Abstract
Objective
To estimate the relative frequency and relative risk of post-traumatic stress disorder (PTSD) attributed to traumatic brain injury (TBI).
Data Sources
PubMed and Embase were searched from database inception until January 26, 2019.
Study Selection
Two independent investigators screened titles, abstracts, and full texts. We selected studies that included subjects presenting with TBI, and where the number of subjects with TBI and PTSD could be extrapolated. There were no restrictions on study design.
Data Extraction and Synthesis
Data were extracted by two independent investigators and results were pooled using random-effects meta-analysis.
Results
In civilian populations, relative frequency of PTSD following TBI was 12.2% after 3 months (CI-95 (7.6 to 16.8%) I2 = 83.1%), 16.3% after 6 months (CI-95 (10.2 to 22.4%), I2 = 88.4%), 18.6% after 12 months (CI-95 (10.2 to 26.9%), I2 = 91.5%), and 11.0% after 24 months (CI-95 (0.0 to 25.8%), I2 = 92.0%). Relative risk was 1.67 after 3 months (CI-95 (1.17 to 2.38), P = 0.011, I2 = 49%), 1.36 after 6 months (CI-95 (0.81 to 2.30), P = 0.189, I2 = 34%), and 1.70 after 12 months (CI-95 (1.16–2.50), P = 0.014, I2 = 89%). In military populations, the relative frequency of associated PTSD was 48.2% (CI-95 (44.3 to 52.1%), I2 = 100%) with a relative risk of 2.33 (CI-95 (2.00 to 2.72), P < 0.0001, I2 = 99.9%).
Conclusions and Relevance
TBI is a risk factor for PTSD in clinic-based civilian populations. There are insufficient data to assess the relative frequency or relative risk of PTSD in moderate to severe TBI. Due to significant between-study heterogeneity, the findings of our study should be interpreted with caution.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Post-traumatic stress disorder (PTSD) and traumatic brain injury (TBI) are both pathological consequences of head trauma. Estimates suggest that the 12-month prevalence of PTSD is 1.1% [1], while 69 million people per year experience TBI worldwide [2]. In addition, patients with PTSD attributed to TBI often display symptoms such as headaches [3], anxiety [4], and depression [5], all of which are among the leading causes of disability worldwide [6]. In veterans from the USA, PTSD and TBI are associated with substantial long-term medical and disability expenses [7], and outpatient healthcare costs are more than doubled for patients with comorbid PTSD and TBI, compared with patients who suffer from only one of the two [7]. However, inconsistent findings on the prevalence of PTSD attributed to TBI means that it is difficult to assess the actual scope of the public health burden imposed by TBI-associated PTSD. An overview of the current literature is therefore highly needed.
We therefore decided to perform a systematic review and meta-analysis of PTSD attributed to TBI in civilian and military populations. Meta-analysis was performed to estimate pooled relative frequencies and relative risk of PTSD attributed to TBI.
Methods
Data sources and study selection
We searched PubMed and Embase for studies on the relative frequency and relative risk of PTSD attributed to TBI. The search was performed January 26, 2019, with the following search string: “(traumatic brain injury OR concussion OR head trauma OR head injury OR brain trauma OR brain injury OR blast injury) AND (PTSD OR post-traumatic stress disorder OR post-traumatic stress disorder OR post-traumatic stress disorder OR shell-shock OR shell shock OR battle fatigue).” Two investigators, A.I. and H.M.K., screened all search results by title and abstract. Full texts were retrieved whenever a publication included subjects with TBI, and it was believed that the publication might present data on PTSD. Following full-text retrieval, the same two authors performed full-text screening and assessed all publications according to pre-defined eligibility criteria (Supplement Table 1). Final study inclusion was decided by consensus between A.I. and H.M.K.
