Abstract
There is no research about age difference in the process of sequential learning in non-human primates. Is there any difference between young and adults in sequential learning process? Six chimpanzees (Pan troglodytes), 3 young and 3 adults, learned the Arabic numeral sequence 1 to 9 by touching the numerals on a touch-screen monitor in ascending order. Initially, the sequence always started with the numeral 1, i.e. ‘start-fixed task’. Training began with the sequence 1–2, 1–2–3, and continued sequentially up to 1–2–3–4–5–6–7–8–9. Later, the subjects were introduced to sequences that started with a random numeral, but always ended with 9, i.e. ‘end-fixed task’. Performance in the end-fixed task was worse relative to the familiar start-fixed task. After training with various sequences of adjacent numerals, the subjects were given a transfer test for the non-adjacent numerals. The results suggested that all chimpanzees indeed mastered sequential ordering, and although there was no fundamental difference in the acquisition process between the two age groups, there was a significant age difference in memory capacity. Based on their knowledge of sequential ordering, the subjects were then asked to perform a masking task in which once a subject touched the lowest numeral, the other numeral(s) turned to white squares. Performance of the masking task by young chimpanzees was better than that of adults in accuracy and degree of difficulty (number of numerals). Taken together, these data clearly demonstrate a similarity among subjects in the way chimpanzees acquire knowledge of sequential order regardless of age differences in sequential learning. Moreover, they reveal that once knowledge of sequential order is established, it can be a good index used to evaluate memory capacity in young and adult chimpanzees.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Sequential learning can be defined as “the ability to encode and represent the order of discrete items occurring in a sequence” (Conway and Christiansen 2001). Sequential learning is ubiquitous in various cognitive domains, such as human speech and language processing, the learning of action sequences, or any task that requires putting items into an ordered series. One of the simplest types of sequential learning involves the learning of an arbitrarily fixed sequence of items, such as remembering a phone number (Conway and Christiansen 2001). In these cases, learning can be as simple as forming associations between items in a sequence.
Sequential learning has been extensively studied in non-human primates (e.g. ring-tailed lemurs: Merritt et al. 2007; capuchin monkeys: D’Amato and Colombo 1988; Japanese monkeys: Ohshiba 1997; and rhesus macaques: Chen et al. 1997; Swartz et al. 1991, 2000; Terrace et al. 2003; Treichler and Raghanti 2009; Treichler and Tilburg 1999, 2002; Treichler et al. 2003; Washburn and Rumbaugh 1991). Chimpanzees, the closest evolutionary neighbours to humans, have also been the focus of these studies (Boysen et al. 1993; Biro and Matsuzawa 1999; Tomonaga and Matsuzawa 2000; Beran et al. 2004). Nonetheless, there are no studies that focus on developmental differences in sequential learning by non-human primates.
The goal of this study is to investigate the sequential learning of Arabic numerals by chimpanzees in different developmental stages, e.g. young versus adults. A female chimpanzee named ‘Ai’ was the first chimpanzee that learned to use Arabic numerals to label sets of real-life objects with a corresponding number (Matsuzawa 1985; Murofushi 1997). Ai later mastered various additional numerical skills in both the cardinal and ordinal domains (Tomonaga and Matsuzawa 2002; Tomonaga 2008), including the use of zero (Biro and Matsuzawa 2001). Moreover, she has shown high working memory ability while using her knowledge of Arabic numerals (Kawai and Matsuzawa 2000). The present study aims to verify and extend what we have learned from Ai by exploring the nature of sequential learning of numerical ordering in multiple chimpanzees.
We introduced a new series of experiments to six naïve chimpanzees involving sequential learning tasks that used Arabic numerals. The subjects belonged to two age groups: young and adults. The young chimpanzee group consisted of three individuals that were approximately 4-years-old at the beginning of this study. The adult chimpanzee group included three individuals that were 20 years or older. These age ranges made it possible to compare differences between the two age groups as well as studying the differences between individuals. Although all six subjects had participated in various kinds of tasks in previous studies (see Matsuzawa 2003; Matsuzawa et al. 2006; Matsuzawa 2009a), they had never experienced the sequential order tasks presented in this study. Our aim in this study was to describe how naïve chimpanzees learned sequential order using numerals. We performed a transfer test of the learned sequence to non-adjacent numerals to determine the nature of the subjects’ knowledge of acquired order. Furthermore, we compared sequential learning between young and adult chimpanzees, as well as their working memory ability based on their knowledge of sequential order.
