Abstract
Multiple sclerosis (MS) is a demyelinating and neurodegenerative disease of the central nervous system (CNS), most probably autoimmune in origin, usually occurring in young adults with a female/male prevalence of approximately 3:1. Women with MS in the reproductive age may face challenging issues in reconciling the desire for parenthood with their condition, owing to the possible influence both on the ongoing or planned treatment with the possible consequences on the disease course and on the potential negative effects of treatments on foetal and pregnancy outcomes. At MS diagnosis, timely counselling should promote informed parenthood, while disease evolution should be assessed before making therapeutic decisions. Current guidelines advise the discontinuation of any treatment during pregnancy, with possible exceptions for some treatments in patients with very active disease. Relapses decline during pregnancy but are more frequent during puerperium, when MS therapy should be promptly resumed in most of the cases. First-line immunomodulatory agents, such as interferon-β (IFN-β) and glatiramer acetate (GA), significantly reduce the post-partum risk of relapse. Due to substantial evidence of safety with the use of GA during pregnancy, a recent change in European marketing authorization removed the pregnancy contraindication for GA. This paper reports a consensus of Italian experts involved in MS management, including neurologists, gynaecologists and psychologists. This consensus, based on a review of the available scientific evidence, promoted an interdisciplinary approach to the management of pregnancy in MS women.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Multiple sclerosis (MS) is an inflammatory and neurodegenerative demyelinating disease of the central nervous system (CNS), most likely autoimmune in origin, usually occurring in young adults, with a female/male ratio of approximately 3:1 [1]. Therefore, in female patients, the disease most often occurs during the childbearing period [2], which highlights the relevance of pregnancy-related issues in the management of MS patients [3].
Whereas 50–60 years ago women with MS were simply discouraged from initiating a pregnancy, better information about the risks of pregnancy has been provided to MS patients in the following decades [4]. In particular, a turning point in the approach to pregnancy resulted from the multicentre observational Pregnancy in Multiple Sclerosis (PRIMS) study that involved 12 European countries in the follow-up of 254 women with MS during their pregnancy and 12 months after. This study provided documentation that the relapse rate declined during pregnancy, particularly in the third trimester, and increased in the post-partum period, particularly in the first [5]. Further studies followed over the two decades since then, which have included (i) follow-up surveys [6], (ii) recent observational studies that either excluded a long-term effect [7] or observed a protective effect of pregnancy on maternal MS [8, 9] and (iii) systematic reviews [10]. In addition, the availability of an increasing number of therapies with specific effects on pregnancy and understanding of the impact of treatment discontinuation because of improved knowledge about the disease in general [1] and pregnancy in particular [11] has contributed to a significant change in the management of MS patients who consider becoming pregnant. The attitude of specialists is currently focused on the importance of both adequate and timely counselling for family planning and therapeutic management before, during and after pregnancy.
A panel of Italian experts in different areas of the management of pregnancy in MS patients examined the available evidence in the field and discussed the different actions to be taken before, during and after pregnancy in MS patients, focusing on planning, counselling and therapeutic choices. This paper reports the consensus reached by this panel and highlights the importance of establishing an interdisciplinary approach.
Planning
MS is now generally considered to be a condition that does not preclude parenthood and pregnancy [10]. However, pregnancy planning is an essential requirement for a responsible maternity. As for contraception, not only there is no evidence that oral contraceptives can negatively affect the clinical course of MS [12], but it has also been suggested that oral contraceptives with high dose oestrogens can provide anti-inflammatory effects. These hormones, when administered in combination with interferon-β (IFN-β) in women with relapsing-remitting MS, are effective in reducing the rate of development of new brain lesions observed at magnetic resonance imaging (MRI) [13]. Therefore, the first key message to be conveyed to a woman affected by MS should be the necessity of planning the pregnancy.
Thus, and similar to any other chronic disease, it is appropriate that proposed parenthood is discussed with the woman and her partner, taking into consideration the activity and severity of the disease. In this context, to achieve a complete picture of the patient condition beyond her neurological status, possible psychosocial, psychiatric or cognitive disturbances as well as the presence of comorbidities should be considered in the counselling and carefully evaluated, with the aid of a psychologist and a psychiatrist when needed [14]. Moreover, the neurologist should discuss with the couple the responsibilities and future commitments related to parenthood, as well as possible issues related to childbearing, child raising and disease prognosis. The ultimate goal of the interdisciplinary medical team is, indeed, to minimize risks to the woman with MS, the newborn and then growing child, as well as the couple itself [15].
Ideally, the time of diagnosis of MS and pregnancy planning should not coincide, because a time window is needed for the neurologist to evaluate disease activity and course in the specific case and moreover the risk of an attack is higher immediately after the first attack. Therefore, information and education of the patient about the need to plan the pregnancy should be established from the beginning of the therapeutic relationship. Moreover, since the diagnosis often coincides with the initiation of a therapy, the therapeutic choice should take into clear consideration the woman’s desire to undertake a pregnancy in the short-term, when it is compatible with the type of disease evolution.
Therefore, optimal planning starts in the pre-pregnancy phase and involves several steps, as outlined in the following sections.
Counselling
In the pre-pregnancy phase, all efforts should be made to reconcile the patient desire for parenthood with the presence of a chronic disease. Therefore, the neurologist should promote informed parenthood though proactive discussion and counselling at diagnosis, including contraception indications. Each case should be examined specifically, in order to adopt the most appropriate approach.
An important point often raised by the patients is the fear of hereditary transmission of the disease, and this issue needs to be clearly addressed. MS is not hereditarily transmissible, since both genetic and environmental factors influence the susceptibility to the disease [1]. There is a higher risk of developing the disease if relatives are affected, and the risk increases proportionally to the genetic sharing. In western countries, the age-adjusted risk in a child having one parent with MS is 2%, a 15-fold higher figure compared to the risk (0.3%) of the general population [1]. Therefore, the risk must be quantified on a case-by-case basis, also considering geographical differences. Indeed, most published data derive from North European patient cohorts, and they may not fit other ethnical entities: for instance, the risk of MS for dizygotic twins is approximately 1:10 in North Europe [1] and 1:30 in Italy [16]. Finally, a strict minority of families may present with three or more cases of MS among relatives and these “multiplex families” may require a specific genetic counselling [17].
