Abstract
Alzheimer’s disease (AD) is a progressive neurodegenerative disease leading to a gradual and irreversible loss of memory, linguistic skills, and perception of time and space, thinking, and behavior. Dietary pattern has been presented as a contributor to the incidence of Alzheimer’s. This study aimed at reviewing the evidence on the relation between dietary pattern and AD. This systematic search was performed on the articles available in PubMed, Scopus, and Web of Sciences databases until May 2019 using keywords, including (diet, food, dietary pattern, food pattern) and (Alzheimer’s disease) among observational studies. After excluding duplicated, and irrelevant studies, 26 studies were eligible for this review study. We categorized the studied dietary patterns into two groups: healthy and unhealthy diet. This study reviewed two case-control, five cross-sectional, and 19 prospective studies. Eight studies assessed unhealthy diet (high-fat diet, high-glycemic diet, sweetened sugary beverage, etc.) and the risk of AD. In addition, the other studies considered the effect of healthy diet such as Mediterranean diet, dietary approaches to stop hypertension (DASH), Mediterranean-DASH intervention for neurodegenerative delay, and seafood-rich diet on AD. This literature review indicated that adherence to a healthy dietary pattern has neuroprotective effects on AD prevention, while unhealthy diet can cause neurodegenerative effects in AD etiology. In conclusion, our findings showed that adherence to healthy diet can decrease oxidative stress and inflammation and accumulation of amyloid-β and consequently can decrease the risk of AD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alzheimer’s disease (AD) is a complex, progressive, and disabling neurodegenerative disease with no cure, leading to cognitive impairment and memory loss. It is also the most common cause of dementia. AD is characterized by the deposition of extracellular amyloid β plaques and the presence of neurofibrillary tangles. This disease affects millions of people around the world [1, 2]. In 2017, it was estimated that 5.5 million Americans of all ages lived with Alzheimer’s dementia [3]. Every 66 s, one person in the USA develops Alzheimer’s dementia, and by 2050, one person in the USA will develop Alzheimer’s dementia every 33 s [2]. Like other common chronic diseases, AD develops as a result of multiple factors rather than a single cause [2]. The greatest risk factors for late-onset Alzheimer’s are older age (being aged 65 or older) [3, 4], having a family history of Alzheimer’s [5, 6], and carrying the APOE e4 gene [7, 8].
Despite the lack of change or modification, the mentioned risk factors to reduce the risk of AD [2], various studies have indicated that factors such as management of cardiovascular risk factors (diabetes, obesity, smoking, and hypertension), regular physical activity, and healthy diet may reduce the risk of dementia [9]. Diet may play an important role in the incidence and prevention of AD and can have considerable potential for nonpharmacological prevention [10]. Some epidemiological evidence has suggested that adherence to a diet with low glycemic index [11], higher consumption of omega-3 polyunsaturated fatty acids [12], calorie restriction [13], and Mediterranean diet (MD) [14] was associated with delay in the onset of dementia and a decrease in AD biomarker burden; however, some studies have showed that a high-glycemic diet and high cholesterol intake as unhealthy diet do not affect the risk of AD [15, 16].
Although many studies have been assessed the association between each of the mentioned dietary patterns alone and AD [17,18,19], no studies have been conducted to summarize findings about this association. According to the high prevalence of AD, assessment of a dietary pattern and its relationship to AD is important, since it can help to decrease the risk of AD. Therefore, the objective of present study was to conduct a systematic review of the findings on dietary pattern and the risk of AD.
Methods
This study was designed based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) protocol [20]. In one review study, we performed a literature search on electronic databases, including PubMed, Scopus, Web of Sciences, and Google Scoular for related publications until May 2019.
Search strategy
A systematic search was performed by two researchers independently using Medical subject headings (MeSH) and non-MeSH keywords as follows: (diet, food, dietary pattern, food pattern) and (Alzheimer’s disease). Furthermore, references of these articles were examined to identify other relevant articles that we had not found them in our initial search. In addition, we did not consider any language, location, and time restrictions in the search strategy.
Inclusion and exclusion criteria
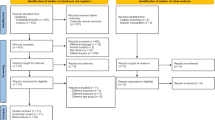
In this review, inclusion criteria were (1) studies examining all dietary patterns using dietary assessment tools, including FFQ, food record and so on; (2) studies conducted on adults (no rat and animal studies); (3) studies assessing the risk of AD; and (4) studies designing observation (prospective or retrospective or cross-sectional or case-control studies). In accordance with the purpose of the study, we considered studies with a long time exposure (dietary pattern). Therefore, we did not include a randomized clinical trial evaluating the effect of diet in a short time. Additionally, we did not include letters, comments, and short communication, since the study method and results were not written completely. Furthermore, we considered only original studies (no review or meta-analysis). Overall, in the initial search, we found 1401 relevant articles that, after removing duplicated studies and screening for interest topic, we selected 119 articles for review with more details. Among these articles, we excluded 30 irrelevant studies. A total of 44 articles were excluded since they did not examine AD and mentioned other cognitive disorders. A total of 18 articles were excluded due to no dietary pattern measured. Furthermore, 19 articles were excluded, since diet was not examined. Finally, 26 studies were included in the current review study (Fig. 1).
