Abstract
The aim of this study was to examine the incidence and prognosis of nontyphoid Salmonella bacteraemia in a well-defined population in which complete follow-up investigations had been performed. All patients with nontyphoid Salmonella bacteraemia from 1994 through 2003 in North Jutland County, Denmark, were eligible for the study. Annual incidence rates were calculated for 10-year age groups. The North Jutland County Bacteraemia Database (inclusion of subjects), medical hospital records, the Prescription Registry (redemption of prescription drugs), and the Central Population Registry (deaths) were used as data sources. The outcomes were mortality within 30 and 180 days of the first nontyphoid-Salmonella-positive blood sample. Cox proportional-hazards regression analysis was performed, first with age and comorbidity as evidenced by Charlson index scores, and second with selected clinical and laboratory prognostic variables potentially related to nontyphoid Salmonella bacteraemia per se. A total of 111 non-typhoid Salmonella bacteraemia patients were included in the study. The incidence rate (mean 2.3/100,000 person-years) increased steadily from 1.9/100,000 person-years in the 40–49-year age group to 14.6/100,000 person-years in those >90 years. Twelve (11%) and 24 (22%) patients died within 30 and 180 days, respectively. Cox regression analyses showed that increasing age and, to a higher degree, increasing levels of comorbidity were independently associated with an unfavourable outcome, whereas none of the clinical or laboratory variables studied were strong independent prognostic factors. In conclusion, the presence of comorbid diseases and old age were independently associated with mortality, whereas clinical and laboratory variables were less important.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The main clinical manifestation of nontyphoid Salmonella (NTS) infection in otherwise healthy humans is a self-limited gastroenteritis [1]. However, NTS bacteraemia may ensue, more commonly in patients who are immunocompromised or have comorbid diseases [1]. Further complications are often related to extraintestinal focal infections after haematogenous spread.
Few prognostic studies of NTS bacteraemia have been based on population, and they often focused on certain age groups [2–13]. In addition, many studies included relatively few patients from only one hospital [4–6, 9, 10, 12]. A number of studies of NTS bacteraemia showed that the combination of increased age and comorbid diseases was associated with a poor outcome [7, 14–16], but as none of these studies applied multivariate analyses, the independent impact of each of these factors could not be assessed. Moreover, few studies of NTS bacteraemia have evaluated Salmonella serotypes (especially not after S. enteritidis became pandemic [17, 18]) or biochemical variables prognostically.
Thus, we believe there is a paucity of population-based NTS bacteraemia studies that incorporate biochemical variables and multivariate analyses in a prognostic assessment. In North Jutland County, Denmark, medical wards in hospitals serve all residents within the county, and all Danish residents have a unique personal identification number used in electronic registries and medical hospital records. This enabled us to conduct a population-based study in which complete follow-up of a substantial number of NTS bacteraemia patients was conducted. The aim of the study was to determine the incidence and prognosis of nontyphoid Salmonella bacteraemia in a well-defined population, using multivariate analyses of variables.
Materials and methods
Setting
The Danish public health system is tax financed and free of charge for all residents. In the study period, North Jutland County had a mean of 492,843 residents (9.3% of Denmark’s population), served by eight public hospitals. All residents of the county for whom infectious diseases were suspected were admitted to the nearest hospital, or in more complicated cases, to the county referral hospital in Aalborg. Thus, the county’s entire population served as the background population.
Microbiological procedures
All blood cultures were carried out by the Department of Clinical Microbiology, Aalborg Hospital, Denmark. During the study period, two procedures were used, both of which included the use of three bottles per sample (except for infants, for whom only one sample bottle was used). Until December 1995, the Colorbact system (Statens Serum Institut, Copenhagen, Denmark) was in use, and in January 1996, the BacT/Alert system (bioMérieux, Durham, NC, USA) was introduced [19]. All Salmonella isolates were serotyped, S. enteritidis and S. typhimurium at the Department of Clinical Microbiology, and other serotypes at Statens Serum Institut, using the Kaufmann–White scheme [20]. Antimicrobial susceptibility testing was performed at the Department of Clinical Microbiology on all isolates by a disk diffusion method (Neo-Sensitabs; Rosco, Taastrup, Denmark) on Danish horse blood agar or Mueller–Hinton II agar [19]. All results were entered in the laboratory information system of the Department of Clinical Microbiology (ADBakt; Autonik, Skoldinge, Sweden) and the North Jutland County Bacteraemia Database [21].
