Abstract
The purpose of this study was to assess diffusion tensor imaging in the diagnosis of mild-moderate carpal tunnel syndrome (CTS) and to correlate fractional anisotropy (FA) and apparent diffusion coefficient (ADC) with nerve conduction study and clinical tests. This prospective study was conducted upon 39 patients (9 males, 30 females; mean age = 33 years) with early stage of CTS. Twenty age- and sex-matched healthy volunteers were included as a control group. All patients underwent clinical tests and nerve conduction studies with calculation of the distal motor latency (DML) and distal sensory latency (DSL). Patients with CTS and volunteers underwent diffusion tensor imaging of the median nerve. The cross-sectional area (CSA), FA, and ADC of the median nerve were calculated. There was significant difference in FA and ADC between patients and controls (P = 0.001 respectively) and between mild and moderate CTS (P = 0.001 respectively). The cutoff FA and ADC of the median nerve used for the diagnosis of CTS were 0.45 and 1.31 × 10−3 mm2/s with areas under the curve (AUC) of 0.985 and 0.954, respectively, and the values used to differentiate mild from moderate CTS were 0.42 and 1.35 × 10−3 mm2/s with AUC of 0.964 and 0.688, respectively. The FA and ADC of the median nerve were correlated with CSA (r = 0.894; −0.769), DML (r = −0.935; 0.781), DSL (r = −0.953; 0.781), Tinel’s test (r = 0.742; 0.684), and Phalen’s test (r = 0.862; 0.742). Diffusion tensor imaging can be used for the diagnosis of mild-moderate CTS and well correlated with nerve conduction studies and clinical tests.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Carpal tunnel syndrome (CTS) is caused by an entrapment of the median nerve at the level of the carpal tunnel. CTS is a common disorder with an estimated 6% annual incidence rate in adults in the general population. Early treatment is important to avoid permanent damage to the median nerve. Patients with mild disease treated with 6 weeks to 3 months of conservative treatment and patients with moderate disease are referred for surgical evaluation [1–4]. Diagnosis of CTS is usually established by characteristic clinical features, clinical tests, and nerve conduction studies. But there is overlap and false results with clinical tests and nerve conduction studies [5, 6]. Routine and advanced musculoskeletal ultrasound has been used for the diagnosis of different joint disorders and CTS, but their results are conflicting and operator dependent [6–8]. Ultrasound elastography and three-dimensional, dynamic ultrasound, and power duplex sonography are used for the assessment of CTS, but their results are preliminary and overlapping [9, 10]. Routine magnetic resonance imaging is used for the assessment of CTS, but its results are qualitative and variable [11].
Diffusion-weighted MR imaging and diffusion tensor imaging allow for the measurement of the tissue microstructure and reflect the random motion of water protons [12, 13]. Diffusion tensor imaging extracts and characterizes diffusion patterns to provide exquisite details of the tissue microstructure and fiber tracking. Diffusion tensor imaging allows for three-dimensional rendering of the peripheral nerves; hence, it has the potential to improve the diagnosis of peripheral nerve disorders, optimize lesion localization, and determine the extent of neural dysfunction [14–24]. The two most common measures of the diffusion tensor are trace and anisotropy. Fractional anisotropy (FA) and apparent diffusion coefficient (ADC) are widely used in diffusion tensor imaging to measure compactness of the tissues and intercellular space and provide estimates independent of fiber directionality. FA estimates the coherence of oriented structures such as myelinated nerve fibers. It is the extent to which water within a voxel diffuses preferentially along one axis rather than exhibiting isotropic diffusion. Diffusion tensor imaging is used for the imaging of peripheral nerves in different parts of the body. Few studies discuss the role of diffusion tensor imaging of CTS [14–28]. Wang et al. [14] is the leading author of previous studies that I refer in this paper as this study discusses meta-analysis of different studies using diffusion tensor imaging in the assessment of CTS. The rationale for considering diffusion tensor imaging for the diagnosis of CTS is that it is a more convenient, rapid, non-invasive, and non-operator-dependent technique [20–26].
The aim of this work is to assess diffusion tensor imaging in the diagnosis of mild-moderate CTS and to correlate FA and ADC with nerve conduction study and clinical tests.