Assessment of the study quality
The Newcastle-Ottawa scale, for assessing the quality of non-randomized studies in review studies [21], was used to assess the study quality. Based on this scale, all the reviewed studies had an appropriate quality.
Data extraction and dietary pattern evaluation
The studies’ information was extracted in a checklist designed for this review study. This checklist included the first author, year of publication, study design, country of origin, sample size, gender, studied age range, dietary pattern assessment tools, kind of diet, and the most important outcome.
The introduced dietary patterns of these studies were categorized by two researchers independently into 2 groups: healthy and unhealthy diet. Healthy diet was tended to contain high intake of fruits, vegetables, fish, poultry, whole grain, and liquid oil, particularly olive oil. Unhealthy diet was characterized by high intake of red and processed meat, Sugar-sweetened beverage (SSB), refined grain, high-fat foods such as high-fat dairy, high intake of egg, and high-sugar foods. Based on these definitions among 26 studies, 8 studies were examined unhealthy diet and 19 studies examined healthy diet. (Table 1).
Results
In this literature review, we found 26 relevant studies. A total of 2 studies were case-control [27, 29], 5 studies [37, 38, 44,45,46] were cross-sectional, and other studies were designed prospectively [24, 26, 31, 35, 36, 41, 42, 47,48,49,50,51,52,53,54,55,56,57,58,59]. There were a total of 52–5395 participants per study. These studies were conducted in Australian [24, 32, 38], America [26, 27, 29, 31, 36, 37, 44, 46,47,48, 50,51,52,53, 57, 58], Asian [42, 45], and European countries [35, 49, 54, 59]. The ages in the studies were 25 years old and older in all of the studies. All of the studies examined sexual distribution except studies by Ylilauri et al. [16] and Olsson et al. [35] that participants in this study were male, and Hill et al. [24] examined only females.
All the studies assessed dietary intake by the food frequency questionnaire (FFQ) except four studies [16, 23, 35, 54]. Dietary assessment tools in the studies conducted by Laitinen [23] and Eskelinen [54] were the dietary habit questionnaire, and in studies conducted by Ylilauri [16] and Olsson [35], they were as food record. A total of 8 studies examined the risk of AD and unhealthy dietary patterns, including high-fat diet [22, 23], high-glycemic diet [15], SSB [25], high-cholesterol diet [16], DIF [26], and Western diet [27] (Table 2). Healthy diet and the risk of AD were assessed by 19 studies consisting of MD [19, 24, 28, 29, 32,33,34,35,36,37, 52, 55], dietary approaches to stop hypertension (DASH) [34], Mediterranean-DASH intervention for neurodegenerative delay (MIND) [34, 37], HEI [35, 54], seafood-rich diet [39,40,41], soy-based food diet [42], capsaicin-rich diet [43], high-protein diet [35, 38], and low-fat diet [24]. (Table 3).
In the literature review, we found that high-fat diet was associated with the risk of AD [22, 23]. Both of studies indicated that saturated fats, especially trans-fat, could increase the risk of AD [22, 23], while Hill et al. [24] did not observe any association between high-fat diet and AD. Luchsinger et al. [15] showed that high-glycemic diet as unhealthy diet did not affect the risk of AD even after being adjusted for potential confounders, including age, sex, diabetic, and APOE-ε4. We observed that, in the study by Pase et al. [25] in the community-based Framingham Heart Study, high intake of total sugary beverages, fruit juice, and sugar-sweetened soft drinks in diet increased with the presence of pre-clinical symptom of AD. In another population-based Kuopio Ischaemic Heart Disease Risk Factor study by Ylilauri et al. [16], high cholesterol intake of diet was not associated with the incidence of AD. However, they recommended moderate consumption of egg and other sources of cholesterol to improve cognitive function. DIF as another unhealthy dietary pattern was introduced by Gu et al. [26] that did not observe any association between adherence to DIF and AD. [27] In the study by Gustaw-Rothenberg et al. [27], patients with Alzheimer’s tended to follow Western dietary patterns (Table 2).
Among healthy dietary patterns, MD was studied more than other dietary patterns [19, 24, 28, 29, 32,33,34,35,36,37, 52, 55]. Each of 12 studies reported that MD was associated with the decrease of AD risk [19, 28, 32,33,34]. Morris et al. [34] examined 3 dietary patterns and the risk of AD that, in this study, higher adherence to MD and DASH and moderate adherence to MIND might decrease incidence of AD. Morris et al. in two independent studies [39, 40] showed that intake of all seafoods could decrease AD risk, while Devore et al. [41] did not observe any association between seafood and AD. Liu et al. [43] reported that capsaicin-rich diet had positive effects on biomarker’s AD and cognitive performance. Furthermore, high intake of soy-based food, high protein, and adherence to healthy dietary patterns showed prevention effects on AD [38, 42, 54] (Table 3).
Discussion
Our findings based on the literature review showed that adherence to healthy diet could decrease the risk of AD, while people with unhealthy diet are at an increased risk of AD. Healthy diet was defined as a dietary pattern that helps to maintain and improve the general health of the body with the least harmful effects [60]. Based on this definition, healthy diet was tended to be a regular intake of fruits, vegetables, whole grains, and white meat (fish and poultry) [61]. The major healthy dietary patterns were studied in relation to the risk of AD, including MD [19, 24, 28, 29, 32,33,34,35,36,37, 52, 55], DASH [34], MIND [34, 37], HEI [35, 54], seafood-rich diet [39,40,41], soy-based food diet [42], capsaicin-rich diet [43], high-protein diet [35, 38], and low-fat diet [24].
