Abstract
Social knowledge beyond one’s direct relationships is a key in successfully manoeuvring the social world. Individuals gather information on the quality of social relationships between their group companions, which has been termed triadic awareness. Evidence of the use of triadic awareness in natural contexts is limited mainly to conflict management. Here we investigated triadic awareness in wild Barbary macaques (Macaca sylvanus) in the context of bridging interactions defined as male–infant–male interactions whereby a male (initiator, holder) presents an infant to another male (receiver, non-holder) in order to initiate an affiliative interaction with that male. Analyses based on 1263 h of focal observations on ten infants of one wild social group in Morocco supported the hypothesis that males use their knowledge of the relationship between infants and other adult males when choosing a male as a partner for bridging interactions. Specifically, (i) the number of bridging interactions among holder–infant–receiver triads was positively affected by the strength of the infant–receiver relationship and (ii) when two males were available as bridging partners, a male was more likely to be chosen as the receiver the stronger his social relationship with the infant relative to the other available male. This demonstrates that non-human primates establish triadic awareness of temporary infant–male relationships and use it in a naturally occurring affiliative context. Our results contribute to the discussion about the mechanism underlying the acquisition of triadic awareness and the benefits of its usage, and lend support to hypotheses linking social complexity to the evolution of complex cognition.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Non-human primates living in stable social groups develop agonistic and affiliative relationships with group members of the same and other age–sex classes (e.g. Cheney et al. 1986; van Hooff and van Schaik 1994; Silk et al. 2006). These social relationships are established by social partners who individually recognize each other and repeatedly interact with one another over time, allowing past interactions to be predictive of future ones (Hinde 1976). This implies that individuals possess knowledge about their own relationships, that they use to modify their own behaviour towards others, and that it may also allow them to predict the behaviour of others. This capacity helps the individual to avoid aggression (e.g. De Waal 1986; van Hooff and van Schaik 1994), to increase fitness (e.g. Silk 2007a, b, 2009; Schülke et al. 2010) and contributes to the stability and cohesion of the group (e.g. Sterck et al. 1997; Lehmann et al. 2007). It has been suggested that the challenges of social life might drive the evolution of complex social knowledge, the so-called triadic awareness defined as knowledge about the relationships among other individuals without assuming actual awareness. The capacity to recognize who outranks whom, who is closely bonded with whom, who is likely to support whom or intervene against whom, and to adjust one’s behaviour accordingly, has been documented in apes (e.g. Tomasello and Call 1997; De Waal 2007), Old World (Cheney et al. 1986; Cheney and Seyfarth 1999), New World monkeys (Perry et al. 2004; but see also Ferreira et al. 2006), other mammals (Engh et al. 2005; Connor 2007; Johnson 2010) and in birds (Peake et al. 2002; Seed et al. 2007).
Evidence for triadic awareness of non-human primates mainly comes from experiments. Male hamadryas baboons Papio hamadryas use knowledge of the quality of male–female relationships when deciding whether to challenge a male for access to females (Bachmann and Kummer 1980). Adult vervet monkeys Cercopithecus aethiops react to playbacks of juvenile distress vocalizations by looking at the juvenile’s mother, indicating triadic awareness of kin relations (Cheney and Seyfarth 1980). Female longtail macaques Macaca fascicularis demonstrate their ability to recognize kinship relationship in a discrimination task (Dasser 1988). Triadic awareness of rank relationships has been inferred from playback experiments using artificial sequences of calls of group members: e.g. in chacma baboons Papio ursinus calls mimicking interactions that are discordant with the current dominance relations between parties elicit stronger reactions in group members than calls in accordance with the hierarchy (Cheney et al. 1995; Kitchen et al. 2005).
Another set of studies used observational data on social behaviour in natural contexts to assess triadic awareness of others’ dominance, kin and affiliative relationships. Individuals engaging in agonistic conflicts solicit support and target solicitations more often from group mates who outrank their opponent (e.g. Silk 1999) and from individuals with whom they are more closely bonded than their opponents (Perry et al. 2004). Support is likely to be offered to the higher ranking of the opponents (Schino et al. 2007), who is more likely to succeed in the conflict and/or represents a more powerful ally in prospective future conflicts (e.g. Bissonnette et al. 2009). After the conflict individuals may discriminate against the opponent’s kin or affiliates; the aggressor directs reconciliatory behaviour at the opponent’s close relatives (Judge 1991) or avoids affiliative interactions with them in expectation of retaliation (Call et al. 2002), while the victim may redirect aggression towards the opponent’s kin (Aureli et al. 1992; Judge 1982; Smuts 1985; Cheney and Seyfarth 1989).
Although these patterns imply the use of triadic awareness, it has been pointed out that some of them may be also the result of alternative, simpler mechanisms, such as recruitment of allies based on an individual’s own affiliative or dominance relationship, or simple rules of the recruitment of the highest-ranking available individual (Silk 1999; Perry et al. 2004; Range and Noë 2005), that do not require triadic awareness. This ambiguity may partly arise from the relative rarity of the interactions suitable for the research question: supporter recruitment only demonstrates the use of triadic awareness if the invitee recruits the higher ranking from both opponents and is not ranking in between them at the same time. Elegant experiments have been designed to rule out such alternative mechanisms and may more effectively demonstrate the cognitive capacity for triadic awareness. Nevertheless, these experiments are less informative about the use of this cognitive capacity in natural context, which limits the interpretations of its biological relevance (De Waal 1991; Schino et al. 2006, 2007). For example, playback experiments, which simulate situations that never or rarely happen (e.g. dominant individual being threatened by subordinate one), cannot demonstrate the development of social cognition as a response to selection pressure, also because the relative frequencies as played out in different social contexts cannot be assessed.
Studying triadic awareness under natural conditions should therefore complement experimental research. New observational studies should focus on underexplored social contexts that provide a more complete understanding of whether and how individuals use triadic awareness in different situations of their daily lives. Here we focus on a frequently occurring behaviour that may allow for an assessment of how triadic awareness is used in a natural affiliative context, specifically a type of polyadic infant handling the so-called bridging interactions (Ogawa 1995a) in male Barbary macaques.