Definition of Traumatic Brain Injury
TBI is defined as an alteration in brain function, or other evidence of brain pathology, caused by an external force [8]. For the purpose of this study, TBI was defined as exposure to a direct or indirect blow to the head, identified by interview with hospital or research staff, record review or self-report on a questionnaire, or by searching databases for validated diagnostic codes. TBI severity was graded according to the parameters presented in Supplement Table 2. Publications that did not report injury severity were designated as “not reported.” Publications that identified any of the symptoms listed in Supplement Table 2, but did not report the boundaries used to categorize injury severity, were designated as “cannot be determined accurately.”
Definition of Post-Traumatic Stress Disorder
PTSD is a trauma- or stressor-related psychiatric disorder that can develop following exposure to actual or threatened traumatic events [9]. A formal diagnosis can be made if patients meet the criteria outlined in the Diagnostic and Statistical Manual of Mental Disorders or the International Classification of Diseases. Patients are designated as screening positive if the diagnosis is made by a structured or semi-structured interview, by record review, by database search for validated diagnostic codes, or by administrating a validated questionnaire. We prioritized interviews in studies that utilized a mixture of interviews and questionnaires. Within questionnaire-based studies, we prioritized results calculated by a combination of symptom clusters and cut-off scores over results calculated using symptom clusters alone, which were again prioritized over results calculated by cut-off score alone. If results for several cut-off scores were presented, the highest score was included.
Data extraction and quality assessment
Data were extracted using a pre-defined form. Quality assessment was performed with a modified Newcastle-Ottawa Scale. The instrument contains four domains with nine questions in total. Each question gets scored as either 0 or 1, with a maximum score of nine. All nine domains were not relevant for all of the included studies. We therefore calculated a total sum score, and a weighted fraction calculated as the total sum score divided by the maximally attainable score. Study quality was rated as low for studies with a weighted fractional score ≤ 25%, medium for scores > 25% and ≤ 50%, high for scores > 50% and ≤ 75%, and very high for scores > 75%. Two investigators, A.I. and H.M.K., independently extracted data and scored each study. Any discrepancies in data extraction or quality assessment were resolved between the two authors.
Statistical analysis
The outcome of interest was the relative frequency of PTSD attributed to TBI and the relative risk of PTSD in TBI compared to controls. A random-effects meta-analysis that accounted for between-study heterogeneity was performed. Effect sizes were calculated using the inverse variance method on untransformed data. Between-study variance was calculated using the restricted maximum likelihood method. Tau was assumed not-equal between studies and the Hartung-Knapp modification was used to adjust test statistics and confidence intervals. Effect sizes were only calculated for groups consisting of two or more studies. The χ2 tests and the I2 statistic were used to assess between-study heterogeneity. Data analyses were conducted separately for military and civilian populations and for population-based and non-population-based studies. We calculated effect sizes for each study population regardless of TBI severity and, when possible, for studies where subjects could be classified by our definition as “mild TBI only” or “moderate to severe TBI.” When two or more studies used overlapping populations, only numbers from the study with the largest number of participants were included. Studies with military populations tend to assess PTSD at a single time point with a varying or unknown time interval between the head injury and the PTSD assessment. Because of that, our analysis was restricted to non-longitudinal studies. Most civilian studies reported the intervals between the TBI and the PTSD assessment. We therefore grouped the results from each study into the following groups based on the time between TBI and PTSD assessment: [1:3] = 3 months, (3:6] = 6 months, (6:12] = 12 months, 12:24] = 24 months. Relative frequency was estimated at [1:3], (3:6], (6:12], and (12:24] months after TBI, and relative risk at [1:3], (3:6], (6–12] months (see Supplement 1 for further details). Sources of heterogeneity were investigated with univariate meta-regression, in order to assess the influence of moderators on effect size. Meta-regression was only performed for analyses with 10 or more studies, and with a minimum of two subgroups in each analysis. The following confounding variables were evaluated: sample source: clinic-based, community- or population-based, and registry- or data-based; military service status; deployment status; TBI assessment method; TBI severity mild only; type of comparison group; DSM version; PTSD instrument type; PTSD as primary outcome and study quality > 75% vs. ≤ 75%. Publication bias was assessed via visual inspection of a funnel plot of standard error against proportion effect size and quantitatively by Egger’s test with significance defined as α ≤ 0.05. This was only done for analyses which included 10 or more studies [10]. All statistical analyses were performed with R version 3.6.0 using the “meta” and “metafor” packages.