Disease characteristics
The characteristics of the disease should be carefully evaluated in making a decision about pregnancy. Even if pregnancy reduces MS disease activity during the last trimester, several studies indicate that pregnancy should not be pursued in the presence of highly active or aggressive disease [18, 19]. When a patient receives a proven diagnosis of MS, the neurologist may ask for a wait of 1 year before making pregnancy plans, to allow a sufficient time interval in order to observe the evolution of the disease.
In fact, the severity of the disease is assessed not only on the basis of the clinical and MRI characteristics at onset but also on the basis of disease evolution over time and response to previous or ongoing therapies. Therefore, the neurologist needs a time window during the course of the disease to perform an appropriate evaluation, before sharing a decision with the woman. Furthermore, as already mentioned, the presence of comorbidities, psychosocial, psychiatric or cognitive issues [20] needs to be included in the integrated evaluation of risks possibly associated with pregnancy in MS patients.
Therapeutic decision
The most appropriate treatment for a patient with MS who is planning a pregnancy should be identified considering the severity of the disease, the potential impact of the drugs on pregnancy and foetal outcomes as well as the risk of relapses in the mother. The availability of large datasets providing solid information on the influences of available medications on fertility, pregnancy and childbirth is therefore of paramount importance for appropriate counselling.
Since 1979, the United States Food and Drug Administration (FDA) has classified the drugs that may represent a risk during pregnancy under a five-letter system. Recently, a progressive replacement of this system with a narrative structure for pregnancy labelling was promoted [21]. A similar classification was issued by the European Medicines Agency (EMA) [22]. Moreover, European consensus guidelines have also given recommendations on this matter [23].
The choice of medications may also involve fertility issues. In MS patients, fecundity can be lower than in non-affected women, both due to possible endocrine and sexual disturbances associated with MS and because of the negative effects of some medications on fertility [24]. Therefore, women with MS who face problems in becoming pregnant may decide to undergo assisted reproductive techniques (ARTs), such as in vitro fertilization (IVF) [24]. While MS does not influence the success of IVF, ART failure may increase the relapse rate in patients with MS. The mechanism is possibly multifactorial, partly because of the decrease in sex hormones, partly due to the use of some drugs for ovarian desensitization. Agonists of gonadotropin-releasing hormone (GnRH) stimulate the immune system and, considering the dosage commonly used, this may explain the observed increase in relapse rate. On the contrary, the use of GnRH antagonists or recombinant gonadotropins does not affect MS relapse rates [25]. The potential risks and benefits of ARTs should be discussed with the patient by the neurologist. Patients should also consider undergoing the ART procedure during a period of quiescent, not active disease, and gynaecologists should discuss the use of GnRH antagonists rather than agonists with the patient [11].
Vitamin D deficiency should be investigated before pregnancy and supplementation provided on a case-by-case basis. Vitamin D supplementation is particularly important when blood concentrations of the vitamin fall below normal levels. In general, there is a large body of evidence suggesting that vitamin D deficiency increases MS risk [26] and women with MS have lower vitamin D levels during pregnancy and puerperium, compared with non-affected women [27]. In particular, the role of maternal vitamin D deficiency during pregnancy was established in a nested case-control study by the Finnish Maternity Cohort [28], where maternal 25(OH)D levels <12.02 ng/mL during early pregnancy were associated with a nearly twofold increased risk of MS in the offspring, compared with women who did not have deficient vitamin levels. Therefore, women with MS in pre-pregnancy should be counselled to take vitamin D at the average dosage of 1000–2000 units/day recommended by the American College of Obstetricians and Gynaecologists [29].
Omega-3 fatty acids and docosahexaenoic acid (DHA), in particular, are essential for the development of the central nervous system (CNS) of the foetus and newborn and are commonly acquired by an adequate diet [30]. In all pregnant women, including those with MS, it is appropriate to evaluate the dosage received from the dietary source and consequently to indicate DHA supplementation if needed.
There is also a large body of evidence of the importance of folic acid, which is commonly administered at 0.4–1.0 g daily doses during the periconceptional period and to all pregnant MS patients until the first trimester of pregnancy, unless the presence of a specific deficit of folic acid requires a longer administration period [10].
A retrospective cohort study demonstrated a higher risk of anaemia during pregnancy in MS patients, compared to healthy individuals, as well as a higher risk for infants to undergo meconium aspiration syndrome [31]. Protocols are available that define blood count thresholds associated with the need for intervention with specific medications, especially based on the integration with bioavailable iron. Whether the screening for anaemia routinely performed in all women at first and third trimester of pregnancy helps to identify subjects at risk among MS patients before and during pregnancy is a debated issue. The implementation of the screening approach for anaemia and the application of the related protocols are nevertheless recommended [31].
Smoking cessation as well as guidance on sleep and accommodations to family or professional life are common recommendations for all women contemplating a pregnancy programme, including MS patients [10].
Management of MS therapy: before pregnancy
MS is not a risk factor for pregnancy per se, and on the contrary, it likely has a protective effect [32] which can be explained by the observation that the high levels of circulating sex hormones associated with pregnancy exert several epigenetic effects, including those on inflammation and immune response genes [12]. Nevertheless, pregnancies are classified as low risk or high risk. In the “treatment era” of MS, due to intervening choices about treatment before, during and after pregnancy, only in particular circumstances will pregnancy be classified as “at risk”. However, although pregnancy in a MS patient is not “at risk” because the outcome is usually that of a “normal” pregnancy, there is a need for more intensive and more “medical” monitoring, performed by specialists in foeto-maternal medicine teamed with gynaecologists and with the aid of more involved sonographic investigations, together with the aim of reassuring the patient [11]. The choice of anaesthesia or delivery method should be based in all cases only on obstetrical indications, as in women not affected by MS [33]. Furthermore, the reassuring message to be conveyed to the woman is that in the majority of cases pregnancy in MS women undergoes a physiological course, as shown by a large database study of pregnancy outcomes in over 10,000 women with MS, revealing that the frequency of adverse outcomes was comparable to that of the general obstetric population [34].
Decisions about pregnancy in MS women should clearly take into account whether the patient is already on therapy or not.