Infant handling is broadly defined as non-maternal manipulation of an infant by individuals other than the infant’s mother and may include different positive, neutral and negative interactions between the infant and its non-maternal caretaker, irrespective of the caretakers’ sex and age class (Hrdy 1976, 2005). Infant handling is found across different taxa (see Riedman 1982 for a review; Clutton-Brock 2002) with pronounced interspecific variation in intensity and type of interactions (Woodroffe and Vincent 1994; Hrdy 2005). In several species of Old World monkeys, males engage in a specific type of polyadic infant handling, variously called bridging interactions (Ogawa 1995a), triadic male–infant interactions (Taub 1980), male–infant–male interactions (Zhao 1996), or agonistic buffering (Deag and Crook 1971). During these interactions, two males simultaneously manipulate one infant, exhibiting a typical series of ritualized behaviours including teeth-chattering, lifting the infant above their heads and inspection of the infant’s genitals (Deag 1980). Bridging has been reported in several papionin primates: Barbary Macaca sylvanus (Deag and Crook 1971), Tibetan M. thibetana (Ogawa 1995a), stumptail M. acrtoides (Estrada and Sandoval 1977), longtail M. fascicularis (de Waal et al. 1976), Assamese M. assamensis (Bernstein and Cooper 1998) and bonnet macaques M. radiata (Silk and Samuels 1984), yellow Papio cynocephalus (Collins 1986), olive P. anubis (Smuts 1985) and chacma baboons P. ursinus (Busse and Hamilton 1981), sooty mangabeys Cercocebus atys (Busse and Gordon 1984), grey-cheeked mangabeys Cercocebus albigena (Chalmers 1968) and geladas Theropithecus gelada (Dunbar 1984).
Dyadic infant handling and bridging interactions are often unequally distributed among infants and potential handlers. Males differ in their general interest in infant handling and also in preferences for particular infants (e.g. Taub 1980). In some baboon species, these preferences may to some extent reflect the likelihood of paternity (Nguyen et al. 2009; Moscovice et al. 2010). In macaques, male preferences for infants appear mostly unrelated either to paternity or to past mating (Paul et al. 1992; Ménard et al. 2001; but see Ménard et al. 1992; Ostner et al. 2013) but may reflect the male’s social relationships with the mother and may be predictive of future mating opportunities (Ménard et al. 2001; Smuts and Gubernick 1992). Males also choose the male partner in the bridging interaction non-randomly (e.g. Estrada and Sandoval 1977; Dunbar 1984; Ogawa 1995a). Males initiate more bridging interactions with relatively higher-ranking males than with lower-ranking males (Paul et al. 1996; Silk and Samuels 1984; Collins 1986; Deag 1980), and/or with males who are relatively close to their own rank (Stein 1984; Paul et al. 1996). It has also been suggested that the male initiating the bridging interaction preferentially uses the infant that is preferred by the receiver to increase the chances of a successful interaction, indicating that males recognize affiliative relationships between other males and infants (Ogawa 1995b). This suggestion implies the use of triadic awareness in bridging interactions. Patterns of interactions in accordance with this mechanism have been found in Barbary macaques (Paul et al. 1996), but the element of partner choice has not yet been systematically studied.
Barbary macaques live in multimale–multifemale groups. Males emigrate from their natal group after reaching sexual maturity, while females remain in the natal group with their offspring. They are seasonal breeders with a mating season in autumn and a birth season in spring (reviewed in Fooden 2007). Females mate with numerous males (Small 1990), and paternal kinship is not recognized (Ménard et al. 2001; Kuester et al. 1994; but see Ménard et al. 1992). Despite such promiscuity selecting against male care for offspring (see van Schaik and Paul 1996), infants may spend exceedingly large proportions of time being carried, cradled and groomed by males, whereas aggression or abuse by males is rare (e.g. Deag 1980; Paul 1999). Most interactions are initiated and maintained by males, but the contact seems voluntary and infants can be responsible for its start or termination (BK personal observation). Male infant handling bouts have been reported to generally last up to 20 min (Deag and Crook 1971), but may take even up to over an hour (BK personal observation), and are often alternated with bridging interactions.
In this study we investigated the relationship between the strength of the infant–male affiliative relationship and the distribution of bridging interactions initiated by the adult male holding the infant. We predicted that the stronger the relationship between the infant and another male the higher would be his chance of being picked as a partner for a bridging interaction by the infant holder. This implies triadic awareness on the part of the infant holder who initiates the interaction (Ogawa 1995b). Unlike the previous study (Ogawa 1995b) we assumed that an infant holder (initiator) chooses a receiving male based on the infant he has instead of searching for an infant that fits his preselected male partner (receiver). This assumption is more plausible for Barbary macaques because bridging interactions are often preceded by extended dyadic infant carrying and handling episodes. More specifically we predicted that the number of interactions of each holder–infant–receiver triad would be positively related to the strength of the affiliative relationship between the infant and the receiver of the interaction. We also predicted that the stronger a male’s relationship with the infant is relative to a second available male (the closest bystander), the more likely he is to be chosen as the receiver of the interaction.
Methods
Field site and subjects
This study was conducted on one group of wild Barbary macaques inhabiting the cedar and oak forest of the Ifrane National Park in the Middle Atlas Mountains of Morocco (33–240°N, 005–120°W). Permission to conduct the research in the park was granted by the Haut Commissariat aux Eaux et Forêts et à la Lutte Contre la Désertification of Morocco.
The data were collected by BK during two field seasons corresponding with two following birth seasons (April–August 2013, April–September 2014). The study group (Green Group) was well habituated to the presence of human observer, and all members were individually recognizable. In both seasons the group consisted of 7 adult (5 years old and older) males and 6 adult females (older than 5 years). There were 19 juveniles in 2013 and 20 in 2014. All adult females gave birth in both seasons resulting in 6 infants in each season (5 female, 1 male in 2013; 3 female, 3 male in 2014).
