Abstract
A brain abscess is a life-threatening infection, frequently with serious sequelae. Culture-based methods present many limitations and do not enable an exhaustive documentation of the bacterial flora. 16S rRNA-based amplification, cloning, and high-throughput sequencing have dramatically increased the number of identified agents of brain abscesses, showing that the causative flora is polymicrobial in up to 40 % of cases, with the presence of at least one anaerobic bacterium. In contrast, real-time polymerase chain reaction (PCR) technology is an appealing alternative to culture-based methods for diagnosing brain abscesses due to its speed, sensitivity, and specificity. Molecular typing is available for several bacterial and fungal genera, and this user-friendly tool is accessible for the clinical microbiology laboratory to diagnose microbes involved in a brain abscess. This article reviews the applications of the currently available tools for the etiological diagnosis of a brain abscess.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A brain abscess is a focal, intracerebral infection that begins as a localized area of cerebritis and develops into a collection of pus surrounded by a well-vascularized capsule [1]. The medical management of this condition remains mostly empirical because of a lack of comprehensive knowledge of the causative microorganisms. Bacteria are the main recognized agents, but fungi and parasites may be involved. Brain abscesses occur at all ages, more frequently in men than in women [2]. In children and adolescents, bacterial intracerebral abscess is a particularly serious condition, often leading to permanent neurological sequelae or death [3]. Sequelae have been reported in 9–36 % of patients [2, 4].
Brain abscesses may develop when the integrity of the central nervous system is compromised (trauma or neurosurgery) in 8–19 % of cases [2], by direct extension from a contiguous suppurative focus (sinuses, middle ear, mastoids, or oral cavity) in 25–50 % of cases [1], or by hematogenous dissemination from a distant focus in 15–30 % of cases [5]. Despite all these potential routes, 20–30 % of cases, for which no obvious source can be identified, are classified as cryptic brain abscesses [1, 5]. Etiologic organisms vary according to the source of infection as well as the immunocompetence of the host [1, 6]. Polybacterial infections are frequent when a brain abscess results from sinusitis or a dental abscess [6], and Streptococcus, Bacteroides, Prevotella, Porphyromonas, and Fusobacterium species as well as Micrococcus micros are frequently associated with brain abscesses in these cases [6]. In addition, many as yet uncultured bacteria were also detected in this situation [6]. Brain abscesses of otogenic origin mostly associate Streptococcus species, Bacteroides species, Pseudomonas aeruginosa, and Enterobacteriaceae. The most commonly reported bacterial agents for brain abscesses are Staphylococcus aureus in the case of metastatic spread; viridans streptococci from endocarditis; Enterobacteriaceae and Pseudomonaceae from urinary tract infections; Streptococcus species, Enterobacteriaceae, and anaerobes from intra-abdominal infections; and Streptococcus, Fusobacterium, and Actinomyces species from lung abscesses. In brain abscesses resulting from a penetrating trauma, S. aureus, Clostridium species, and Enterobacteriaceae are the most common causative agents. The most common agents associated with brain abscesses following neurosurgical procedures are S. aureus, Staphylococcus epidermidis, Enterobacteriaceae, and Pseudomonaceae. Immunocompromised patients with human immunodeficiency virus (HIV) infections, organ transplantation, chemotherapy, or steroid use suffer infections caused by Nocardia, Staphylococcus, Streptococcus, and Pseudomonas species [6].
Using culture, polymicrobial infections are reported to represent only 11–18 % of brain abscesses [6]. However, molecular techniques showed that these infections represent 40 % of all brain abscess samples, with at least one anaerobic bacterium being isolated in every case of polybacterial brain abscess [6]. Currently, more than 147 bacterial taxa have been reported to cause brain abscess, including 29 reported for the first time using a metagenomic approach [6–8]. These include 129 known species from 48 genera and 18 as yet unclassified and uncultured bacteria (Table 1).
Overall, the most common bacterial species causing brain abscesses in the literature was Streptococcus intermedius, detected in 35 % of brain abscess samples [9]. In the past 20 years, major advances in the diagnosis and management of brain abscesses have contributed to the decrease in associated morbidity and mortality [9]. These include progresses in radiographic scanning, the availability of new antimicrobials, and the development of novel surgical techniques [9]. However, brain abscesses remain associated with a high mortality and a clinically significant neurological morbidity [6].
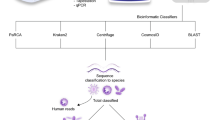
A rapid and accurate identification of an infectious source of a brain abscess is required in order to rationally and appropriately utilize an antibiotic therapy to reduce mortality and sequelae. Therefore, a close coordination between neurosurgeons, infectious diseases specialists, and microbiologists is increasingly important in the management of this disease [1]. In this review, we survey the various diagnostic methods used for brain abscesses (Fig. 1) and discuss their advantages and pitfalls.
