aureus bacteremia (SAB) in the United States is 4–38 per 100,000 person-years. Increased frequency of invasive surgery, use of intravascular devices and immunosuppression has led to a sharp increase in the incidence of SAB in recent years. SAB is a fatal disease with 30-day all-cause mortality of 20–30% [1–3], 90-day mortality of 36% [4, 5] and increased mortality rates thereafter [6].
Several studies reported risk factors for mortality in SAB. Most of them reported in hospital mortality, or 30 days and up to 90 days mortality. Few studies reported 1-year mortality and longer [6–8]. Risk factors for 30–90 days mortality include age, female gender, comorbidities (diabetes and malignancy), functional status [3], residence in a nursing home [9], pulmonary source or endocarditis [3], unknown source [5], severe sepsis or septic shock at presentation [3, 5], metastatic or complicated SAB infection, methicillin resistance [3, 4], inappropriate empiric antibiotic [10], and higher vancomycin MICs [11].
However, data on long-term survival beyond 90 days are scarce. Thus, we aimed to define risk factors for long-term mortality in patients followed for up to 22 years.
Methods
Data collection
The study was conducted at Rabin Medical Center, Beilinson Hospital, a 900-bed primary and tertiary care university hospital. The hospital has four main intensive care units (ICUs) and small ICUs in which patients undergo mechanical ventilation within the six internal medicine and two surgical departments.
All consecutive adult patients who had been diagnosed with S. aureus bacteremia were identified. Data were collected retrospectively between January 1, 1999 and December 31, 2010 by reviewing patients’ medical records and were merged with data prospectively collected between April 1, 1988 and September 30, 1994. Results from these databases have been previously described [10, 12]. The study was approved by the local ethical committee.
Contaminants (S. aureus isolated in a single set of blood cultures with no evidence of systemic inflammatory response syndrome within 48 h of blood culture, in which the patient was not treated and did not die within 30 days) were excluded. Patients were included only once, for the first episode of S. aureus bacteremia. Patients were followed up until October 2010. Survival data were obtained from the national health of ministry database.
Microbiology
Two separate sets of blood cultures were obtained routinely (up to 6 sets if endocarditis was suspected), with 10 mL of blood collected in one aerobic and one anaerobic bottle. The Bactec 460 was used between 1988 and 1992 and the Bactec 9240 microbial system (Becton Dickinson, Franklin Lakes, NJ) after 1992. Isolates were identified on the basis of morphologic features and API system. Susceptibility to antibiotics was tested by the disk diffusion method on Mueller-Hinton agar, according to Clinical and Laboratory Standards Institute (CLSI) procedures. Repeated blood cultures were obtained until results were negative.
Definitions
Health care associated infections were defined as any of the following: previous hospitalization of ≥2 days during previous 90 days; clinic visit during previous 30 days; home IV therapy or chemotherapy or wound treatment during previous 30 days; or patients arriving from long-term care facilities. Hospital-acquired infections were defined as presentation of infection after >48 h of hospitalization. Other infections were considered community acquired. Central line associated infections were defined as any SAB without an obvious source other than the catheter and if the catheter was removed then positive culture of the catheter tip. An infectious diseases expert determined the source of infection per each episode. Appropriate empirical antibiotic treatment was defined as matching in vitro susceptibility of the S. aureus isolate and given within 48 h after obtaining blood culture, except for single use of aminoglycoside or rifampicin treatment.
Statistical analysis
Analyses were performed using the Statistical Package for the Social Sciences (SPSS Inc., Chicago, IL, USA). For survival analysis we used the Kaplan Meier procedure. Variables significantly associated with mortality in Kaplan Meier were analyzed using the Cox regression model. Goodness of fit of the model was evaluated using the chi-square test.
Results
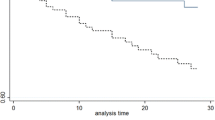
We included 1,692 patients in the analysis. The mean age at onset of bacteremia was 69.7 years (standard deviation 16.4; median 73 years, interquartile range 61–81); 59.5% (1,006 patients) were males. During the entire follow up, 1276 patients (75%) died. Within one year, 1050 patients (62%) died and within 5 years of follow up 1,215 patients (72%) died (Fig. 1).
