Abstract
Laboratory-based surveillance for bacterial meningitis was conducted in a network of infectious disease hospitals in Egypt to better understand the epidemiology of this infection. Healthcare and laboratory personnel were trained in basic surveillance and microbiologic processing of cerebrospinal fluid (CSF) specimens. All bacterial isolates from CSF were confirmed and tested for antimicrobial susceptibility. PCR testing was performed on a random subset of purulent, culture-negative CSF specimens. Of 11,070 patients who met criteria for the case definition, 843 (8%) were culture positive (42% positive for Streptococcus pneumoniae, 20% for Haemophilus influenzae serotype b, 17% for each of Neisseria meningitidis and Mycobacterium tuberculosis, and 6% for other bacteria). Of 1,784 (46%) CSF specimens tested by PCR, 232 (13%) were positive for the first three major pathogens. Of N. meningitidis isolates, 52% belonged to serogroup A, 35% to serogroup B, and 4% to serogroup W135. S. pneumoniae isolates comprised 46 different serotypes, of which 6B, 1, 19A, 23F, and 6A were the most predominant. The overall case-fatality rate for culture-positive cases was 26% and was highest among patients with M. tuberculosis (47%). Factors significantly associated with death (p < 0.05) included admission to rural hospitals, long prodromal period, referral from other hospitals, antibiotic treatment prior to admission, and clear CSF (<100 cells/mm3). Susceptibility to ampicillin and ceftriaxone was observed in 44 and 100% of H. influenzae serotype b isolates and in 52 and 94% of S. pneumoniae isolates, respectively. This surveillance highlights the significant mortality and morbidity associated with bacterial meningitis in Egypt. Decision makers need to review current treatment guidelines and introduce appropriate vaccines for prevention and control of the disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bacterial meningitis remains an important public health problem worldwide [1]. Even with antimicrobial therapy and the availability of advanced intensive care, case-fatality rates (CFRs) are ∼25% in industrialized countries [2, 3] and are much higher in the developing world [1]. Between 10 and 20% of survivors develop permanent disabling neurologic sequelae such as epilepsy, mental retardation, or deafness. In the Middle East, localized epidemics of bacterial meningitis have been reported from various countries [4]. Saudi Arabia experienced large meningococcal meningitis epidemics during the Hajj seasons [5, 6]. Sudan falls within the African meningitis belt and has experienced large-scale meningitis epidemics, the latest of which occurred in 2001–2002 [4].
Worldwide, the major causes of bacterial meningitis are Streptococcus pneumoniae, Haemophilus influenzae serotype b, and Neisseria meningitidis [7, 8]. In Egypt, most published meningitis studies were conducted at one large infectious disease hospital in Cairo [9], and available data describing the epidemiology of bacterial meningitis country-wide is minimal. However, since determining the etiology and antimicrobial susceptibility of meningitis pathogens is necessary for improving the clinical management of cases, for guiding therapeutic decisions, and for designing preventive strategies, the Egyptian Ministry of Health and Population and the U.S. Naval Medical Research Unit #3 (NAMRU-3) initiated laboratory-based surveillance in 1998 to better describe the epidemiologic and clinical characteristics of patients with bacterial meningitis in a network of 14 infectious disease hospitals throughout the country. Some findings in children under 6 years old, obtained during the first 2 years of this surveillance (1998–2000), have been partly described [10].
Since isolation of fastidious organisms from the cerebrospinal fluid (CSF) is challenging and requires the use of special media, standard bacteriologic isolation methods, and careful handling, polymerase chain reaction (PCR) testing was used on a random subset of CSF specimens to maximize the pathogen yield. Antibiotic resistance of identified pathogens was evaluated according to the latest guidelines of the Clinical and Laboratory Standards Institute (CLSI, formerly NCCLS). The present report describes the results of this surveillance, as well as the lessons learned during the process.
Materials and methods
Surveillance setup and case identification
Active, laboratory-based surveillance for meningitis was initiated in 1998 in six infectious disease hospitals (also known as “fever” hospitals), including two in urban Cairo, two in Lower Egypt, and two in Upper Egypt. Personnel at additional sites were gradually trained, and those sites were included in the first 3 years of study to form a surveillance network of 14 hospitals throughout the country. In Egypt, patients with suspected meningitis are required by law to be referred to these fever hospitals. Lumbar puncture for patients with suspected meningitis is a routine diagnostic procedure in these hospitals in Egypt. All patients with suspected meningitis (see definitions below) admitted to the participating hospitals were evaluated.
Prior to the implementation of this laboratory-based surveillance activity, a baseline assessment of the microbiology laboratories in participating hospitals revealed that they have variable capacities to perform basic microbiologic functions. Laboratory training was conducted to standardize techniques, and upgrading was performed through the provision of key equipment, reagents, and supplies. A standardized, simple form for collecting information on basic demographic and clinical characteristics of enrolled patients was designed. Clinical training for the participating clinicians was conducted on standard case definition criteria, aseptic collection of CSF from patients with suspected meningitis, and completion of the standardized data collection forms. In this report, a total of 11,070 patients were enrolled in the surveillance network during the period from June 1998 to June 2004.