Methods used for the diagnosis of brain abscess are mainly based on refined traditional methods, such as: (i) culture and microscopy; (ii) matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF): spectra generated directly from organisms are compared to a reference database of spectra from known organisms and the results are reported based on the confidence of the spectral match; (iii) 16S ribosomal DNA (rDNA)-based identification: this method increased the number of bacterial species isolated from brain abscesses as compared with standard culture; (iv) DNA fingerprinting approaches [chromosomal restriction fragment length polymorphism (RFLP), randomly amplified polymorphic DNA (RAPD), multilocus sequence typing (MLST), and repetitive sequence-based polymerase chain reaction (rep-PCR)] that depend on the combination of rapid nucleic acid amplification and detection methods to facilitate real-time analysis; and, more recently, on the specific and high-sensitive detection methods such as (v) real-time PCR and multiplex PCR that are able to detect and differentiate a wide range of Gram-negative and Gram-positive bacteria involved in brain abscesses
Culture-based identification of brain abscess pathogens
Culture remains widely used as a first-line tool for the diagnosis of infectious diseases, despite significant limitations, such as issues surrounding the transportation of samples, indeterminate and presumptive quantification and identification, and demanding techniques [10, 11]. In brain abscesses, culture is mostly performed from pus collected by neurosurgical drainage [12]. However, culture from brain abscess pus has been reported to be negative in 9–63 % of cases [4, 10]. A comparative study has been conducted by using both culture- and molecular-based techniques to evaluate the bacterial spectrum of brain abscesses [6]. The results demonstrated that culture was unable to detect a substantial proportion of the bacterial population that is fastidious or nonculturable. In addition, culture may be impaired by one of the following problems: (i) administration of an empirical antibiotic therapy prior to specimen collection may prevent bacterial growth; culture can yield misleading answers in polymicrobial infections if the administered antibiotics have affected the involved bacteria unequally, permitting some to grow but not others. In previous studies, molecular methods have detected a significantly larger number of species than culture-based methods for patients who had received antibiotics prior to sample collection [6, 13]. (ii) The nutritional media used for sample transportation and the general conditions of transportation may also influence culture results. Species that grow well on the medium used during transport can continue to grow until the sample is finally plated for culture. This growth then results in a significant change in the balance of microbes identified, since some species can actively grow at the expense of others. Another factor is the atmosphere used to incubate the pus specimen. This is especially the case for anaerobic bacteria, as they require adequate transport conditions and special care in the laboratory, which are not systematically achieved [6]. In many studies, anaerobic bacteria have only been detected by molecular methods [6, 13]. Most bacteria grow within 24 to 72 h. However, some fastidious bacteria need more than 5 days to be cultivated [7]. As an example, Nocardia isolation and susceptibility testing may take weeks and, due to time-consuming phenotypic methods, nocardiosis is sometimes diagnosed after the dissemination of the strain to other organs or after the patient’s death [14]. In addition, in polymicrobial infections, fastidious bacteria such as Mycoplasma spp. may be overgrown in culture by faster-growing organisms. It is also widely accepted that plate culturing techniques may only reveal a small fraction of the true microbial population, primarily due to an inability to detect organisms that might not be cultivable with existing media, requirement of a specific temperature, phases of metabolic activity, or are viable but enter a noncultivable state [15]. A recent study of the digestive flora using more than 200 culture conditions through ‘culturomics’ enabled the identification of more than 30 new bacterial species [16] and could be of benefit to the study of brain abscess pathogens. (iii) An additional problem related to culture methods is the isolation and identification of predominant bacteria and not the other species which are present in lower concentrations. This may also be due to difficulties in differentiating colonies on agar plates. Contamination of the culture medium is one of the most frustrating problems faced by laboratories and physicians [17]. Poor plating techniques or the use of an improper device for plating the collected samples can contaminate the culture medium by external bacteria or fungi. Such contamination can result in a misleading documentation of brain abscess samples [17].
Regarding the identification of cultivated pathogens, in most brain abscess studies, bacterial identification was obtained using manual or automated biochemical tests. However, conventional methods sometimes fall short when microorganisms exhibit unusual phenotypic profiles or express few of the phenotypic criteria characterized by these methods, as is the case for Mycoplasma species. In contrast, recent advances in matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF) mass spectrometry (MS) bacterial identification and DNA sequencing technology have greatly enhanced the ability of microbiologists to precisely identify a bacterial isolate [18]. Arends et al. used this technique to report a brain abscess case attributable to Nocardia veterana infection [19]. Currently, intriguing MALDI-TOF MS developments are being made regarding the phenotypic detection of certain antibiotic resistance mechanisms and could serve as a promising approach in brain abscess cases. Molecular biology-based diagnosis seems to be a modern research tool that may become the technique of choice in the diagnosis of brain abscesses due to the inevitable limitations of culture-based detection methods.