Inclusion criteria
The North Jutland County Bacteraemia Database [21] was used to retrieve patients with NTS bacteraemia. All subjects from whom NTS was detected in a blood sample from 1994 through 2003 (i.e., a 10-year period) were eligible for the study.
Data retrieval
The Danish personal identification number was the unique key used for each patient and linkage between registries. Data were retrieved electronically by linkage to the laboratory information system (antimicrobial susceptibility from 1995 through 2003), the North Jutland Prescription Registry (drugs on prescriptions redeemed from pharmacies and subsidised by the National Health Service) [22], and the Central Population Registry (status on living or dead, and, for the latter, date of death). Antibiotic resistance data from 1994 and data from the medical records were entered manually in the study database.
We reviewed all medical records to obtain data on comorbidity, clinical and laboratory variables, and medicine given during hospitalisation. The Charlson index [23] was used to categorise comorbid diseases, as this index has proved its validity in several prognostic studies [24, 25]. In addition, major gastrointestinal conditions (major gastrointestinal surgery, Crohn’s disease, ulcerative colitis, or diverticulitis) were recorded separately.
For calculations of incidence rates, numbers of residents in North Jutland County were retrieved from [26], using population categorised by age (integer years) as per 1 January for each of the years 1994–2003.
Statistical analysis
Stata (Intercooled Stata 8.0 for Windows) was used for all analyses.
Populations within 10-year age groups (ranging from 0–9 to >90 years) were calculated by summarising populations over the period 1994–2003. Annual incidence rates within age groups were then calculated as numbers of NTS bacteraemia patients per 100,000 person-years.
The sampling date (i.e., the date of taking the first NTS-positive blood sample) was the start event. The outcomes were death within 30 and 180 days from the sampling date.
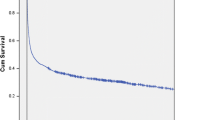
All continuous variables were grouped into 2–4 categories and treated as categorical. Firstly, we estimated the association between the study variables and outcomes by counts and percentages in contingency tables. Secondly, a few variables [27] that were associated with higher mortality were selected for further analyses. For all variables, we made Kaplan–Meyer survival curve estimates. Cox proportional-hazards regression analysis was used for the estimation of 30-day and 180-day mortality rate ratios (MRRs) and 95% confidence intervals, in both crude and multivariate analyses. Variables were grouped into characteristics prior to and related to the NTS bacteraemia episode. Amongst characteristics prior to the NTS bacteraemia episode, age and the presence of comorbid diseases seemed to be highly associated with mortality. Therefore, these were used in a first-step multivariate analysis to assess their independent impact on mortality. Variables related to the NTS bacteraemia episode that tended to be associated with higher mortality were included in a second-step multivariate analysis to deduce their impact, after taking putative confounding from age and comorbid diseases into consideration. The assumptions for the model were assessed graphically [28].
Ethical considerations
The study was conducted according to guidelines of the Danish National Committee on Biomedical Research Ethics and approved by the Danish Data Protection Agency (record no. 2004-41-4004). The study complies with the current laws of Denmark.
Results
General patient characteristics
A total of 113 patients had NTS detected by blood culture from 1994 through 2003. All were hospitalised, but one was a non-Danish resident without a personal identification number and another had no retrievable medical records. Thus, the study population comprised 112 patients for the incidence rate calculations and 111 patients for the prognostic assessment studies. Only 10 of 111 (9%) patients were believed to have acquired their NTS infection nosocomially [29]. Seventeen (15%) patients with community-acquired NTS gastroenteritis reported contact with other patients with gastroenteritis, but no bacteraemia episodes per se were clustered. One hundred (90%) patients had their NTS-positive blood sample taken within 5 days after hospital admission, 82 (74%) of these within 1 day. The remaining 11 patients had NTS bacteraemia detected from 6 to 45 days after admission. This group of patients was typically admitted to hospital due to symptoms from comorbid diseases such as cancer or heart disease, and few presented with a typical clinical picture of bacteraemia or NTS infection (e.g., gastroenteritis) at admission (data not shown).