The study protocol was approved by the U.S. Naval Medical Research Unit No.3 Institutional Review Board, Protocol #30968, in compliance with all federal regulations governing the protection of human subjects. The experiments comply with the current laws of Egypt.
Case definitions
Criteria described previously by Youssef et al. [10] have been used throughout this study. Case patients with a CSF cell count of ≥100 cells/mm3 were classified as having purulent meningitis on the basis of WHO definitions [11].
CSF processing and bacterial culture
Methods used in this study have been described previously [10]. Briefly, CSF was inspected for appearance and processed for differential leukocytic cell count and glucose/protein levels. Microbiologic analysis included Gram stain and bacterial culture on blood, MacConkey, and chocolate agar plates [8]. Samples collected after hours were inoculated into Trans-Isolate Medium (TIM) [12] and cultured on plates on the next day [12]. For Mycobacterium tuberculosis, CSF samples were inoculated into Lowenstein–Jensen medium (Becton Dickinson, Franklin Lakes, NJ, USA). The decision to perform M. tuberculosis cultures was made at the discretion of the admitting physician [13].
Monthly hospital visits
On a monthly basis, investigators conducted site visits to all participating hospitals and met with clinicians and laboratory personnel to follow up on progress, solve problems, evaluate quality control measures, perform refresher training as needed, and replenish media, reagents, and supplies. In addition, they collected the following: surveillance data, recovered isolates, TIM bottles (preserved at room temperature), aliquots of frozen CSF (preserved at >20°C), and M. tuberculosis cultures. Confirmation of bacterial isolates, evaluation of antimicrobial sensitivity patterns, and PCR of a subset of culture-negative CSF specimens were all conducted at NAMRU-3.
Antimicrobial susceptibility testing
For N. meningitidis and S. pneumoniae, antimicrobial susceptibility testing was performed by the E-test method according to the manufacturer’s instructions (AB Biodisk, Solna, Sweden). Inocula were prepared in 0.85% saline, and turbidity was adjusted to the equivalent of a 0.5 McFarland standard. Results were interpreted as recommended by the CLSI (formerly NCCLS) [14]. The Kirby–Bauer disk diffusion method [15] was used only for H. influenzae serotype b isolates. Six antibiotics were used: penicillin (10 units), ampicillin (10 μg), chloramphenicol (30 μg), tetracycline (30 μg), trimethoprim/sulfamethoxazole (5 μg) (TMP/SMX), and ceftriaxone. Beta-lactamase testing was conducted for ampicillin-resistant isolates of H. influenzae and N. meningitidis.
Serotyping
N. meningitidis and H. influenzae serotype b were serogrouped by slide agglutination using specific antisera (Becton–Dickenson). Pneumococcal isolates were serotyped using latex agglutination and the Quellung reaction for confirmation as previously described [16].
PCR screening of culture-negative CSF specimens
A total of 1,784 purulent culture-negative specimens were selected for PCR testing. To evaluate sensitivity and specificity of the assay, CSF specimens from 105 patients with culture-confirmed disease by any of the three major pathogens were included. Bacterial DNA was extracted using the QIAamp DNA Blood Mini Kit (Qiagen, Valencia, CA, USA) according to the manufacturer’s instructions. The primers used to screen for S. pneumoniae (ply gene) and N. meningitidis (ctrA gene) were published previously by Corless et al. [17]. Primers for H. influenzae serotype b (bexA gene) were published by Falla et al. [18].
PCR reaction mixtures for all three bacteria were prepared separately using the same amount of DNA template and reagents. Briefly, 5 μl of an extracted CSF specimen was added to a 20-μl volume of PCR reaction mix containing the following: 1 U of Taq DNA polymerase (Promega, Madison, WI); 200 μM dATP, dGTP, dTTP, and dCTP; 2.5 μl of 10× buffer (Promega, Madison, WI, USA); 0.4 μM of each primer, and 2 mM MgCl2. In every PCR run, a negative control (no DNA template) and a positive control (bacterial DNA from ATCC strains) for each pathogen were included. S. pneumoniae ATCC 49619, H. influenzae ATCC 10211, and N. meningitidis ATCC 13090 were used as positive controls.
DNA was amplified using a GeneAmp PCR System 9700 thermocycler (Applied Biosystems, Foster City, CA, USA) using the following parameters: for S. pneumoniae, initial heating at 94°C for 5 min followed by 30 cycles of denaturation at 94°C for 30 sec, annealing at 63°C for 30 sec, and extension at 72°C for 30 sec; for H. influenzae, serotype b, initial heating at 94°C for 5 min followed by 25 cycles of denaturation at 94°C for 60 sec, annealing at 55°C for 30 sec, and extension at 72°C for 30 sec; for N. meningitidis, initial heating at 94°C for 5 min followed by 35 cycles of denaturation at 94°C for 30 sec, annealing at 55°C for 30 sec, and extension at 72°C for 30 sec. After completion of all cycles, a final extension of 7 min at 72°C was performed for each run. PCR products (8 μl) were electrophoresed through 2% agarose in 1× TBS buffer and made visible by ethidium bromide staining. All PCR-positive results for CSF were repeated for confirmation, and proportions of reactive pathogens were extrapolated to express the respective rates in total purulent culture-negative specimens.