The presence of stable disease is the optimal condition allowing the patient to tolerate the pregnancy. Therefore, the appropriate timing for planning a pregnancy cannot be established during active disease but only after response to therapy has been validated with a proven condition of no evidence of disease activity (NEDA), as determined by clinical and MRI parameters [35]. In the optimal situation, this condition of inactivity needs to persist for at least 2 years, which seems to be the most appropriate period to take the risk of therapy discontinuation, as shown by recent NEDA evaluations for determining therapy strategies [36, 37]. However, this period may be reduced to 1 year due to practical reasons and, in general, the duration of the observation should be individualized and adapted to the type of treatment and disease evolution. Moreover, the specific type of ongoing therapy should always be considered before taking a decision.
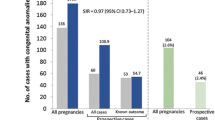
Regarding the discontinuation of ongoing therapies, both the EFNS guidelines [38] and the recent European guidelines on MS pharmacological management, jointly issued by the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) and the European Academy of Neurology (EAN) [39], clearly indicate that any type of treatment should be discontinued during pregnancy based on previous practical recommendations of EAN [40]. In clinical practice, however, based on post-marketing studies [41] in patients who are at high risk of disease reactivation after treatment suspension, some drugs, like IFN-β, glatiramer acetate (GA; Copaxone®) or natalizumab (Tysabri®), are continued until pregnancy is confirmed. Furthermore, in women with high activity and at risk of relapses, some therapies can be maintained. In particular, patients who discontinue natalizumab because they are planning a pregnancy may experience severe relapses during pregnancy [42]. Because of this risk, in patients with active disease, it is suggested that natalizumab could be continued until the second trimester of gestation. The maintenance of GA and IFN-β was not considered in the current guidelines, even though GA was not associated with any teratogenic effect in a prospective observational multicentric study [43], in a study focusing on paternal exposure to GA [44] and in a comprehensive systematic review on the use of disease-modifying drugs in pregnant MS patients [45]. Notably, following a comprehensive examination of pregnancy cases exposed to GA, the United Kingdom Medicine & Healthcare products Regulatory Agency (MHRA) issued a recent change of the European marketing authorisation of this drug, by removing the pregnancy contraindication for GA 20 mg/mL [46] and 40 mg/mL [47]. It was determined that, in the light of current data not indicating teratogenicity or foeto-neonatal toxicity, use in pregnancy is now supported where the benefit to the mother outweighs the risk to the foetus.
In general, the practical approach of neurologists is based on three possible attitudes: (i) in patients with a prolonged period of NEDA, a drug washout period is indicated before discontinuing contraceptive treatment, which also applies to medications such as GA or IFN-β; (ii) in patients with recent disease activity or with a previous high disease activity, treatment with some drugs (GA, IFN-β and natalizumab) can be continued until conception even if for different reasons; and (iii) in selected patients with highly active disease, and after a careful evaluation of the risk-benefit ratio, GA and IFN-β can be administered throughout pregnancy [4]. For GA, this approach is supported both by the absence of negative pregnancy and foetal outcomes, established by the large Italian multicentre study [43], and by the recent changes of the European marketing authorisation [46, 47]. As few safety data are available about dimethyl fumarate (Tecfidera®), fingolimod (Gilenya®) and teriflunomide (Aubagio®), these drugs should be discontinued and contraception continued for an appropriate period of time before undergoing pregnancy [11].
In the case of natalizumab, the risk for relapses upon withdrawal [42] has already been noted. However, in a clinical series in women with very active disease who had discontinued the drug due to pregnancy, early resumption of natalizumab soon after delivery allowed control of disease activity to be regained and relapses at puerperium prevented [48]. Moreover, natalizumab has been demonstrated to be superior to fingolimod in patients with relapsing-remitting MS [37]. On the other hand, the safety of this drug in pregnancy, particularly relative to spontaneous abortion and newborn haematological alterations, is currently debated [49,50,51]. Recently, two studies on a large number of cases found that neither IFN-β nor natalizumab was significantly associated with abortions or intrauterine deaths [51, 52]. Despite controversial evidence, the option of natalizumab therapy in a naïve patient with active disease might be considered to prevent rebound of disease activity and disease worsening.
Guidelines recommend that, in the case of planned pregnancy in a woman who previously experienced a relevant disease activity, treatments based on either IFN-β or GA should be maintained as long as possible. The reason is that these drugs have no documented negative impact on the health of the mother and child, while natalizumab should be excluded in this context. However, in case of increased disease activity even during pregnancy, the risk-benefit ratio favours the initiation or maintenance of natalizumab treatment to protect the mother from the risk of damaging the foetus [39]. This choice should follow discussion with the patient about the known and potential risks to the mother (in terms of relapse) and to the foetus (in terms of foetal defects or spontaneous abortions or haematological abnormalities), with the ultimate decision to be taken by the patient.
Notably, the FDA has classified most of the drugs for MS therapy in the C category (Table 1) (only mitoxantrone is categorized as D). On the other hand, GA, previously classified as FDA Class B, no longer has a pregnancy contraindication (according to the modified European marketing authorization) and can be suggested for bridging therapy in women who are planning a pregnancy and are receiving treatments requiring a washout period, such as dimethyl fumarate or fingolimod. This approach would avoid exposing women to a prolonged washout period between the discontinuation of contraception and the moment of conception [53].
Finally, pregnancy is usually confirmed in the interval of weeks 6 to 10, when organogenesis occurs. Since there is no evidence of defects of organogenesis for the above mentioned drugs, this could support the possibility of maintaining or initiating these therapies in specific groups of pregnant women with active disease. On the other hand, in patients on first-line therapies with disease stabilized for many years, pregnancy can be planned and therapies can be discontinued at conception.
Management of MS therapy: during pregnancy
Once the pregnancy has started, the following issues need to be considered. Among the different therapeutic options, available data indicate that foetal haematological alterations (anaemia and thrombocytopenia) may be induced by natalizumab administration during the third trimester of pregnancy [49]. Therefore, the use of this drug should be carefully monitored, and it is strongly advised that the newborn assessment is conducted by a paediatric haematologist. Since natalizumab withdrawal promotes a rebound effect with possible relapses during pregnancy [42, 54], its discontinuation should be considered, along with discussion of a suitable alternative, only on a case-by-case basis.