Age distribution
Figure 1 shows the age distribution of the patients. The numbers of patients decreased until the age group 30–39 years, after which they increased until a decrease from 80 years onwards, i.e., a bimodal distribution was seen. The mean incidence rate was 2.3 per 100,000 person-years. It decreased from the age group 0–9 years until the age group 30–39 years, after which it increased steadily from the age group 40–49 years until beyond 90 years. The peak in the age group 0–29 years was more conspicuous when looking at numbers of patients compared to the incidence rates.
Cumulative mortality
Twelve (11%) and 24 (22%) patients died within 30 and 180 days, respectively. Of the 12 patients who died within 30 days, seven died within 2 days and 11 during the hospitalisation for NTS bacteraemia. Ten of the 12 patients who died within the 30–180-day interval died after discharge, and two died while still hospitalised, after 35 and 36 days. Of these 12 patients, four had recurrent Salmonella bacteraemia diagnosed before death, three had respiratory tract infection of undetermined microbial etiology, and one died with biochemical signs of severe infection. None of the 87 survivors were readmitted with NTS infection within the 180-day study period, indicating they had all recovered from their NTS infection.
Patient characteristics prior to hospitalisation
Mortality generally rose with increasing age (Table 1). None of the patients had a diagnosis of HIV or AIDS (data not shown). There was generally higher mortality amongst patients with comorbid diseases compared to those without. Higher 180-day mortality was also recorded if patients had been hospitalised within the last month, had undergone previous cancer therapy, or had received antibiotics or immunosuppressive drugs 180 or 365 days, respectively, prior to the NTS sampling date. No other characteristics prior to admission were related to increased mortality.
Patient characteristics related to hospitalisation
Lower mortality, especially at 180 days, was related to the occurrence of serotypes other than S. enteritidis or S. typhimurium (Table 2). Amongst the 105 patients from whom three bottles per sample were taken, numbers of NTS-positive sample bottles were positively related to mortality, especially at 180 days. Patients without gastroenteritis had higher mortality than patients with gastroenteritis, after both 30 and 180 days. Altogether, 77 patients were >50 years old, and seven (9%) of these (age range 52–87 years) were diagnosed with an intravascular focus of infection (aortic aneurism in five, endocarditis in two). Dialysis or treatment in an intensive care unit marked a poor outcome, but as there were only 12 such patients, this should be interpreted with caution. The initiation of empirical antibiotic treatment, regardless of whether it was appropriate for Salmonella infection, was associated with higher mortality. Very few isolates were resistant to antibiotics normally effective for Salmonella, e.g., 110 of 111 were ciprofloxacin sensitive (data not shown). The few patients with low temperatures on the sampling date had a poor outcome. No major deviations in mortality were seen when patients with and without missing laboratory variables were compared. Lower albumin and haemoglobin levels were associated with higher mortality. Neither C-reactive protein nor the remaining laboratory variables marked a poor outcome.
Multivariate analyses
Crude 30-day and 180-day MRRs declined for both age and comorbidity after adjustment for each other in Model 1 (Table 3). Both crude and multivariate MRRs were consistently higher for comorbid diseases than for age groups. The inclusion of any of the clinical variables in Model 2 did not alter MRRs for either age or comorbidity. Moreover, the clinical variable MRRs did not exhibit conspicuous changes due to their inclusion in Model 2, except those for empirical antibiotic treatment and gastroenteritis, which approached 1. Overall, age groups and comorbidity levels showed consistently high MRRs, whereas the other model variables had MRRs much closer to 1.
Discussion
The age-related bimodal distribution of NTS bacteraemia, calculated from sheer numbers of patients, has been reported from other studies that often included a high proportion of infants [11, 16, 30, 31]. However, incidence rates in our study showed a less clear bimodal distribution of NTS bacteraemia cases and a considerable occurrence amongst the elderly, a tendency also reported from one of the few other studies that calculated incidence rates [7].
With regard to prognostic factors, we encountered noticeably few differences between 30-day and 180-day mortality. It is difficult to determine specific death causes retrospectively in debilitated patients [32]. The impacts of bacteraemia per se and comorbid diseases on the prognosis of the individual patient are often interrelated and probably exacerbated in debilitated patients.