Results
Search results
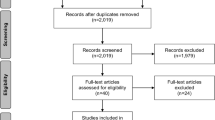
The database searches yielded 3385 hits in PubMed and 3162 hits in Embase (Fig. 1). Seven additional publications were identified through a manual search of the reference lists from the included publications. We found 5309 unique records after removal of duplicates. We retrieved 579 full texts, from which 193 full texts were initially included. Removal of publications describing duplicate cohorts reduced the total number to 174 publications. Fifty-one included civilian populations and 123 included military populations.
Thirty-one civilian publications and 104 military publications were included in the meta-analysis. A full reference list of the 193 identified publications is presented in Supplement Appendix 1. Summary of study characteristics is presented in Table 1.
Relative frequency of PTSD in civilian populations
For non-population-based studies with TBI of any severity, relative frequency of PTSD was 12.2% after 3 months (n = 1572, CI-95 = 7.6 to 16.8%, I2 = 83.1%), 16.3% after 6 months (n = 1817, CI-95 = 10.2 to 22.4%, I2 = 88.4%), 18.6% after 12 months (n = 2876, CI-95 = 10.2 to 26.9%, I2 = 91.5%), and 11.0% after 24 months (n = 575, CI-95 = 0.0 to 25.8%, I2 = 92.0%) (Table 2). The difference in relative risk between time points was not statistically significant (Q = 3.32, P = 0.3455). When analysis was restricted to populations defined as mild TBI only, relative frequency was 10.8% after 3 months (n = 1257, CI-95 = 7.4 to 14.2%, I2 = 40.5%), 14.6% after 6 months (n = 993, CI-95 = 7.8 to 21.5%, I2 = 63.1%), 21.5% after 12 months (n = 964, CI-95 = 9.3 to 33.6%, I2 = 88.8%), and 16.8% after 24 months (n = 407, CI-95 = 0.0 to 38.8%, I2 = 0.0%) (Table 2). The difference in relative frequency between time points was statistically significant (Q = 10.85, P = 0.0126). The current review and meta-analysis only identified five studies with moderate to severe TBI, with less than 275 participants per time point. The results are therefore not presented here, but the forest plot can be found in the supplementary material
We were only able to identify one population-based study in civilians and thus, meta-analysis was not performed.
Relative risk of PTSD in civilian populations
For non-population-based studies with TBI of any severity, relative risk of PTSD was calculated to 1.67 after 3 months (TBI = 1465, controls = 2070, CI-95 = 1.17 to 2.38, P = 0.011, I2 = 49%), 1.36 after 6 months ((TBI = 325, controls = 538, CI-95 = 0.81 to 2.30, P = 0.189, I2 = 34%), and 1.70 after 12 months (TBI = 2271, controls = 3641, CI-95 = 1.16 to 2.50, P = 0.014, I2 = 89%) (Table 3). The difference between time points was not significant (P = 0.6598). For populations defined as mild TBI only, relative risk of PTSD was calculated to 1.66 after 3 months (TBI = 1257, controls = 1644, CI-95 = 0.88 to 3.13, P = 0.093, I2 = 69%), 0.92 after 6 months (TBI = 109, controls = 138, CI-95 = 0.14 to 6.22, P = 0.691, I2 = 0%), and 1.84 after 12 months (TBI = 689, controls = 1794, CI-95 = 1.81 to 1.86, P < 0.0001, I2 = 0%) (Table 3). The difference between time points was statistically significant (P < 0.0001).
Meta-analysis was not performed for moderate to severe TBI, or for population-based studies, due to a lack of eligible studies.
Meta-regression in civilian populations
Meta-regression was performed for studies reporting the relative frequency of TBI of any severity at 3, 6, and 12 months in non-population-based studies. None of the predictors were significantly associated with the relative frequency of PTSD.