The safety of steroid use during pregnancy varies according to the type of drug, the period of pregnancy during which the treatment is administered and the duration and dosage of the treatment. Patients with MS during pregnancy may benefit from short courses (3 to 5 days) of prednisolone or methylprednisolone, which can be safely administered during the second and third trimesters. These drugs are actively metabolized by the placenta and their extremely low concentration in foetal blood limits the risk of hampered foetal growth associated with long-term dexamethasone or betamethasone exposure in late pregnancy [55]. Whenever possible, steroid administration should be avoided during the first trimester of pregnancy, due to the possible, albeit rare, teratogenic effects, such as cleft palate [19, 55, 56].
The neurological condition of all pregnant patients with MS should be ideally checked every 3 months. Cases with possible subtle disease reactivation can be safely monitored with no-contrast low-field-strength MRI (1.5 Tesla) throughout pregnancy [57].
Notwithstanding multiple reassuring evidence, a second level ultrasound evaluation of all foetuses exposed to MS medications should be scheduled at 20–22 weeks of gestation.
Obstetric anaesthesia is not influenced by MS; specifically, epidural anaesthesia has no effect on disease progression and can be safely performed during vaginal delivery or caesarean section, as evidenced by a prospective multicentre study that included 21 primary Italian MS centres [33]. MS does not preclude spontaneous delivery [10]; however, the rate of either caesarean or operative deliveries may be increased in presence of major motor disability, due to the rapid exhaustion of maternal pushing efforts combined to a cautious attitude of the attending physician.
Management of MS therapy: post-pregnancy
In MS, the role of breastfeeding with respect to disease activity remains controversial. Although all drugs licenced for MS treatment are contraindicated during breastfeeding [10], the patient should be counselled before getting pregnant or early in pregnancy that the putative transfer of MS medications to the newborn is uncertain and that the maternal desire to breastfeed, together with the known benefits of long-standing (6 months or more) lactation [58], should be balanced with the conflicting data that relate breastfeeding to MS pathophysiology. The puerperium is a moment of high risk for MS flares and therapy should resume soon after delivery in cases with high periconceptional disease activity [59]. Indeed, while a meta-analysis [60] and other recent studies [61,62,63,64] showed that exclusive breastfeeding has a protective effect on the clinical activity of MS, other findings favour a more neutral role and suggest a “reverse causality”, linking the choice to breastfeed to less active disease before and during pregnancy [65].
Overall, in case of very active disease before pregnancy, therapy should be resumed as soon as possible, as early as 3 days after delivery [11]. In this regard, two large prospective studies revealed that GA may reduce the risk of reactivation by 50% [66] and that early administration (within 3 months post-partum) of interferons or GA significantly reduced the risk of relapses during puerperium and over a follow-up of at least 1 year [67]. This supports the early resumption of these therapies, especially in MS patients with active disease. Patients with low disease activity at conception and during pregnancy who decide to breastfeed should receive a MRI evaluation within the first month after delivery. Any sign of MRI-established activity should prompt the immediate cessation of breastfeeding as well as therapy resumption. As before, planning is essential, and agreement should be established with the patient during pregnancy planning, not delayed until after delivery.
A summary of recommendations for the use of disease-modifying drugs in women with multiple sclerosis considering pregnancy is presented in Table 2.
Conclusions
Pregnancy is a major concern for patients with MS. A survey of almost 6000 women with MS in their childbearing years revealed that 79% of them chose not to become pregnant after diagnosis. In 34.5% of cases, the choice was related to disease-related issues, especially possible interference with child raising, burdening the partner or fear of transmitting MS to their offspring [68]. This was also reflected in another recent study, where disease-related issues, including fear of current or future disability, fear of passing on the disease, concerns over treatment options and discouragement by physicians, were among the most frequently reported reasons for childlessness in women with MS [69]. Therefore, women with MS and their partners should have the opportunity of discussing parenthood in an unbiased manner.
Pregnancy-related issues should be discussed as early as possible, preferably at the moment of diagnosis and in any case when a therapeutic choice has to be made. All health professionals involved are committed to informing MS patients and their families about the potential impact of MS on pregnancy. They should also actively address the fears and concerns of women and their partners, delivering the key messages that MS is not a specific contraindication to pregnancy, that pregnancies in women with MS generally have a physiological course and that the children of mothers with MS most often have a normal development [11].
Moreover, sexual dysfunctions possibly occurring in MS patients should be carefully considered by the neurologist during the counselling for family planning, with referral of the patient to the appropriate specialists where indicated [65].
The pharmacological management of MS in pregnant women involves pre-pregnancy, pregnancy and post-pregnancy periods and, until now, comprehensive and detailed guidelines have been lacking.
Due to limited information, for most of the available drugs, potential damage to the foetus cannot be excluded, so that discontinuation of treatments before conception is recommended. On the other hand, the potential harm to the mother because of drug discontinuation should also be considered. The disease characteristics and the evidence on the safety of a few medications available should be carefully considered on a case-by-case basis before making a decision. As noted, GA, previously assigned to Pregnancy Category B by the FDA [21], has been recently re-evaluated by the UK MHRA authority [46, 47] to remove pregnancy as a strict contraindication. Therefore, therapy with GA no longer needs to be discontinued in women with MS when planning a pregnancy.
The MS relapse rate decreases during pregnancy, while early after delivery patients are exposed to an increased risk of relapses [59], so that the resumption of any therapy discontinued during pregnancy is highly advisable. However, this poses the issue of choices related to breastfeeding, since all available therapies are contraindicated during breastfeeding. Evidence about the possible protective role of breastfeeding remains controversial. Even if breastfeeding is possible in women with MS, in the presence of established MS activity that requires prompt therapy resumption breastfeeding should be avoided. Again, breastfeeding choice should be discussed in advance, during pre-conception counselling.
Finally, the management of MS during pregnancy can best be attained by the integrated efforts of an interdisciplinary team including neurologists, gynaecologists and psychologists. This interdisciplinary team is generally well accepted and even demanded by patients, who may also need psychological support at different moments of their reproductive course, before, during and after pregnancy [14].
