Abstract
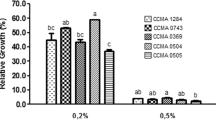
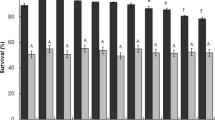
The aim of this study was to evaluate the probiotic nature, antimutagenicity, and mutagen-binding ability of the bacterial strain JG22 isolated from pepper-leaf jangajji. The strain, which was identified as Lactobacillus paracasei subsp. tolerans JG22, retained a high survival rate when exposed to pepsin-containing gastric juice and intestinal fluid supplemented with oxgall. This strain adhered to Caco-2 cells and exhibited resistance to ciprofloxacin, rifampicin, erythromycin, and cephalothin. Furthermore, against the 3 mutagens tested, live cells of strain JG22 displayed the greatest antimutagenicity against 2-nitroflourene (2-NF), with 51.37% inhibition, but exhibited relatively low antimutagenicity (11.07%) against 4-nitroquinoline-1-oxide (4-NQO). The antimutagenic effect of this strain was somewhat reduced after heat treatment for 30 min at 80°C. The most efficient mutagen-binding was observed with live cells of strain JG22, which bound 69.5% of the 2-NF. Thus, the putative probiotic strain JG22 could play a vital role in reducing the risk of cancer by absorbing mutagens and suppressing mutagenesis. Therefore, we consider the strain a good candidate for functional cultures and food system development.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Shuker DE. The enemy at the gates? DNA adducts as biomarkers of exposure to exogenous and endogenous genotoxic agents. Toxicol. Lett. 134: 51–56 (2002)
Burns AJ, Rowland IR. Antigenotoxicity of probiotics and prebiotics on faecal water-induced DNA damage in human colon adenocarcinoma cells. Mutat. Res. 551: 233–243 (2004)
Pool-Zobel BL, Neudeker C, Domizlaff I, Ji S, Schillinger U, Rumney C, Moretti M, Villarini M, Scassellati-Sforzolini G, Rowland I. Lactobacillus- and Bifidobacterium-mediated antigenotoxicity in the colon of rats. Nutr. Cancer 26: 365–380 (1996)
Vorobieva LI, Khodjaev EY, Cherdinceva TA. The study of induced antimutagenesis of propionic acid bacteria. J. Microbiol. Methods 24: 249–258 (1996)
Park KY, Kim SH, Son TJ. Antimutagenic activities of cell wall and cytosol fractions of lactic acid bacteria isolated from kimchi. J. Food Sci. Nutr. 3: 329–333 (1998)
Lankaputhra WEV, Shah NP. Antimutagenic properties of probiotic bacteria and of organic acids. Mutat. Res. 39: 169–182 (1998)
Waters MD, Stack HF, Jackson MA, Brockman HE, de Flora S. Activity profiles of antimutagens: In vitro and in vivo data. Mutat. Res. 350: 109–129 (1996)
Hosono A. Functional of lactic acid bacteria for the health. Bull. Dairy Tech. Assoc. 50: 92–116 (2000)
Saarela M, Mogensen G, Fonden R, Matto J, Mattila-Sandholm T. Probitoic bacteria: Safety, functional and technological properties. J. Biotechnol. 84: 197–215 (2000)
Maragkoudakis PA, Zoumpopoulou G, Christos M, Kalantzopoulos G, Pot B, Tsakalidou E. Probiotic potential of Lactobacillus strains isolated from dairy products. Int. Dairy J. 16: 189–199 (2006)
Tanaka H, Hashiba H, Kok J, Mierau I. Bile salt hydrolase of Bifidobacterium longum-biochemical and genetic characterization. Appl. Environ. Microbiol. 66: 2502–2512 (2000)
Tuomola EM, Salminen SJ. Adhesion of some probiotic and diary Lactobacillus strains to Caco-2 cell cultures. Int. J. Food Microbiol. 41: 45–51 (1998)
Maron DM, Ames BN. Revised methods for the Salmonella mutagenicity test. Mutat. Res. 113: 173–215 (1983)
Srinivasan P, Sabitha KE, Shyamaladevi CS. Attenuation of 4-nitroquinoline 1-oxide induced in vitro lipid peroxidation by green tea polyphenols. Life Sci. 80: 1080–1086 (2007)