Data analysis
Data collection forms were entered into a computerized database (Epi Info version 6.04). Data were analyzed by age, etiology, locality, and outcome. The chi-square test was used to detect significant associations between variables. A p value of ≤0.05 was considered statistically significant.
Results
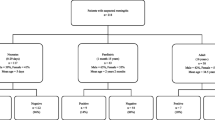
Between June 1998 and June 2004, 11,070 patients with suspected meningitis were admitted to the participating hospitals. Laboratory findings showed that 843 (8%) of them had culture-confirmed bacterial meningitis, although 185 (22%) had <100 leukocytes/mm3. Cases with low leukocyte counts were associated with S. pneumoniae (37%), M. tuberculosis (19%), H. influenzae serotype b (17%), N. meningitidis (15%), and others (12%). Overall, S. pneumoniae was the leading cause of bacterial meningitis (42%), followed by H. influenzae serotype b (20%), N. meningitidis (16%), and M. tuberculosis (16%). Other less frequently identified pathogens included Staphylococcus aureus (4%), Cryptococcus neoformans (0.7%), alpha-hemolytic streptococci (0.4%), and Brucella spp. (0.2%).
Among children <5 years old (n = 334), H. influenzae serotype b was the most commonly isolated bacterium (48%), followed by S. pneumoniae (36%), N. meningitidis (9%), and M. tuberculosis (3%). The distribution of meningitis pathogens by age is presented in Fig. 1.
Most patients (68%) presented at four major hospitals in three big governorates: Cairo, Giza, and Menofiya. Culture-positive rates at these hospitals varied between 2 and 11% (mean, 6%), while rates at those receiving fewer patients ranged between 3 and 31% (mean, 9.6%; Table 1).
Epidemiologic characteristics
The median age of culture-confirmed patients was 11 years for those with S. pneumoniae, 7 months for those with H. influenzae serotype b, 14 years for those with N. meningitidis, and 23 years for those with M. tuberculosis (Table 2). Pneumococcal meningitis, meningococcal meningitis, and tuberculous meningitis occurred in all age groups, while 48% of meningitis cases caused by H. influenzae serotype b occurred in patients <5 years of age. The case-fatality ratio (CFR) ranged from 14% for patients with N. meningitidis to 47% for patients with M. tuberculosis. The CFR was relatively higher among patients <5 years of age and those >40 years of age (Fig. 2). Univariate analyses showed that factors associated with death during hospitalization included admission to hospitals in remote or rural areas (p < 0.05), longer prodromal phase (interval between disease onset and hospital admission), previous hospital admission for the same disease (p = 0.05), tuberculous meningitis (p < 0.01), antibiotic treatment prior to admission (p < 0.05), and clear CSF (<100 cells/mm3) (p < 0.01). After stratification by prodromal periods of <5 days or ≥5 days, the above risk factors (except for the use of antibiotics) remained significantly associated with mortality (Table 3).
The classic triad of meningitis symptoms (fever, headache, and stiff neck) was observed in 47% of patients with laboratory-confirmed disease. Skin rash was highest among patients with meningococcal meningitis. Neurologic complications occurred in 6% of cases during hospitalization and included epilepsy, nerve palsy, muscle paralysis, hydrocephalus, brain infarction, mental retardation, and deafness. These complications were significantly associated with a longer prodromal period (p < 0.001), use of antibiotics prior to admission (p < 0.05), and pathogen type (46% in S. pneumoniae, 27% in H. influenzae serotype b, and 7% in M. tuberculosis).
PCR results
Of the 1,784 purulent culture-negative CSF samples tested by PCR, 232 (13%) were positive, 108 (6%) with S. pneumoniae, 90 (5%) with N. meningitidis, and 36 (2%) with H. influenzae serotype b. Using culture-confirmed cases as controls, the specificity of the assay was 100% for all three pathogens, while sensitivity was 97% for S. pneumoniae, 96% for H. influenzae serotype b, and 95% for N. meningitidis. If we extrapolate the PCR results and combine them with the culture results, the overall proportion of pathogen confirmation increases to 11% (1,220/11,070) and the proportion among patients with purulent meningitis to 42% (n = 2,903).
Bacterial serotyping
Of the N. meningitidis isolates (n = 135), 51% were typed as B, 35% as A, and 4% as W-135. While only 2% belonged to serogroups D and Y, 8% were untypeable. S. pneumoniae (n = 205) isolates comprised 46 serotypes, with the five most common (>6% each) being 6B, 1, 19A, 23F, and 6A [16]. The remaining serotypes occurred in a frequency ranging between 1 and 4%. Ten isolates (5%) were untypeable.
Antimicrobial resistance
Of all H. influenzae serotype b isolates, 45% were resistant to ampicillin, of which 37% (17/52) demonstrated beta-lactamase production. Resistance rates for TMP-SMX and chloramphenicol were 40 and 37%, respectively. Of all S. pneumoniae isolates tested, 49% were nonsusceptible to penicillin (46% intermediate, MIC range 0.12–1.0 μg/ml; 3% resistant, MIC 2.0 μg/ml), and 6% were nonsusceptible to ceftriaxone (5% intermediate, MIC 1.0 μg/ml; 1% resistant, MIC >2 μg/ml; Table 4).