Publication bias in civilian populations
Assessment of publication bias was only performed for studies reporting on relative frequency for any severity at 3, 6, and 12 months. Funnel plots can be seen in supplementary material and were asymmetric at visual inspection at all time points, indicating publication bias. Egger’s test showed statistically significant bias at 3 months (P = 0.03) and 6 months (0.03) but not for 12 months (P = 0.42).
Relative frequency of PTSD in military populations
For TBI of any severity, relative frequency of associated PTSD was estimated to be 48.2% (n = 563,264, CI-95 = 44.3 to 52.1%, I2 = 100%). For mild TBI only, relative frequency of associated PTSD was estimated to be 48.8% (n = 25,521, CI-95 = 43.1 to 54.6%, I2 = 99.4%) (Table 2). Due to a lack of eligible studies, meta-analysis was not performed for moderate to severe TBI.
Relative risk of PTSD in military populations
For TBI of any severity, relative risk of associated PTSD was calculated to be 2.33 (TBI = 517074, controls = 5480874, CI-95 = 2.00 to 2.72, P < 0.0001, I2 = 99.9%). For mild TBI only, relative risk of associated PTSD was calculated to be 2.32 (TBI = 16078, controls = 37761, CI-95 = 1.67 to 3.23, P < 0.0001, I2 = 98%) (Table 3). Due to a lack of eligible studies, meta-analysis was not performed for moderate to severe TBI.
Meta-regression in military populations
Meta-regression was performed separately for relative frequency and relative risk of TBI-associated PTSD, for TBI of any severity, and mild TBI only.
An association was found between the factors sampling source, service status, deployment status, and relative frequency. For sampling source, relative frequency was significantly lower in survey-based studies (reference variable: clinic-based). For service status, relative frequency was significantly lower in samples with servicemembers on active duty and in mixed samples of active-duty servicemembers and veterans, compared to studies with veterans alone (reference variable: veterans). For deployment status, relative frequency was significantly higher in studies that assessed subjects more than 12 months after return from deployment, including samples with subjects where the time of return from deployment was not reported, in samples with deployed and non-deployed subjects, and in samples where deployment status was unknown (reference variable: within 12 months). Sampling source, service status, and deployment status explained 11%, 27%, and 20% of the heterogeneity respectively in TBI of any severity, and 25%, 50%, and 33% in mild TBI only.
Only sampling source was associated with relative risk, with relative risk being higher in survey-based studies with TBI of any severity. Sampling source explained 16% of the heterogeneity in a univariate model for relative risk in TBI of any severity.
Publication bias in military populations
Funnel plots for studies reporting relative frequency and relative risk of TBI-associated PTSD in TBI of any severity and mild TBI only, are presented in the supplementary material. Funnel plots were not asymmetric at visual inspection and Egger’s test did not reveal any statistically significant asymmetry.
Discussion
After pooling results from 31 civilian studies and 104 military studies, we have demonstrated that 11.0 to 18.6% of civilians develop PTSD within a 2-year period following a TBI, while 48.2% of servicemembers or veterans have TBI-associated PTSD. In comparison, the relative frequency of PTSD is estimated to be 23.1% in the combined population of previously deployed, current, and former servicemembers [11], and 1.1% in the general population over a 1-year period [1], making PTSD much more prevalent among individuals with TBI. When compared to individuals with no TBI, the risk of PTSD was 1.7 times higher in civilians with TBI and 2.3 times higher in military personnel. When we restricted the analysis to subjects with mild TBI only, the results did not change. Thus, our findings show that even a mild TBI increases the risk for developing PTSD.
TBI-associated PTSD poses a significant problem for both civilian and military populations. In the USA alone, TBI results in 2.87 million emergency department visits each year [12], and our findings support that a substantial proportion will develop clinically significant signs of PTSD. Patients with TBI and PTSD have higher rates of other psychiatric comorbidities [13], more post-concussive symptoms [14], and have poorer recovery and treatment response for other comorbidities, such as headaches [15]. Furthermore, administrative data from veterans’ health encounters show that healthcare costs are doubled for veterans with comorbid TBI and PTSD, compared to veterans who only present with TBI [7]. PTSD therefore greatly contributes to the overall burden of TBI, and our findings emphasize the need for better control of TBI and more interventions in the period following TBI.
