Abstract
The objective of this study is to analyze clinical manifestations, features of imaging, and laboratory assessment of patients with neuropsychiatric SLE (NPSLE) for better diagnosis and outcome prediction. One hundred eighteen NPSLE patients admitted to the Anhui Provincial Hospital in Hefei, China, between January 2006 and December 2016 were enrolled and analyzed retrospectively. All patients fulfilled the American College of Rheumatology revised classification criteria for SLE. Patients with NPSLE fulfilled the American College of Rheumatology (ACR) nomenclature and case definitions. All NPSLE patients underwent neurological investigations including MRI of nervous system, electroencephalograms, or CSF examination as part of the diagnostic evaluation of nervous system involvement. All statistical analyses were performed. According to different types of data, different statistical methods were used to determine factors associated with abnormal MRI among NPSLE patients. Statistical significance was defined as P value < 0.05(two-tailed). Twelve different neurological manifestations of NPSLE patients were shown, in which headache was most common symptom (25.95%, 34/131), followed by seizures (25.19%, 33/131), cerebrovascular disease (18.32%, 24/131), psychosis (8.40%, 11/131), and others including mood disorder, cognitive dysfunction, plexopathy, cranial neuropathy, movement disorder, myelopathy, acute confusional state, and anxiety disorder. Thirteen patients have two neurological symptoms at the same time. Cerebrospinal fluid was assessed in 76 NPSLE patients, in which 29 patients had higher pressure of cerebrospinal fluid and 66 patients had abnormal immunoglobulin in cerebrospinal fluid, predominantly with an increase of IgG (84.21%, 64/76), followed by an increase of IgA (69.74%, 53/76), and IgM accounted for 47.74% (34/76). The MRI taken by 66.10% (78/118) patients have shown abnormal lesions and/or ischemic changes in the bilateral cerebral hemisphere, thalamus, pons, brainstem, and cerebellum. The abnormal changes in MRI were correlated with antiphospholipid antibody (APL) and C3 (P = 0.026 and 0.040, respectively). The most common clinical manifestation of NPSLE is headache, followed by seizures and cerebrovascular accident. The test of cerebrospinal fluid and MRI plays an important role in the assessment of NPSLE. The abnormal intracranial lesions were correlated with the level of anti-cardiolipin antibodies (ACL) and C3.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Systemic lupus erythematosus (SLE) is a relapsing-remitting female-predominant chronic inflammatory disease characterized by the production of a variety of autoantibodies, involving multiple organs [1,2,3]. Neuropsychiatric manifestation in systemic lupus erythematosus (NPSLE) is one of the most serious complications of the disease. It occurs in 37–95% of SLE patients, which is the major cause of morbidity and mortality in patients with SLE [4, 5]. Because of the diversity of clinical manifestations including headaches, mood disorders, seizures, and psychosis ranging from mild to serious, the early diagnosis of NPSLE is still a challenge for clinicians. The reasons for this variability are attributable to the low specificity of the American College of Rheumatology (ACR) case definitions for SLE-attributed neuropsychiatric (NP) syndromes [6] and its complex interrelated pathogenetic mechanisms, including genetic factors, vasculopathy, neuroendocrine-immune imbalance, tissue and neuronal damage mediated by autoantibodies, inflammatory mediators, and blood–brain barrier dysfunction [7].
The diagnosis of NPSLE is based on a global assessment (such as patient’s illness history, clinical manifestations, imaging, and serum indexes) and judicious exclusion of a wide range of mimics as systemic and infectious conditions [7]. Various antibodies are used for the diagnosis of SLE, including anti-double-stranded DNA (anti-dsDNA), anti-smith antibody (anti-Sm), anti-ribonucleoprotein (anti-RNP), and ribosomal P protein antibodies (anti-P) [8]. But the pathogenic role of autoantibodies and their association with NPSLE is still a matter of debate. Among all autoantibodies, anti-P seems to be closely associated with NPSLE [9,10,11,12]. An association between depression and anti-P antibodies has been reported in SLE patients [13]. Animal studies show that circulating human anti-P autoantibodies impair flexible memory task performance in mice when the blood–brain barrier is disrupted, and it might contribute to the cognitive impairment that is frequently observed in SLE [14]. But a meta-analysis of Karassa et al. found that anti-P antibody testing has insignificant diagnostic utility for NPSLE overall and in particular for SLE neuropsychiatric presentations [15]. Moreover, with the popularity of magnetic resonance imaging (MRI), it has emerged as a promising technique to study NPSLE, as T2-weighted and fluid-attenuated inversion recovery (FLAIR) scans have detected focal hyperintense lesions, which are present in deep, periventricular and subcortical white matter in up to 70% of NPSLE patients [16].