Data collection
Behavioural data were collected using handheld HP iPAQ 114 series pocket PCs loaded with Pendragon Forms Version 5.1 (© Pendragon Software Cooperation, U.S.A.). We used continuous focal sampling (Altmann 1974) with infants as focal subjects. In both seasons, the data collection started when at least 4 infants were born. We observed all infants until the end of the field season, except one male infant (born in 2014), who disappeared approximately 2 months after his birth. Infants were followed during observation sessions lasting 2 h, during which we recorded all social interactions between the focal infant and other group members. We pseudorandomized the order in which we observed infants to ensure that all infants were observed equally often at the different times of the day. The data collection on the 12 infants yielded a total of 1430 h of observation. We excluded two infants that never interacted with adult males from the analyses. Thus the analyses were based on 1263 h of focal observation of 10 infants (hours of focal data per infant mean ± SD = 126.3 ± 12.5).
We defined dyadic infant handling as an interaction between an adult male and an infant. During the interaction the adult male and infant were in body contact that included cradling, dorsal carrying, ventral carrying, grooming, resting in body contact and “contact crawling” defined as an infant crawling in body contact with a male or playfully climbing over a male’s body (see Thierry et al. 2000; Deag 1980, for a more detailed description of the behaviours). We also included polyadic interactions if they involved only one adult male and one or more females or non-adult males (e.g. a male manipulates an infant together with a juvenile individual or the mother of the infant). For each dyadic infant handling interaction, we recorded the start and end time, and the identity (ID) of the male involved.
We defined bridging as an interaction involving (at least) two adult males who simultaneously manipulated one infant, exhibiting a series of ritualized behaviours including teeth-chattering or lip smacking, inspection of the infant’s genitalia and lifting the infant above their heads (Deag 1980). We scored the start of the interaction once both males (being already in body contact with the infant and to each other) displayed teeth-chattering in a way typical for bridging behaviour. We classified the initiator as the male that approached the other to start the body contact, and the receiver as the male being approached. For interactions in which males approached at the same time, the initiator was classified as unknown. The male who was in body contact with the infant before the bridging interaction started was classified as the infant holder. We scored the end of the interaction once the males stopped being in mutual body contact with one another. A new independent bridging interaction was scored after a >2 min break in body contact between males.
We sorted bridging interactions into three types (see also Paul et al. 1996; Zhao 1996; Ogawa 1995a): (1) interactions initiated by a male that is in dyadic interaction with the infant (initiator = holder) and that approached another male without an infant (receiver = non-holder); (2) interactions initiated by a male without an infant (initiator = non-holder) that approached a male that was already interacting with an infant (receiver = holder); (3) other cases, i.e. interactions with unknown initiator, interactions initiated by both males, and interactions that were not preceded by dyadic handling between the infant and any of the males participating in the following bridging interaction. Only interactions belonging to the first category (bridging initiated by the infant holder) were considered suitable for the analysis of triadic awareness in this study because the other two options could not reliably discriminate the target of the interaction (infant or adult male) or the role of the initiator. In the second field season we expanded the data collection to obtain additional information about the choice of male partner and recorded the ID of the nearest male present within 10 m at the beginning of a bridging interaction and classified him as a bystander.
We used an ad libitum method (Altmann 1974) to record all dyadic agonistic interactions. For each season we entered the recorded data into a winner–loser dominance matrix and built a hierarchy based on the standardized normalized David’s score (Schmid and De Vries 2013).
Data analysis
We used a composite sociality index (CSI; Silk et al. 2006) to assess the strength of dyadic affiliative relationships between infants and males. The CSI was based on: (1) the duration and (2) frequency of body contact (including ventral carrying, cradling, grooming and also infant directed polyadic behaviour that involved one male and female or juvenile individual/s) and (3) the duration of crawling body contact as defined above. The three behaviours were highly correlated in row-wise matric correlations (rhorw,av ranged from 0.80–0.88) run with MatMan 1.1.4 (De Vries et al. 1993). For each dyad, we divided the value for each behaviour by the average across all dyads this infant formed with all males in the group and averaged the resulting relative value of all three behaviours. Thus, the index expresses the relative strength of the bond of the infant–male dyad compared to bonds this infant had with all males. Any infant–male interaction was excluded, if being a part of bridging according to the definition, so that these two variables were independent from one another.
To test the predictions of our hypothesis, that holders choose receivers based on the strength of the relationship the receiver has with the infant we used two generalized linear mixed-effect models (GLMM) using the lme4 package (Bates et al. 2015) in R 3.1.1 (R Core Team 2014). Fitted models were assessed for over-dispersion and model stability (see Quinn and Keough 2002).
To test prediction 1, that the number of interactions of each holder–infant–receiver triad is predicted by the strength of the bond between the infant and receiver, we used a GLMM with assumed Poisson distribution and the number of bridging interactions among each holder–infant–receiver triad as the response variable (N = 654; for more information about the distribution of interactions see Table S1 in Online Resource 1). The logarithm of the total observation time of each infant was entered as an offset term (which standardized the number of bridging interactions for durations of focal observation). We included as the two predictors of interest the CSI between holder and infant (CSIh), and the CSI between infant and receiver (CSIr) and as the random factors the identities of the infant, initiating holder and receiver to avoid pseudoreplication, and a factor distinguishing each unique holder–infant–receiver triad to account for over-dispersion. We also included several variables, one by one, to control for the effects of birth season (2013 or 2014), the David’s score of the receiver (DSr) and the rank distance between holder and receiver, computed as an absolute value of the rank difference between holder and receiver (|ΔDShr|). We dropped these controls if they did not improve the Bayesian Information Criterion (BIC, Schwarz 1978) of the model by >2.
To test prediction 2, that the holder bases the choice of receiver between two available males on their CSI with the infant we used a GLMM with assumed binomial distribution. The binomial response was scored as one if the male was selected to be the receiver and zero if the male remained a bystander to the bridging interaction (two lines for each interaction; N = 224). We entered as predictor of interest the difference between the CSI of the infant–receiver and the infant–bystander dyad (ΔCSIrb) with positive values indicating the selection of the male with the stronger relationship with the infant, and negative values indicating the selection of the male with the weaker relationship with the infant. Similar to the first model, we included as random factors in our null model the identities of the infant, the holder, the receiver and the bystander and the identity of the interaction. We also controlled for the effect of the rank distance between the holder and the involved male (receiver or bystander), calculated as an absolute difference of their David’s score (|ΔDShm|) and the rank distance between receiver and bystander, calculated as a difference between their David’s score (ΔDSrb), by assessing the BIC of the model as these controls were included one by one.