In general, the occurrence of comorbid diseases also increases in the elderly, i.e., age and comorbidity are highly associated. A number of NTS bacteraemia studies showed that the combination of increased age and comorbid diseases was associated with a poor outcome [7, 14–16], but multivariate analyses were not used. In our study, increasing age and severity of comorbid diseases were consistently related to higher mortality. Their reduced MRRs in Model 1 (Table 3) illustrated their mutual association. However, as MRRs remained high in both multivariate models, age and, to a higher degree, comorbid diseases were independently associated with higher mortality. The association between increasing age per se and higher mortality has also been reported from general bacteraemia studies [33, 34]. Interestingly, comorbidity was independently related to a poorer outcome than that reported from other studies on community-acquired bacteraemia using Charlson index points in which MRRs pertaining to >0 points did not exceed 3 [25, 34, 35].
The clinical variables generally had MRRs close to 1, and they did not alter the MRR for either age or comorbidity (Table 3, Model 2). Other NTS bacteraemia studies reported the occurrence of gastroenteritis to be higher in children than in adults [14–16]. This was also seen in our study (data not shown) and partly explains the more favourable outcome for patients with gastroenteritis (Table 3, Model 2 in which crude MRR approached 1). Interestingly, MRRs for serotypes other than S. enteritidis and S. typhimurium did not change in Model 2. Much attention has been paid to extraintestinal infections caused by rare serotypes, whereas there has been less focus on S. enteritidis and S. typhimurium [30, 36, 37], maybe because most studies were conducted before the present pandemic S. enteritidis period [17, 18], so more studies are needed to explore this.
To our knowledge, no other NTS bacteraemia study has used the number of NTS-positive sample bottles in a prognostic assessment. In our study, a tendency towards increased MRRs was seen when comparing two NTS-positive sample bottles to one, a finding in agreement with studies of other bacteria [38, 39], but more studies are needed to assess its validity as a prognostic determinant.
The 9% occurrence of intravascular foci among >50-year-old patients is in agreement with other studies [40, 41]. Low temperature at admission, rather than fever, predicted a poor outcome in our study, and this is a well-documented prognostic aspect of sepsis [42, 43]. We also found low albumin and haemoglobin levels to be markers of an unfavourable outcome. Albumin has been reported as an unfavourable prognostic marker in numerous studies involving various diseases [44, 45], whereas haemoglobin has been studied less. In the few NTS bacteraemia studies that analysed blood variables, only leukopenia was related to an unfavourable outcome [8, 12, 14], but we did not confirm this in our study. However, as we did not use adjusted MRRs for temperature, albumin, or haemoglobin, these results should be interpreted with caution.
In this population-based study, information and complete follow-up were available for all but two patients, and most NTS infections were community acquired. Thus, the biases often encountered in general studies of bacteraemia, such as different infection acquisitions, primary foci, and bacterial species, were minimised. In most of our cases, there were minimal time spans between the admission and sampling dates, indicating that the suspicion of bacteraemia was an inherent part of the clinical picture at admission. Patients with longer time spans between these two dates presented with a less typical clinical picture at admission. We therefore omitted the 11 patients whose time span between the dates was >5 days and reiterated all statistical tests. This, however, only resulted in minor MRR changes (data not shown), so all 111 patients were included in the final analysis. In addition, the use of the admission date versus the sampling date as the start event only caused minimal MRR changes (data not shown).
This study also had certain limitations. Although most other clinical studies of NTS bacteraemia had fewer than 111 patients, the statistical precision of our study is still low. In addition, our study had the same setbacks encountered in all historical studies based on information gathered in real-life clinical situations without a specific research purpose, of which confounding by indication is probably the most important. As an example, empirical antibiotic treatment was related to a poor prognosis in the univariate analysis, which we believe was due mainly to the initiation of this treatment in the most severely ill patients. This was an important reason for applying a separate analysis of variables prior to and related to hospitalisation, as the former were not prone to confounding by indication.
In conclusion, we found that incidence rates of NTS bacteraemia encountered in elderly patients were especially high. Moreover, old age and, to a higher degree, the presence of comorbid diseases, were independently related to unfavourable outcome, and no clinical or laboratory variables altered this general tendency.