General method
Subjects
A total of 6 chimpanzees (Pan troglodytes) served as subjects. The three young chimpanzees were born in 2000 and included a male named Ayumu (3 years and 11-months-old), and two females named Cleo (3 years and 10-months-old) and Pal (3 years and 8-months-old). These ages were recorded at the beginning of the experiments in this study, which began in April 2004. At that time the young chimpanzees were still suckling, and the average weaning time of the chimpanzees is 4–5-years-old. Young need their mothers as a safe base from which to explore their surrounding environment. The three adult chimpanzees included a male named Akira (28-years-old and the biological father of both Ayumu and Pal), and two females named Chloe (23-years-old and the mother of Cleo) and Pan (20-years-old and the mother of Pal). The subjects are members of a community of 14 chimpanzees living in a semi-natural enriched environment at the Primate Research Institute of Kyoto University (Matsuzawa et al. 2006). Care and use of the chimpanzees adhered to the 2002 version of the “Guide for care and use of laboratory primates” of the Primate Research Institute, Kyoto University. The research design was approved by the Animal Welfare and Animal Care Committee of the Institute.
Apparatus
Experiments were conducted in a testing room with twin test booths (2.0 m in width, 3.2 m/3.2 m in depth, and 2.0 m in height) made of transparent glass walls and a vertical sliding door connecting the booths. The twin booths are at opposite sides of the test room and are used to concurrently test two subjects on identical tasks. Each booth was equipped with a touch-screen monitor (Pro-Tect, PD-105TP15; TORiSAN, LMU-TK15A4TJ; MEE, TSD-FT157-MN) controlled by a personal computer (A-ONE, VXM-GLC). Correct trials were followed by food rewards consisting of a small 8-mm cube of apple or half a raisin delivered automatically by a universal feeder (Biomedica, BUF-310-P50). The number of apple pieces delivered for a correct trial was usually only 1 but was increased to a maximum of 5 depending on the motivation and performance of each subject. In this study, three mother–offspring pairs participated in the experiments at the same time in the twin booths, and included Ai and Ayumu, Chloe and Cleo, and Pan and Pal. Ai participated alongside her son Ayumu, but because she had previous experience on numerical tasks (see Matsuzawa 1985, 2009b; Kawai and Matsuzawa 2000), her data were excluded from the analyses in this study. The adult male Akira was tested alone.
Stimuli
The stimuli used were the Arabic numerals 1 through 9 presented as white Gothic typeface 3 cm in height against a black background on the monitor. The numerals appeared in different positions in each trial within an 8 by 5 invisible matrix on the touch-screen monitor.
General procedure
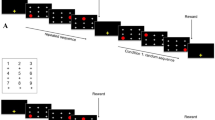
We invited each chimpanzee to the booth from their outdoor compound by calling their name, and they walked through a corridor to the test room and booth on their own free will. A trial began when the subject touched a start key (white circle) presented on the monitor. This was followed by the appearance of the numerical stimuli, which the subject was required to touch one by one in ascending order (Fig. 1). Numerals correctly selected disappeared from the monitor immediately with a concurrent click sound as feedback. When the subject touched all the numerals in the correct ascending order, a chime sounded and it was followed by a food reward. If a numeral was selected incorrectly, the screen was cleared, a buzzer sounded, and no food was delivered. The computer-controlled task was conducted, on average, for 30 min a day. All subjects, except Akira, participated in the experiment 5–6 days a week; Akira participated 1–2 days a week.
Experiment 1
To extend and verify results obtained from studies involving Ai in the past (see Biro and Matsuzawa 1999; Kawai and Matsuzawa 2000; Tomonaga and Matsuzawa 2002; Tomonaga 2008; Matsuzawa 2009b), we started a new series of experiments consisting of a sequential learning task using Arabic numerals with the six naïve-chimpanzee subjects.
Methods
Procedure
One numeral in the range of 1 to 9 was randomly presented on the monitor as part of pre-training. This easy task was aimed at habituating the subjects to the apparatus and testing booth. The location of the numeral on the screen was randomized. A trial was as follows. When the subject touched the start-key (white circle), it disappeared and was followed by the numeral. Touching the numeral then made the monitor turn off with a chime informing the subject that it was a correct trial and rewarding him/her with food: this was a type of errorless training. One session consisted of 50 trials. The subjects received two to six sessions of pre-training touching a numeral on the screen.