The rates of PTSD following a military-related TBI are higher than those seen following civilian-related TBI. This is likely due to differences in the manner in which the TBI is acquired and the frequency of traumatic exposures. TBIs in military populations are mainly acquired during deployment. A recent meta-analysis in military populations found that the number and length of deployments, being in a combat role, as well as experiencing prior trauma, including witnessing someone being wounded or killed, were all associated with an increased relative frequency of PTSD [16]. Thus, military personnel, especially those assigned to combat roles in conflict zones, are more likely to have repeated exposure to traumatic events, which increases the odds of developing PTSD. On the contrary, individuals in civilian studies are more likely to experience fewer traumatic exposures.
Our study has some limitations that need to be addressed. We observed substantial heterogeneity within studies on both civilian and military populations. Our pooled relative frequencies and relative risk estimates should therefore be interpreted with caution. The heterogeneity could arise from several factors and was investigated through meta-regression of study-level characteristics. In civilian populations, none of the study-level characteristics were associated with the relative frequency of PTSD attributed to TBI. However, the analysis was limited by small sample sizes. Furthermore, a majority of civilian studies were clinic-based. One survey-based study found that 42% of individuals with TBI do not seek medical care, and that the odds of seeking medical care are reduced in patients with mild TBI compared to those with moderate or severe TBI [17]. Our findings may thus not be generalizable to representative samples of civilians with TBI. In military populations, lower relative frequencies were observed for the following study-level characteristics: survey-based samples, status as active-duty servicemembers, and TBI subjects assessed within 12 months of return from deployment. These findings could be explained by a large overlap in studies characterized as survey-based, active-duty, and return from deployment within 12 months [18,19,20,21,22,23,24,25,26,27,28,29,30]. Subjects on active duty were sampled regardless of healthcare status, whereas veteran populations were sampled from Veteran Affairs clinics in clinic-based studies, and from healthcare administrative databases in database-based studies. Thus, these military studies could have a selection bias towards TBI subjects, who are more likely to present with PTSD.
The observed heterogeneity could also be explained by a lack of robust and standardized epidemiological assessment tools of both PTSD and TBI. First, the relative frequencies of PTSD were mainly determined by diagnostic interviews or questionnaires, which varied considerably. In civilian studies, eight different interviews and 12 different questionnaires were used, while in military studies, seven different interviews and nine different questionnaires were used. In addition, the most frequently used questionnaire, the PTSD Checklist (PCL) and its subtypes, have been shown to have a pooled positive predictive value of 72% [31]. PTSD questionnaires are therefore likely to overestimate the number of PTSD cases relative to structured interviews such as the Clinician-Administered PTSD Scale (CAPS), the Composite International Diagnostic Interview (CIDI), and the Structured Clinical Interview (SCID). Second, differences in enrollment criteria and assessment of baseline characteristics, could also contribute to heterogeneity. For example, 10 of 174 studies included participants with just one episode of TBI. Of the remaining 164 studies, only 13 studies reported the number of life-time TBIs. Third, we found notable differences in study methodology between control populations. In military studies, controls without combat-related TBI could still have sustained a non-combat TBI, while controls without military-related TBI could have sustained a TBI in civilian circumstances. Fourth, previous history of PTSD, or the rate of pre-injury PTSD, was not accounted for in 161 of the 174 included studies. Fifth, case definitions of TBI varied considerably among studies. One recent meta-analysis of PTSD following TBI, concluded that injury severity did not affect the relative frequency and relative risk of PTSD [32]. We, however, do not believe the influence of injury severity can be reliably assessed. As mentioned above, we found considerable variations in TBI case definitions, and few studies reporting on PTSD following moderate to severe TBI. Hence, we were unable to perform a meta-analysis for military populations with moderate to severe TBI. Moreover, the number of studies and number of study participants in civilian studies were insufficient for making meaningful conclusions about relative frequency or relative risk. Lastly, some studies relied on certain trauma-related characteristics (i.e., duration of loss of consciousness (LOC), blast-related TBI vs. non-blast TBI) for study inclusion. It has been suggested that subjects with TBI and LOC [21], or blast-related TBI [33], experience higher rates of PTSD compared to subjects with TBI without LOC or non-blast TBI. Because of that, differences in the reported relative frequencies of PTSD could reflect disparities in certain trauma-related characteristics.