While high rates of resistance to tetracycline (52%) and TMP-SMX (60%) were detected in S. pneumoniae, resistance to erythromycin (11%) and chloramphenicol (9%) was considerably lower (Table 3). Multidrug resistance to >3 antibiotics was observed in 4% of S. pneumoniae isolates, and 50% of these were characterized as serotypes 23F, 6B, and 6A. Resistance of N. meningitidis to TMP-SMX, ampicillin, and penicillin was 86, 5, and 1%, respectively. More than 40% of N. meningitidis isolates showed intermediate resistance to either ampicillin (MIC 0.25–1.0) or penicillin (MIC 0.12–0.25), while intermediate resistance to both penicillin and TMP-SMX was observed in 34% of all isolates. A single N. meningitidis isolate belonging to group B (with intermediate resistance to penicillin) was also positive for beta-lactamase production. Another isolate from group D was resistant to both penicillin and TMP-SMX. Of the three W-135 isolates, two exhibited intermediate resistance to penicillin.
Discussion
This surveillance study highlights the significant mortality and morbidity of bacterial meningitis in Egypt and offers key information to assist national health authorities in designing prevention and management strategies. It also demonstrates the importance of strengthening laboratory and surveillance capacities to provide a solid infrastructure for monitoring trends of bacterial meningitis in the country, characterizing antimicrobial resistance patterns, detecting outbreaks, and monitoring the impact of prevention strategies.
Our results revealed a relatively low rate of culture-positive disease (8% by culture only, 11% by culture and PCR), which may be due to multiple factors, including the use of antibiotics before admission, a common problem in Egypt and many other developing countries [19]. In addition, our use of a broad clinical case definition in order to increase the sensitivity of the surveillance allowed the inclusion of many patients with suspected disease, viral meningitis, or other syndromes such as febrile seizures. Furthermore, the yield of CSF culture may have been significantly impaired by some of the logistical difficulties in rapidly processing CSF cultures. Interestingly, 185 of 843 (22%) culture-positive patients had a CSF cell count of <100 leukocytes/mm3, likely reflecting the early stage of clinical disease. However, as we found in our study, those cases were associated with high mortality rates (26%), similar to rates observed with high cell counts [20, 21], stressing the importance of performing routine cultures despite CSF appearance or consistency.
Our findings show that isolation rates of S. pneumoniae, H. influenzae serotype b, and N. meningitidis were similar to those found in neighboring countries, including the United Arab Emirates [22], Kuwait [23], Libya [24], and Israel [25]. However, results differed from findings in Saudi Arabia [26] and Sudan [27], where N. meningitidis, rather than H. influenzae type b, was the second most prevalent pathogen, and in children in Jordan, where N. meningitidis was the main cause of meningitis (33%) [28]. H. influenzae serotype b is a fastidious organism and can be difficult to isolate from CSF, especially if specimens are not processed quickly and if adequate media and supplements are not used (e.g. X and V growth factors) [29, 30]. Although S. pneumoniae was the leading cause of bacterial meningitis in this study, an earlier investigation [9] showed this organism to be less predominant in Cairo (12%) between 1966 and 1989 compared to N. meningitidis (43%; data adjusted in relation to culture results). This observation may be attributed to the broader geographical representation of hospitals recruited in this study, and may also reflect a real change in the epidemiology of bacterial meningitis in Egypt, reflecting the increased use of polysaccharide meningococcal vaccines in some sectors [31].
When PCR and culture results were combined, the detection rates of S. pneumoniae and H. influenzae serotype b remained fairly unchanged, but those of N. meningitidis increased (from 16 to 23%), and the organism became the second most frequently identified pathogen, supporting a more frequent presence in purulent CSF at culturally undetectable concentrations, dead or live. The higher number of meningococcal meningitis cases positive by PCR could be explained by delays in processing or culturing CSF specimens and reflects the fragility of N. meningitidis. The reported high sensitivity and specificity of PCR concurs with the previous findings of Corless et al. [17], Tzanakaki et al. [32], and Gray et al. [33]. Usefulness of PCR for the diagnosis of bacterial meningitis in the field has been recently shown to be valuable in sub-Saharan African countries, known to have frequent epidemics of meningococcal meningitis and limited reliable laboratories able to culture N. meningitidis, S. pneumoniae, and H. influenzae serotype b [34, 35]. In addition, the technique shows a potential to influence decisions regarding antimicrobial therapy [36].
The proportion of tuberculous meningitis cases in the present study, along with the high corresponding CFRs, is alarming and requires additional studies to better describe the epidemiology of this infection and its risk factors. Our M. tuberculosis isolation rates (16%) are relatively high, possibly due to the presence of predisposing socioeconomic or immunocompromising factors in the affected communities [9, 37]. However, most tuberculous meningitis cases were identified at four major infectious disease hospitals in Cairo, Assuit, Alexandria, and Menofia, signifying more vigilant case management and heavy referral of refractory cases to these hospitals.