After the pre-training phase, the first stimulus pair consisting of numeral 1 and 2 was introduced. The two numerals appeared in random positions on the monitor. The subject was required to touch the two numerals in ascending order. If the subject touched 2 instead of 1 first, a buzzer sounded and the trial ended. A learning criterion was set based on the average performance by Ai who had already acquired knowledge of numerals 1 through 9. For the learning criterion we ran 200 trials, with 4 sessions each consisting of 50 trials, for each sequence from 1 through 9 from Ai. When the naïve subjects reached the ‘1–2’ learning criteria, a new numeral, 3, was introduced (Table 1). Eventually, the subjects learned to touch the numerals 1–2, 1–2–3, 1–2–3–4 in ascending order until they learned the sequence 1–2–3–4–5–6–7–8–9. Each subject received four sessions per day, and a session on average consisted of 50 trials.
During this stage of training, we introduced the following parameters to facilitate learning: (1) we adopted a correction method by repeating the same trial until the correct response occurred; performance on correction trials was excluded in the assessment learning based on criterion, (2) we manipulated the Inter-Trial-Interval within a range of 1–5 s, (3) we manipulated Time Out within a range of 0–3 s, (4) we mixed new sequences with old, familiar, and easier sequences that had been already learned by the subjects in previous phases, and (5) we introduced an easy sequence session, with one numeral less than the new sequence, as a baseline before the training session.
Results and discussion
All of the subjects successfully mastered the skill of touching numerals 1 to 9 sequentially (Table 1). Chi-square tests showed significant differences for each individual subject between the observed percent of correct trials and the percent predicted by chance for each task based on the number of possible numeral combinations in each task (all p < 0.05). It took between 551 to 944 sessions in total (including baseline session) for each subject to learn to correctly touch the 9 numerals in ascending order. There was no statistical difference in the learning speed between age groups (mean number of sessions for young group ± SD: 778 ± 159; adult group: 765 ± 199; t-test: t = 0.14, df = 4, p = 0.93). This result clearly demonstrated that all chimpanzees, regardless of age, could learn the sequence 1 through 9 as Ai and other primates have (Terrace 2005; Terrace et al. 2003).
Experiment 2
In experiment 1 the subjects learned to touch sequences starting from 1 to 2 and eventually finishing with 1 to 9. In that experiment the first numeral of the sequences was always 1. In experiment 2 we tested the flexibility of the acquired fixed sequences by introducing a series of transfer tests. These tests consisted of new sequences that could not only start from numeral 1, but also from other numerals. Experiment 2 began immediately after the last phase of experiment 1 finished.
Methods
Procedure
In this experiment a session included different sequence lengths. In a session, individuals were presented with one of two tasks: a task familiar to them from experiment 1 or a new task. We called the familiar task used in experiment 1 “the start-fixed task” because it consisted of sequences that always began with the numeral 1; there were a total of eight sequence patterns starting with 1–2, 1–2–3, all the way up to 1–2–3–4–5–6–7–8 and 1–2–3–4–5–6–7–8–9. We called the new task introduced in this experiment “the end-fixed task” because it consisted of sequences that always ended with 9. Here too there were eight patterns starting with 8–9, 7–8–9, all the way up to 2–3–4–5–6–7–8–9 and 1–2–3–4–5–6–7–8–9. Each of the 8 sequences randomly appeared five times in a session and each session consisted of 40 trials. The two tasks (the start-fixed task and the end-fixed task) were not mixed within a session, and each task was conducted alternately. Each subject received two or four sessions per day. The data collected were from 10 sessions for each task.