Conclusions and future directions
PTSD is a common sequela of TBI, and even a mild TBI is associated with an increased risk of PTSD. We found that almost one-fifth of civilians with concussions develop PTSD attributed to TBI, while a staggering 48.2% of military personnel present with PTSD after TBI. However, due to methodological concerns, accurate data on the prevalence of PTSD is limited and should be interpreted with caution. In order to standardize assessments of PTSD and TBI in research settings, epidemiological studies with rigorous methodology and an international consensus are needed. This will provide accurate measurements and facilitate comparative analyses in the future. Furthermore, study-level characteristics (i.e., TBI severity, LOC, GCS, PTA, pre-trauma psychiatric history) should be systematically recorded to enable analysis of PTSD predictors in TBI patients, including the impact of TBI severity.
In conclusion, TBI-related PTSD constitutes a large-scale public health burden and a concerted effort is much warranted to address the devastating impact on patients and their families. Considerable heterogeneity in the relative frequency of TBI-related PTSD underscores the need for an international standardization of epidemiological studies, so to better elucidate the enormous burden and societal costs for both civilian and military populations.
References
Karam EG, Friedman MJ, Hill ED et al (2014) Cumulative traumas and risk thresholds: 12-month PTSD in the world mental health (WMH) surveys. Depress Anxiety. 31(2):130–142
Dewan MC, Rattani A, Gupta S et al (2019) Estimating the global incidence of traumatic brain injury. J Neurosurg. 130(4):1080–1097
Afari N, Harder LH, Madra NJ et al (2009) PTSD, combat injury, and headache in veterans returning from Iraq/Afghanistan. Headache J Head Face Pain. 49(9):1267–1276
Pietrzak RH, Goldstein RB, Southwick SM et al (2011 Apr) Prevalence and axis I comorbidity of full and partial posttraumatic stress disorder in the United States: results from Wave 2 of the National Epidemiologic Survey on Alcohol and Related Conditions. J Anxiety Disord. 25(3):456–465
Rytwinski NK, Scur MD, Feeny NC et al (2013 Jun) The co-occurrence of major depressive disorder among individuals with posttraumatic stress disorder: a meta-analysis. J Trauma Stress. 26(3):299–309
(2018) Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet:1789–1858. https://doi.org/10.1016/S0140-6736(18)32279-7
Kehle-Forbes SM, Campbell EH, Taylor BC et al (2017) Does co-occurring traumatic brain injury affect VHA outpatient health service utilization and associated costs among veterans with posttraumatic stress disorder? An examination based on VHA administrative data. J Head Trauma Rehabil. 32(1):E16–E23
Menon DK, Schwab K, Wright DW et al (2010) Position statement: definition of traumatic brain injury. Arch Phys Med Rehabil. 91(11):1637–1640
American Psychiatric Association. Diagnostic and statistical manual of mental disorders (5th). American Psychiatric Association; 2013. Arlington. 265–290. https://doi.org/10.1176/appi.books.9780890425596
Sterne JAC, Sutton AJ, Ioannidis JPA et al (2011) Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ. 343(7818):0–8
Fulton JJ, Calhoun PS, Wagner HR et al (2015) The prevalence of posttraumatic stress disorder in operation enduring freedom/operation Iraqi Freedom ( OEF / OIF ) veterans: a meta-analysis. J Anxiety Disord. 31:98–107
Centers for Disease Control and Prevention. Surveillance report of traumatic brain injury-related emergency department visits, hospitalizations, and deaths—United States, 2014.; Available from: www.cdc.gov/TraumaticBrainInjury
Alway Y, McKay A, Gould KR et al (2016) Factors associated with posttraumatic stress disorder following moderate to severe traumatic brain injury: a prospective study. Depress Anxiety. 33(1):19–26