Our final models include the predictors of interest, all random effects and those controls that improved the BIC. Collinearity of the selected predictors was assessed by variance inflation factor (VIF < 10; Bowerman and O’Connell 1990). Significance of our predictors of interest was determined by comparing a model with the predictor to a reduced model without this predictor using anova function.
Results
Distribution of interactions
In 2013 we assessed the rank of 7 adult males based on 124 interactions. The David’s score ranged from −14.5 to 11.8 (median = 0.8) with 3 (14.3 %) dyads with unknown and 1 (4.8 %) with a two-way relationship. In 2014 the David’s score was based on 114 interactions and showed the same range as in the previous season (median = 0.7) with 2 (9.5 %) dyads with unknown and 4 (19 %) dyads with two-way relationship. The change of David’s score between seasons (in absolute values) ranged between 0 and 9.4 (median = 5.4) for each male. All males engaged in dyadic infant handling and bridging interactions. The ten infants that were included in the analysis spent between 3.7 and 26.3 % of focal observation time in dyadic infant handling interactions with males (mean ± SD = 16.2 ± 7.2). The durations of dyadic infant handling interactions (continuous body contact uninterrupted by bridging interaction) varied between 0.03 and 84.5 min (mean ± SD = 2.7 ± 5.2). The values of infant–male CSI based on these dyadic interactions ranged between 0 and 5.8 (median = 0.4; see Fig. S1 in Online Resource 2).
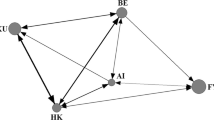
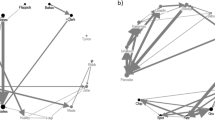
The dataset included 1873 male bridging interactions (between 10 and 368 for each infant, mean ± SD = 187.3 ± 122.1). Of these, 654 (between 6 and 148 for each infant, mean ± SD = 65.4 ± 46.2) were initiated by a male holding the infant and were used for testing the first prediction. These interactions were distributed among 155 holder–infant–receiver triads (from 420 possible). The distribution of interactions is shown in Fig. 1 and Table S1 in Online Resource 1. We recorded the ID of the bystander for 209 of these interactions. A bystander was present in 112 cases (between 4 and 38 cases for each of 6 infants followed during the second season) which we used to test prediction 2. The dataset included interactions with all males participating as holders (range = 5–36), receivers (range = 9–36) and bystanders (range = 12–18). For more details about the distribution of interactions see Fig. 2 and Table S2 in Online Resource 1.
Relationship between the strength of the social relationship (measured as composite sociality index, CSI) between the infant and the receiver (CSIr) and between the infant and the bystander (CSIb). Data points below the diagonal indicate that the receiver had a higher CSI with the infant than the bystander. The size of data points indicates the number of interactions among same infant–receiver–bystander triad. In 72 % of cases the holder chose the male with the stronger bond with the infant as a partner for a bridging interaction
The model selection results for the model testing prediction 1 are summarized in Table S3 in Online Resource 1. The CSI between infant and holder (CSIh), and between infant and receiver (CSIr) both increased the model’s quality. Season, rank of the receiver (DSr), and the difference between rank of the holder and receiver (|ΔDShr|) did not improve either the null model or the models with CSIh and/or CSIr. We thus fitted our final model only with CSIh and CSIr as predictor variables. The frequency of bridging interactions among the holder–infant–receiver triad significantly increased with increasing CSIh and increasing CSIr (ranging between 0 and 5.8; see Table 1; Figs. 3, 4). The model predicted that an increase of CSIh by 1.0 increased the expected frequency of interaction 2.3–3.1 times, and each increase of CSIr by 1.0 increased the expected frequency of interactions 1.7–2.3 times (95 % CI). There was no substantial collinearity between the two predictors (VIF = 1.21).
The model selection results for the model testing prediction 2 are summarized in Table S4 in Online Resource 1. The rank distance between holder and involved male (receiver or bystander; |∆DShm|) and between receiver and bystander (ΔDSrb) did not improve the null model. Adding the difference between the CSI of the infant–receiver and the infant–bystander dyad (ΔCSIrb) improved the model (Table 2; Fig. 5) and was retained as the only predictor in the final model. An increase of ΔCSIrb (ranging between −3.68 and 3.68) by 1.0 increased the probability that a male was selected 1.7–2.7 times (95 % CI).
Discussion
Our results support the hypothesis that males choose their partners based on the relative strength of the affiliative relationship the partner has with the infant. First, the number of bridging interactions of a holder–infant–receiver triad was predicted by the strength of the relationship between the infant and the receiver of the interaction after controlling for the effect of the relationship between the infant and its holder. Second, an infant holder’s choice between two males in proximity of the interaction was predicted by the relative strength of their affiliative relationships with the infant. The stronger a male’s relationship to the infant, relative to the strength of the other male’s relationship, the more likely he was chosen as a receiver instead of being left as a bystander to the interaction. Neither relative nor absolute rank of the receiver was a significant predictor of the distribution of bridging interactions. These patterns in male bridging interactions indicate the use of triadic awareness. Males as initiators of the interactions use their knowledge of the relationships that other males have with an infant they are currently holding when choosing the receiver of the interaction. The use of triadic awareness in the context of infant handling has also been suggested in Tibetan macaques (Ogawa 1995b): in most bridging interactions that were initiated by infant holders, the receiver was provided with the infant he handled the most often, his “affiliated infant”. These results led to the conclusion that the male holding an infant chooses a specific infant based on his knowledge of the preferences of potential receivers. Similarly, our results suggest that Barbary macaque males use knowledge of the relationships between infants and other males when they select partners for bridging interactions. However, we based our study on a slightly different assumption and methodology than the previous study and provide new details that were not previously considered.