Results and discussion
We found that there were significant differences in the subjects’ performance of the start-fixed task versus the end-fixed task (Fig. 2). Their performance in the unfamiliar end-fixed task was significantly worse than that of the familiar start-fixed task. A two-way ANOVA revealed that both main effects (tasks and stimuli as number of numerals) were significant in all of the subjects and that there was also an interaction between the main effects; Ayumu (tasks: F 1,144 = 290.00, p < 0.001; stimuli: F 7, 144 = 31.59, p < 0.001; interaction: F 7, 144 = 14.02, p < 0.001), Cleo (tasks: F 1,144 = 82.03, p < 0.001; stimuli: F 7, 144 = 20.71, p < 0.001; interaction: F 7,144 = 4.83, p < 0.001), Pal (tasks: F 1,144 = 72.10, p < 0.001; stimuli: F 7,144 = 14.99, p < 0.001; interaction: F 7,144 = 3.87, p < 0.001), Akira (tasks: F 1,144 = 63.32, p < 0.001; stimuli: F 7,144 = 29.40, p < 0.001; interaction: F 7,144 = 3.93, p < 0.001), Chloe (tasks: F 1,144 = 97.31, p < 0.001; stimuli: F 7,144 = 21.01, p < 0.001; interaction: F 7,144 = 6.70, p < 0.001), and Pan (tasks: F 1,144 = 97.30, p < 0.001; stimuli: F 7,144 = 25.81, p < 0.001; interaction: F 7,144 = 3.89, p < 0.001), respectively. Moreover, the response time of the first touch was much longer in the end-fixed task than in the start-fixed task (Fig. 3); there was a significant main effect of tasks among the subjects except for Akira in a two-way ANOVA; Ayumu (tasks: F 1,437 = 72.79, p < 0.001; stimuli: F 7,437 = 2.94, p < 0.01; interaction: F 7,437 = 2.27, p < 0.05), Cleo (tasks: F 1,381 = 249.24, p < 0.001; stimuli: F 7,381 = 12.51, p < 0.001; interaction: F 7,381 = 9.96, p < 0.001), Pal (tasks: F 1,505 = 6.98, p < 0.01; stimuli: F 7,505 = 1.47, p = 0.17; interaction: F 7,505 = 1.47, p = 0.18), Akira (tasks: F 1,474 = 3.27, p = 0.07; stimuli: F 7,474 = 0.69, p = 0.68; interaction: F 7,474 = 1.25, p = 0.27), Chloe (tasks: F 1,495 = 377.64, p < 0.001; stimuli: F 7,495 = 6.93, p < 0.001; interaction: F 7,495 = 9.18, p < 0.001), and Pan (tasks: F 1,484 = 115.93, p < 0.001; stimuli: F 7,484 = 6.69, p < 0.001; interaction: F 7,484 = 4.17, p < 0.001).
There was a significant effect of task and age but there was no significant effect of interaction on performance (two-way ANOVA; age: F 1,956 = 8.50, p < 0.01; task: F 1,956 = 274.02, p < 0.001; interaction: F 1,956 = 2.76, p = 0.10). There was a significant effect of task, age and their interaction on response time (two-way ANOVA; age: F 1,2869 = 17.20, p < 0.001; task: F 1,2869 = 108.39, p < 0.001; interaction: F 1,2869 = 8.46, p = 0.003). Despite there was no difference in response time in the start-fixed task between age groups (post-hoc pair wise contrasts, F 1,2869 = 1.04, p = 0.31), adults had a longer response time than young in the end-fixed task (post-hoc pair wise contrasts, F 1,2869 = 19.75, p < 0.001). Differences in performance and response time between age groups appear only when presented with the novel (end-fixed) task.
Even though the sequences in the end-fixed task had appeared as part of the previously learned sequence ‘1 to 9’ in experiment 1, all chimpanzees, young and adults, showed difficulty in this task. The sequences were similar to list learning (D’Amato and Colombo 1988; Terrace 1993), yet previous knowledge of ‘1 to 9’ did not transfer to the sequences in experiment 2. Why this task was so difficult remains unclear, but one plausible explanation is the variation in the starting point. The sequences of the start-fixed task always started from 1. In contrast, the end-fixed task did not always start with 1 but with other numerals. The results suggest that the numeral 1 was always a cue for initiating touch in the start-fixed task. However, the subjects did not have this fixed cue of the same starting numeral in the end-fixed task.
Experiment 3
The first two experiments required the chimpanzees to touch the numerals 1 to 9 in ascending order. This was true for both the start-fixed task and end-fixed task. Up to this point the numerals displayed on the screen were always sequentially adjacent. Now we wanted to assess the subjects’ performance with non-adjacent numerals. Thus, in experiment 3 we asked whether the subjects acquired a flexible knowledge of order, which is something more complex than a simple fixed sequence. Our aim was to answer this question by providing an opportunity for the subjects to transfer their knowledge of previously learned sequences of numerals to unfamiliar combinations of non-adjacent numerals.
Methods
Procedure
Prior to testing the subjects’ ability to learn non-adjacent numerals we performed a limited series of preliminary training tasks with adjacent numerals, i.e. two, three, and four adjacent numerals. For example, in the two adjacent numeral tasks, the subjects were asked to choose the sequence of 1–2, 2–3, 3–4, 4–5, 5–6, 6–7, 7–8 or 8–9; the three adjacent numerals consisted of 1–2–3, 2–3–4, 3–4–5, 4–5–6, 5–6–7, 6–7–8, or 7–8–9; the four adjacent numerals consisted of 1–2–3–4, 2–3–4–5, 3–4–5–6, 4–5–6–7, 5–6–7–8, or 6–7–8–9. These preliminary training tasks served as a control condition for how the subjects would perform when given varying lengths of adjacent numeral sequences containing varying least numerals. During this preliminary training we found that the chimpanzees’ performance deteriorated when the fixed length of the sequence got longer, e.g. 84.3% average accuracy in the two numerals condition, 66.3% average in the three numeral condition, and 59% average in the four numeral condition for the 6 subjects.