In this study, we retrospectively analyzed the data of NPSLE patients in our center, aiming to analyze the clinical features of NPSLE.
Materials and methods
Patients
One hundred eighteen newly onset NPSLE patients admitted to the Anhui Provincial Hospital in Hefei, China, between January 2006 and December 2016 were enrolled and analyzed retrospectively. All patients fulfilled the American College of Rheumatology revised classification criteria for SLE [17]. Patients with NPSLE fulfilled the ACR nomenclature and case definitions published in 1999 [18], excluding the comorbid conditions and concomitant factors of offending drugs, central nervous system infection, primary neurological conditions (primary neurological conditions (cerebral hemorrhage, cerebral infarction, disseminated sclerosis, etc.)), tumor, and known metabolic derangements, such as hypoglycemia, ketoacidosis, hypoxemia, uremia, or electrolyte imbalance. All NPSLE patients underwent neurological investigations including MRI of nervous system, electroencephalograms, or CSF examination as part of the diagnostic evaluation of nervous system involvement. The identification and classification of NPSLE were confirmed by at least two neurologists or psychiatrists independently. We analyzed the data of demographic information, clinical presentations, laboratory findings, and imaging during hospitalization.
Laboratory test and MRI acquisition
The laboratory data included a standard hematological and immunological profile: white blood count, antinuclear antibodies (ANA), double-stranded (ds) DNA antibodies, anti-Smith (Sm), anti-ribonucleoprotein (RNP) antibodies, anti-ribosomal phosphoprotein (anti-P), antiphospholipid antibody (APL), serum complement levels, serum immune globulin, and erythrocyte sedimentation rate (ESR). These antibodies were screened by standard enzyme-linked immunosorbent assay (ELISA), and serum complement levels were detected by immunoturbidimetry.
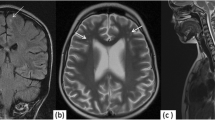
All MRI scans were performed by 3.0 Tesla MR systems (Siemens Medical System, Munich, Germany) and included T1-weighted, T2-weighted, FLAIR images, and diffusion-weighted images (DWI). In some patients, gadolinium enhancement was performed on T1-weighted or FLAIR images. All the MRI scans were evaluated by expert neuroradiologists blinded to the clinical and immunological data. Several types of MRI findings were defined: inflammatory-like lesions, brain infarcts, atrophy, demyelination, and bleeds. Inflammatory-like lesions were defined as T2/FLAIR hyperintense lesions involving gray or white matter, generally medium large, ill-defined, without vascular territory distribution, with possible mass effect.
Statistical analysis
Results with continuous data were presented as mean ± standard deviation (SD). Categorical data were presented as the absolute count and percentage. For comparison of categorical data, chi-square test with continuity correction was applied. Student’s t test was used for comparison of continuous data with a normal distribution; otherwise, Mann–Whitney U test was applied. Risk factors related to NPSLE were analyzed by using univariate analysis and multiple logistic regressions. Pearson correlation analysis was used in the correlation of continuous variables. All statistical analyses were performed by using SPSS 19.0 (IBM® SPSS® Statistics). Statistical significance was defined as P value < 0.05 (two-tailed).
Results
Demographic characteristics
This study consisted of 118 patients with newly onset NPSLE (Table 1). There were 109 females (92.37%) and 9 males (7.63%). The mean age of the patients was 33.77 ± 12.68 years, ranging from 14 to 69. The duration of the disease was 41.3 ± 65.3 months, ranging from 7 days to 20 years. In all patients, 12 patients suffer from hypertension, 3 patients suffer from diabetes mellitus, and 1 patient suffers from hypothyroidism.