We did not assume that holders chose specific infants based on the relationship that an available male (potential receiver) had with different infants (e.g. Ogawa 1995b for Tibetan macaques), but that the holder chooses specific males (as receivers) based on the infant he currently has access to. This adjustment is based on the patterns of infant handling in Barbary macaques, in which bridging interactions are typically preceded by, or alternate with, long dyadic handling periods between the infant and one of the males later involved in the bridging interaction (see Deag and Crook 1971). The low availability of infants leads to long handling episodes, making it rather unlikely that males would be able to find a particular infant (or be motivated to “give up” one infant for another) based on their choice of a receiver male. We suggest that males rather keep one infant for a long time and search for a suitable receiver. This is also in accord with the earlier study on Barbary macaques, which also assumes, that males take infants to specific males rather than to those who just happen to be close (Deag 1980). This view is supported by our result that males were more likely to be chosen as receivers the stronger their relationship to the infant relative to the second available male. We cannot completely rule out, however, that both mechanisms—selection of particular infant and a particular receiving male depending on their relationships—act in concert. Future research should assess in more detail the availability of other potential receivers and other infants to quantify constraints on both mechanisms.
In our study we used a composite sociality index to measure the strength of infant–male relationships instead of using the frequency of interactions (Ogawa 1995b). Due to long periods of dyadic infant handling it is likely that males mainly consider the duration of the interactions between other males and infants when assessing infant–male relationship strength, rather than the number of separate interactions. Thus, a composite index that combines frequency and duration of different behaviours might be better suited for the assessment of infant–male relationships in this species. Our CSI quantified how strong the relationship was between a particular infant and male, relative to the average strength of the relationship between the specific infant and all other males. The distribution of CSI values shows that each infant realizes a number of relationships that vary in strength, rather than affiliating almost exclusively with a single male. Thus, knowledge of third-party relationships may not be restricted to the ability to distinguish between two categories of individuals (affiliated vs. non-affiliated), but might reflect continuous variation in the strength of different relationships. Future studies could benefit from an investigation of the effect of the chosen method of relationship assessment on the results, and explain in more detail how males evaluate infant–male relationships (e.g. whether the duration or frequency of interactions factors most strongly in their assessment).
As mentioned in the introduction, some previous studies struggled to distinguish whether the individual used triadic awareness or acted based on an egocentric view of the world and the strength of his own relationships (see e.g. Perry et al. 2004). In order to address this problem we controlled the holder’s relationships with the infant and absolute and relative dominance rank of the receiver which may affect the holder’s choice of receiver, according to previous studies (Deag and Crook 1971; Paul et al. 1996). We suggest that the study of triadic awareness of infant–male relationships might be less vulnerable to the described problem of ambiguity, compared to the studies based on dominance relationships: where individuals are part of the same hierarchy, they may base their knowledge of others’ dominance relationships either on monitoring the interactions of others (triadic awareness), or on comparing their own dominance relationships with each of other individuals (e.g. the individual who ranks in between two others may recruit the higher ranking from both opponents based on own position; Range and Noë 2005; Bissonnette et al. 2009). In the case presented here, however, it is clear that the relationship that other males have with an infant cannot be easily deduced from one’s own relationships. Holders, whose awareness we assess, have a relationship with the infant they hold; the strength of this relationship influences how often the infant is available for other males, but does not affect how the time is distributed among them. Infant–male relationships also differ from the relationships that females establish among each other. In male dispersal species female relationships are strongly affected by maternal relatedness (Hamilton 1964; Ruiter and Geffen 1998; Silk et al. 2006), which allows one to predict certain aspects of a female’s behaviour from the behaviour of her relative to some degree. Unlike females in matrilineal societies the more individualistic males can also be expected to be independent of each other in developing preferences for certain infants. Thus an understanding of a certain infant–male relationship needs to be based on the monitoring of the interactions of that dyad.
The considerable difference between infant–male and most other affiliative relationships is their ephemerality. Infant handling is very seasonal; male interest in infants peaks shortly after birth and rapidly decreases (see Berghänel et al. 2011). As a consequence of such seasonality, relationships are transient and males have very little time to assess the quality of others’ infant–male relationships and to implement this knowledge during bridging interactions. The fact that males establish triadic awareness of quickly emerging and quickly fading relationships indicates their ability to update their knowledge of others’ relationships quite quickly.
Seasonality of infant handling also implies that males invest in monitoring third-party relationships despite the fact that the information is quickly outdated and needs to be gathered again every year. In light of these investments it seems relevant to ask how males benefit from using knowledge about third-party relationships during bridging, and how these interactions are linked to dyadic infant handling. It has been suggested that bridging interactions mainly serve to establish and maintain bonds among males (Deag and Crook 1971; Paul et al. 1996). The agonistic buffering hypothesis (Deag and Crook 1971) proposes that when holding an infant, males can approach higher-ranking males without being attacked and have a chance to improve and/or re-establish disturbed relationships and reduce stress. The relationship management hypothesis (Paul et al. 1996) emphasizes that bridging gives males the opportunity to interact peacefully in general, not only after a conflict, and that the interactions may contribute to male–male bonding that is profitable in various ways (Kümmerli and Martin 2008) even long term, e.g. via coalition formation (Widdig et al. 2000; Young et al. 2014a). Being provided with his preferred infant the approached male may be more likely to establish an affiliative relationship with the holder, which may become beneficial in terms of coalitionary support as shown in the study species (Berghänel et al. 2011; Young et al. 2014a). The infant preferred by the receiver may be a more effective “buffer” against aggression because the approached male may tend to avoid a conflict that could harm his favourite infant. If males handle infants to regulate their relationship with the mother (e.g. Ménard et al. 2001; Smuts 1985) the receiver should also avoid aggression towards the infant holder because it could disrupt his own relationship with the infant’s mother (Ogawa 1995b). This means, that the choice of receiver might also be influenced by the holder’s previous experience with aggression by specific male when holding specific infant. However, in our study, the rate of observed aggression was too rare to be responsible for observed patterns of the receiver choice suggesting that the choice is rather based on observed male–infant interactions.