To improve the performance of sequential learning of adjacent numerals regardless of the length of sequences, we added an extra-phase to the preliminary training of all the subjects. We sequentially introduced intensive training of the end-fixed sequences starting from 7–8–9, then 6–7–8–9 all the way up to 2–3–4–5–6–7–8–9. Each session consisted of 50 trials, and a maximum of 40 sessions were conducted.
After intensive training with adjacent numerals, we proceeded to conduct a transfer test for non-adjacent numerals. We performed eight kinds of test sequences ranging from a two-numeral-length to a nine-numeral-length. For each numeral-length condition, all possible combinations of numeral sequences were used. For example, the task for two-numeral-length condition included combinations, such as 2–5, 4–9, 3–6, etc, and consisted of 36 kinds of trials (9C2). A seven-numeral-length condition used combinations like 1–2–4–5–6–7–9, 2–3–4–6–7–8–9, or 1–3–4–5–6–7–8, and consisted of 36 kinds of trials (9C7). The task for three- (9C3) and six-numeral-length (9C6) conditions consisted of 84 kinds of trials, while the task for four- (9C4) and five-numeral-length (9C5) conditions consisted of 126 kinds of trials. In the eight-numeral-length condition there are only 9 possible sequence variations, therefore a session consisted of 54 trials (6 repetitions of 9 kinds of trials). Finally, the nine-numeral-length condition is always 1–2–3–4–5–6–7–8–9, and a session consisted of 50 trials.
All possible combinations of numerals were tested in the transfer test, and therefore sequences contained both adjacent and non-adjacent numerals within a session. The transfer test of non-adjacent numerals began with the two numeral tasks and proceeded to the nine numeral tasks. To cancel out the order of the tasks, the test continued by starting with the nine numeral tasks and down to the two numerals tasks. Each numeral length task, except for the two and seven numerals length, was tested twice a day and was conducted during a total of 4 sessions. The two- and seven-numeral conditions were tested three times a day and conducted in a total of 6 sessions. In summation, there were between 200 and 252 trials for each numeral length.
Results and discussion
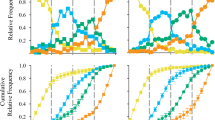
Results of intensive training with the end-fixed sequences and the non-adjacent test were obtained from five of the subjects and not from the adult male, Akira, who did not proceed to this test. It took between 25 and 82 sessions in total for each subject to complete the intensive training of the end-fixed sequences (Table 2). All five subjects performed well in the transfer test, and similar to that in the adjacent sequence tasks; Chi-square tests showed significant difference between observed percent correct and percent predicted by chance for each individual and each task (p < 0.05; Fig. 4), even though the subjects had no previous experience with non-adjacent sequences. The results from experiment 2 initially demonstrated that the numeral 1 appeared to be a special cue for initiating touches on the screen. However, after the training with sequences that start from numerals other than 1, the subjects succeeded in identifying the smallest numeral. Previous research (Treichler and Tilburg 2002; Terrace et al. 2003) reported that macaques had flexible knowledge from possible novel combinations of acquired sequence. Our data not only confirm these previous results, but also show that there were no fundamental differences between adjacent and non-adjacent numeral sequences by introducing this kind of careful control in the training of adjacent numerals, and that this occurs regardless of age.
The performance of the non-adjacent task by five chimpanzee subjects. All subjects showed high performance in the task. CL in the caption refers to chance level for each number of numerals sequence. The x-axis shows the number of the numerals in the sequence; percentage of trials correctly completed during each session is shown on the y-axis
Experiment 4
Experiments 1 to 3 showed that the chimpanzee subjects learned sequential order using numerals in ascending order from 1 to 9. For them it was not a mere fixed sequence but a more flexible sequence order. However, this raises another question, and that is namely how do chimpanzees mentally perform the task of touching numerals in ascending order? It is possible that they compare the numerals one by one at once (Ohshiba 1997), or that they grasp the scattered numerals at a glance and make a pre-plan of the consecutive touches (e.g. only the next selection, Beran et al. 2004; entire sequence, Biro and Matsuzawa 1999). To determine the strategy for touching the numerals, we asked the subjects to perform a masking task.