Mediterranean diet in relation to risk of AD
The expression “Mediterranean diet” refers to the dietary habits of the Mediterranean countries that are characterized by high intake of fruits, vegetables, whole grains, legume, nuts, olive, and olive oil, as well as moderate consumption of fish and fat-free and low-fat dairy products and a decrease of red and processed meat. In this pattern, it is acceptable to have a moderate intake of alcohol and wine, but it is forbidden in some religions [17]. MD has been associated with decrease of cardiovascular disease (CVD), diabetes, metabolic syndrome, cancer, and cognitive disorders [62,63,64,65]. An investigation of the reviewed articles showed that greater adherence to MD was associated with a decrease of AD [19, 24, 28, 29, 32,33,34,35,36,37, 52, 55]. Results of the ATTICA study after 10-year follow-up showed that greater adherence to MD could decrease the level of interleukin 6, human tumor necrosis factor-α, C-reactive protein, and oxy low-density lipoprotein (ox LDL) [66]. High intake of antioxidant foods in this dietary pattern plays a key role in the induction of oxidative stress and inflammatory disorders [28, 30]. High levels of reactive oxygen species and reactive nitrogen were seen in the brain of patients with AD. Inflammation and oxidative stress play a role in AD pathogenesis [32, 67] (Fig. 2). Therefore, MD is known as a neuroprotective dietary pattern due to high intake of fruits, vegetables, polyunsaturated fat, and low alcohol and wine intake that can lead to decrease of inflammation and oxidative stress.
DASH diet in relation to risk of AD
DASH diet is effective in the treatment of hypertension and is recommended for CVDs [68]. This diet emphasizes low consumption of saturated fat, total fat, and red and processed meat and promotion of intake of fruits, vegetables, low-fat dairy, and whole grains [68, 69]. In other words, this approach can increase intake of potassium, calcium, and magnesium and fiber, and simultaneously decrease intake of sodium, refined grains, and saturated fat [69]. Similarly to MD, it seems that DASH diet with effects on inflammatory pathways and oxidative stress can prevent AD (Fig. 2) [70].
High intake of antioxidants, including vitamin C, vitamin E, beta carotene, and flavonoids from fruits, vegetables, and vegetable oils, can enhance antioxidant capacity and reduce inflammation [71]. On the other hand, results from a Québec Longitudinal Study on nutrition and successful aging (NuAge Study) showed that high intake of sodium was associated with the risk of cognitive maintenance in elders [72]. Inconsistent with the results, the study “Health, Aging and Body Composition (Health ABC)” (2018) did not confirm the finding that decrease of sodium and increase of potassium intake were associated with the risk of cognitive disorders [73]. Although no studies have examined the effect of dietary sodium and potassium on AD in elderly population, DASH diet is an appropriate approach to prevent AD due to high intake of dietary antioxidants and anti-inflammatories.
MIND in relation to risk of AD
This dietary pattern has been developed by Morris in Chicago for the first time emphasizing the intake of natural and plant-based foods and limitation of animal food source and saturated fat intake [74]. This healthy diet has 15 components, including 10 brain healthy food groups (green leafy vegetables, other vegetables, nuts, berries, beans, whole grains, white meat (fish and poultry), olive oil instead of other oil, and wine intake limitation) as well as decrease of 5 unhealthy food groups, including red meats, butter and stick margarine, cheese, pastries and sweets, and fried/fast food [34]. The possible mechanisms for the effect of this diet on AD were associated with the high intake of n-3 fatty acids from fish consumption, leading to decrease of amyloid-β formation, oxidative stress, and inflammation [75] (Fig. 2). In addition, these complications related to AD were improved with a high intake of flavonoids, beta carotene, folate, and carotenoids from green leafy vegetables and other vegetables [34].
Other healthy dietary patterns in relation to risk of AD
Other healthy dietary patterns were examined in this review, including seafood, soy-based foods, capsaicin-rich, and high-protein diet that can reduce the risk of AD [38, 39, 42, 43]. Seafood is a source of n-3 fatty acids that can decrease inflammation, oxidative stress, and formation of amyloid-β [75,76,77]. Similarly, capsaicin, as an active ingredient in chili pepper, has effective beneficial effects on AD, which was mediated by a decrease of inflammation and oxidative stress, and neuroprotective effects [78, 79] (Fig. 2). Liu et al. showed that capsaicin-rich diet leads to decrease of amyloid-β formation and can delay AD [43]. Therefore, it seems that adherence to this healthy diet to decrease AD has beneficial effects; however, further studies are required in this field.