Variables found to be significant risk factors for long-term mortality in Kaplan–Meyer analysis (Table 1) were advanced age, female sex, inappropriate empirical antibiotic therapy in the first 48 h, source of infection pneumonia or primary/ unknown, diabetes, dementia, oxacillin resistance, higher Charlson score, shock at presentation and arrival to current hospitalization from an institution (another hospital or long-term care facility). On multivariate analysis variables remaining significant were age, female gender, pneumonia or primary/ unknown source of infection, dementia, Charlson score, shock at onset and arrival to hospitalization from an institution (Table 2). Oxacillin resistance and inappropriate empirical antibiotics were both inserted into the analysis, because no significant correlation was found between these two variables using Pearson correlation test. However, both of them were non-significant on multivariate analysis. Goodness of fit of the model was acceptable based on the chi-square test.
Among 1,018 patients who were alive at day 30, univariate analysis demonstrated similar risk factors as in the entire group, excluding age and presentation with shock, which were non-significant in this analysis (Table 1). Multivariate analysis showed age, Charlson score and arrival from an institution to be significantly associated with long-term mortality in multivariate analysis (Table 2).
Mortality rates among patients aged 65 and older were significantly higher compared to younger patients at any time point. One year mortality and 5 years mortality in older patients reached 71.8% and 81.9%, respectively, compared to 41.6% and 50.0%, in younger patients (p < 0.001).
We sought for an explanation of the differences between men and women. Baseline and infection characteristics between men and women were similar except for arrival from home and independent functional capacity, which were significantly more frequent in men. Women were less likely to undergo transesophageal echocardiography (TEE) than men (Table 3). Other management variables were not significantly different between females and males (including appropriate antibiotic therapy, infectious diseases consultation (Table 3), surgical procedures, drainage and debridement procedures, valve replacement or any foreign body removal and performance of transthoracic echo (data not shown).
Discussion
In a cohort of 1,692 patients with SAB the mortality rate was 62% at one year and 72% at 5 years. We found significantly higher mortality in females compared to males at any time point of follow up (Fig. 2) and in patients 65 years and older compared to younger patients (Fig. 3).
Significant risk factors for long-term mortality in multivariate analysis included age, female gender, pneumonia or primary/ unknown source of infection, dementia, Charlson score, shock at onset and arrival to hospitalization from an institution. In patients surviving 30 days the infection-related risk factors no longer mattered, and significant risk factors for mortality were age, Charlson score and arrival from an institution.
High mortality rates beyond 30 days have been previously reported in SA infections: one year mortality in SA infections in general was reported 32% for MSSA and 51% for MRSA [13]; one-year mortality in SAB was reported 38–48% [7, 8]; 5-year mortality was reported as 67% in ICU patients with MRSA bacteremia [14]; and 14 years mortality reached 66% in a study conducted by Yaw et al. [6].
Our results concerning risk factors for long-term mortality are consistent with previous studies evaluating either long- or short-term mortality in SAB [3]. Background conditions previously descried as risk factors for long-term mortality include older age [6, 7], malignancy [7, 8], higher Charlson score [6] and low BMI [8]. Infection-related risk factors include inappropriate therapy [8], pulmonary or unknown source of infection [7], higher SAPS II score, community-acquired infection and MRSA infection [6].
Female gender has been previously reported to be associated with higher short-term mortality rates in SAB, either due to MRSA [10, 15–17] or any SAB [18, 19], in spite of the higher longevity of females in the general population. Possible reasons have been suggested to explain the increased mortality in females with SAB. These include different health-seeking behaviors among women, higher MRSA rates (not shown in our cohort) and hormonal differences [3]. The low rates of use of cardiac echo in women in our cohort should promote searching for other management variables that might not have been captured in our study.
This study had several limitations. It is a single center retrospective study. We did not collect data on the time interval from initiation of infection until admission to hospital, thus we cannot assess the relation between health-seeking behaviors to mortality. Explanations for the higher mortality in women were searched post-hoc and were not a part of the hypotheses that drove collection of data.
In conclusion, patient-related risk factors for long-term mortality in our cohort of SAB patients were older age, female gender, higher comorbidity score, dementia and arrival from an institution. Infection-related risk factors that drove long-term mortality in these patients with SAB were septic shock at presentation and pneumonia as source of infection, or unknown source of infection. These patients merit special attention.
Women had a worse long-term prognosis than men, in contrast with their better survival in the population at large. Dedicated data collection should address the explanation of this phenomenon, especially if management of infection is different.
SAB in people older than 65 is a sign of a dire 3-year survival. These patients should be under close surveillance even if they have survived the acute episode, and the long-term trajectory of elderly survivors of SAB should be better described and investigated.