Bourdichon F, Casaregola S, Farrokh C, Frisvad JC, Gerds ML, Hammes WP, Harnett J, Huys G, Laulund S, Ouwehand A, Powell IB, Prajapati JB, Seto Y, Schure ET, Boven AV, Vankerckhoven V, Zgoda A, Tuijtelaars S, Hansen EB. Food fermentations: Microorganisms with technological beneficial use. Int. J. Food Microbiol. 154: 87–97 (2012)
Peres CM, Peres C, Hernandez-Mendoza A, Malcata FX. Review on fermented plant materials as carriers and sources of potentially probiotic lactic acid bacteria-With an emphasis on table olives. Trends Food Sci. 26: 31–42 (2012)
Lim SM. Bile salts degreadation and cholesterol assimilation ability of Pediococcus pentosaceus MLK67 isolated from mustard leaf kimchi. Korean J. Microbiol. 47: 231–240 (2011)
Lim SM, Ahn DH. Factors affecting adhesion of lactic acid bacteria to Caco-2 cells and inhibitory effect on infection of Salmonella Typhimurium. J. Microbiol. Biotechnol. 22: 1731–1739 (2012)
Merrell DS, Camilli A. Acid tolerance of gastrointestinal pathogens. Curr. Opin. Microbiol. 5: 51–55 (2002)
Cotter PD, Hill C. Surviving the acid test: Responses of Gram-positive bacteria to low pH. Microbiol. Mol. Biol. Rev. 67: 429–453 (2003)
De Smet I, Van Hoorde L, Vande Woestyne M, Cristiances H, Verstraete W. Significance of bile salt hydrolytic activities of lactobacilli. J. Appl. Bacteriol. 79: 292–301 (1995)
Burns P, Vinderola G, Binetti A, Quiberoni A, de Los Reyes-Gavilan CG, Reinheimer J. Bile-resistant derivatives obtained from non-intestinal dairy lactobacilli. Int. Dairy J. 18: 377–385 (2008)
Corzo G, Gilliland SE. Measurement of bile salt hydrolase activity from Lactobacillus acidophilus based on disappearance of conjugated of bile salts. J. Dairy Sci. 82: 466–471 (1999)
Moser SA, Savage DC. Bile salt hydrolase activity and resistance to toxicity of conjugated bile salts are unrelated properties in Lactobacilli. Appl. Environ. Microbiol. 67: 3476–3480 (2001)
Vidhyasagar V, Jeevaratnam K. Evaluation of Pediococcus pentosaceus strains isolated from idly batter for probiotic properties in vitro. J. Funct. Foods 5: 235–243 (2013)
Granato D, Perotti F, Masserey I, Rouvet M, Golliard M, Servin A, Brassart D. Cell surface-associated lipoteichoic acid acts as an adhesion factor for attachment of Lactobacillus johnsonii La1 to human enterocyte-like Caco-2 cells. Appl. Environ. Microbiol. 65: 1071–1077 (1999)
Kirtzalidou E, Pramateftaki P, Kotsou M, Kyriacou A. Screening for Lactobacilli with probiotic properties in the infant gut microbiota. Clin. Microbiol. 17: 440–433 (2011)
Wright, GD. Bacterial resistance to antibiotics: Enzymatic degradation and modification. Adv. Drug Delivery Rev. 57: 1451–1470 (2005)
Nadathur SR, Gould SJ, Baklynski AT. Antimutagenicity of fermented milk. J. Dairy Sci. 77: 3287–3295 (1994)
Cassand P, Abdelli H, Bouley C, Denariaz G, Narbonne JF. Inhibitory effect of dairy products on the mutagenicities of chemicals and dietary mutagens. J. Dairy Res. 61: 545–552 (1994)
Zhang XB, Ohta Y. Binding of mutagens by fractions of the cell wall skeleton of lactic acid bacteria on mutagens. J. Dairy Sci. 74: 1477–1481 (1991)
Sreekumar O, Hosono A. The antimutagenic properties of a polysaccharide produced by Bifidobacterium longum and its cultured milk against some heterocyclic amines. Can. J. Microbiol. 44: 1029–1036 (1998)
Sreekumar O, Hosono A. Antimutagenicity and influence of physical factors in binding Lactobacillus gasseri and Bifidobacterium longum cells to amino acid pyrolysates. J. Dairy Sci. 81: 1508–1516 (1998)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lim, SM. Antimutagenicity activity of the putative probiotic strain Lactobacillus paracasei subsp. tolerans JG22 isolated from pepper leaves Jangajji. Food Sci Biotechnol 23, 141–150 (2014). https://doi.org/10.1007/s10068-014-0019-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-014-0019-2