Unhealthy diet, Western diet in relation to risk of AD
In the literature review, we observed that 8 studies examined unhealthy dietary patterns and the risk of AD [15, 16, 22,23,24,25,26,27]. These unhealthy dietary patterns included dietary high-fat diet [22, 23], high-glycemic diet [15], SSB [25], high-cholesterol diet [16], DIF [26], and Western diet [27]. In the literature, a high intake of total fat, saturated fat, cholesterol, sodium, processed foods, refined grains, and simple carbohydrates and high sugar intake were considered Western diet [68, 80]. Manzel et al. [81] in their review study stated that Western diet led to increase in inflammatory diseases consisting of obesity, metabolic syndrome, and cardiovascular disease. In another review study, we observed that Western diet can degenerate the blood-brain barrier and a subsequent risk of dementia is mediated by damaging the hippocampus in the brain [82]. To the best of our knowledge, no study has assessed Western diet in relation to the risk of AD and its biological mechanism is still unknown. However, some studies have suggested that a higher intake of saturated and trans fat can increase inflammation and oxidative stress. As stated in healthy dietary patterns, inflammation and oxidative stress play a key role in etiology of AD [22, 23]. Gustaw-Rothenberg et al. [27] evaluated a high intake of processed meat, butter, high-fat dairy products, eggs, and refined sugar and the risk of AD. These foods are the components of Western diet that had harmful effects on AD. Therefore, based on these results, consumption of unsaturated fat, especially n-3 fatty acids instead of saturated and trans fat, to promote neuroprotective effects is recommended (Fig. 2).
Furthermore, despite the role of dietary cholesterol in inflammatory disorders [83], Ylilauri et al. [16] did not observe a significant association between dietary cholesterol and the risk of AD. In his opinion, dietary cholesterol has no effect on the human brain and subsequently cognitive diseases. The other components of Western diet are SSB and increase of glycemic load, which were examined by Pase [25], Luchsinger [15], and Hill [24]. In animal studies, a high intake of sugar enhances the accumulation of amyloid-β and damage of the hippocampus, leading to an increased risk of AD [18, 84]. In the study by Pase et al. [25], a higher SSB intake could cause brain atrophy and increased preclinical AD. While Luchsinger et al. [15] did not find a significant association between high glycemic load diet and the risk of AD, they reported that the lack of an association may be due to the measurement error of the glycemic index and the under-reporting of carbohydrate intake by the studied population. Hill [24] introduced these foods as “junk foods,” which were significantly associated with amyloid-β deposition.
Conclusion
In conclusion, our findings based on the literature review showed that adherence to a healthy dietary pattern has neuroprotective effects on AD prevention. These major healthy dietary patterns include MD, DASH, MIND, HEI, dietary seafood, soy-based foods, capsaicin-rich, high-protein, and low-fat diet that can decrease oxidative stress and inflammation and accumulation of amyloid-β, while, among these reviewed studies, we observed that unhealthy dietary patterns, particularly in a high intake of fat and SSB, can promote oxidative stress and inflammation and accumulation of amyloid-β and subsequent development of AD. However, more studies are required in other components of dietary patterns in relation to AD. Overall, according to the limited number of studies on AD and dietary patterns, the definition of a healthy and unhealthy diet is too little specified. Therefore, further well-designed and controlled studies seem to be needed to identify a healthy diet in this population.
References
Blennow K, de Leon MJ, Zetterberg H (2006) Alzheimer’s disease. Lancet (London, England) 368(9533):387–403. https://doi.org/10.1016/s0140-6736(06)69113-7
Association As (2017) 2017 Alzheimer’s disease facts and figures. Alzheimers Dement 13(4):325–373
Hebert LE, Weuve J, Scherr PA, Evans DA (2013) Alzheimer disease in the United States (2010–2050) estimated using the 2010 census. Neurology 80(19):1778–1783
Hebert L, Bienias J, Aggarwal N, Wilson R, Bennett D, Shah R, Evans D (2010) Change in risk of Alzheimer disease over time. Neurology 75(9):786–791