Aase DM, Babione JM, Proescher E et al (2018) Impact of PTSD on post-concussive symptoms, neuropsychological functioning, and pain in post-9/11 veterans with mild traumatic brain injury. Psychiatry Res. 268(June):460–466
Sawyer K, Bell KR, Ehde DM et al (2015) Longitudinal study of headache trajectories in the year after mild traumatic brain injury: relation to posttraumatic stress disorder symptoms. Arch Phys Med Rehabil. 96(11):2000–2006
Xue C, Ge Y, Tang B et al (2015) A meta-analysis of risk factors for combat-related PTSD among military personnel and veterans. PLoS One. 10(3):1–21
Setnik L, Bazarian JJ (2007) The characteristics of patients who do not seek medical treatment for traumatic brain injury. Brain Inj. 21(1):1–9
Adams RS, Larson MJ, Corrigan JD et al (2012) Frequent binge drinking after combat-acquired traumatic brain injury among active-duty military personnel with a past year combat deployment. J Head Trauma Rehabil. 27(5):349–360
Aralis HJ, Macera CA, Rauh MJ et al (2014) Traumatic brain injury and PTSD screening efforts evaluated using latent class analysis. Rehabil Psychol. 59(1):68–78
Arbisi PA, Polusny MA, Erbes CR et al (2011) The Minnesota Multiphasic Personality Inventory-2 Restructured Form in National Guard soldiers screening positive for posttraumatic stress disorder and mild traumatic brain injury. Psychol Assess. 23(1):203–214
Hoge CW, McGurk DM, Thomas JL et al (2008) Mild traumatic brain injury in U.S. soldiers returning from Iraq. N Engl J Med. 358(5):453–463
Macera CA, Aralis HJ, MacGregor AJ et al (2012) Postdeployment symptom changes and traumatic brain injury and/or posttraumatic stress disorder in men. J Rehabil Res Dev. 49(8):1197
MacGregor AJ, Dougherty AL, Tang JJ et al (2013) Postconcussive symptom reporting among US combat veterans with mild traumatic brain injury from operation Iraqi Freedom. J Head Trauma Rehabil. 28(1):59–67
Polusny MA, Nelson NW, Erbes CR et al (2011) Longitudinal effects of mild traumatic brain injury and posttraumatic stress disorder comorbidity on postdeployment outcomes in national guard soldiers deployed to Iraq. Arch Gen Psychiatry. 68(1):79–89
Rona RJ, Jones M, Fear NT et al (2012) Mild traumatic brain injury in UK military personnel returning from Afghanistan and Iraq: cohort and cross-sectional analyses. J Head Trauma Rehabil. 27(1):33–44
Schwab K, Terrio HP, Brenner LA et al (2017) Epidemiology and prognosis of mild traumatic brain injury in returning soldiers. Neurology. 88(16):1571–1579
Vasterling JJ, Brailey K, Proctor SP et al (2012) Neuropsychological outcomes of mild traumatic brain injury, post-traumatic stress disorder and depression in Iraq-deployed US Army soldiers. Br J Psychiatry. 201(3):186–192
Wilk JE, Herrell RK, Wynn GH et al (2012) Mild traumatic brain injury (concussion), posttraumatic stress disorder, and depression in U.S. soldiers involved in combat deployments: association with postdeployment symptoms. Psychosom Med. 74(3):249–257
Wilk JE, Thomas JL, McGurk DM et al (2010) Mild traumatic brain injury (concussion) during combat: lack of association of blast mechanism with persistent postconcussive symptoms. J Head Trauma Rehabil. 25(1):9–14
Yurgil KA, Barkauskas DA, Vasterling JJ et al (2014) Association between traumatic brain injury and risk of posttraumatic stress disorder in active-duty marines. JAMA Psychiatry. 71(2):149–157
Tiet QQ, Schutte KK, Leyva YE (2013) Diagnostic accuracy of brief PTSD screening instruments in military veterans. J Subst Abuse Treat. 45(1):134–142
Van Praag DLG, Cnossen MC, Polinder S et al (2019) Post-traumatic stress disorder after civilian traumatic brain injury: a systematic review and meta-analysis of prevalence rates. J Neurotrauma. 13:1–13
Lippa SM, Pastorek NJ, Benge JF et al (2010) Postconcussive symptoms after blast and nonblast-related mild traumatic brain injuries in afghanistan and iraq war veterans. J Int Neuropsychol Soc. 16(5):856–866
Acknowledgments
We would like to express our gratitude to Caroline Augusta Richelsen for helping with editing the manuscript.