Neuropsychiatric characteristics
A total of 131 NP events occurred in 118 patients. Twelve of the 19 ACR syndromes were identified in our study. One hundred five patients (88.98%) presented with one set of NP symptoms, while 13 (11.02%) had two kinds of mental symptoms. The most frequent manifestation was headache, followed by seizure disorder, cerebrovascular disease, and psychosis (Table 2).
Cerebrospinal fluid characteristics
Cerebrospinal fluid was assessed in 76 patients with NPSLE, of which 30 (39.47%) patients had higher cerebrospinal fluid pressure and 66 (86.84%) patients had elevated cerebrospinal fluid immunoglobulin (Ig) (Table 3). IgG rise mainly accounted for 84.21% (64/76), followed by IgA (69.74%, 53/76) and IgM (47.74%, 34/76). There was no relevance of immunoglobulin level between serum and cerebrospinal fluid. We also did not find any association between cerebrospinal fluid abnormalities and manifestations of NPSLE.
Imaging characteristics
Magnetic resonance imaging (MRI) of the nervous system was the most widely used in NPSLE patients, with 78 (66.10%) patients showing abnormal findings. The lesions involved the bilateral cerebral hemisphere, thalamus, pons, brainstem, and cerebellum and characterized by inflammatory, infarct, demyelination, or atrophy change. In the MRI findings, periventricular hyperintensity (34/78, 43.59%) was the most frequent MRI abnormality, followed by cortical lesions (26/78, 33.33%) and basal ganglia lesions (19/78, 24.36%), while 16 patients had more than one MRI lesion (Table 4).
Imaging and laboratory tests
The hematological and immunological abnormalities are shown in Table 5. All patients (100%) had positive ANA, 50 (42.37%) patients had elevated anti-dsDNA, 34 (28.81%) patients had elevated anti-Sm, and 28 (23.72%) patients had elevated anti-P. We compared the clinical manifestation and hematological and immunological indexes of NPSLE patients with MRI findings (ruled out the 4 patients whose MRI abnormality may be not directly related to SLE). The frequency of ANA, anti-dsDNA, and anti-Sm was compared between MRI normal and abnormal groups. It also happened in anti-ribosomal phosphoprotein, Anua (anti-nucleusome antibodies), anti-C1q, and anti-RNP. However, we found statistical significance between two group patients in APL, mucocutaneous involvement, and duration (P = 0.026, 0.036, 0.030, respectively). In hematological, the complement 3 (C3) of the two groups showed significance statistically (P = 0.040). We did not find a difference of other parameters such as white blood cells (WBC), ESR, albumin (ALB), immune globulin (Ig), and complement 4 (C4) between the two groups. In the subsequent multivariate logistic regression analyses of laboratory tests, APL (P = 0.045) and C3 (P = 0.042) showed statistically significant differences between MRI abnormal and normal groups (Table 6).
Discussion
At present, some researchers believe that the inflammation in SLE tends to reduce along with the extension of the course in SLE, as patients with a course of less than 2 years had a higher morbidity of NPSLE [19], but other had different views. Toledano P found that the occurrence of NPSLE was not correlated with the course of SLE [20]. Kohut SA examined the prevalence of depressive symptoms in young adults with NPSLE and found no correlation with age [21]. In our study, we could find that there was no obvious regularity of NP symptom onset, supporting the idea that NPSLE may happen at any time of the disease [22, 23], which is consistent with Toledano’s findings.
Just as in previous studies [24, 25], the most common neurological symptom in NPSLE patients was headache. Hilda Fragoso-Loyo et al. [26] reported that higher levels of inflammatory molecules were found in patients with headache from intracranial hypertension and intractable non-specific headache than in patients with migraine. Also, all the elevated molecule levels had decreased significantly when the headache had resolved. It suggested that headache, but not migraine, shares the intracranial inflammatory profile in CSF observed in other NPSLE syndromes. There was a high prevalence of seizures in our series compared with studies of other ethnicity [4, 27]. But similar result was found in the report of Mok et al. [28], whose patients were also Chinese (28%). Besides the heterogeneity of NP events, whether Chinese NPSLE patients are more likely to have seizure needs further study.