Green RC, Cupples LA, Go R, Benke KS, Edeki T, Griffith PA, Williams M, Hipps Y, Graff-Radford N, Bachman D (2002) Risk of dementia among white and African American relatives of patients with Alzheimer disease. Jama 287(3):329–336
Tyas SL, Manfreda J, Strain LA, Montgomery PR (2001) Risk factors for Alzheimer’s disease: a population-based, longitudinal study in Manitoba, Canada. Int J Epidemiol 30(3):590–597
Saunders AM, Strittmatter WJ, Schmechel D, George-Hyslop PS, Pericak-Vance M, Joo S, Rosi B, Gusella J, Crapper-MacLachlan D, Alberts M (1993) Association of apolipoprotein E allele ϵ4 with late-onset familial and sporadic Alzheimer’s disease. Neurology 43(8):1467–1467
Farrer LA, Cupples LA, Haines JL, Hyman B, Kukull WA, Mayeux R, Myers RH, Pericak-Vance MA, Risch N, Van Duijn CM (1997) Effects of age, sex, and ethnicity on the association between apolipoprotein E genotype and Alzheimer disease: a meta-analysis. Jama 278(16):1349–1356
Baumgart M, Snyder HM, Carrillo MC, Fazio S, Kim H, Johns H (2015) Summary of the evidence on modifiable risk factors for cognitive decline and dementia: a population-based perspective. Alzheimers Dement 11(6):718–726
Hill E, Clifton P, Goodwill AM, Dennerstein L, Campbell S, Szoeke C (2018) Dietary patterns and β-amyloid deposition in aging Australian women. Alzheimer’s & Dementia: Translational Research & Clinical Interventions
Taylor MK, Sullivan DK, Swerdlow RH, Vidoni ED, Morris JK, Mahnken JD, Burns JM (2017) A high-glycemic diet is associated with cerebral amyloid burden in cognitively normal older adults. Am J Clin Nutr 106(6):1463–1470
Gu Y, Schupf N, Cosentino S, Luchsinger J, Scarmeas N (2012) Nutrient intake and plasma β-amyloid. Neurology 78(23):1832–1840
Wang J, Ho L, Qin W, Rocher AB, Seror I, Humala N, Maniar K, Dolios G, Wang R, Hof PR (2005) Caloric restriction attenuates β-amyloid neuropathology in a mouse model of Alzheimer’s disease. FASEB J 19(6):659–661
Merrill DA, Siddarth P, Raji CA, Emerson ND, Rueda F, Ercoli LM, Miller KJ, Lavretsky H, Harris LM, Burggren AC (2016) Modifiable risk factors and brain positron emission tomography measures of amyloid and tau in nondemented adults with memory complaints. Am J Geriatr Psychiatry 24(9):729–737
Luchsinger J, Tang M, Mayeux R (2007) Glycemic load and risk of Alzheimer’s disease. The Journal of Nutrition, Health & Aging 11(3):238
Ylilauri MP, Voutilainen S, Lönnroos E, Mursu J, Virtanen HE, Koskinen TT, Salonen JT, Tuomainen T-P, Virtanen JK (2017) Association of dietary cholesterol and egg intakes with the risk of incident dementia or Alzheimer disease: the Kuopio Ischaemic Heart Disease Risk Factor Study, 2. Am J Clin Nutr 105(2):476–484
Dernini S, Berry EM (2015) Mediterranean diet: from a healthy diet to a sustainable dietary pattern. Frontiers in nutrition 2:15
Francis HM, Stevenson RJ (2011) Higher reported saturated fat and refined sugar intake is associated with reduced hippocampal-dependent memory and sensitivity to interoceptive signals. Behav Neurosci 125(6):943–955
Gu Y, Luchsinger JA, Stern Y, Scarmeas N (2010) Mediterranean diet, inflammatory and metabolic biomarkers, and risk of Alzheimer’s disease. J Alzheimers Dis 22(2):483–492
Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, Shekelle P, Stewart LA (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Systematic Reviews 4(1). https://doi.org/10.1186/2046-4053-4-1
Scale N-O, Wells G, Shea B, O’Connell D, Peterson J, Welch V, Losos M, al e (2014) The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses
Morris MC, Evans DA, Bienias JL, Tangney CC, Bennett DA, Aggarwal N, Schneider J, Wilson RS (2003) Dietary fats and the risk of incident Alzheimer disease. Arch Neurol 60(2):194–200
Laitinen M, Ngandu T, Rovio S, Helkala E-L, Uusitalo U, Viitanen M, Nissinen A, Tuomilehto J, Soininen H, Kivipelto M (2006) Fat intake at midlife and risk of dementia and Alzheimer’s disease: a population-based study. Dement Geriatr Cogn Disord 22(1):99–107
Hill E, Clifton P, Goodwill AM, Dennerstein L, Campbell S, Szoeke C (2018) Dietary patterns and β-amyloid deposition in aging Australian women. Alzheimer’s and Dementia: Translational Research and Clinical Interventions 4:535–541. https://doi.org/10.1016/j.trci.2018.09.007
Pase MP, Himali JJ, Jacques PF, DeCarli C, Satizabal CL, Aparicio H, Vasan RS, Beiser AS, Seshadri S (2017) Sugary beverage intake and preclinical Alzheimer’s disease in the community. Alzheimers Dement 13(9):955–964
Gu Y, Nieves JW, Luchsinger JA, Scarmeas N (2011) Dietary inflammation factor rating system and risk of Alzheimer disease in elders. Alzheimer Dis Assoc Disord 25(2):149–154. https://doi.org/10.1097/WAD.0b013e3181ff3c6a