Overall, the interdisciplinary team can more effectively minimize the maternal and foetal risks, monitor drug safety and effectiveness and identify the therapeutic strategy most appropriate for the individual patient, based on available scientific evidence, clinical experience and integration of different specific competences.
Change history
26 December 2017
The original version contained a mistake. The authors have specified only in a few paragraphs that all the contents of the paper are meant for Copaxone but not for unbranded glatiramer acetate, Authors ask to add the specification of Copaxone or branded glatiramer acetate everytime.
References
Compston A, Coles A (2008) Multiple sclerosis. Lancet 372(9648):1502–1517. doi:10.1016/S0140-6736(08)61620-7
Bove R, Chitnis T (2014) The role of gender and sex hormones in determining the onset and outcome of multiple sclerosis. Mult Scler 20(5):520–526. doi:10.1177/1352458513519181
Miller DH, Fazekas F, Montalban X, Reingold SC, Trojano M (2014) Pregnancy, sex and hormonal factors in multiple sclerosis. Mult Scler 20(5):527–536. doi:10.1177/1352458513519840
Pozzilli C, Pugliatti M, Paradig M. S. Group (2015) An overview of pregnancy-related issues in patients with multiple sclerosis. Eur J Neurol 22(Suppl 2):34–39. doi:10.1111/ene.12797
Confavreux C, Hutchinson M, Hours MM, Cortinovis-Tourniaire P, Moreau T (1998) Rate of pregnancy-related relapse in multiple sclerosis. Pregnancy in Multiple Sclerosis Group. N Engl J Med 339(5):285–291. doi:10.1056/NEJM199807303390501
Vukusic S, Hutchinson M, Hours M, Moreau T, Cortinovis-Tourniaire P, Adeleine P, Confavreux C, Pregnancy In Multiple Sclerosis Group (2004) Pregnancy and multiple sclerosis (the PRIMS study): clinical predictors of post-partum relapse. Brain 127(Pt 6):1353–1360. doi:10.1093/brain/awh152
Karp I, Manganas A, Sylvestre MP, Ho A, Roger E, Duquette P (2014) Does pregnancy alter the long-term course of multiple sclerosis? Ann Epidemiol 24(7):504–508 e502. doi:10.1016/j.annepidem.2014.04.007
Masera S, Cavalla P, Prosperini L, Mattioda A, Mancinelli CR, Superti G, Chiavazza C, Vercellino M, Pinessi L, Pozzilli C (2015) Parity is associated with a longer time to reach irreversible disability milestones in women with multiple sclerosis. Mult Scler 21(10):1291–1297. doi:10.1177/1352458514561907
Jokubaitis VG, Spelman T, Kalincik T, Lorscheider J, Havrdova E, Horakova D, Duquette P, Girard M, Prat A, Izquierdo G, Grammond P, Van Pesch V, Pucci E, Grand'Maison F, Hupperts R, Granella F, Sola P, Bergamaschi R, Iuliano G, Spitaleri D, Boz C, Hodgkinson S, Olascoaga J, Verheul F, McCombe P, Petersen T, Rozsa C, Lechner-Scott J, Saladino ML, Farina D, Iaffaldano P, Paolicelli D, Butzkueven H, Lugaresi A, Trojano M, Group MSS (2016) Predictors of long-term disability accrual in relapse-onset multiple sclerosis. Ann Neurol 80(1):89–100. doi:10.1002/ana.24682
Bove R, Alwan S, Friedman JM, Hellwig K, Houtchens M, Koren G, Lu E, McElrath TF, Smyth P, Tremlett H, Sadovnick AD (2014) Management of multiple sclerosis during pregnancy and the reproductive years: a systematic review. Obstet Gynecol 124(6):1157–1168. doi:10.1097/AOG.0000000000000541
Vukusic S, Marignier R (2015) Multiple sclerosis and pregnancy in the 'treatment era'. Nat Rev Neurol 11(5):280–289. doi:10.1038/nrneurol.2015.53
McCombe PA, Greer JM (2013) Female reproductive issues in multiple sclerosis. Mult Scler 19(4):392–402. doi:10.1177/1352458512452331
Pozzilli C, De Giglio L, Barletta VT, Marinelli F, Angelis FD, Gallo V, Pagano VA, Marini S, Piattella MC, Tomassini V, Pantano P (2015) Oral contraceptives combined with interferon beta in multiple sclerosis. Neurol Neuroimmunol Neuroinflamm 2(4):e120. doi:10.1212/NXI.0000000000000120
Payne D, McPherson KM (2010) Becoming mothers. Multiple sclerosis and motherhood: a qualitative study. Disabil Rehabil 32(8):629–638. doi:10.3109/09638280903204708
Coyle PK (2016) Management of women with multiple sclerosis through pregnancy and after childbirth. Ther Adv Neurol Disord 9(3):198–210. doi:10.1177/1756285616631897
Ristori G, Cannoni S, Stazi MA, Vanacore N, Cotichini R, Alfo M, Pugliatti M, Sotgiu S, Solaro C, Bomprezzi R, Di Giovanni S, Figa Talamanca L, Nistico L, Fagnani C, Neale MC, Cascino I, Giorgi G, Battaglia MA, Buttinelli C, Tosi R, Salvetti M (2006) Multiple sclerosis in twins from continental Italy and Sardinia: a nationwide study. Ann Neurol 59(1):27–34. doi:10.1002/ana.20683
Sadovnick AD, Macleod PM (1981) The familial nature of multiple sclerosis: empiric recurrence risks for first, second-, and third-degree relatives of patients. Neurology 31(8):1039–1041
Coyle PK (2014) Multiple sclerosis and pregnancy prescriptions. Expert Opin Drug Saf 13(12):1565–1568. doi:10.1517/14740338.2014.973848
Ferrero S, Pretta S, Ragni N (2004) Multiple sclerosis: management issues during pregnancy. Eur J Obstet Gynecol Reprod Biol 115(1):3–9. doi:10.1016/j.ejogrb.2003.10.020
Culpepper WJ 2nd (2015) The incidence and prevalence of comorbidity in multiple sclerosis. Mult Scler 21(3):261–262. doi:10.1177/1352458515574151