MRI, as a specialized and sensitive investigation in NPSLE, was normally used in the recognition of structural abnormalities of the nervous system. Although MRI is helping in precise diagnosis, its relevance to the present NP manifestations is still limited. Essam A. Abda et al. [29] found that NPSLE patients with MRI abnormalities were older and had longer duration of illness. Furthermore, other studies have found a correlation between accumulated SLE damage and the time of evolution of the disease with the presence of white matter lesions [30, 31]. However, in Pilar Toledano et al.’s study [20], no similar results were founded, but their data suggest that the presence of certain abnormalities, such as SLEDAI and low complement levels, seems to correlate with the presence of an inflammatory pattern on MRI. In this study, we investigated the relationships between clinical, immunological features and MRI abnormalities in patients with NPSLE. The most frequent MRI abnormality was white matter involvement; the most common site was found in periventricular white matter. The role of white matter involvement in NPSLE was not clear. In contrast to some research, which found an association between periventricular hyperintensity and cognitive impairment [32], cerebrovascular disease [33], or other NP manifestations, we have found an association of APL between normal MRI and abnormal MRI patients. It suggests that APL plays an important role in intracranial inflammation and ischemic changes. Clinically, the presence of APL, which target anionic phospholipids and protein-phospholipid complexes, has been associated with a variety of NP syndromes such as stroke, transient ischemic attacks, chorea, dementia, and transverse myelitis, while lupus anticoagulant was associated with cognitive disorders [4, 34, 35]. These APL antibodies have been shown to upregulate soluble adhesion, leading to an enhanced inflammatory response and subsequent thromboembolic events [36]. Other authors also found an association between white matter hyperintensities and antiphospholipid antibodies [32]. In our study, we also found that low C3 levels were associated with the presence of MRI lesions, but there was no association with age and disease duration, which is similar to Pilar Toledano’s study. The associations between NPSLE and C3 may be explained by disease activity. The role of complement activation and complement components in lupus needs further research.
Previous study has shown that CSF anti-U1RNP antibody positivity is associated with increased level of CSF IFN-α and MCP-1 levels [37,38,39,40]. It seems that the autoantibody was associated with inflammation levels. In our study, most of the patients showed a high level of CSF immunoglobulin reflecting an occurrence of intracranial immune response. Immunoglobulin in CSF was examined in 76 patients, of which 66 cases showed elevated levels, while only 43 patients showed magnetic resonance anomalies in brain MRI, suggesting that the detection of inflammation by assessment of immunoglobulin in CSF is sensitive than the detection by brain MRI of structural changes which usually occur in later stage with cerebral vascular occlusion and brain cell necrosis.
In summary, we found that the most common clinical manifestation of NPSLE is headache, followed by epilepsy and cerebrovascular accident. APL and low C3 level are associated with abnormal MRI signal in NPSLE, while CSF and abnormal MRI are common in NPSLE.
References
Ruiz-Irastorza G, Khamashta MA, Castellino G et al (2001) Systemic lupus erythematosus. Lancet 357:1027–1032
Lauvsnes MB, Omdal R (2012) Systemic lupus erythematosus, the brain, and anti-NR2 antibodies. J Neurol 259(4):622–629
Jeltsch-David H, Muller S (2014) Neuropsychiatric systemic lupus erythematosus: pathogenesis and biomarkers. Nat Rev Neurol 10(10):579–596
Sanna G, Bertolaccini ML, Cuadrado MJ et al (2003) Neuropsychiatric manifestations in systemic lupus erythematosus: prevalence and association with antiphospholipid antibodies. J Rheumatol 30:985–992