Gustaw-Rothenberg K (2009) Dietary patterns associated with Alzheimer’s disease: population based study. Int J Environ Res Public Health 6(4):1335–1340. https://doi.org/10.3390/ijerph6041335
Scarmeas N, Stern Y, Tang MX, Mayeux R, Luchsinger JA (2006) Mediterranean diet and risk for Alzheimer’s disease. Ann Neurol 59(6):912–921
Scarmeas N, Stern Y, Mayeux R, Luchsinger JA (2006) Mediterranean diet, Alzheimer disease, and vascular mediation. Arch Neurol 63(12):1709–1717. https://doi.org/10.1001/archneur.63.12.noc60109
Scarmeas N, Luchsinger JA, Schupf N, Brickman AM, Cosentino S, Tang MX, Stern Y (2009) Physical activity, diet, and risk of Alzheimer disease. Jama 302(6):627–637
Gu Y, Luchsinger JA, Stern Y, Scarmeas N (2010) Mediterranean diet, inflammatory and metabolic biomarkers, and risk of Alzheimer’s disease. J Alzheimer Dis 22(2):483–492. https://doi.org/10.3233/jad-2010-100897
Gardener S, Gu Y, Rainey-Smith SR, Keogh JB, Clifton PM, Mathieson S, Taddei K, Mondal A, Ward VK, Scarmeas N (2012) Adherence to a Mediterranean diet and Alzheimer’s disease risk in an Australian population. Transl Psychiatry 2(10):e164
Mosconi L, Murray J, Tsui W, Li Y, Davies M, Williams S, Pirraglia E, Spector N, Osorio R, Glodzik L (2014) Mediterranean diet and magnetic resonance imaging-assessed brain atrophy in cognitively normal individuals at risk for Alzheimer’s disease. J Prev Alzheimers Dis 1(1):23
Morris MC, Tangney CC, Wang Y, Sacks FM, Bennett DA, Aggarwal NT (2015) MIND diet associated with reduced incidence of Alzheimer’s disease. Alzheimers Dement 11(9):1007–1014
Olsson E, Karlstrom B, Kilander L, Byberg L, Cederholm T, Sjogren P (2015) Dietary patterns and cognitive dysfunction in a 12-year follow-up study of 70 year old men. J Alzheimer Dis 43(1):109–119. https://doi.org/10.3233/jad-140867
Vassilaki M, Aakre JA, Syrjanen JA, Mielke MM, Geda YE, Kremers WK, Machulda MM, Alhurani RE, Staubo SC, Knopman DS, Petersen RC, Lowe VJ, Jack CR, Roberts RO (2018) Mediterranean diet, its components, and amyloid imaging biomarkers. J Alzheimers Dis 64(1):281–290. https://doi.org/10.3233/JAD-171121
Calil SRB, Brucki SMD, Nitrini R, Yassuda MS (2018) Adherence to the Mediterranean and MIND diets is associated with better cognition in healthy seniors but not in MCI or AD. Clinical Nutrition ESPEN 28:201–207. https://doi.org/10.1016/j.clnesp.2018.08.001
Fernando W, Rainey-Smith SR, Gardener SL, Villemagne VL, Burnham SC, Macaulay SL, Brown BM, Gupta VB, Sohrabi HR, Weinborn M, Taddei K, Laws SM, Goozee K, Ames D, Fowler C, Maruff P, Masters CL, Salvado O, Rowe CC, Martins RN (2018) Associations of dietary protein and fiber intake with brain and blood amyloid-beta. J Alzheimer Dis 61(4):1589–1598. https://doi.org/10.3233/jad-170742
Morris MC, Evans DA, Bienias JL, Tangney CC, Bennett DA, Wilson RS, Aggarwal N, Schneider J (2003) Consumption of fish and n-3 fatty acids and risk of incident Alzheimer disease. Arch Neurol 60(7):940–946
Morris MC, Brockman J, Schneider JA, Wang Y, Bennett DA, Tangney CC, van de Rest O (2016) Association of seafood consumption, brain mercury level, and APOE ε4 status with brain neuropathology in older adults. Jama 315(5):489–497
Devore EE, Grodstein F, van Rooij FJ, Hofman A, Rosner B, Stampfer MJ, Witteman JC, Breteler MM (2009) Dietary intake of fish and omega-3 fatty acids in relation to long-term dementia risk. Am J Clin Nutr 90(1):170–176. https://doi.org/10.3945/ajcn.2008.27037
Ozawa M, Ninomiya T, Ohara T, Doi Y, Uchida K, Shirota T, Yonemoto K, Kitazono T, Kiyohara Y (2013) Dietary patterns and risk of dementia in an elderly Japanese population: the Hisayama Study. Am J Clin Nutr 97(5):1076–1082. https://doi.org/10.3945/ajcn.112.045575
Liu C-H, Bu X-L, Wang J, Zhang T, Xiang Y, Shen L-L, Wang Q-H, Deng B, Wang X, Zhu C (2016) The associations between a capsaicin-rich diet and blood amyloid-β levels and cognitive function. J Alzheimers Dis 52(3):1081–1088
Mosconi L, Murray J, Davies M, Williams S, Pirraglia E, Spector N, Tsui WH, Li Y, Butler T, Osorio RS, Glodzik L, Vallabhajosula S, McHugh P, Marmar CR, de Leon MJ (2014) Nutrient intake and brain biomarkers of Alzheimer’s disease in at-risk cognitively normal individuals: a cross-sectional neuroimaging pilot study. BMJ Open 4(6):e004850. https://doi.org/10.1136/bmjopen-2014-004850
Liu CH, Bu XL, Wang J, Zhang T, Xiang Y, Shen LL, Wang QH, Deng B, Wang X, Zhu C, Yao XQ, Zhang M, Zhou HD, Wang YJ (2016) The associations between a capsaicin-rich diet and blood amyloid-β levels and cognitive function. J Alzheimers Dis 52(3):1081–1088. https://doi.org/10.3233/JAD-151079