The classic meningitis symptoms were seen in variable proportions of patients, demonstrating the limited value of clinical criteria in disease diagnosis unless combined with confirmatory laboratory investigations. While this study was not designed to fully evaluate complications or predictors of mortality, longer prodromal period and rural residence were found to be associated with poor outcomes (p = 0.05). Male gender, being married, and being employed were probably indirect markers of older age and of delay in seeking diagnosis. Likewise, receiving antibiotics prior to admission was associated with higher mortality rates (p = 0.05), possibly due to delayed presentation or delayed diagnosis. These factors may also explain the increased mortality among patients reporting previous admission to hospitals for the same disease and among those showing clear CSF (<100 cells/mm3). Clear CSF has been attributed to previous partial treatment, sepsis, systemic complications, or a blunted response to infection [20, 21]. These findings are in agreement with previous reports [38–40] and can guide public health officials to take appropriate measures for improving the outcome of meningitis by increasing population awareness as to the seriousness of this disease and the importance of having rapid access to healthcare facilities.
Our data suggest a valuable role for the current conjugate H. influenzae serotype b vaccine in the control of meningitis in children. H. influenzae serotype b was detected in 48% of CSF collected from children under 5 years of age. Though available in the private market, the vaccine is not routinely used in Egypt but is currently being considered for introduction in routine immunization schedules by the Egyptian Ministry of Health and Population. In recent years, the epidemiology of H. influenzae serotype b meningitis in developed and developing countries has witnessed major shifts, with a dramatic decrease in the incidence of H. influenzae serotype b disease in young children after the introduction of H. influenzae serotype b vaccine [40–42].
The majority of N. meningitidis isolates in the present study belonged to serogroup B (51%). This may be explained by the fact that polysaccharide meningococcal vaccines against serogroups A and C have been used in Egypt since 1992 [31] as part of vaccination programs for schoolchildren and other government sectors. A few isolates of the epidemic N. meningitidis group W-135 (4%) were also recovered from discrete locations in Egypt during 1999–2000. The patients may have been exposed to concurrent epidemics due to circulation of this strain in Africa or Saudi Arabia during the Hajj [6].
The majority of S. pneumoniae isolates recovered from meningitis patients in Egypt (43% of total purulent specimens identified by culture and PCR) comprised serotypes 6B, 1, 19A, 23F, and 6A [16]. This distribution is different from that found in the USA [43], Europe [44], and neighboring countries such as Israel [25, 45] and Saudi Arabia [46]. None of the available S. pneumoniae vaccines is used in Egypt, since only 29 and 42% of the local serotypes are represented in the current 7- and 11-valent conjugate vaccines, respectively [47]. Details of S. pneumoniae serotype distribution and temporal changes in Egypt have been reported earlier [16]. Our data suggest a need to collect additional information on serotype distribution for other forms of invasive pneumococcal disease and to consider more representative serotypes in the pneumococcal vaccines under development.
Until recently, antibiotic resistance profiles of N. meningitidis isolates were not routinely evaluated at all laboratories due to the lack of standard criteria for the interpretation of antibiotic sensitivity data for this pathogen. In accordance with the latest CLSI standard criteria guidelines [14], our results show that the majority of N. meningitidis isolates were highly resistant to TMP-SMX (86%) and that more than 40% were intermediately resistant to ampicillin or penicillin (with or without TMP-SMX). This is an early report of these resistance profile findings from Egypt and the Middle East region. Similar resistance data for penicillin have been observed in different European countries [48].
Similarly, almost half of the H. influenzae serotype b isolates were resistant to ampicillin, and S. pneumoniae isolates showed intermediate resistance to penicillin, in agreement with reports from many parts of the world [49–51]. The Egyptian isolates showed a higher resistance to erythromycin than those from Saudi Arabia (11 vs. 4%). In comparison with isolates from the USA [43], resistance profiles of Egyptian isolates for tetracycline and TMP-SMX were two to three times higher, but resistance to erythromycin and chloramphenicol was in the same range [13]. Rates of multidrug resistance in S. pneumoniae serotypes were higher among isolates from Egypt than among those from the USA [43]. In Egypt, the most common antibiotics currently used for the empirical treatment of bacterial meningitis still include ampicillin and penicillin. Because antibiotics are available over the counter in Egypt, this may have resulted in the observed higher resistance to commonly used antibiotics. Our findings suggest that physicians should consider third-generation cephalosporins for the empiric treatment of this infection in Egypt. If adequate microbiologic capacity is available, the antibiotic therapy can be changed once the organism is identified and its antimicrobial susceptibility pattern is determined.