The use of triadic awareness may also be guided by mechanisms including hormonal regulation and stress reduction. According to the social buffering hypothesis (not to be confused with the agonistic buffering hypothesis) any affiliative interaction with a closely bonded individual may decrease the physiological stress response, which consequently increases individual health (Cohen and Wills 1985; Hennessy et al. 2009). The hormonal response to social contact (social buffering) depends on the emotional state of the interacting individuals (Kikusui et al. 2006). Hence, the strength of the relationship between the infant and the receiver may predict not only the behavioural responses of the receiver, but also his hormonally regulated attitude towards the initiator (which also feeds back on the hormonal response of the initiator). This suggests that choosing a receiver based on the infant’s relationships may drive a hormonally mediated positive loop (Nagasawa et al. 2015) that benefits both individuals. Future research will have to show how levels of physiological stress, aggression-related hormones and bonding-related hormones are linked (see, e.g. Wingfield et al. 1990; Young et al. 2014b).
Conclusion
Our results indicate that Barbary macaque males recognize the affiliative relationships between infants and other males and make use of this triadic awareness when choosing male partners for bridging interactions. The capacity to monitor, memorize and act upon the social relationships of others has already been documented, but previous studies usually focused on different types of relationships and different contexts of use. Here we provide evidence for the use of triadic awareness that is not related to aggression and is based on temporary and dynamic affiliative infant–male relationships.
References
Altmann J (1974) Observational study of behavior: sampling methods. Behaviour 49:227–266
Aureli F, Cozzolino R, Cordischi C, Scucchi S (1992) Kin-oriented redirection among Japanese macaques: an expression of a revenge system? Anim Behav 44:283–291
Bachmann C, Kummer H (1980) Male assessment of female choice in hamadryas baboons. Behav Ecol Sociobiol 6:315–321. doi:10.1007/BF00292774
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw. doi:10.18637/jss.v067.i01
Berghänel A, Ostner J, Schröder U, Schülke O (2011) Social bonds predict future cooperation in male Barbary macaques, Macaca sylvanus. Anim Behav 81:1109–1116
Bernstein IS, Cooper MA (1998) Ambiguities in the behavior of Assamese macaques. Am J Primatol 45:170–171
Bissonnette A, De Vries H, van Schaik CP (2009) Coalitions in male Barbary macaques, Macaca sylvanus: strength, success and rules of thumb. Anim Behav 78:329–335. doi:10.1016/j.anbehav.2009.05.010
Bowerman BL, O’Connell RT (1990) Linear statistical models: an applied approach. PWS-Kent Publishing Company, Boston
Busse CD, Gordon TP (1984) Infant carrying by adult male mangabeys (Cercocebus atys). Am J Primatol 6:133–141
Busse C, Hamilton WJ (1981) Infant carrying by male chacma baboons. Science 212:1281–1283. doi:10.1126/science.212.4500.1281
Call J, Aureli F, De Waal FBM (2002) Postconflict third-party affiliation in stumptailed macaques. Anim Behav 63:209–216
Chalmers NR (1968) The social behaviour of free living mangabeys in Uganda. Folia Primatol 8:263–281
Cheney DL, Seyfarth RM (1980) Vocal recognition in free-ranging vervet monkeys. Anim Behav 28:362–367
Cheney DL, Seyfarth RM (1989) Redirected aggression and reconciliation among vervet monkeys, Cercopithecus aethiops. Behaviour 110:258–275
Cheney DL, Seyfarth RM (1999) Recognition of other individuals’ social relationships by female baboons. Anim Behav 58:67–75
Cheney DL, Seyfarth RM, Smuts B (1986) Social relationships and social cognition in nonhuman primates. Science 234:1361–1366
Cheney DL, Seyfarth RM, Silk JB (1995) The responses of female baboons (Papio cynocephalus ursinus) to anomalous social interactions: evidence for causal reasoning? J Comp Psychol 109:134
Clutton-Brock T (2002) Breeding together: kin selection and mutualism in cooperative vertebrates. Science 296:69–72
Cohen S, Wills TA (1985) Stress, social support, and the buffering hypothesis. Psychol Bull 98:310
Collins DA (1986) Interactions between adult male and infant yellow baboons (Papio c. cynocephalus) in Tanzania. Anim Behav 34:430–443
Connor RC (2007) Dolphin social intelligence: complex alliance relationships in bottlenose dolphins and a consideration of selective environments for extreme brain size evolution in mammals. Philos Trans R Soc Lond B Biol Sci 362:587–602
Dasser V (1988) A social concept in Java monkeys. Anim Behav 36:225–230
De Vries H, Netto WJ, Hanegraaf PL (1993) Matman: a program for the analysis of sociometric matrices and behavioural transition matrices. Behaviour 125:157–175
De Waal FBM (1986) The integration of dominance and social bonding in primates. Q Rev Biol 61:459–479
De Waal FBM (1991) Complementary methods and convergent evidence in the study of primate social cognition 1). Behaviour 118:297–320. doi:10.1163/156853991X00337
De Waal FBM (2007) Chimpanzee politics: power and sex among apes. JHU Press, Baltimore
De Waal FBM, van Hooff JA, Netto WJ (1976) An ethological analysis of types of agonistic interaction in a captive group of Java-monkeys (Macaca fascicularis). Primates 17:257–290
Deag JM (1980) Interactions between males and unweaned Barbary macaques: testing the agonistic buffering hypothesis. Behaviour 75(1):54–81
Deag JM, Crook JH (1971) Social behaviour and “agonistic buffering” in the wild Barbary macaque Macaca sylvana L. Folia Primatol 15:183–200
Dunbar RIM (1984) Infant-use by male gelada in agonistic contexts: agonistic buffering, progeny protection or soliciting support? Primates 25:28–35