References
Holland TL, Arnold C, Fowler VG Jr (2014) Clinical management of Staphylococcus aureus bacteremia: a review. JAMA 312:1330–1341
Khatib R, Sharma M (2013) Echocardiography is dispensable in uncomplicated Staphylococcus aureus bacteremia. Medicine (Baltimore) 92:182–188
van Hal SJ, Jensen SO, Vaska VL et al (2012) Predictors of mortality in Staphylococcus aureus Bacteremia. Clin Microbiol Rev 25:362–386
Kobayashi D, Yokota K, Takahashi O et al (2014) A predictive rule for mortality of inpatients with Staphylococcus aureus bacteraemia: A classification and regression tree analysis. Eur J Intern Med 25:914–918
Paulsen J, Mehl A, Askim A et al (2015) Epidemiology and outcome of Staphylococcus aureus bloodstream infection and sepsis in a Norwegian county 1996–2011: an observational study. BMC Infect Dis 15:116
Yaw LK, Robinson JO, Ho KM (2014) A comparison of long-term outcomes after methicillin-resistant and methicillin-sensitive Staphylococcus aureus bacteraemia: an observational cohort study. Lancet Infect Dis 14:967–975
Fatkenheuer G, Preuss M, Salzberger B et al (2004) Long-term outcome and quality of care of patients with Staphylococcus aureus bacteremia. Eur J Clin Microbiol Infect Dis 23:157–162
Hanses F, Spaeth C, Ehrenstein BP et al (2010) Risk factors associated with long-term prognosis of patients with Staphylococcus aureus bacteremia. Infection 38:465–470
Pastagia M, Kleinman LC, Lacerda de la Cruz EG et al (2012) Predicting risk for death from MRSA bacteremia. Emerg Infect Dis 18:1072–1080
Paul M, Kariv G, Goldberg E et al (2010) Importance of appropriate empirical antibiotic therapy for methicillin-resistant Staphylococcus aureus bacteraemia. J Antimicrob Chemother 65:2658–2665
Hidayat LK, Hsu DI, Quist R et al (2006) High-dose vancomycin therapy for methicillin-resistant Staphylococcus aureus infections: efficacy and toxicity. Arch Intern Med 166:2138–2144
Paul M, Zemer-Wassercug N, Talker O et al (2011) Are all beta-lactams similarly effective in the treatment of methicillin-sensitive Staphylococcus aureus bacteraemia? Clin Microbiol Infect 17:1581–1586
Haessler S, Mackenzie T, Kirkland KB (2008) Long-term outcomes following infection with meticillin-resistant or meticillin-susceptible Staphylococcus aureus. J Hosp Infect 69:39–45
Ho KM, Robinson JO (2009) Risk factors and outcomes of methicillin-resistant Staphylococcus aureus bacteraemia in critically ill patients: a case control study. Anaesth Intensive Care 37:457–463
Lamagni TL, Potz N, Powell D et al (2011) Mortality in patients with methicillin-resistant Staphylococcus aureus bacteraemia, England 2004–2005. J Hosp Infect 77:16–20
Maor Y, Hagin M, Belausov N et al (2009) Clinical features of heteroresistant vancomycin-intermediate Staphylococcus aureus bacteremia versus those of methicillin-resistant S. aureus bacteremia. J Infect Dis 199:619–624
Seybold U, Kourbatova EV, Johnson JG et al (2006) Emergence of community-associated methicillin-resistant Staphylococcus aureus USA300 genotype as a major cause of health care-associated blood stream infections. Clin Infect Dis 42:647–656
Allard C, Carignan A, Bergevin M et al (2008) Secular changes in incidence and mortality associated with Staphylococcus aureus bacteraemia in Quebec, Canada, 1991–2005. Clin Microbiol Infect 14:421–428
Mansur N, Hazzan R, Paul M et al (2012) Does sex affect 30-day mortality in Staphylococcus aureus bacteremia? Gend Med 9:463–470
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The current analysis was supported in part by a grant from the Israeli Ministry of Science, Technology and Space GA 3–12075. MP and LL are members of the European Society of Clinical Microbiology and Infectious Diseases—Study Group for Infections in the Elderly (ESGIE), and they would like to thank ESGIE members for their stimulating support to write this manuscript.
Conflict of interest
All authors declare no competing interests.
Ethical approval
The study was approved by the local ethical committee.
Informed consent
No informed consent was required for this retrospective study.
Rights and permissions
About this article
Cite this article
Yahav, D., Yassin, S., Shaked, H. et al. Risk factors for long-term mortality of Staphylococcus aureus bacteremia. Eur J Clin Microbiol Infect Dis 35, 785–790 (2016). https://doi.org/10.1007/s10096-016-2598-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-016-2598-8