This study demonstrates the high morbidity and mortality of bacterial meningitis in Egypt. Temporal changes in the distribution of S. pneumoniae and N. meningitidis serotypes have been observed, highlighting the need to conduct ongoing surveillance and to maintain laboratory capacity. The data is crucial for clinicians to recommend proper treatment as well as for the public health community to design preventive measures and to introduce appropriate meningitis vaccines and monitor their efficacy. Currently, the Ministry of Health and Population in Egypt recommends empiric treatment with penicillin and chloramphenicol for patients with meningitis. On the basis of the antimicrobial resistance data and the high morbidity and mortality observed in this study, we recommend that the Ministry of Health and Population in Egypt revise the national treatment guidelines for bacterial meningitis to include first-line therapy with ceftriaxone. Providers should be made aware of the importance of M. tuberculosis as a common cause of meningitis, particularly among older children and adults. Moreover, providers are encouraged to use laboratory-based surveillance as a key tool in the clinical management of disease. Studies are ongoing to evaluate other etiologies of meningitis known to occur in Egypt, such as viruses and leptospirosis.
References
World Health Organization (2003) State of the art of new vaccines; research & development initiative for vaccine research. WHO, Geneva, pp 34–35
Durand ML, Calderwood SB, Weber DJ, Miller SI, Southwick FS, Caviness VS Jr, Swartz MN (1993) Acute bacterial meningitis in adults: a review of 493 episodes. N Engl J Med 328:21–28
Aronin SI, Peduzzi P, Quagliarello VJ (1998) Community-acquired bacterial meningitis: risk stratification for adverse clinical outcome and effect of antibiotic timing. Ann Intern Med 129:862–869
World Health Organization (2002) Technical consultation report on the prevention and control of epidemic meningococcal disease in Africa. WHO, Burkina Faso, pp 23–24
Centers for Disease Control and Prevention (1987) Meningococcal disease among travelers returning from Saudi Arabia. Morb Mortal Wkly Rep 36:559
Lingappa JR, Al-Rabeah AM, Hajjeh R, Mustafa T, Fatani A, Al-Bassam T, Badukhan A, Turkistani A, Makki S, Al-Hamdan A, Al-Jeffri A, Al Mazrou Y, Perkins BA, Popovic T, Mayer LW, Rosenstein NE (2000) Serogroup W-135 meningococcal disease during the Hajj. Emerg Infect Dis 9:665–671
World Health Organization (1998) Control of epidemic meningococcal disease, WHO practical guidelines, 2nd edn. WHO, Geneva (WHO/EMC/BAC/98.3)
World Health Organization (1999) Communicable disease surveillance and response; laboratory methods for the diagnosis of meningitis caused by Neisseria meningitidis, Streptococcus pneumoniae, and Haemophilus influenzae. WHO, Geneva (WHO/CDS/CSR/EDC/99.7)
Girgis NI, Sippel JE, Kilpatrick ME, Sanborn WR, Mikhail IA, Cross E, Erian MW, Sultan Y, Farid Z (1993) Meningitis and encephalitis at the Abbassia Fever Hospital, Cairo, Egypt, from 1966 to 1989. Am J Trop Med Hyg 48:97–107
Youssef FG, El-Sakka H, Azab A, Eloun S, Chapman GD, Ismail T, Mansour H, Hallaj Z, Mahoney F (2004) Etiology, antimicrobial susceptibility profiles, and mortality associated with bacterial meningitis among children in Egypt. Ann Epidemiol 14:44–48
World Health Organization (2004) Communicable disease case definitions toolkit. WHO, Geneva (WHO/CDS/2004.24)
Ajello GW, Feeley JC, Hayes PS, Reingold AL, Bolan G, Broome CV, Phillips CJ (1984) Trans-isolate medium: a new medium for primary culturing and transport of Neisseria meningitidis, Streptococcus pneumoniae, and Haemophilus influenzae. J Clin Microbiol 20:55–58
Youssef FG, Afifi SA, Azab AM, Wasfy MM, Abdel-Aziz KM, Parker TM, Oun SA, Jobanputra NN, Hajjeh RA (2006) Differentiation of tuberculous meningitis from acute bacterial meningitis using simple clinical and laboratory parameters. Diagn Microbiol Infect Dis 55:275–278
Clinical and Laboratory Standards Institute (2005) Performance standards for antimicrobial susceptibility testing, 15th informational supplement. Approved standard M100-S15. CLSI, Wayne, PA
Esser VM, Elefson DE (1970) Experiences with the Kirby–Bauer method of antibiotic susceptibility testing. Am J Clin Pathol 54:193–198
Wasfy MO, Pimentel G, Abdel-Maksoud M, Russell KL, Barrozo CP, Klena JD, Earhart K, Hajjeh R (2005) Antimicrobial susceptibility and serotype distribution of Streptococcus pneumoniae causing meningitis in Egypt, 1998–2003. J Antimicrob Chemother 55:958–964