Food and Drug Administration (FDA) (2014) Content and format of labeling for human prescription drug and biological products; requirements for pregnancy and lactation labeling. Available from: http://www.federalregister.gov/a/2014-28241. Accessed 26 November, 2016
European Medicines Agency (EMA) (2016) Quality review of documents human product-information annotated template (English) version 9. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/Template_or_form/2009/10/WC500004368.pdf. Accessed 26 November, 2016
National Institute for Health Care and Excellence (NICE) (2014) Multiple sclerosis in adults: management. Clinical guideline. Available from: https://www.nice.org.uk/guidance/cg186. Accessed 26 November 2016
Cavalla P, Rovei V, Masera S, Vercellino M, Massobrio M, Mutani R, Revelli A (2006) Fertility in patients with multiple sclerosis: current knowledge and future perspectives. Neurol Sci 27(4):231–239. doi:10.1007/s10072-006-0676-x
Michel L, Foucher Y, Vukusic S, Confavreux C, de Seze J, Brassat D, Clanet M, Clavelou P, Ouallet JC, Brochet B, Pelletier J, Labauge P, Lebrun C, Lepage E, Le Frere F, Jacq-Foucher M, Barriere P, Wiertlewski S, Laplaud DA, Club Francophone de la Sclerose En Plaques (2012) Increased risk of multiple sclerosis relapse after in vitro fertilisation. J Neurol Neurosurg Psychiatry 83(8):796–802. doi:10.1136/jnnp-2012-302235
Amato MP, Derfuss T, Hemmer B, Liblau R, Montalban X, Soelberg Sørensen P, Miller DH, for the 2016 ECTRIMS Focused Workshop Group (2017) Environmental modifiable risk factors for multiple sclerosis: report from the 2016 ECTRIMS focused workshop [epub ahead of print]. Multiple Sclerosis Journal:135245851668684. doi:10.1177/1352458516686847
Jalkanen A, Kauko T, Turpeinen U, Hamalainen E, Airas L (2015) Multiple sclerosis and vitamin D during pregnancy and lactation. Acta Neurol Scand 131(1):64–67. doi:10.1111/ane.12306
Munger KL, Aivo J, Hongell K, Soilu-Hanninen M, Surcel HM, Ascherio A (2016) Vitamin D status during pregnancy and risk of multiple sclerosis in offspring of women in the Finnish Maternity Cohort. JAMA Neurol 73(5):515–519. doi:10.1001/jamaneurol.2015.4800
ACOG American College of Obstetricians and Gynecologists (2011) Committee opinion no. 495: vitamin D: screening and supplementation during pregnancy. Obstet Gynecol 118(1):197–198
Greenberg JA, Bell SJ, Ausdal WV (2008) Omega-3 fatty acid supplementation during pregnancy. Rev Obstet Gynecol 1(4):162–169
Mueller BA, Zhang J, Critchlow CW (2002) Birth outcomes and need for hospitalization after delivery among women with multiple sclerosis. Am J Obstet Gynecol 186(3):446–452
Ponsonby AL, Lucas RM, van der Mei IA, Dear K, Valery PC, Pender MP, Taylor BV, Kilpatrick TJ, Coulthard A, Chapman C, Williams D, McMichael AJ, Dwyer T (2012) Offspring number, pregnancy, and risk of a first clinical demyelinating event: the AusImmune Study. Neurology 78(12):867–874. doi:10.1212/WNL.0b013e31824c4648
Pasto L, Portaccio E, Ghezzi A, Hakiki B, Giannini M, Razzolini L, Piscolla E, De Giglio L, Pozzilli C, Paolicelli D, Trojano M, Marrosu MG, Patti F, La Mantia L, Mancardi GL, Solaro C, Totaro R, Tola MR, Di Tommaso V, Lugaresi A, Moiola L, Martinelli V, Comi G, Amato MP, M. S. Study Group of the Italian Neurological Society (2012) Epidural analgesia and cesarean delivery in multiple sclerosis post-partum relapses: the Italian cohort study. BMC Neurol 12:165. doi:10.1186/1471-2377-12-165
Kelly VM, Nelson LM, Chakravarty EF (2009) Obstetric outcomes in women with multiple sclerosis and epilepsy. Neurology 73(22):1831–1836. doi:10.1212/WNL.0b013e3181c3f27d
Banwell B, Giovannoni G, Hawkes C, Lublin F (2013) Editors' welcome and a working definition for a multiple sclerosis cure. Mult Scler Relat Disord 2(2):65–67. doi:10.1016/j.msard.2012.12.001
Kappos L, De Stefano N, Freedman MS, Cree BA, Radue EW, Sprenger T, Sormani MP, Smith T, Haring DA, Piani Meier D, Tomic D (2016) Inclusion of brain volume loss in a revised measure of 'no evidence of disease activity' (NEDA-4) in relapsing-remitting multiple sclerosis. Mult Scler 22(10):1297–1305. doi:10.1177/1352458515616701
Baroncini D, Ghezzi A, Annovazzi PO, Colombo B, Martinelli V, Minonzio G, Moiola L, Rodegher M, Zaffaroni M, Comi G (2016) Natalizumab versus fingolimod in patients with relapsing-remitting multiple sclerosis non-responding to first-line injectable therapies. Mult Scler 22(10):1315–1326. doi:10.1177/1352458516650736
Sellebjerg F, Barnes D, Filippini G, Midgard R, Montalban X, Rieckmann P, Selmaj K, Visser LH, Sorensen PS, Relapses ETFoToMS (2005) EFNS guideline on treatment of multiple sclerosis relapses: report of an EFNS task force on treatment of multiple sclerosis relapses. Eur J Neurol 12(12):939–946. doi:10.1111/j.1468-1331.2005.01352.x
Otero-Romero S, Amato MP, Chandraratna D, Clanet M, Comi G, Derfuss T, Fazekas F, Hemmer B, Hartung HP, Havrdova E, Kappos L, Liblau R, Lubetzki C, Miller DH, Olsson T, Selmaj C, Siva A, Sorensen PS, Thalheim C, Wiendl H, Zipp F, Thompson AJ, Gold R, Montalban X (2016) ECTRIMS-EAN clinical practice guideline on pharmacological management of multiple sclerosis [abstract] . Multiple sclerosis journal 22(abstract 255):834