Morris MC, Brockman J, Schneider JA, Wang Y, Bennett DA, Tangney CC, van de Rest O (2016) Association of seafood consumption, brain mercury level, and APOE epsilon4 status with brain neuropathology in older adults. Jama 315(5):489–497. https://doi.org/10.1001/jama.2015.19451
Morris MC, Evans DA, Bienias JL, Tangney CC, Bennett DA, Aggarwal N, Schneider J, Wilson RS (2003) Dietary fats and the risk of incident Alzheimer disease. Arch Neurol 60(2):194–200
Morris MC, Evans DA, Bienias JL, Tangney CC, Bennett DA, Wilson RS, Aggarwal N, Schneider J (2003) Consumption of fish and n-3 fatty acids and risk of incident Alzheimer disease. Arch Neurol 60(7):940–946. https://doi.org/10.1001/archneur.60.7.940
Laitinen MH, Ngandu T, Rovio S, Helkala EL, Uusitalo U, Viitanen M, Nissinen A, Tuomilehto J, Soininen H, Kivipelto M (2006) Fat intake at midlife and risk of dementia and Alzheimer’s disease: a population-based study. Dement Geriatr Cogn Disord 22(1):99–107. https://doi.org/10.1159/000093478
Scarmeas N, Stern Y, Tang MX, Mayeux R, Luchsinger JA (2006) Mediterranean diet and risk for Alzheimer’s disease. Ann Neurol 59(6):912–921. https://doi.org/10.1002/ana.20854
Luchsinger JA, Tang MX, Mayeux R (2007) Glycemic load and risk of Alzheimer’s disease. J Nutr Health Aging 11(3):238–241
Scarmeas N, Stern Y, Mayeux R, Manly JJ, Schupf N, Luchsinger JA (2009) Mediterranean diet and mild cognitive impairment. Arch Neurol 66(2):216–225. https://doi.org/10.1001/archneurol.2008.536
Gu YA, Nieves JW, Stern Y, Luchsinger JA, Scarmeas N (2010) Food combination and Alzheimer disease risk a protective diet. Arch Neurol 67(6):699–706. https://doi.org/10.1001/archneurol.2010.84
Eskelinen MH, Ngandu T, Tuomilehto J, Soininen H, Kivipelto M (2011) Midlife healthy-diet index and late-life dementia and Alzheimer’s disease. Dement Geriatr Cogn Dis Extra 1(1):103–112
Gardener S, Gu Y, Rainey-Smith SR, Keogh JB, Clifton PM, Mathieson SL, Taddei K, Mondal A, Ward VK, Scarmeas N, Barnes M, Ellis KA, Head R, Masters CL, Ames D, Macaulay SL, Rowe CC, Szoeke C, Martins RN (2012) Adherence to a Mediterranean diet and Alzheimer’s disease risk in an Australian population. Transl Psychiatry 2:e164. https://doi.org/10.1038/tp.2012.91
Berti V, Murray J, Davies M, Spector N, Tsui WH, Li Y, Williams S, Pirraglia E, Vallabhajosula S, McHugh P, Pupi A, de Leon MJ, Mosconi L (2015) Nutrient patterns and brain biomarkers of Alzheimer’s disease in cognitively normal individuals. J Nutr Health Aging 19(4):413–423. https://doi.org/10.1007/s12603-014-0534-0
Morris MC, Tangney CC, Wang YM, Sacks FM, Bennett DA, Aggarwal NT (2015) MIND diet associated with reduced incidence of Alzheimer’s disease. Alzheimers Dement 11(9):1007–1014. https://doi.org/10.1016/j.jalz.2014.11.009
Pase MP, Himali JJ, Jacques PF, DeCarli C, Satizabal CL, Aparicio H, Vasan RS, Beiser AS, Seshadri S (2017) Sugary beverage intake and preclinical Alzheimer’s disease in the community. Alzheimer Dementia 13(9):955–964. https://doi.org/10.1016/j.jalz.2017.01.024
Ylilauri MPT, Voutilainen S, Lönnroos E, Mursu J, Virtanen HEK, Koskinen TT, Salonen JT, Tuomainen TP, Virtanen JK (2017) Association of dietary cholesterol and egg intakes with the risk of incident dementia or Alzheimer disease: the Kuopio Ischaemic Heart Disease Risk Factor Study. Am J Clin Nutr 105(2):476–484. https://doi.org/10.3945/ajcn.116.146753
de Ridder D, Kroese F, Evers C, Adriaanse M, Gillebaart M (2017) Healthy diet: health impact, prevalence, correlates, and interventions. Psychol Health 32(8):907–941
Lv N, Xiao L, Ma J (2014) Dietary pattern and asthma: a systematic review and meta-analysis. J Asthma Allergy 7:105
Schwingshackl L, Hoffmann G (2015) Adherence to Mediterranean diet and risk of cancer: an updated systematic review and meta-analysis of observational studies. Cancer Med 4(12):1933–1947
Sleiman D, Al-Badri MR, Azar ST (2015) Effect of Mediterranean diet in diabetes control and cardiovascular risk modification: a systematic review. Front Public Health 3:69
Widmer RJ, Flammer AJ, Lerman LO, Lerman A (2015) The Mediterranean diet, its components, and cardiovascular disease. Am J Med 128(3):229–238
Hardman RJ, Kennedy G, Macpherson H, Scholey AB, Pipingas A (2016) Adherence to a Mediterranean-style diet and effects on cognition in adults: a qualitative evaluation and systematic review of longitudinal and prospective trials. Front Nutr 3:22