Molecular detection of brain abscess pathogens
Direct 16S ribosomal DNA (rDNA) gene amplification and sequencing
In recent years, molecular methods have been increasingly used to detect microorganisms in clinical samples, including those from patients with central nervous system infections. In particular, polymerase chain reaction (PCR) assays targeting conserved regions of the 16S rDNA gene were useful in identifying fastidious, uncultivable, and novel microorganisms from various clinical specimens [20]. Several studies suggested that broad-range bacterial PCR may be the only method to yield an etiological diagnosis of brain abscesses in several situations, including the administration of antibiotics prior to sampling or the presence of fastidious pathogens [21–24].
This technology also offers a more rapid and accurate identification of microbes causing brain abscesses than standard phenotypic methods that depend on bacterial growth [21–24]. As an example, rDNA PCR-based diagnosis of brain abscess caused by S. intermedius, Nocardia spp. or mycobacteria infections [22], and fastidious organisms, such as Mycoplasma hominis, Gemella morbillorum, Aggregatibacter aphrophilus, and Fusobacterium necrophorum, can be achieved in one day, compared with microbiological isolation that may take weeks [24]. In addition, molecular detection identified streptococci in children who received antibiotics prior to brain abscess puncture [22].
However, a major drawback of conventional 16S rDNA sequencing is that it is inadequate to discriminate among taxa in clinical specimens that contain mixed floras. The limited sensitivity of direct 16S rDNA gene sequencing and identification of microbes involved in brain abscesses is another drawback that can never exclude the presence of bacterial DNA in a brain abscess pus sample [6, 25]. In addition, some samples produce false-negative results due to the low concentration of bacteria and the presence of inhibitory substances. Beta-globin gene amplification has been recommended to check the efficiency of DNA extraction and the possible presence of inhibitors in the brain abscess samples that exhibit negative PCR amplification [8].
16S rDNA gene cloning and sequencing
PCR amplification of the 16S rDNA gene, followed by cloning and sequencing, has resulted in improved identification of the bacterial diversity from clinical and environmental samples [26–30]. When applied to brain abscess specimens, this approach dramatically increased the number of identified agents of cerebral abscesses, showing that the flora is significantly more variable than that characterized by culture or regular PCR [6, 7]. High-throughput analyses of the bacteria associated with brain abscesses showed that the flora was even more variable than expected, including 27 bacterial species found in cerebral abscesses that had not been previously reported [6]. Moreover, this approach permitted the identification of two novel bacteria, the first being a novel Staphylococcus species [8] and the second being a novel anaerobic bacterium that represents a novel species in a new genus within the phylum Bacteroidetes [7]. The observed increase in the repertoire of brain abscess-causative organisms can be linked to molecular evidence for fastidious, but yet uncultured, organisms. Fifteen sequences matched contemporarily uncultured bacteria that had previously been characterized among anaerobic periodontal and intestinal flora [6].
The results of 16S rDNA gene cloning and sequencing could be influenced by many factors. Amplification of DNA from mixed bacterial templates may result in the formation of molecular artifacts that do not truly represent any valid organism [31]. In addition, chimeric molecules may be created by broad-range amplification of DNA extracts containing multiple bacterial species. The presence of such molecules can compromise the integrity of sequence databases and cause misinterpretation of the phylogenetic status by the creation of false genetic diversity within prokaryotes. The percentage of chimeric insert in 16S rDNA gene libraries generated from brain abscess samples ranged from 0 to 5 %, depending on the sample [6], and such sequences should be excluded from the libraries using the CHIMERA_ CHECK program of the Ribosomal Database Project II [31]. Another important factor is the number of clones which are necessary to represent the bacterial population present in the sample. Al Masalma et al. suggested that 100 clones might not be enough to enable the identification of all bacterial species present in the samples [6].
Genotyping
Genotyping is an important epidemiological technique for recognizing outbreaks of infection, detecting the cross-transmission of nosocomial pathogens, determining the source of the infection, and recognizing particularly virulent strains of organisms. Outbreaks of infectious disease often result from exposure to a common source of an etiologic agent. Generally, the etiologic agent causing an outbreak is derived from a single cell whose progeny are genetically identical or closely related to the source organism. However, there is sufficient diversity at the species level that organisms isolated from different sources at different times and in different geographical regions may be differentiated or classified into subtype strains. Fingerprinting of bacteria associated with brain abscesses is usually accomplished by the following approaches.