Leone MA, Keindl M, Schapira AH, Deuschl G, Federico A (2015) Practical recommendations for the process of proposing, planning and writing a neurological management guideline by EAN task forces. Eur J Neurol 22(12):1505–1510. doi:10.1111/ene.12818
Amato MP, Portaccio E, Ghezzi A, Hakiki B, Zipoli V, Martinelli V, Moiola L, Patti F, La Mantia L, Mancardi GL, Solaro C, Tola MR, Pozzilli C, De Giglio L, Totaro R, Lugaresi A, Di Tommaso V, Paolicelli D, Marrosu MG, Comi G, Pellegrini F, Trojano M, Society MSSGotIN (2010) Pregnancy and fetal outcomes after interferon-beta exposure in multiple sclerosis. Neurology 75(20):1794–1802. doi:10.1212/WNL.0b013e3181fd62bb
De Giglio L, Gasperini C, Tortorella C, Trojano M, Pozzilli C (2015) Natalizumab discontinuation and disease restart in pregnancy: a case series. Acta Neurol Scand 131(5):336–340. doi:10.1111/ane.12364
Giannini M, Portaccio E, Ghezzi A, Hakiki B, Pasto L, Razzolini L, Piscolla E, De Giglio L, Pozzilli C, Paolicelli D, Trojano M, Marrosu MG, Patti F, La Mantia L, Mancardi G, Solaro C, Totaro R, Tola MR, De Luca G, Lugaresi A, Moiola L, Martinelli V, Comi G, Amato MP (2012) Pregnancy and fetal outcomes after glatiramer acetate exposure in patients with multiple sclerosis: a prospective observational multicentric study. BMC Neurol 12:124. doi:10.1186/1471-2377-12-124
Pecori C, Giannini M, Portaccio E, Ghezzi A, Hakiki B, Pasto L, Razzolini L, Sturchio A, De Giglio L, Pozzilli C, Paolicelli D, Trojano M, Marrosu MG, Patti F, Mancardi GL, Solaro C, Totaro R, Tola MR, De Luca G, Lugaresi A, Moiola L, Martinelli V, Comi G, Amato MP, Society MSSGotIN (2014) Paternal therapy with disease modifying drugs in multiple sclerosis and pregnancy outcomes: a prospective observational multicentric study. BMC Neurol 14:114. doi:10.1186/1471-2377-14-114
Lu E, Wang BW, Guimond C, Synnes A, Sadovnick D, Tremlett H (2012) Disease-modifying drugs for multiple sclerosis in pregnancy: a systematic review. Neurology 79(11):1130–1135. doi:10.1212/WNL.0b013e3182698c64
MHRA-Medicine & Health products Regulatory Agency (2016) Summary of product characteristics, Copaxone 20 mg/ml solution for injection, pre-filled syringe, revision 02/12/2016. Available from: http://www.mhra.gov.uk/. Accessed 31 January, 2017
MHRA-Medicine & Health products Regulatory Agency (2017) Summary of product characteristics, Copaxone 40 mg/ml solution for injection, pre-filled syringe, revision 30/03/2017. Available from: http://www.mhra.gov.uk/. Accessed 04 April, 2017
Vukusic S, Durand-Dubief F, Benoit A, Marignier R, Frangoulis B, Confavreux C (2015) Natalizumab for the prevention of post-partum relapses in women with multiple sclerosis. Mult Scler 21(7):953–955. doi:10.1177/1352458514554056
Haghikia A, Langer-Gould A, Rellensmann G, Schneider H, Tenenbaum T, Elias-Hamp B, Menck S, Zimmermann J, Herbstritt S, Marziniak M, Kumpfel T, Meinl I, Plavina T, Gold R, Hellwig K (2014) Natalizumab use during the third trimester of pregnancy. JAMA Neurol 71(7):891–895. doi:10.1001/jamaneurol.2014.209
Gandhi S, Jakimovski D, Ahmed R, Hojnacki D, Kolb C, Weinstock-Guttman B, Zivadinov R (2016) Use of natalizumab in multiple sclerosis: current perspectives. Expert Opin Biol Ther 16(9):1151–1162. doi:10.1080/14712598.2016.1213810
Friend S, Richman S, Bloomgren G, Cristiano LM, Wenten M (2016) Evaluation of pregnancy outcomes from the Tysabri(R) (natalizumab) pregnancy exposure registry: a global, observational, follow-up study. BMC Neurol 16(1):150. doi:10.1186/s12883-016-0674-4
Thiel S, Langer-Gould A, Rockhoff M, Haghikia A, Queisser-Wahrendorf A, Gold R, Hellwig K (2016) Interferon-beta exposure during first trimester is safe in women with multiple sclerosis—a prospective cohort study from the German Multiple Sclerosis and Pregnancy Registry. Mult Scler 22(6):801–809. doi:10.1177/1352458516634872
Comi G, Amato MP, Bertolotto A, Centonze D, De Stefano N, Farina C, Gallo P, Ghezzi A, Grimaldi LM, Mancardi G, Marrosu MG, Montanari E, Patti F, Pozzilli C, Provinciali L, Salvetti M, Tedeschi G, Trojano M (2016) The heritage of glatiramer acetate and its use in multiple sclerosis. Multiple Sclerosis and Demyelinating Disorders 1(1):6. doi:10.1186/s40893-016-0010-2
Martinelli V, Colombo B, Dalla Costa G, Dalla Libera D, Moiola L, Falini A, Comi G, Filippi M (2016) Recurrent disease-activity rebound in a patient with multiple sclerosis after natalizumab discontinuations for pregnancy planning. Mult Scler 22(11):1506–1508. doi:10.1177/1352458513492246
Houtchens MK, Kolb CM (2013) Multiple sclerosis and pregnancy: therapeutic considerations. J Neurol 260(5):1202–1214. doi:10.1007/s00415-012-6653-9
Carmichael SL, Shaw GM, Ma C, Werler MM, Rasmussen SA, Lammer EJ, National Birth Defects Prevention Study (2007) Maternal corticosteroid use and orofacial clefts. Am J Obstet Gynecol 197(6):585 e581–585 e587. doi:10.1016/j.ajog.2007.05.046
Simon JH, Li D, Traboulsee A, Coyle PK, Arnold DL, Barkhof F, Frank JA, Grossman R, Paty DW, Radue EW, Wolinsky JS (2006) Standardized MR imaging protocol for multiple sclerosis: Consortium of MS Centers consensus guidelines. AJNR Am J Neuroradiol 27(2):455–461