Corless CE, Guiver M, Borrow R, Edwards-Jones V, Fox AJ, Kaczmarski EB (2001) Simultaneous detection of Neisseria meningitidis, Haemophilus influenzae, and Streptococcus pneumoniae in suspected cases of meningitis and septicemia using real-time PCR. J Clin Microbiol 39:1553–1558
Falla TJ, Crook DW, Brophy LN, Maskell D, Kroll JS, Moxon ER (1994) PCR for capsular typing of Haemophilus influenzae. J Clin Microbiol 32:2382–2386
Kilpatrick ME, Mikhail IA, Girgis NI (1987) Negative cultures of cerebrospinal fluid in partially treated bacterial meningitis. Trop Geogr Med 39:345–349
McMillan DA, Lin CY, Aronin SI, Quagliarello VJ (2001) Community-acquired bacterial meningitis in adults: categorization of causes and timing of death. Clin Infect Dis 33:969–975
Weisfelt M, Van de Beek D, Spanjaard L, Reitsma JB, Gans J (2006) Attenuated cerebrospinal fluid leukocyte count and sepsis in adults with pneumococcal meningitis: a prospective cohort study. BMC Infect Dis 6:149
Mahmoud R, Mahmoud M, Badrinath P, Sheek-Hussein M, Alwash R, Nicol AG (2002) Pattern of meningitis in Al-Ain medical district, United Arab Emirates: a decadal experience (1990–99). J Infect 44:22–25
Zaki M, Daoud AS, el Saleh Q, West PW (1990) Childhood bacterial meningitis in Kuwait. J Trop Med Hyg 93:7–11
Shembesh NM, El Bargathy SM, Rao BN, Kashbur IM (1998) A prospective study of bacterial meningitis in children from north-eastern Libya. Ann Trop Paediatr 18:203–207
Dagan R (1994) Epidemiology of pediatric meningitis caused by Haemophilus influenzae B, Streptococcus pneumoniae and Neisseria meningitidis, Israel. Isr J Med Sci 30:351–355
Al-Mazrou YY, Musa EK, Abdalla MN, Al-Jeffri MH, Al-Hajjar SH, Mohamed OM (2003) Disease burden and case management of bacterial meningitis among children under 5 years of age in Saudi Arabia. Saudi Med J 24:1300–1307
Salih MA, Khaleefa OH, Bushara M, Taha ZB, Musa ZA, Kamil I, Hofvander Y, Olcen P (1991) Long-term sequelae of childhood acute bacterial meningitis in a developing country. A study from the Sudan. Scand J Infect Dis 23:175–182
Daoud AS, al-Sheyyab M, Abu-Ekteish F, Obeidat A, Ali AA, el-Shanti H (1996) Neonatal meningitis in northern Jordan. J Trop Pediatr 1:267–270
Kilian, M, Biberstein EL (1984) Genus: Haemophilus. In: Krieg NR, Holt J (eds) Bergey’s manual of systematic bacteriology, vol. 1. Williams & Wilkins, Baltimore, MD, pp 558–569
Sell SH (1987) Haemophilus influenzae type b meningitis: manifestations and long-term sequelae. Pediatr Infect Dis J 6:758–775
Nakhla I, Frenck RW Jr, Teleb NA, Oun SE, Sultan Y, Mansour H, Mahoney F (2005) The changing epidemiology of meningococcal meningitis after introduction of bivalent A/C polysaccharide vaccine into school-based vaccination programs in Egypt. Vaccine 23:3288–3293
Tzanakaki G, Tsopanomichalou M, Kesanopoulos K, Matzourani R, Sioumala M, Tabaki A, Kremastinou J (2005) Simultaneous single-tube PCR assay for the detection of Neisseria meningitidis, Haemophilus influenzae type b and Streptococcus pneumoniae. Clin Microbiol Infect 11:386–390
Gray SJ, Trotter CL, Ramsay ME, Guiver M, Fox AJ, Borrow R, Mallard RH, Kaczmarski EB (2006) Epidemiology of meningococcal disease in England and Wales 1993/94 to 2003/02: contribution and experiences of the Meningococcal Reference Unit. J Med Microbiol 55:887–896
Parent du Chatelet I, Traore Y, Gessner BD, Antignac A, Naccro B, Njanpop-Lafourcade BM, Ouedraogo MS, Tiendrebeogo SR, Varon E, Taha MK (2005) Bacterial meningitis in Burkina Faso: surveillance using field-based polymerase chain reaction testing. Clin Infect Dis 40:17–25
Chanteau S, Sidikou F, Djibo S, Moussa A, Mindadou H, Boisier P (2006) Scaling up of PCR-based surveillance of bacterial meningitis in the African meningitis belt: indisputable benefits of multiplex PCR assay in Niger. Trans R Soc Trop Med Hyg 100:677–680
Van Haeften R, Palladino S, Kay I, Keil T, Heath C, Waterer GW (2003) A quantitative LightCycler PCR to detect Streptococcus pneumoniae in blood and CSF. Diagn Microbiol Infect Dis 47:407–414
Schoeman J, Wait J, Burger M, van Zyl F, Fertig G, van Rensburg AJ, Springer P, Donald P (2002) Long-term follow up of childhood tuberculous meningitis. Dev Med Child Neurol 44:522–526
Quagliarello VJ, Scheld WM (1997) Treatment of bacterial meningitis. N Engl J Med 336:708–716
Bonsu B, Harper M (2001) Fever interval before diagnosis, prior antibiotic treatment, and clinical outcome for young children with bacterial meningitis. Clin Infect Dis 32:566–572
Adegbola RA, Secka O, Lahai G, Lloyd-Evans N, Njie A, Usen S, Oluwalana C, Obaro S, Weber M, Corrah T, Mulholland K, McAdam K, Greenwood B, Milligan PJ (2005) Elimination of Haemophilus influenzae type b disease from The Gambia after the introduction of routine immunization with a Haemophilus influenzae serotype b conjugate vaccine: a prospective study. Lancet 366:144–150