Repetitive sequence-based PCR (rep-PCR)
rep-PCR typing is based on the presence of interspersed multiple copies of short repetitive specific conserved sequences [including repetitive extragenic palindromic (REP) sequences, enterobacterial repetitive intergenic consensus (ERIC) sequences, and BOX elements] found in noncoding regions of microbial genomes, commonly used to genotype isolates and to differentiate highly related strains [32, 33].
rep-PCR typing has been used in several studies to detect brain abscess pathogens. Citrobacter diversus causes sporadic sepsis and meningitis in neonates and young infants, which is usually associated to brain abscesses. Harvey et al. used rep-PCR-generated DNA fingerprints to show that C. diversus causes brain abscesses in infants as a result of vertical transmission from mother to infant in utero [34]. rep-PCR analysis was also used for the identification and subtyping of Bartonella henselae and Bartonella quintana isolates associated with brain abscesses [35].
As commercial typing systems for rep-PCR are available for several bacterial and fungal genera, the use of molecular typing has become a user-friendly tool accessible to the clinical microbiology laboratory. The DiversiLab system (bioMérieux, Marcy l’Etoile, France) offers a complete package for typing and data analysis, incorporating database libraries for organisms. This system could be used for the detection of microbes such as methicillin-resistant S. aureus, Enterococcus faecium, and Clostridium difficile involved in brain abscesses.
PCR-restriction fragment length polymorphism (PCR-RFLP)
RFLP analysis has been used successfully to establish the taxonomic relatedness and identification of several bacteria in clinical specimens [36]. PCR-RFLP was used in few brain abscess studies, including cases of Nocardia otitidiscaviarum [37], Tsukamurella tyrosinosolvens [38], and Streptococcus constellatus infections [39]. The study conducted by Pelaez et al. was based on RFLP analysis of 16S rDNA genes, which enabled the identification and comparison of the different isolates of N. otitidiscaviarum from the brain abscess patient [37]. Accurate identification of Tsukamurella by phenotypic methods is difficult, as it may lead to the diagnosis of Corynebacterium, Rhodococcus, Nocardia, Gordonia, or Mycobacterium species [38]. Moreover, 16S rDNA gene sequences have not been found to be discriminative enough for the correct identification of Tsukamurella species, because of small differences in the 16S rDNA gene sequence. Sheng et al. showed that RFLP analysis of the hsp65 gene of Tsukamurella is useful for the identification of T. tyrosinosolvens involved in brain abscesses [38]. Even though PCR-RFLP is cheap and accessible to many laboratories, it has some drawbacks, including a lack of discriminatory power, databases to be compared to, and it has low reproducibility and interlaboratory comparability.
Randomly amplified polymorphic DNA-PCR (RAPD-PCR)
RAPD assays are based on the use of short random sequence primers which hybridize with sufficient affinity to chromosomal DNA sequences at low stringency conditions. The number and location of these random primer sites vary for different bacterial species or strains, and the banding pattern reflects the particular bacterial species or strain [40].
Marques da Silva et al. [39] reported a brain abscess case caused by S. constellatus in an immunocompromised patient by using RAPD and RFLP techniques. The source of infection was suspected to be oral infection (periodontitis). This study also demonstrated that gene transfer by genetic recombination in the periodontal pocket was responsible for the emergence of a strain variant of S. constellatus that had the potential to cause an abscess at a distant site (brain). RAPD was used to compare this strain to Nocardia species and isolates recovered from different brain abscess patients for epidemiological study, investigating the sources of outbreaks and the relatedness of isolates from the same patient [41].
Multilocus sequence typing (MLST)
MLST is a genotyping method that detects allelic variation at multiple housekeeping loci that are accumulating very slowly in the bacterial population [42]. The procedure characterizes isolates of bacterial species using the DNA sequences of internal fragments of multiple (usually six) housekeeping genes. Approximately 450–500-bp internal fragments of each gene are used, as these can be accurately sequenced on both strands using an automated DNA sequencer [42]. For each housekeeping gene, the different sequences present within a bacterial species are assigned as distinct alleles and, for each isolate, the alleles at each of the loci define the allelic profile or sequence type (ST) [43]. MLST is highly unambiguous, portable, and provides good discriminatory power to differentiate isolates. Several bacterial species involved in brain abscesses have been analyzed by this method [44].