World Health Organization (2006) Pregnancy, childbirth, postpartum and newborn care. A guide for essential practice, World Health Organization (WHO) Department of Making Pregnancy Safer Geneva, Switzerland
Finkelsztejn A, Brooks JB, Paschoal FM Jr, Fragoso YD (2011) What can we really tell women with multiple sclerosis regarding pregnancy? A systematic review and meta-analysis of the literature. BJOG 118(7):790–797. doi:10.1111/j.1471-0528.2011.02931.x
Pakpoor J, Disanto G, Lacey MV, Hellwig K, Giovannoni G, Ramagopalan SV (2012) Breastfeeding and multiple sclerosis relapses: a meta-analysis. J Neurol 259(10):2246–2248. doi:10.1007/s00415-012-6553-z
Conradi S, Malzahn U, Paul F, Quill S, Harms L, Then Bergh F, Ditzenbach A, Georgi T, Heuschmann P, Rosche B (2013) Breastfeeding is associated with lower risk for multiple sclerosis. Mult Scler 19(5):553–558. doi:10.1177/1352458512459683
Fares J, Nassar AH, Gebeily S, Kobeissy F, Fares Y (2016) Pregnancy outcomes in Lebanese women with multiple sclerosis (the LeMS study): a prospective multicentre study. BMJ Open 6(5):e011210. doi:10.1136/bmjopen-2016-011210
Hellwig K, Rockhoff M, Herbstritt S, Borisow N, Haghikia A, Elias-Hamp B, Menck S, Gold R, Langer-Gould A (2015) Exclusive breastfeeding and the effect on postpartum multiple sclerosis relapses. JAMA Neurol 72(10):1132–1138. doi:10.1001/jamaneurol.2015.1806
Ragnedda G, Leoni S, Parpinel M, Casetta I, Riise T, Myhr KM, Wolfson C, Pugliatti M (2015) Reduced duration of breastfeeding is associated with a higher risk of multiple sclerosis in both Italian and Norwegian adult males: the EnvIMS study. J Neurol 262(5):1271–1277. doi:10.1007/s00415-015-7704-9
Amato MP, Portaccio E (2015) Fertility, pregnancy and childbirth in patients with multiple sclerosis: impact of disease-modifying drugs. CNS Drugs 29(3):207–220. doi:10.1007/s40263-015-0238-y
Ford CC, Johnson KP, Lisak RP, Panitch HS, Shifronis G, Wolinsky JS, Copaxone Study Group (2006) A prospective open-label study of glatiramer acetate: over a decade of continuous use in multiple sclerosis patients. Mult Scler 12(3):309–320. doi:10.1191/135248506ms1318oa
Portaccio E, Ghezzi A, Hakiki B, Sturchio A, Martinelli V, Moiola L, Patti F, Mancardi GL, Solaro C, Tola MR, Pozzilli C, De Giglio L, Totaro R, Lugaresi A, De Luca G, Paolicelli D, Marrosu MG, Comi G, Trojano M, Amato MP, M. S. Study Group of the Italian Neurological Society (2014) Postpartum relapses increase the risk of disability progression in multiple sclerosis: the role of disease modifying drugs. J Neurol Neurosurg Psychiatry 85(8):845–850. doi:10.1136/jnnp-2013-306054
Alwan S, Yee IM, Dybalski M, Guimond C, Dwosh E, Greenwood TM, Butler R, Sadovnick AD (2013) Reproductive decision making after the diagnosis of multiple sclerosis (MS). Mult Scler 19(3):351–358. doi:10.1177/1352458512452920
Ferraro D, Simone AM, Adani G, Vitetta F, Mauri C, Strumia S, Senesi C, Curti E, Baldi E, Santangelo M, Montepietra S, Immovilli P, Guareschi A, Neri W, Granella F, Caniatti L, Tola MR, Motti L, Pesci I, Montanari E, Sola P (2017) Definitive childlessness in women with multiple sclerosis: a multicenter study. Neurol Sci. doi:10.1007/s10072-017-2999-1
Acknowledgements
We thank Luisa Granziero and Ray Hill, independent medical writers, who provided editorial assistance, respectively, in manuscript drafting and in English-language editing and journal styling prior to submission. This assistance was funded by Teva.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Maria Pia Amato has received research grants and honoraria as a speaker and member of advisory boards from Bayer, Biogen, Merck, Novartis, Roche, Sanofi Genzyme and Teva. Paola Cavalla has received speaker honoraria and honoraria for serving on advisory board activities from Almirall, Biogen, Merck Serono, Novartis, Sanofi Genzyme and Teva. Giancarlo Comi has received compensation for consulting services and/or speaking activities from Teva, Novartis, Sanofi, Genzyme, Merck, Biogen, Roche, Almirall, Receptos, Celgene, Forward Pharma and Excemed. Maria Giovanna Marrosu has received speaker honoraria and honoraria for serving on advisory board activities from Bayer Schering, Biogen Idec, Merck Serono, Novartis, Genzyme, Sanofi Aventis and Teva and research grants from Merck Serono and Novartis. Francesco Patti has received fees for consulting and/or advisory board activities from Almirall, Bayer, Biogen, Celgene, Merck, Novartis, Roche, Sanofi Genzyme and Teva. All other authors declare no conflict of interest.
Additional information
A correction to this article is available online at https://doi.org/10.1007/s10072-017-3226-9.
Rights and permissions
About this article
Cite this article
Amato, M.P., Bertolotto, A., Brunelli, R. et al. Management of pregnancy-related issues in multiple sclerosis patients: the need for an interdisciplinary approach. Neurol Sci 38, 1849–1858 (2017). https://doi.org/10.1007/s10072-017-3081-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-017-3081-8