Engh AL, Siebert ER, Greenberg DA, Holekamp KE (2005) Patterns of alliance formation and postconflict aggression indicate spotted hyaenas recognize third-party relationships. Anim Behav 69:209–217
Estrada A, Sandoval JM (1977) Social relations in a free-ranging troop of stumptail macaques (Macaca arctoides): male-care behaviour I. Primates 18:793–813
Ferreira RG, Izar P, Lee PC (2006) Exchange, affiliation, and protective interventions in semifree-ranging brown capuchin monkeys (Cebus apella). Am J Primatol 68:765–776. doi:10.1002/ajp.20277
Fooden J (2007) Systematic review of the Barbary macaque, Macaca sylvanus (Linnaeus, 1758). Fieldiana Zool 113:1–60
Hamilton WD (1964) The genetical evolution of social behavior, I & II. J Theor Biol 104:451–471
Hennessy MB, Kaiser S, Sachser N (2009) Social buffering of the stress response: diversity, mechanisms, and functions. Front Neuroendocrinol 30:470–482
Hinde RA (1976) Interactions, relationships and social structure. Man 11:1–17
Hrdy SB (1976) Care and exploitation of nonhuman primate infants by conspecifics other than the mother. Adv Study Behav 6:101–158
Hrdy SB (2005) Evolutionary context of human development: the cooperative breeding model. In: Carter CS, Anhert L, Grossmann KE, Hrdy SB, Lamb ME, Porges SW, Sachser N (eds) Attachment and bonding: a new synthesis. MIT Press, Cambridge, MA, pp 9–32
Johnson CM (2010) Observing cognitive complexity in primates and cetaceans. J Comp Psychol 23:587–624
Judge P (1982) Redirection of aggression based on kinship in a captive group of pigtail macaques. Int J Primatol 3:225–237
Judge PG (1991) Dyadic and triadic reconciliation in pigtail macaques (Macaca nemestrina). Am J Primatol 23:225–237
Kikusui T, Winslow JT, Mori Y (2006) Social buffering: relief from stress and anxiety. Philos Trans R Soc Lond B Biol Sci 361:2215–2228
Kitchen DM, Cheney DL, Seyfarth RM (2005) Male chacma baboons (Papio hamadryas ursinus) discriminate loud call contests between rivals of different relative ranks. Anim Cogn 8:1–6
Kuester J, Paul A, Arnemann J (1994) Kinship, familiarity and mating avoidance in Barbary macaques, Macaca sylvanus. Anim Behav 48:1183–1194
Kümmerli R, Martin RD (2008) Patterns of infant handling and relatedness in Barbary macaques (Macaca sylvanus) on Gibraltar. Primates 49:271–282
Lehmann J, Korstjens AH, Dunbar RIM (2007) Group size, grooming and social cohesion in primates. Anim Behav 74:1617–1629. doi:10.1016/j.anbehav.2006.10.025
Ménard N, Scheffrahn W, Vallet D, Zidane C, Reber C (1992) Application of blood protein electrophoresis and DNA fingerprinting to the analysis of paternity and social characteristics of wild Barbary macaques. In: Martin RD, Dixson AF, Wickings EJ (eds) Paternity in primates: genetic tests and theories. Karger, Basel, pp 155–174
Ménard N, von Segesser F, Scheffrahn W, Pastorini J, Vallet D, Gaci B, Martin RD, Gautier-Hion A (2001) Is male–infant caretaking related to paternity and/or mating activities in wild Barbary macaques (Macaca sylvanus)? Comptes Rendus Académie Sci Sér III Sci Vie 324:601
Moscovice LR, Di Fiore A, Crockford C, Kitchen DM, Wittig R, Seyfarth RM, Cheney DL (2010) Hedging their bets? Male and female chacma baboons form friendships based on likelihood of paternity. Anim Behav 79:1007–1015
Nagasawa M, Mitsui S, En S, Ohtani N, Ohta M, Sakuma Y, Onaka T, Mogi K, Kikusui T (2015) Oxytocin-gaze positive loop and the coevolution of human-dog bonds. Science 348:333–336. doi:10.1126/science.1261022
Nguyen N, van Horn RC, Alberts SC, Altmann J (2009) “Friendships” between new mothers and adult males: adaptive benefits and determinants in wild baboons (Papio cynocephalus). Behav Ecol Sociobiol 63:1331–1344
Ogawa H (1995a) Bridging behavior and other affiliative interactions among male Tibetan macaques (Macaca thibetana). Int J Primatol 16:707–729
Ogawa H (1995b) Recognition of social relationships in bridging behavior among Tibetan macaques (Macaca thibetana). Am J Primatol 35:305–310
Ostner J, Vigilant L, Bhagavatula J, Franz M, Schülke O (2013) Stable heterosexual associations in a promiscuous primate. Anim Behav 86:623–631. doi:10.1016/j.anbehav.2013.07.004
Paul A (1999) The socioecology of infant handling in primates: Is the current model convincing? Primates 40:33–46
Paul A, Kuester J, Arnemann J (1992) DNA fingerprinting reveals that infant care by male Barbary macaques (Macaca sylvanus) is not paternal investment. Folia Primatol 58:93–98
Paul A, Kuester J, Arnemann J (1996) The sociobiology of male-infant interactions in Barbary macaques, Macaca sylvanus. Anim Behav 51:155–170
Peake TM, Terry AMR, McGregor PK, Dabelsteen T (2002) Do great tits assess rivals by combining direct experience with information gathered by eavesdropping? Proc R Soc Lond B Biol Sci 269:1925–1929
Perry S, Barrett HC, Manson JH (2004) White-faced capuchin monkeys show triadic awareness in their choice of allies. Anim Behav 67:165–170
Quinn GP, Keough MJ (2002) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge
R Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, p 2013
Range F, Noë R (2005) Can simple rules account for the pattern of triadic interactions in juvenile and adult female sooty mangabeys? Anim Behav 69:445–452
Riedman ML (1982) The evolution of alloparental care and adoption in mammals and birds. Q Rev Biol 57:405–435
Ruiter JD, Geffen E (1998) Relatedness of matrilines, dispersing males and social groups in long-tailed macaques (Macaca fascicularis). Proc R Soc Lond B Biol Sci 265:79–87
Schino G, Tiddi B, Di Sorrentino EP (2006) Simultaneous classification by rank and kinship in Japanese macaques. Anim Behav 71:1069–1074
Schino G, Tiddi B, Di Sorrentino EP (2007) Agonistic support in juvenile Japanese macaques: cognitive and functional implications. Ethology 113:1151–1157