Cronobacter species are rare causes of infant brain abscess, resulting in death in approximately 40 % of cases [45]. Due to the relatedness among Cronobacter species, it is difficult to distinguish them by 16S rDNA sequence analysis. MLST has the ability to identify the species, and also reveals a better understanding of the genomic variation between Cronobacter spp. Accumulating evidence shows that the periodontal pathogen Porphyromonas gingivalis [46] causes extra-oral infections, such as lung and brain abscesses [47, 48]. MLST was developed for P. gingivalis using internal 310- to 420-bp DNA fragments of the eight ubiquitous chromosomal genes. Likewise, MLST was developed for S. aureus using seven housekeeping genes [49]. The devised method was used in a fatal case of a brain abscess caused by a community-associated, methicillin-resistant S. aureus strain (USA300) in a 37-year-old incarcerated woman with a history of furunculosis and intravenous drug use [49]. McDowell et al. [50] developed a novel MLST methodology and database (http://pubmlst.org/pacnes/) for Propionibacterium acnes based on the analysis of seven core housekeeping genes for future epidemiological and evolutionary studies of P. acnes associated with brain abscesses. Besides these, MLST methods have been developed for common pathogens of brain abscesses, i.e., Neisseria spp., Salmonella, Burkholderia cepacia complex, and P. aeruginosa, among others.
The deposit of MLST sequence data in an internet-based repository greatly enhances the global understanding of epidemiological trends, prognostic factors, and outcomes of several bacterial diseases, and its implication in brain abscesses could improve the therapeutic strategy for this disease.
Multiplex PCR assays
The multiplex PCR assay technique is widely employed in clinical laboratories because it is capable of providing a detailed picture of a bacterial community. Moreover, this approach commonly targets the 16S rDNA gene and is especially useful in the identification of slow-growing and fastidious organisms when applied to brain abscesses. A Streptococcus-specific multiplex PCR analysis targeting the 16S rDNA could detect various Streptococcus species correctly in one step [23]. In another study, a multiplex PCR assay was used to detect and identify S. aureus, Haemophilus influenzae, Streptococcus spp., and P. aeruginosa in clinical specimens, including brain abscesses [51]. This assay was able to detect as little as 5 to 50 pg of bacterial DNA. The sensitivity of the assay was evaluated as 100 % for P. aeruginosa, S. aureus, and Streptococcus spp., and 94.3 % for H. influenzae; the specificity was 100 % for all four microorganisms (positive predictive value, 100 %; negative predictive value, 98.2 %) [51].
Cerebral abscesses caused by amoebae are uncommon and are rarely diagnosed antemortem. Morphologically, Balamuthia and Acanthamoeba are difficult to distinguish, which is problematic clinically, given the differences in antimicrobial susceptibility [52]. A multiplex PCR assay for Acanthamoeba, Balamuthia, and Naegleria targeting the mitochondrial 18S rDNA gene was developed with high specificity and sensitivity (96 %, 98 %, and 99 %, respectively) [53]. This assay can identify and distinguish the presence of different amoeba in brain abscess specimens with a detection limit of one amoeba per sample.
Real-time PCR
Over the past ten years, several studies have evaluated the application of real-time PCR for both the detection and quantification of pathogens associated with brain abscesses by either the TaqMan probe or SYBR Green techniques [54, 55]. Adaptation of these approaches to real-time PCR offers a closed system of detection requiring no post-PCR processing that permits the simultaneous examination of several agents involved in acute bacterial cerebral abscesses. Discrimination between species-specific amplicons can be achieved by the analysis of amplicon melting curves and inferring the corresponding melting temperature of each amplicon. Probe hybridization can also be used to discriminate between amplicons.
Several studies have demonstrated that real-time PCR is a powerful tool for the microbiological examination of brain abscesses. This technique was used for the detection of F. necrophorum in aspirates from brain and renal abscesses, resolved diagnostic confusion to prevent unnecessary investigation, and to direct specific antimicrobial treatment [54]. In another study, Pascual et al. highlighted the importance of real-time PCR for the detection of M. hominis, which is not routinely detected on axenic media and by Gram-staining due to a lack of a cell wall [55]. Real-time PCR represents an efficient tool for the diagnosis of M. hominis infections and, thus, contributes to better defining the prevalence and pathogenicity of M. hominis. Moreover, unique species-specific conserved targets have been reported for TaqMan-based real-time PCR detection of brain abscess pathogens. The pneumolysin gene (ply) and the autolysin gene (lytA) are targeted for the identification of S. pneumoniae [56]. The gene bexA encoding a critical component of the polysaccharide exporter [57] is frequently used for the identification of H. influenzae. The invasion-associated protein gene (iap), the listeriolysin O gene (hlyA), and the metalloprotease gene (mpl) have been targeted for the identification of Listeria monocytogenes [58]. The coagulase gene (coa) for S. aureus and the hyaluronidase gene (hylB) for S. agalactiae have also been reported [59].