Koloverou E, Panagiotakos D, Pitsavos C, Chrysohoou C, Georgousopoulou E, Grekas A, Christou A, Chatzigeorgiou M, Skoumas I, Tousoulis D (2016) Adherence to Mediterranean diet and 10-year incidence (2002–2012) of diabetes: correlations with inflammatory and oxidative stress biomarkers in the ATTICA cohort study. Diabetes Metab Res Rev 32(1):73–81
Singh B, Parsaik AK, Mielke MM, Erwin PJ, Knopman DS, Petersen RC, Roberts RO (2014) Association of mediterranean diet with mild cognitive impairment and Alzheimer’s disease: a systematic review and meta-analysis. J Alzheimers Dis 39(2):271–282
Rai SK, Fung TT, Lu N, Keller SF, Curhan GC, Choi HK (2017) The Dietary Approaches to Stop Hypertension (DASH) diet, Western diet, and risk of gout in men: prospective cohort study. bmj 357:j1794
Siervo M, Lara J, Chowdhury S, Ashor A, Oggioni C, Mathers JC (2015) Effects of the Dietary Approach to Stop Hypertension (DASH) diet on cardiovascular risk factors: a systematic review and meta-analysis. Br J Nutr 113(1):1–15
Tangney CC, Li H, Wang Y, Barnes L, Schneider JA, Bennett DA, Morris MC (2014) Relation of DASH-and Mediterranean-like dietary patterns to cognitive decline in older persons. Neurology 83(16):1410–1416
Siti HN, Kamisah Y, Kamsiah J (2015) The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (a review). Vasc Pharmacol 71:40–56
Fiocco AJ, Shatenstein B, Ferland G, Payette H, Belleville S, Kergoat M-J, Morais JA, Greenwood CE (2012) Sodium intake and physical activity impact cognitive maintenance in older adults: the NuAge Study. Neurobiol Aging 33(4):829. e821–829. e828
Nowak KL, Fried L, Jovanovich A, Ix J, Yaffe K, You Z, Chonchol M (2018) Dietary sodium/potassium intake does not affect cognitive function or brain imaging indices. Am J Nephrol 47(1):57–65
Marcason W (2015) What are the components to the MIND diet? J Acad Nutr Diet 115(10):1744
Berendsen AM, Kang J, Feskens E, de Groot C, Grodstein F, Van de Rest O (2018) Association of long-term adherence to the mind diet with cognitive function and cognitive decline in American women. J Nutr Health Aging 22(2):222–229
Samieri C, Morris M-C, Bennett DA, Berr C, Amouyel P, Dartigues J-F, Tzourio C, Chasman DI, Grodstein F (2017) Fish intake, genetic predisposition to Alzheimer disease, and decline in global cognition and memory in 5 cohorts of older persons. Am J Epidemiol 187(5):933–940
Connor WE, Connor SL (2007) The importance of fish and docosahexaenoic acid in Alzheimer disease. Oxford University Press,
Avraham Y, Grigoriadis N, Magen I, Poutahidis T, Vorobiav L, Zolotarev O, Ilan Y, Mechoulam R, Berry E (2009) Capsaicin affects brain function in a model of hepatic encephalopathy associated with fulminant hepatic failure in mice. Br J Pharmacol 158(3):896–906
Kang C, Wang B, Kaliannan K, Wang X, Lang H, Hui S, Huang L, Zhang Y, Zhou M, Chen M (2017) Gut microbiota mediates the protective effects of dietary capsaicin against chronic low-grade inflammation and associated obesity induced by high-fat diet. MBio 8(3):e00470–e00417
Hariharan D, Vellanki K, Kramer H (2015) The Western diet and chronic kidney disease. Curr Hypertens Rep 17(3):16. https://doi.org/10.1007/s11906-014-0529-6
Manzel A, Muller DN, Hafler DA, Erdman SE, Linker RA, Kleinewietfeld M (2014) Role of “Western diet” in inflammatory autoimmune diseases. Curr Allergy Asthma Rep 14(1):404
Hsu TM, Kanoski SE (2014) Blood-brain barrier disruption: mechanistic links between Western diet consumption and dementia. Front Aging Neurosci 6:88
Kleemann R, Verschuren L, van Erk MJ, Nikolsky Y, Cnubben NH, Verheij ER, Smilde AK, Hendriks HF, Zadelaar S, Smith GJ (2007) Atherosclerosis and liver inflammation induced by increased dietary cholesterol intake: a combined transcriptomics and metabolomics analysis. Genome Biol 8(9):R200
Cao D, Lu H, Lewis TL, Li L (2007) Intake of sucrose-sweetened water induces insulin resistance and exacerbates memory deficits and amyloidosis in a transgenic mouse model of Alzheimer disease. J Biol Chem 282(50):36275–36282
Acknowledgments
We appreciate the Department of Nutrition, Kermanshah University of Medical Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 24 kb)
Rights and permissions
About this article
Cite this article
Samadi, M., Moradi, S., Moradinazar, M. et al. Dietary pattern in relation to the risk of Alzheimer’s disease: a systematic review. Neurol Sci 40, 2031–2043 (2019). https://doi.org/10.1007/s10072-019-03976-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-019-03976-3