Once the subjects had mastered the order of nine numerals in all kinds of combinations of 1 through 9, both adjacent and non-adjacent, we introduced a memory task named “the masking task” (Kawai and Matsuzawa 2000). In this task, the numeral(s) on the screen turned to white squares immediately after the subject touched the smallest numeral. This tested working memory based on the knowledge of the ascending order of the numerals. This masking task was tested around the time the three young chimpanzees reached the age of 5 years.
Methods
Subjects
The subjects were the five chimpanzees who participated in experiments 1–3. The adult male Akira was not included.
Procedure
After touching the first numeral, all other numerals were replaced by white squares, or a checker-pattern square to completely suppress the possible afterimage. Subjects were required to touch the white squares in the order that the numerals were originally shown (Fig. 5). The masking task began with only two numerals. All possible combinations from 1 to 9, of which there were 36 variants, were tested. Next, the test proceeded to a three numerals masking task (84 variants), and then a four numerals masking task (126 variants). The number of trials in a session was kept constant at 50 trials regardless of the conditions. The performance in the masking task was compared with the corresponding non-masking task (Figs. 5, 6, 7).
A comparison of performance of three young subjects (closed bars) and two adults (open bars) in the non-masking task (a) and the masking task (b). The data for the non-masking task were the average of 252 trials from 2 successive sessions. The data for the masking task were the average of 250 trials from 5 successive sessions. The x-axis shows the subjects, while the percentage of trials correctly completed during each session is shown on the y-axis
A comparison of response time by three young (closed symbols) and two adult chimpanzees (open symbols) of the non-masking task (a) and the masking task (b). The data for the non-masking task were the median response time in the correct trials from 2 successive sessions. The data of the masking task were the median of the response time in the correct trials from 5 successive sessions. The x-axis shows the touch in a sequence, while the median response time (ms) during correct trials is shown on the y-axis
Results and discussion
All five chimpanzees mastered the masking task to some extent. Although the same amount of training was given, there was an age group difference. Ayumu mastered the task with two numerals, three numerals, four numerals, and all the way to the entire nine-numeral masking task. The young females Cleo and Pal learned up to the seven and six masked numeral tasks, respectively. In contrast, the two adults, Chloe and Pan, reached the level of training the task with four masked numerals.
We compared adults and young in their performance of the four-numeral condition, i.e. the maximum length reached by the adults in the non-masking task, versus the masking task. In this comparison, there was also a significant difference in performance between the two age groups, with young performing better than adults. All subjects had knowledge of sequences from the previous non-masking task experiments (see Fig. 6a). There was no significant difference between age groups or between individuals (age group: F 1,5 = 0.14, p = 0.73; individuals: F 1,5 = 4.61, p = 0.07). However, the performance of adults was lower than that of the young in the masking task (see Fig. 6b, age group: F 1,20 = 77.61, p < 0.001).
There was a significant effect of touch order, but not age or the interaction between age and touch order in the response time pattern for the non-masking task (see Fig. 7, age groups: F 1,4160 = 0.002, p = 0.96; touch: F 3,4160 = 25.00, p < 0.001; interaction: F 3,4160 = 1.50, p = 0.21) Likewise, there was a significant effect of touch order, but not age or the interaction between age and touch order in the masking task (age groups: F 1,3472 = 0.53, p = 0.47; touch: F 3,3472 = 75.87, p < 0.001). The response time to the first numeral was much longer than to the remaining numerals in the sequence, suggesting that the subjects judged whole numerals presented on the monitor before making their first touch. These results support the idea that the long latency to the first touch may be due to pre-planning of how an individual will touch the multiple numerals on the screen (Biro and Matsuzawa 1999). Thus, although the strategy of memorizing and touching numerals may be common to all of the subjects, these data demonstrated that young chimpanzees were better than adults in their memory capacity.
General discussion
We have been able to provide some answers to the question on whether there are any developmental differences in sequential learning. Our data shows that all of the six naïve-chimpanzees succeeded to learn the sequence of Arabic numeral from 1 to 9 regardless of age. Furthermore, all subjects showed similarities in the acquisition process: a similar speed to complete the training phase learning numerals 1 through 9 (Exp. 1), similar difficulty with the end-fixed task except response time (Exp. 2), and a similar clear transfer from adjacent sequences to non-adjacent sequences (Exp. 3). We also found that the strategy for touching the numerals was common to all chimpanzee subjects (see Fig. 7). All subjects had longer response times to the 1st touch in a sequence, and the response time to remaining numerals on the screen was nearly equal regardless of the length of the sequence. This suggests that both young and adult chimpanzees have similar strategies of pre-planning and this is consistent with results from previous studies (e.g. Biro and Matsuzawa 1999). Clearly, the acquisition of sequence 1 to 9 is not different among chimpanzees in different age groups.