Sibbitt WL Jr, Brandt JR, Johnson CR et al (2002) The incidence and prevalence of neuropsychiatric syndromes in pediatric onset systemic lupus erythematosus. J Rheumatol 29:1536–1542
Tay SH (2016) Diagnosing and attributing neuropsychiatric events to systemic lupus erythematosus: time to untie the Gordian knot? Rheumatology (Oxford). https://doi.org/10.1093/rheumatology/kew338
Govoni M, Bortoluzzi A, Padovan M et al (2016) The diagnosis and clinical management of the neuropsychiatric manifestations of lupus. J Autoimmun 74:41–72
Schur PH (2009) Know your labs: a review of state-of-the-art testing for SLE and connective tissue disease. The Rheumatologist. February
C armona-Fernandes D, Santos MJ, Canh Hão H, Fonseca JE (2013) Anti-ribosomal P protein IgG autoantibodies in patients with systemic lupus erythematosus: diagnostic performance and clinical profile. BMC Med 11:98
Gerli R, Caponi L, Tincani A, Scorza R, Sabbadini MG, Danieli MG, De Angelis V, Cesarotti M, Piccirilli M, Quartesan R, Moretti P, Cantoni C, Franceschini F, Cavazzana I, Origgi L, Vanoli M, Bozzolo E, Ferrario L, Padovani A, Gambini O, Vanzulli L, Croce D, Bombardieri S (2002) Clinical and serological associations of ribosomal P autoantibodies in systemic lupus erythematosus: prospective evaluation in a large cohort of Italian patients. Rheumatology (Oxford) 41(12):1357–1366
Heinlen LL, Ritterhouse LL, McClain MT, Keith MP, Neas BR, Harley JB, James JA (2010) Ribosomal P autoantibodies are present before SLE onset and are directed against non-C-terminal peptides. J Mol Med 88(7):719–727
Koscec M, Koren E, Wolfson-Reichlin M, Fugate RD, Trieu E, Targoff IN, Reichlin M (1997) Autoantibodies to ribosomal P proteins penetrate into live hepatocytes and cause cellular dysfunction in culture. J Immunol 159(4):2033
Eber T, Chapman J, Shoenfeld Y (2005) Anti-ribosomal P-protein and its role in psychiatric manifestations of systemic lupus erythematosus: myth or reality? Lupus 14:571–575
Bravo-Zehnder M, Toledo EM, Segovia-Miranda F, Serrano FG, Benito MJ, Metz C, Retamal C, Álvarez A, Massardo L, Inestrosa NC, González A (2015) Anti-ribosomal P protein autoantibodies from patients with neuropsychiatric lupus impair memory in mice. Arthritis Rheumatol 67(1):204–214. https://doi.org/10.1002/art.38900
Karassa FB, Afeltra A, Ambrozic A et al (2006) Accuracy of anti-ribosomal P protein antibody testing for the diagnosis of neuropsychiatric systemic lupus erythematosus: an international meta-analysis. Arthritis Rheum 54:312
Luyendijk J, Steens SC, Ouwendijk WJ, Steup-Beekman GM, Bollen EL, van der Grond J, Huizinga TW, Emmer BJ, van Buchem MA (2011) Neuropsychiatric systemic lupus erythematosus: lessons learned from magnetic resonance imaging. Arthritis Rheum 63(3):722–732
Hochberg MC (1997) Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 40(9):1725
The American College of Rheumatology nomenclature and case definitions for neuropsychiatric lupus syndromes (1999) Arthritis Rheum; 42:599–608
Cervera R, Khamashta MA, Font J et al (2003) Morbidity and mortality in systemic lupus erythematosus during a 10-year period: a comparison of early and late manifestations in a cohort of 1,000 patients. Medicine 82(5):299–308
Toledano P, Sarbu N, Espinosa G, Bargalló N, Cervera R (2013) Neuropsychiatric systemic lupus erythematosus: magnetic resonance imaging findings and correlation with clinical and immunological features. Autoimmun Rev 12(12):1166–1170
Kohut SA, Williams TS, Jayanthikumar J (2013) Depressive symptoms are prevalent in childhood-onset systemic lupus erythematosus (cSLE). Lupus 22(7):712–720
Csepany T, Bereczki D, Kollar J, Sikula J, Kiss E, Csiba L (2003) MRI findings in central nervous system systemic lupus erythematosus are associated with immunoserological parameters and hypertension. J Neurol 250:1348–1354