Concluding remarks
A brain abscess is a life-threatening infection with frequent serious sequelae, whose medical management remains mostly empirical due to a lack of comprehensive knowledge of the organisms responsible for this condition and delays in microbiological results. The identification of microbes involved in brain abscesses using culture-based techniques is time-consuming and extremely biased in their evaluation of microbial genetic diversity. Molecular approaches for the diagnosis and typing of strains involved in brain abscesses have greatly improved the management of brain abscess. However, most studies have relied on the use of broad-range 16S ribosomal DNA (rDNA) as the target, with limited discriminatory power, in particular in polymicrobial infection. In addition, molecular methods do not provide any indication of the potential antibiotic resistance of the involved bacterial infection. In contrast, 16S rDNA-based polymerase chain reaction (PCR), cloning, and sequencing unveiled an elevated proportion of polymicrobial infection and identified a large number of causative agents that were uncultivated, including several putative new species. Systematic diagnosis of brain abscesses using 16S and 18S rDNA-based PCR for the identification of bacterial and fungal pathogens and species-specific PCR to identify Streptococcus intermedius, Mycoplasma faucium, and Mycoplasma hominis are currently being employed in our laboratory to detect fastidious bacteria for further therapeutic approaches.
The diagnostic features of both broad-range and multiplex approaches could be exponentially improved, in terms of specificity and the number of identifiable pathogens, with the development of DNA microarrays, that is, microchips coated with a great number of bacterial targets. In addition, the use of antibiotic resistance genetic markers (to be adapted according to the geographic distribution of resistance genes) and virulence markers in microarrays may provide new insights about microbial resistance to antimicrobials and host immune defenses, as well as potential avenues of therapeutic treatment in brain abscesses. Microarrays containing expressed sequence tags would allow the identification of host response to infection, hopefully to reveal markers of infection, mechanisms of disease pathogenesis, and therapeutic avenues. Moreover, the development of rapid microarray assays would make the diagnosis of brain abscess infections more comprehensive and would permit ‘real-time’ analysis of a tremendous number of samples using several typing approaches simultaneously.
Change history
06 October 2020
An erratum to this paper has been published: https://doi.org/10.1007/s10096-020-04047-8.
References
Mathisen GE, Johnson JP (1997) Brain abscess. Clin Infect Dis 25:763–779
Carpenter J, Stapleton S, Holliman R (2007) Retrospective analysis of 49 cases of brain abscess and review of the literature. Eur J Clin Microbiol Infect Dis 26:1–11
Goodkin HP, Harper MB, Pomeroy SL (2004) Intracerebral abscess in children: historical trends at Children’s Hospital Boston. Pediatrics 113:1765–1770
Tseng JH, Tseng MY (2006) Brain abscess in 142 patients: factors influencing outcome and mortality. Surg Neurol 65:557–562
Xiao F, Tseng MY, Teng LJ et al (2005) Brain abscess: clinical experience and analysis of prognostic factors. Surg Neurol 63:442–449
Al Masalma M, Armougom F, Scheld WM et al (2009) The expansion of the microbiological spectrum of brain abscesses with use of multiple 16S ribosomal DNA sequencing. Clin Infect Dis 48:1169–1178
Al Masalma M, Raoult D, Roux V (2009) Phocaeicola abscessus gen. nov., sp. nov., an anaerobic bacterium isolated from a human brain abscess sample. Int J Syst Evol Microbiol 59:2232–2237
Al Masalma M, Raoult D, Roux V (2010) Staphylococcus massiliensis sp. nov., isolated from a human brain abscess. Int J Syst Evol Microbiol 60:1066–1072
Mishra AK, Fournier PE (2013) The role of Streptococcus intermedius in brain abscess. Eur J Clin Microbiol Infect Dis 32:477–483
Prasad KN, Mishra AM, Gupta D et al (2006) Analysis of microbial etiology and mortality in patients with brain abscess. J Infect 53:221–227
Tonon E, Scotton PG, Gallucci M et al (2006) Brain abscess: clinical aspects of 100 patients. Int J Infect Dis 10:103–109
Lu CH, Chang WN, Lin YC et al (2002) Bacterial brain abscess: microbiological features, epidemiological trends and therapeutic outcomes. QJM 95:501–509