Although individuals can learn the sequence of numerals in a similar way, our data show that there were significant differences between young and adult chimpanzees in the memory task. Young chimpanzees were much better than adult chimpanzees in the masking task. This difference cannot be due to a possible difference in strategy. Adult subjects could answer correctly all the way to four numerals in the masking task and this does not support a strategy of comparing the numerals one by one all at once (Ohshiba 1997) or planning only the next selection (Beran et al. 2004).
The strategy “pre-planning at a glance” should be the same among all the subjects including Ai who was experienced with these tasks (Biro and Matsuzawa 1999; Kawai and Matsuzawa 2000). As mentioned in the introduction, chimpanzee Ai learned a variety of tasks that involved knowledge of numbers. However, when she was introduced to the masking task with numerals, she was already 20-years-old (Kawai and Matsuzawa 2000). In that sense, Ai should be grouped with the adults in this study, and her performance in the masking task (see Kawai and Matsuzawa 2000) was actually below all of the three young chimpanzees. Previous studies by our colleagues and us (Kawai and Matsuzawa 2000; Inoue and Matsuzawa 2007) fit well with the present results, i.e. the experiences of the tasks using same stimuli did not influence working memory capability. But the difference of performance age correlates with working memory capability. This result, however, leads to the obvious question of why young chimpanzees have better working memory than adult chimpanzees?
In studies on humans, young children can be better than adults in certain memory tasks (Hayes and Heit 2004; Sloutsky and Fisher 2004). For example, response strategies gradually change in the different developmental stages of children, such as 5, 7, and 11-year-olds (Fisher and Sloutsky 2005). This kind of developmental difference in learning strategy could be explained as a sort of developmental trade-off. In the course of cognitive development, children may acquire linguistic skills, such as category-based induction, while losing their perceptual photographic memory that appears in physical similarity-based induction. This kind of trade-off may also work in the chimpanzee, a non-verbal animal, as demonstrated by their performance in memory tasks, which was actually better in young. If the developmental trade-off can be applied to non-verbal chimpanzees, performance of memory task in young chimpanzees might decrease gradually as they age to adulthood. This leads to the prediction that young chimpanzees may not be good at category-based induction task. Testing this prediction will be the focus of future studies, and they should help us to better understand cognitive development in chimpanzees.
References
Beran MJ, Pate JL, Washburn DA, Rumbaugh DM (2004) Sequential responding and planning in chimpanzees (Pan troglodytes) and rhesus macaques (Macaca mulatta). J Exp Psychol Anim Behav Proc 30:203–212
Biro D, Matsuzawa T (1999) Numerical ordering in a chimpanzee (Pan troglodytes): planning, executing and monitoring. J Comp Psychol 113:178–185
Biro D, Matsuzawa T (2001) Use of numerical symbols by the chimpanzee (Pan troglodytes): cardinals, ordinals, and the introduction of zero. Anim Cogn 4:193–199
Boysen ST, Berntson GG, Shreyer TA, Quigley KS (1993) Processing of ordinality and transitivity by chimpanzees (Pan troglodytes). J Comp Psychol 107:208–215
Chen S, Swartz KB, Terrace HS (1997) Knowledge of the ordinal position of list items in rhesus monkeys. Psychol Sci 8:80–86
Conway CM, Christiansen MH (2001) Sequential learning in non-human primates. Trends Cognit Sci 5:539–546
D’Amato MR, Colombo M (1988) Representation of serial order in monkeys (Cebus apella). J Exp Psychol Anim Behav Proc 14:131–139
Fisher AV, Sloutsky VM (2005) When induction meets memory: evidence for a gradual transition from similarity-based to category-based induction. Child Dev 76:583–597
Hayes BK, Heit E (2004) Why learning and development can lead to poorer recognition memory. Trends Cognit Sci 8:337–339
Inoue S, Matsuzawa T (2007) Working memory of numerals in chimpanzees. Curr Biol 17:1004–1005