References
Hohmann EL (2001) Nontyphoidal salmonellosis. Clin Infect Dis 32:263–269
Cherubin CE, Neu HC, Imperato PJ, Harvey RP, Bellen N (1974) Septicemia with non-typhoid Salmonella. Medicine (Baltimore) 53:365–376
Hyams JS, Durbin WA, Grand RJ, Goldmann DA (1980) Salmonella bacteremia in the first year of life. J Pediatr 96:57–59
Nelson SJ, Granoff D (1982) Salmonella gastroenteritis in the first three months of life. A review of management and complications. Clin Pediatr (Philadelphia) 21:709–712
Meadow WL, Schneider H, Beem MO (1985) Salmonella enteritidis bacteremia in childhood. J Infect Dis 152:185–189
Wittler RR, Bass JW (1989) Nontyphoidal Salmonella enteric infections and bacteremia. Pediatr Infect Dis J 8:364–367
Lester A, Eriksen NH, Nielsen H et al (1991) Non-typhoid Salmonella bacteraemia in Greater Copenhagen 1984 to 1988. Eur J Clin Microbiol Infect Dis 10:486–490
Galofre J, Moreno A, Mensa J et al (1994) Analysis of factors influencing the outcome and development of septic metastasis or relapse in Salmonella bacteremia. Clin Infect Dis 18:873–878
Schutze GE, Schutze SE, Kirby RS (1997) Extraintestinal salmonellosis in a children’s hospital. Pediatr Infect Dis J 16:482–485
Zaidi E, Bachur R, Harper M (1999) Non-typhi Salmonella bacteremia in children. Pediatr Infect Dis J 18:1073–1077
Ispahani P, Slack RC (2000) Enteric fever and other extraintestinal salmonellosis in University Hospital, Nottingham, UK, between 1980 and 1997. Eur J Clin Microbiol Infect Dis 19:679–687
Rosanova MT, Paganini H, Bologna R, Lopardo H, Ensinck G (2002) Risk factors for mortality caused by nontyphoidal Salmonella sp. in children. Int J Infect Dis 6:187–190
Papaevangelou V, Syriopoulou V, Charissiadou A, Pangalis A, Mostrou G, Theodoridou M (2004) Salmonella bacteraemia in a tertiary children’s hospital. Scand J Infect Dis 36:547–551
Lee SC, Yang PH, Shieh WB, Lasserre R (1994) Bacteremia due to non-typhi Salmonella: analysis of 64 cases and review. Clin Infect Dis 19:693–696
Ramos JM, Garcia-Corbeira P, Aguado JM, Arjona R, Ales JM, Soriano F (1994) Clinical significance of primary vs. secondary bacteremia due to nontyphoid Salmonella in patients without AIDS. Clin Infect Dis 19:777–780
Shimoni Z, Pitlik S, Leibovici L et al (1999) Nontyphoid Salmonella bacteremia: age-related differences in clinical presentation, bacteriology, and outcome. Clin Infect Dis 28:822–827
Rodrigue DC, Tauxe RV, Rowe B (1990) International increase in Salmonella enteritidis: a new pandemic? Epidemiol Infect 105:21–27
Cogan TA, Humphrey TJ (2003) The rise and fall of Salmonella enteritidis in the UK. J Appl Microbiol 94(Suppl):114S–119S
Pedersen G, Schønheyder HC, Kristensen B, Sørensen HT (2000) Community-acquired bacteraemia and antibiotic resistance. Trends during a 17-year period in a Danish county. Dan Med Bull 47:296–300
Popoff MY, Le Minor L (1997) Antigenic formulas of the Salmonella serovars, 7th revision. WHO Collaborating Centre for Reference and Research on Salmonella, Insitut Pasteur, Paris, France
Schønheyder HC (2000) [Two thousand seven hundred and thirty-nine episodes of bacteremia in the county of Northern Jutland, 1996–1998. Presentation of a regional clinical database]. Ugeskr Laeger 162:2886–2891
Nielsen GL, Sørensen HT, Zhou W, Steffensen FH, Olsen J (1997) The pharmacoepidemiologic prescription database of North Jutland—a valid tool in pharmacoepidemiological research. Int J Risk Safety Med 10:203–205
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40:373–383
de Groot V, Beckerman H, Lankhorst GJ, Bouter LM (2003) How to measure comorbidity. A critical review of available methods. J Clin Epidemiol 56:221–229
Lesens O, Methlin C, Hansmann Y et al (2003) Role of comorbidity in mortality related to Staphylococcus aureus bacteremia: a prospective study using the Charlson weighted index of comorbidity. Infect Control Hosp Epidemiol 24:890–896