Kommedal Ø, Kvello K, Skjåstad R et al (2009) Direct 16S rRNA gene sequencing from clinical specimens, with special focus on polybacterial samples and interpretation of mixed DNA chromatograms. J Clin Microbiol 47:3562–3568
Koffi N, Aka-Danguy E, Ngom A et al (1998) Prevalence of nocardiosis in an area of endemic tuberculosis. Rev Mal Respir 15:643–647
Giraffa G, Neviani E (2001) DNA-based, culture-independent strategies for evaluating microbial communities in food-associated ecosystems. Int J Food Microbiol 67:19–34
Lagier JC, Armougom F, Million M et al (2012) Microbial culturomics: paradigm shift in the human gut microbiome study. Clin Microbiol Infect 18:1185–1193
Hall KK, Lyman JA (2006) Updated review of blood culture contamination. Clin Microbiol Rev 19:788–802
Seng P, Drancourt M, Gouriet F et al (2009) Ongoing revolution in bacteriology: routine identification of bacteria by matrix-assisted laser desorption ionization time-of-flight mass spectrometry. Clin Infect Dis 49:543–551
Arends JE, Stemerding AM, Vorst SP et al (2011) First report of a brain abscess caused by Nocardia veterana. J Clin Microbiol 12:4364–4365
Relman DA, Loutit JS, Schmidt TM et al (1990) The agent of bacillary angiomatosis. An approach to the identification of uncultured pathogens. N Engl J Med 323:1573–1580
Nikkari S, Lopez FA, Lepp PW et al (2002) Broad-range bacterial detection and the analysis of unexplained death and critical illness. Emerg Infect Dis 8:188–194
Petti CA, Simmon KE, Bender J et al (2008) Culture-negative intracerebral abscesses in children and adolescents from Streptococcus anginosus group infection: a case series. Clin Infect Dis 46:1578–1580
Tsai JC, Teng LJ, Hsueh PR (2008) Direct detection of bacterial pathogens in brain abscesses by polymerase chain reaction amplification and sequencing of partial 16S ribosomal deoxyribonucleic acid fragments. Neurosurgery 62:547–555
Kupila L, Rantakokko-Jalava K, Jalava J et al (2003) Aetiological diagnosis of brain abscesses and spinal infections: application of broad range bacterial polymerase chain reaction analysis. J Neurol Neurosurg Psychiatry 74:728–733
Harris KA, Hartley JC (2003) Development of broad-range 16S rDNA PCR for use in the routine diagnostic clinical microbiology service. J Med Microbiol 52:685–691
Bittar F, Richet H, Dubus JC et al (2008) Molecular detection of multiple emerging pathogens in sputa from cystic fibrosis patients. PLoS One 3:e2908
Fredricks DN, Fiedler TL, Marrazzo JM (2005) Molecular identification of bacteria associated with bacterial vaginosis. N Engl J Med 353:1899–1911
Siala M, Gdoura R, Fourati H et al (2009) Broad-range PCR, cloning and sequencing of the full 16S rRNA gene for detection of bacterial DNA in synovial fluid samples of Tunisian patients with reactive and undifferentiated arthritis. Arthritis Res Ther 11:R102
Ribeiro AC, Matarazzo F, Faveri M et al (2011) Exploring bacterial diversity of endodontic microbiota by cloning and sequencing 16S rRNA. J Endod 37:922–926
Eckburg PB, Bik EM, Bernstein CN et al (2005) Diversity of the human intestinal microbial flora. Science 308:1635–1638
Cole JR, Chai B, Farris RJ et al (2005) The Ribosomal Database Project (RDP-II): sequences and tools for high-throughput rRNA analysis. Nucleic Acids Res 33:D294–D296
Rasschaert G, Houf K, Imberechts H et al (2005) Comparison of five repetitive-sequence-based PCR typing methods for molecular discrimination of Salmonella enterica isolates. J Clin Microbiol 43:3615–3623
Borneman AR, Desany BA, Riches D et al (2011) Whole-genome comparison reveals novel genetic elements that characterize the genome of industrial strains of Saccharomyces cerevisiae. PLoS Genet 7:e1001287
Harvey BS, Koeuth T, Versalovic J et al (1995) Vertical transmission of Citrobacter diversus documented by DNA fingerprinting. Infect Control Hosp Epidemiol 16:564–569
Rodriguez-Barradas MC, Hamill RJ, Houston ED et al (1995) Genomic fingerprinting of Bartonella species by repetitive element PCR for distinguishing species and isolates. J Clin Microbiol 33:1089–1093
Pukall R, Brambilla E, Stackebrandt E (1998) Automated fragment length analysis of fluorescently-labeled 16S rDNA after digestion with 4-base cutting restriction enzymes. J Microbiol Methods 32:55–63