Sibbitt WL Jr, Sibbitt RR, Brooks WM (1999) Neuroimaging in neuropsychiatric systemic lupus erythematosus. Arthritis Rheum 42:2026–2038
Sciascia S, Bertolaccini ML, Baldovino S, Roccatello D, Khamashta MA, Sanna G (2013) Central nervous system involvement in systemic lupus erythematosus: overview on classification criteria. Autoimmun Rev 12:426–429
Tjensvoll AB, Harboe E, Goransson LG, Beyer MK, Greve OJ, Herigstad A et al (2013) Migraine is frequent in patients with systemic lupus erythematosus: a case-control study. Cephalalgia 31:401–408
Fragoso-Loyo H, Atisha-Fregoso Y, Llorente L, Sánchez-Guerrero J (2013) Inflammatory profile in cerebrospinal fluid of patients with headache as a manifestation of neuropsychiatric systemic lupus erythematosus. Rheumatology (Oxford) 52(12):2218–2222
Hanly JG, McCurdy G, Fougere L, Douglas J, Thompson K (2005) Neuropsychiatric events in systemic lupus erythematosus: attribution and clinical significance. J Rheumatol 32:1459–1456
Mok CC, Lau CS, Wong RWS (2001) Neuropsychiatric manifestations and their clinical associations in southern Chinese patients with systemic lupus erythematosus. J Rheumatol 28:766–771
Abda EA, Selim ZI, Radwan ME et al (2013) Markers of acute neuropsychiatric systemic lupus erythematosus: a multidisciplinary evaluation. Rheumatol Int 33(5):1243–1253
Ainiala H, Dastidar P, Loukkola J, Lehtimaki T, Korpela M, Peltola J et al (2005) Cerebral MRI abnormalities and their association with neuropsychiatric manifestations in SLE: a population-based study. Scand J Rheumatol 34:376–382
Appenzeller S, Bonilha L, Rio PA, Min Li L, Costallat LT, Cendes F (2007) Longitudinal analysis of gray and white matter loss in patients with systemic lupus erythematosus. NeuroImage 34:694–701
Appenzeller S, Vasconcelos Faria A, Li LM, Costallat LTL, Cendes F (2008) Quantitative magnetic resonance imaging analyses and clinical significance of hyperintense white matter lesions in systemic lupus erythematosus patients. Ann Neurol 64:635–643
Ainiala H, Dastidar P, Loukkola J, Lehtimäki T, Korpela M, Peltola J et al (2005) Cerebral MRI abnormalities and their association with neuropsychiatric manifestations in SLE: a population-based study. Scand J Rheumatol 34:376–382
Mikdashi J, Handwerger B (2004) Predictors of neuropsychiatric damage in systemic lupus erythematosus: data from the Maryland lupus cohort. Rheumatology (Oxford) 43:1555–1560
Karassa FB, Ioannidis JP, Touloumi G, Boki KA, Moutsopoulos HM (2000) Risk factors for central nervous system involvement in systemic lupus erythematosus. QJM 93:169–174
Zaccagni H, Fried J, Cornell J, Padilla P, Brey RL (2004) Soluble adhesion molecule levels, neuropsychiatric lupus and lupusrelated damage. Front Biosci 9:1654–1659
Yokoyama T, Fujii T, Kondo-Ishikawa S et al (2014) Association between anti-U1 ribonucleoprotein antibodies and inflammatory mediators in cerebrospinal fluid of patients with neuropsychiatric systemic lupus erythematosus. Lupus 23(7):635–642
Shiozawa S, Kuroki Y, Kim M, Hirohata S, Ogino T (1992) Interferonalpha in lupus psychosis. Arthritis Rheum 35:417–422
Fragoso-Loyo H, Richaud-Patin Y, Orozco-Narvaez A et al (2007) Interleukin-6 and chemokines in the neuropsychiatric manifestations of systemic lupus erythematosus. Arthritis Rheum 56:1242–1250
Iikuni N, Okamoto H, Yoshio T et al (2006) Raised monocyte chemotactic protein-1 (MCP-1)/CCL2 in cerebrospinal fluid of patients with neuropsychiatric lupus. Ann Rheum Dis 65:253–256
Financial support and sponsorship
This study was supported by the National Natural Science Foundation of China (No. 81373187).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None.
Rights and permissions
About this article
Cite this article
Tan, Z., Zhou, Y., Li, X. et al. Brain magnetic resonance imaging, cerebrospinal fluid, and autoantibody profile in 118 patients with neuropsychiatric lupus. Clin Rheumatol 37, 227–233 (2018). https://doi.org/10.1007/s10067-017-3891-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-017-3891-3