Peltola H, Salo E, Saxen H (2005) Incidence of Haemophilus influenzae type b meningitis during 18 years of vaccine use: observational study using routine hospital data. BMJ 330:18–19
Martin M, Casellas JM, Madhi SA, Urquhart TJ, Delport SD, Ferrero F, Chamany S, Dayan GH, Rose CE, Levine OS, Klugman KP, Feikin DR (2004) Impact of Haemophilus influenzae type b conjugate vaccine in South Africa and Argentina. Pediatr Infect Dis J 23:842–847
Doern GV, Heilmann KP, Huynh HK, Rhomberg PR, Coffman SL, Brueggemann AB (2001) Antimicrobial resistance among clinical isolates of Streptococcus pneumoniae in the United States during 1999–2000, including a comparison of resistance rates since 1994–1995. J Antimicrob Chemother 45:1721–1729
Jefferson T, Ferroni E, Curtale F, Giorgi Rossi P, Borgia P (2006) Streptococcus pneumoniae in western Europe: serotype distribution and incidence in children less than 2 years old. Lancet Infect Dis 6:405–410
Rahav G, Toledano Y, Engelhard D, Simhon A, Moses AE, Sacks T, Shapiro M (1997) Invasive pneumococcal infections: a comparison between adults and children. Medicine 76:295–303
Shibl AM, Hussein SS (1992) Surveillance of Streptococcus pneumoniae serotypes in Riyadh and their susceptibility to penicillin and other commonly prescribed antibiotics. J Antimicrob Chemother 29:149–157
Choo S, Finn A (2001) New pneumococcal vaccines for children. Arch Dis Child 84:289–294
Trotter CL, Fox AJ, Ramsay ME, Sadler F, Gray SJ, Mallard R, Kaczmarski EB (2002) Fatal outcome from meningococcal disease—an association with meningococcal phenotype but not with reduced susceptibility to benzylpenicillin. J Med Microbiol 10:855–860
Benbachir M, Benredjeb S, Boye CS, Dosso M, Belabbes H, Kamoun A, Kaire O, Elmdaghri N (2001) Two-year surveillance of antibiotic resistance in Streptococcus pneumoniae in four African cities. Antimicrob Agents Chemother 45:627–629
Gwanzura L, Pasi C, Nathoo KJ, Hakim J, Gangaidzo I, Mielke J, Robertson VJ, Heyderman RS, Mason PR (2003) Rapid emergence of resistance to penicillin and trimethoprim-sulfamethoxazole. Int J Antimicrob Agents 21:557–561
Bradley JS, Connor JD (1991) Ceftriaxone failure in meningitis caused by Streptococcus pneumoniae with reduced susceptibility to beta-lactam antibiotics. Pediatr Infect Dis J 10:871–873
Acknowledgements
We express our sincere appreciation and thanks to the many physicians, nurses, and clinical laboratory staff of the infectious disease hospitals involved in this study. Thanks are extended to the following infectious disease hospital directors: Dr. Yehia Sultan, Dr. Atef Sadaka, Dr. Abdel Halim Abdel Hamid, Dr. Bothaina Bakry, Dr. Mohamed El Ragaby, Dr. Ragaa Sakr, Dr. Mohamed Abdel Hady, Dr. Mofreh Ramzy, Dr. Ahmed Ibrahim, Dr. Said Ibrahim, Dr. Mokhtar Wahba, and Dr. Abdel Fattah Ibrahim. We also thankfully acknowledge support from the staff of the Preventive Sector and the Central Public Health Laboratory of the Ministry of Health and Population who assisted with training and monitoring the activities within the infectious disease hospitals. Appreciation is extended as well to the staff at USAID for their guidance, especially that of Dr. Emad Yanni and Mr. Mark White. We are especially grateful to NAMRU-3 staff members, Mr. Walid William and Mr. Mohamed Abdel Maksoud, and Dr. Maha Talaat for her technical assistance. The present study covers the full surveillance period and all age groups in the population under surveillance. We have published earlier parts of the results from the first 2 years of surveillance in children less than 6 years, because we thought that information was important for decision makers in Egypt who are considering the introduction of Hib vaccine, given the limited data in this country and the whole region. This work was supported by the U.S. Agency for International Development (USAID) and the U.S. Department of Defense Global Emerging Infectious Surveillance System (GEIS) work unit #E022.
Disclaimer
The opinions and assertions contained herein are the private ones of the authors and are not to be construed as official or reflecting the views of the U.S. Department of the Navy or the naval service at large, or the Egyptian Ministry of Health and Population.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Afifi, S., Wasfy, M.O., Azab, M.A. et al. Laboratory-based surveillance of patients with bacterial meningitis in Egypt (1998–2004). Eur J Clin Microbiol Infect Dis 26, 331–340 (2007). https://doi.org/10.1007/s10096-007-0280-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-007-0280-x