Pelaez AI, del Mar Garcia-Suarez M, Manteca A et al (2009) A fatal case of Nocardia otitidiscaviarum pulmonary infection and brain abscess: taxonomic characterization by molecular techniques. Ann Clin Microbiol Antimicrob 8:11
Sheng WH, Huang YT, Chang SC et al (2009) Brain abscess caused by Tsukamurella tyrosinosolvens in an immunocompetent patient. J Clin Microbiol 47:1602–1604
Marques da Silva R, Caugant DA, Josefsen R et al (2004) Characterization of Streptococcus constellatus strains recovered from a brain abscess and periodontal pockets in an immunocompromised patient. J Periodontol 75:1720–1723
Olive DM, Bean P (1999) Principles and applications of methods for DNA-based typing of microbial organisms. J Clin Microbiol 37:1661–1669
Brown-Elliott BA, Brown JM, Conville PS et al (2006) Clinical and laboratory features of the Nocardia spp. based on current molecular taxonomy. Clin Microbiol Rev 19:259–282
Enersen M, Olsen I, van Winkelhoff AJ et al (2006) Multilocus sequence typing of Porphyromonas gingivalis strains from different geographic origins. J Clin Microbiol 44:35–41
Feil EJ, Spratt BG (2001) Recombination and the population structures of bacterial pathogens. Annu Rev Microbiol 55:561–590
Bowen AB, Braden CR (2006) Invasive Enterobacter sakazakii disease in infants. Emerg Infect Dis 12:1185–1189
Nishihara T, Koseki T (2004) Microbial etiology of periodontitis. Periodontol 2000 36:14–26
Iida Y, Honda K, Suzuki T et al (2004) Brain abscess in which Porphyromonas gingivalis was detected in cerebrospinal fluid. Br J Oral Maxillofac Surg 42:180
Scannapieco FA, Bush RB, Paju S (2003) Associations between periodontal disease and risk for nosocomial bacterial pneumonia and chronic obstructive pulmonary disease. A systematic review. Ann Periodontol 8:54–69
Enright MC, Day NP, Davies CE et al (2000) Multilocus sequence typing for characterization of methicillin-resistant and methicillin-susceptible clones of Staphylococcus aureus. J Clin Microbiol 38:1008–1015
Sifri CD, Park J, Helm GA et al (2007) Fatal brain abscess due to community-associated methicillin-resistant Staphylococcus aureus strain USA300. Clin Infect Dis 45:e113–e117
McDowell A, Gao A, Barnard E et al (2011) A novel multilocus sequence typing scheme for the opportunistic pathogen Propionibacterium acnes and characterization of type I cell surface-associated antigens. Microbiology 157:1990–2003
Xirogianni A, Tzanakaki G, Karagianni E et al (2009) Development of a single-tube polymerase chain reaction assay for the simultaneous detection of Haemophilus influenzae, Pseudomonas aeruginosa, Staphylococcus aureus, and Streptococcus spp. directly in clinical samples. Diagn Microbiol Infect Dis 63:121–126
Pritzker AS, Kim BK, Agrawal D et al (2004) Fatal granulomatous amebic encephalitis caused by Balamuthia mandrillaris presenting as a skin lesion. J Am Acad Dermatol 50:S38–S41
Qvarnstrom Y, Visvesvara GS, Sriram R et al (2006) Multiplex real-time PCR assay for simultaneous detection of Acanthamoeba spp., Balamuthia mandrillaris, and Naegleria fowleri. J Clin Microbiol 44:3589–3595
Aliyu SH, Yong PKF, Newport MJ et al (2005) Molecular diagnosis of Fusobacterium necrophorum infection (Lemierre’s syndrome). Eur J Clin Microbiol Infect Dis 24:226–229
Pascual A, Jaton K, Ninet B et al (2010) New diagnostic real-time PCR for specific detection of Mycoplasma hominis DNA. Int J Microbiol 2010:317512
van Haeften R, Palladino S, Kay I et al (2003) A quantitative LightCycler PCR to detect Streptococcus pneumoniae in blood and CSF. Diagn Microbiol Infect Dis 47:407–414
Kroll JS, Loynds BM, Moxon ER et al (1991) The Haemophilus influenzae capsulation gene cluster: a compound transposon. Mol Microbiol 5:1549–1560
Jaton K, Sahli R, Bille J (1992) Development of polymerase chain reaction assays for detection of Listeria monocytogenes in clinical cerebrospinal fluid samples. J Clin Microbiol 30:1931–1936
Dore N, Bennett D, Kaliszer M et al (2003) Molecular epidemiology of group B streptococci in Ireland: associations between serotype, invasive status and presence of genes encoding putative virulence factors. Epidemiol Infect 131:823–833
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mishra, A.K., Dufour, H., Roche, PH. et al. Molecular revolution in the diagnosis of microbial brain abscesses. Eur J Clin Microbiol Infect Dis 33, 2083–2093 (2014). https://doi.org/10.1007/s10096-014-2166-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-014-2166-z