Kawai N, Matsuzawa T (2000) Numeric memory span in a chimpanzee. Nature 403:39–40
Matsuzawa T (1985) Use of numbers by a chimpanzee. Nature 315:57–59
Matsuzawa T (2003) The Ai project: historical and ecological contexts. Anim Cogn 6:199–211
Matsuzawa T (2009a) Chimpanzee mind: looking for the evolutionary roots of the human mind. Anim Cogn. doi:10.1007/s10071-009-0277-1
Matsuzawa T (2009b) Symbolic representation of number in chimpanzees. Curr Opin Neurobiol 19:92–98
Matsuzawa T, Tomonaga M, Tanaka M (eds) (2006) Cognitive development in chimpanzees. Springer, Tokyo
Merritt D, MacLean EL, Jaffe S, Brannon EM (2007) A comparative analysis of serial ordering in ring-tailed lemurs (Lemur catta). J Comp Psychol 121:363–371
Murofushi K (1997) Numerical matching behavior by a chimpanzee (Pan troglodytes): subitizing and analogue magnitude estimation. Jpn Psychol Res 39:140–153
Ohshiba N (1997) Memorization of serial items by Japanese monkeys, a chimpanzee, and humans. Jpn Psychol Res 39:236–252
Sloutsky VM, Fisher AV (2004) When development and learning decrease memory: evidence against category-based induction in children. Psychol Sci 15:553–558
Swartz KB, Chen S, Terrace HS (1991) Serial learning by rhesus monkeys: I. Acquisition and retention of multiple four-item lists. J Exp Psychol Anim Behav Proc 17:396–410
Swartz KB, Chen S, Terrace HS (2000) Serial learning by rhesus monkeys: II. Learning four-item lists by trial and error. J Exp Psychol Anim Behav Proc 26:274–285
Terrace HS (1993) The phylogeny and ontogeny of serial memory: List learning by pigeons and monkeys. Psychol Sci 4:162–169
Terrace HS (2005) The simultaneous chain: a new approach to serial learning. Trends Cognit Sci 9:202–210
Terrace HS, Son LK, Brannon EM (2003) Serial expertise of rhesus macaques. Psychol Sci 14:66–73
Tomonaga M (2008) Relative numerosity discrimination by chimpanzees (Pan troglodytes): evidence for approximate numerical representations. Anim Cogn 11:43–57
Tomonaga M, Matsuzawa T (2000) Sequential responding to arabic numerals with wild cards by the chimpanzee (Pan troglodytes). Anim Cogn 3:1–11
Tomonaga M, Matsuzawa T (2002) Enumeration of briefly presented items by the chimpanzee (Pan troglodytes) and humans (Homo sapiens). Anim Learn Behav 30:143–157
Treichler FR, Raghanti MA (2009) Serial list combination by monkeys (Macaca mulatta): test cues and linking. Anim Cogn. doi:10.1007/s10071-009-0251-y
Treichler FR, Van Tilburg D (1999) Training requirements and retention characteristics of serial list organization by macaque monkeys. Anim Cogn 2:235–244
Treichler FR, Van Tilburg D (2002) Premise-pair training for valid tests of serial list organization in macaques. Anim Cogn 5:97–105
Treichler FR, Raghanti MA, Van Tilburg DN (2003) Linking of serially ordered lists by macaque monkeys (Macaca mulatta): list position influences. J Exp Psychol Anim Behav Proc 29:211–221
Washburn DA, Rumbaugh DM (1991) Ordinal judgments of numerical symbols by macaques (Macaca mulatta). Psychol Sci 2:190–193
Acknowledgments
This study was supported by grants MEXT16002001 and 20002001, JSPS-GCOE (A06, Biodiversity), and JSPS-HOPE to TM. This is a part of PhD thesis of SI. Thanks are due to Sumiharu Nagumo for his help with computer programming, Masaki Tomonaga, Masayuki Tanaka, Misato Hayashi, Dora Biro, Etsuko Nogami and Tomoko Takashima for their support, Armand Jacobs for his advice on statistical analyses, Christopher Martin for his English revision, and Alexander D. Hernandez for his English revision and great advice and help to the manuscript. Thanks are also due to the veterinary staffs and caregivers of the KUPRI chimpanzees.
Author information
Authors and Affiliations
Corresponding author
Additional information
This contribution is part of the Supplement Issue “The Chimpanzee Mind” (Matsuzawa 2009).
Rights and permissions
About this article
Cite this article
Inoue, S., Matsuzawa, T. Acquisition and memory of sequence order in young and adult chimpanzees (Pan troglodytes). Anim Cogn 12 (Suppl 1), 59–69 (2009). https://doi.org/10.1007/s10071-009-0274-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10071-009-0274-4