Author information
Authors and Affiliations
Contributions
SA, HWS, and HA initiated the study. AI, HA, HWS, and SA contributed to the study design. AI and HMK carried out the search and data extraction. AI, RBL, and SA contributed to statistical analysis. All authors contributed to interpreting the results. AI and HA wrote the first draft of the manuscript while CAR, HMK, MS, RBL, HWS, and SA contributed significantly with wording and approving the final manuscript. The corresponding author attests that all listed authors meet authorship criteria and that no other individuals meeting the criteria have been omitted.
Corresponding author
Ethics declarations
Competing interests
All authors have completed the ICMJE uniform disclosure form at www.icmje.org/coi_disclosure.pdf and declare: Afrim Iljazi, Hakan Ashina, and Haidar Muhsen Al-Khazali have no support from any organization for the submitted work; no financial relationships with any organizations that might have an interest in the submitted work in the previous 3 years; no other relationships or activities that could appear to have influenced the submitted work. Richard B. Lipton is the Edwin S. Lowe Professor of Neurology at the Albert Einstein College of Medicine in New York. He receives research support from the NIH: 2PO1 AG003949 (Multiple Principal Investigator), 5U10 NS077308 (Principal Investigator), RO1 NS082432 (Investigator), 1RF1 AG057531 (Site Principal Investigator), RF1 AG054548 (Investigator), 1RO1 AG048642 (Investigator), R56 AG057548 (Investigator), K23 NS09610 (Mentor), K23AG049466 (Mentor), and 1K01AG054700 (Mentor). He also receives support from the Migraine Research Foundation and the National Headache Foundation. He serves on the editorial board of Neurology, senior advisor to Headache, and associate editor to Cephalalgia. He has reviewed for the NIA and NINDS; holds stock options in eNeura Therapeutics and Biohaven Holdings; and serves as consultant, advisory board member, or has received honoraria from the American Academy of Neurology, Alder, Allergan, American Headache Society, Amgen, Autonomic Technologies, Avanir, Biohaven, Biovision, Boston Scientific, Dr Reddy’s, Electrocore, Eli Lilly, eNeura Therapeutics, GlaxoSmithKline, Merck, Pernix, Pfizer, Supernus, Teva, Trigemina, Vector, and Vedanta. He receives royalties from Wolff’s Headache, 7th and 8th Edition, Oxford Press University, 2009, Wiley and Informa. Messoud Ashina is a consultant, speaker, or scientific adviser for Alder, Allergan, Amgen, Eli Lilly, Novartis, and Teva. Henrik Winther Schytz has received consultant fees from Teva, Novartis, and BalancAir and received grants from Novartis. Sait Ashina has received consulting fees from Novartis, Amgen, Allergan, Elly Lilly, Supernus, Satsuma, Percept Promius, and Theranica.
Ethical approval
None
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Iljazi, A., Ashina, H., Al-Khazali, H.M. et al. Post-Traumatic Stress Disorder After Traumatic Brain Injury—A Systematic Review and Meta-Analysis. Neurol Sci 41, 2737–2746 (2020). https://doi.org/10.1007/s10072-020-04458-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-020-04458-7