Schmid VS, De Vries H (2013) Finding a dominance order most consistent with a linear hierarchy: an improved algorithm for the I&SI method. Anim Behav 86:1097–1105
Schülke O, Bhagavatula J, Vigilant L, Ostner J (2010) Social bonds enhance reproductive success in male macaques. Curr Biol 20:2207–2210. doi:10.1016/j.cub.2010.10.058
Schwarz G (1978) Estimating the dimension of a model. Ann Stat 6:461–464
Seed AM, Clayton NS, Emery NJ (2007) Postconflict third-party affiliation in rooks, Corvus frugilegus. Curr Biol 17:152–158. doi:10.1016/j.cub.2006.11.025
Silk JB (1999) Male bonnet macaques use information about third-party rank relationships to recruit allies. Anim Behav 58:45–51
Silk JB (2007a) Social components of fitness in primate groups. Science 317:1347–1351
Silk JB (2007b) The adaptive value of sociality in mammalian groups. Philos Trans R Soc Lond B Biol Sci 362:539–559
Silk JB, Samuels A (1984) Triadic interactions among Macaca radiata: passports and buffers. Am J Primatol 6:373–376
Silk JB, Altmann J, Alberts SC (2006) Social relationships among adult female baboons (Papio cynocephalus) I. Variation in the strength of social bonds. Behav Ecol Sociobiol 61:183–195
Silk JB, Beehner JC, Bergman TJ, Crockford C, Engh AL, Moscovice LR, Wittig RM, Seyfarth RM, Cheney DL (2009) The benefits of social capital: close social bonds among female baboons enhance offspring survival. Proc R Soc B: Biol Sci 276:3099–3104
Small MF (1990) Promiscuity in Barbary macaques (Macaca sylvanus). Am J Primatol 20:267–282
Smuts BB (1985) Sex and friendship in baboons. Aldine, New York
Smuts BB, Gubernick DJ (1992) Male–infant relationships in nonhuman primates: paternal investment or mating effort? In: Hewlett BS (ed) Father–child relations: cultural and biosocial contexts. Aldine de Gruyter, New York, pp 1–30
Stein DM (1984) The sociobiology of infant and adult male baboons. Ablex Publishing Corporation, Norwood
Sterck EHM, Watts DP, van Schaik CP (1997) The evolution of female social relationships in nonhuman primates. Behav Ecol Sociobiol 41:291–309
Taub DM (1980) Testing the “agonistic buffering” hypothesis. Behav Ecol Sociobiol 6:187–197
Thierry B, Bynum E, Baker S, Kinnaird MF, Matsumura S, Muroyama Y, O’Brien TG, Petit O, Watanabe K (2000) The social repertoire of Sulawesi macaques. Primate Res 16:203–226
Tomasello M, Call J (1997) Primate cognition. Oxford University Press, Oxford
van Hooff JARAM, van Schaik CP (1994) Male bonds: affiliative relationships among nonhuman primate males. Behaviour 130:309–337. doi:10.1163/156853994X00587
van Schaik CP, Paul A (1996) Male care in primates: does it ever reflect paternity? Evol Anthropol Issues News Rev 5:152–156
Widdig A, Streich WJ, Tembrock G (2000) Coalition formation among male Barbary macaques (Macaca sylvanus). Am J Primatol 50:37–51
Wingfield JC, Hegner RE, Dufty Jr AM, Ball GF (1990) The “challenge hypothesis”: theoretical implications for patterns of testosterone secretion, mating systems, and breeding strategies. Am Nat 136:829–846
Woodroffe R, Vincent A (1994) Mother’s little helpers: patterns of male care in mammals. Trends Ecol Evol 9:294–297. doi:10.1016/0169-5347(94)90033-7
Young C, Majolo B, Schülke O, Ostner J (2014a) Male social bonds and rank predict supporter selection in cooperative aggression in wild Barbary macaques. Anim Behav 95:23–32
Young C, Majolo B, Heistermann M, Schülke O, Ostner J (2014b) Responses to social and environmental stress are attenuated by strong male bonds in wild macaques. Proc Natl Acad Sci 111:18195–18200
Zhao Q-K (1996) Male-infant-male interactions in Tibetan macaques. Primates 37:135–143. doi:10.1007/BF02381401
Acknowledgments
This study was supported by grant 009/2014/P and 04-151/2016/P provided by the Grant Agency of the University of South Bohemia, Christian-Vogel Fond for Field Research of the Gesellschaft für Primatologie and by the scholarship of the German Academic Exchange Service DAAD. We are grateful to the Haut Commissariat aux Eaux et Forêts et à la Lutte Contre la Désertification of Morocco for research permission and professor Mohamed Mouna (Institute Scientifique, Rabat, Morocco) and professor Mohamed Quarro (Ecole Nationale Forestière d’Ingénieurs, Salé, Morocco) for their invaluable support and cooperation during the field work. We are thankful for the support and comments of all our collaborators and colleagues that contributed to the realization of the study and preparation of the manuscript. We particularly appreciate advices and support from James Waterman, Josephine Kalbitz, Adeelia Goffe, Andreas Berghänel, Christopher Young, Christina Haunhorst and Stanislav Lhota. Special thank goes to Els van Lavieren, the director of the Moroccan Primate Conservation Foundation, for her help and effort related to the protection of wild populations of Barbary macaques. We also want to thank Dr. Sarah Boysen and two anonymous referees for their thoughtful and insightful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Our study was observational and non-invasive. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in this study were in accordance with the standards of the International Primate Society for the use of non-human primates in research.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10071_2016_1041_MOESM2_ESM.tiff
Fig. S1 Distribution of the strength of the relationship (measured as composite sociality index, CSI) between infant–male dyads (TIFF 686 kb)
Rights and permissions
About this article
Cite this article
Kubenova, B., Konecna, M., Majolo, B. et al. Triadic awareness predicts partner choice in male–infant–male interactions in Barbary macaques. Anim Cogn 20, 221–232 (2017). https://doi.org/10.1007/s10071-016-1041-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10071-016-1041-y