Danmarks Statistik (2004) Population statistics, Denmark. http://www.statistikbanken.dk. Cited 11 December 2004
Greenland S (1989) Modeling and variable selection in epidemiologic analysis. Am J Public Health 79:340–349
Hosmer DW Jr, Lemeshow S (1999) Regression models for survival data. In: Hosmer DW Jr, Lemeshow S (eds) Applied survival analysis. John Wiley & Sons, New York 87–112
Garner JS, Jarvis WR, Emori TG, Horan TC, Hughes JM (1988) CDC definitions for nosocomial infections, 1988. Am J Infect Control 16:128–140
Blaser MJ, Feldman RA (1981) Salmonella bacteremia: reports to the Centers for Disease Control, 1968–1979. J Infect Dis 143:743–746
Vugia DJ, Samuel M, Farley MM et al (2004) Invasive Salmonella infections in the United States, FoodNet, 1996–1999: incidence, serotype distribution, and outcome. Clin Infect Dis 38(Suppl 3):S149–S156
Pedersen G, Schønheyder HC (1997) Patients with bacteremia dying before notification of positive blood cultures: a 3-year clinical study. Scand J Infect Dis 29:169–173
Leibovici L, Samra Z, Konigsberger H, Drucker M, Ashkenazi S, Pitlik SD (1995) Long-term survival following bacteremia or fungemia. JAMA 274:807–812
Pedersen G, Schønheyder HC, Sørensen HT (2003) Source of infection and other factors associated with case fatality in community-acquired bacteremia—a Danish population-based cohort study from 1992 to 1997. Clin Microbiol Infect 9:793–802
Thomsen RW, Hundborg HH, Lervang HH, Johnsen SP, Sørensen HT, Schønheyder HC (2004) Diabetes and outcome of community-acquired pneumococcal bacteremia: a 10-year population-based cohort study. Diabetes Care 27:70–76
Mandal BK, Brennand J (1988) Bacteraemia in salmonellosis: a 15-year retrospective study from a regional infectious diseases unit. BMJ 297:1242–1243
Fang FC, Fierer J (1991) Human infection with Salmonella dublin. Medicine (Baltimore) 70:198–207
Schønheyder HC, Gottschau A, Friland A, Rosdahl VT (1995) Mortality rate and magnitude of Staphylococcus aureus bacteremia as assessed by a semiquantitative blood culture system. Scand J Infect Dis 27:19–21
Hanon FX, Monnet DL, Sørensen TL, Mølbak K, Pedersen G, Schønheyder HC (2002) Survival of patients with bacteraemia in relation to initial empirical antimicrobial treatment. Scand J Infect Dis 34:520–528
Cohen PS, O’Brien TF, Schoenbaum SC, Medeiros AA (1978) The risk of endothelial infection in adults with Salmonella bacteremia. Ann Intern Med 89:931–932
Benenson S, Raveh D, Schlesinger Y et al (2001) The risk of vascular infection in adult patients with nontyphi Salmonella bacteremia. Am J Med 110:60–63
Weinstein MP, Murphy JR, Reller LB, Lichtenstein KA (1983) The clinical significance of positive blood cultures: a comprehensive analysis of 500 episodes of bacteremia and fungemia in adults. II. Clinical observations, with special reference to factors influencing prognosis. Rev Infect Dis 5:54–70
Young LS (2000) Sepsis syndrome. In: Mandell GL, Bennett JE, Dolin R (eds) Principles and practice of infectious diseases. Churchill Livingstone, Philadelphia 806–819
Phillips A, Shaper AG, Whincup PH (1989) Association between serum albumin and mortality from cardiovascular disease, cancer, and other causes. Lancet ii:1434–1436
Corti MC, Guralnik JM, Salive ME, Sorkin JD (1994) Serum albumin level and physical disability as predictors of mortality in older persons. JAMA 272:1036–1042
Acknowledgement
Ms. Hanne K. Sørensen, Department of Infectious Diseases, Aalborg University Hospital, is thanked for excellent assistance with the retrieval of medical records.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gradel, K.O., Schønheyder, H.C., Pedersen, L. et al. Incidence and prognosis of nontyphoid Salmonella bacteraemia in Denmark: a 10-year county-based follow-up study. Eur J Clin Microbiol Infect Dis 25, 151–158 (2006). https://doi.org/10.1007/s10096-006-0110-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-006-0110-6