Material and Methods
Patients
Institutional review board approval was obtained, and informed consents were obtained from the patients and controls. This prospective study was conducted upon 45 patients with clinically suspected idiopathic CTS. The inclusion criteria were adult patients with clinical diagnosis of CTS based on the guidelines of the American Academy of Neurology [29] and mild to moderate disease on the nerve conduction study. We excluded patients with underlying medical diseases as diabetes mellitus (n = 4) and renal failure (n = 2). The final patients in this study were 39 patients (9 male and 30 female, age range from 28 to 41 years with mean age of 33 years). Age- and sex-matched healthy controls were recruited from the hospital staff and volunteers. All patients and controls underwent diffusion tensor imaging of the median nerve, and a nerve conduction study was done only for patients. Patients presented with paresthesia within the median nerve distribution (n = 39), dull aching discomfort in the hand (n = 30), weakness in the hand muscle (n = 10), numbness within the median nerve distribution (n = 24).
Clinical tests
The patients underwent a full clinical examination, including Tinel’s and Phalen’s test. Phalen’s test was performed by fully flexing the patient’s wrist for 60 s. The test was positive if the patient’s symptoms in the median nerve distribution were reproduced [5]. Tinel’s sign was tested by tapping the median nerve along its course across the wrist. The test was positive if the patient experienced parenthesis in at least one of three radial digits [5]. Phalen’s sign was positive in 24 patients and Tinel’s sign was positive in 12 patients.
Nerve conduction study
Nerve conduction studies were performed by stander surface stimulating and recording techniques using a portable digital electromyography EBNeuro apparatus with four channels and an appropriate analysis program. Nerve conduction studies were carried for all patients according to the protocol proposed by the American Academy of Neurology [6]. The median and ulnar nerves were stimulated at the wrist and elbow at a distance of 8 cm from the wrist to the active electrode. The motor nerve distal latency (MNDL) and motor and sensory nerve conduction velocities (MNCV and SNCV) were calculated. The severity of CTS was graded as follows: grade 0, normal; grade 1 (very mild), CTS demonstrable only with most sensitive tests; grade 2 (mild), slow median DSL, normal terminal motor latency; grade 3 (moderate), sensory potential preserved with motor slowing, distal motor latency to abductor pollicis brevis (APB) <6.5 ms; grade 4 (severe), sensory potentials absent but motor response preserved, distal motor latency to APB <6.5 ms; grade 5 (very severe), terminal latency to APB >6.5 ms; and grade 6 (extremely severe), sensory and motor potentials effectively un-recordable (surface motor potential from APB <0.2 mV amplitude) [30]. The study included patients with mild and moderate CTS based on the above grading system.
MR imaging
MR imaging was done with a 1.5-T (Achieva, Philips, Best, Netherland) scanner, with a dedicated eight-channel wrist coil (Achieva Sense; Philips, Best, Netherlands). Patients were scanned in the prone arm-overhead position. T2-weighted images were acquired using a turbo spin-echo sequence with the following parameters: TR/TE = 4000/80 ms, turbo spin-echo factor = 2, field of view (FOV) = 15 cm × 15 cm, matrix = 272 × 264, slice thickness = 3 mm, slice gap = 0.3 mm, and scan time = 150 s. The diffusion tensor imaging scan was acquired using a diffusion-weighted single-shot echo-planar imaging pulse sequence with the following parameters: TR/TE = 7049/91 ms, b value = 1000 mm/s2, matrix size = 92 × 88, FOV = 15 cm × 15 cm, slice thickness = 2 mm, slice gap = 0, slices = 45, acquired voxel size = 1.1 mm × 1.1 mm × 2.0 mm, reconstructed voxel size = 1 mm × 1 mm × 2 mm, number of gradient directions = 15, acquisition of five signals, fat suppression = spectral pre-saturation with inversion recovery, echo-planar imaging factor = 47, sensitivity encoding factor = 2, and acquisition time = 240 s.
Image analysis
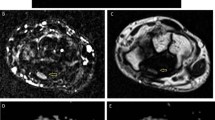
Image analysis was performed by only one radiologist with a 25-year experience in MR imaging, and he was blinded to the clinical tests and nerve conduction studies. The FiberTracking software package (Extended MR WorkSpace, version 2.5.3.0; Philips Medical Systems) was used for image analysis. A region of interest (ROI) was drawn manually at the median nerve at the level of hook of the hamate bone for calculation of FA and ADC (Fig. 1). The ROI was carefully plotted to avoid partial volume artifacts and to exclude vessels, tendons, or adjacent fat. The diffusion tensor tractography of the median nerve was generated, and the T1-weighted 3D fast-field echo image was used for anatomic reference. The cross-sectional area (CSA) of the carpal tunnel was calculated [26].
Statistical analysis
The statistical analysis of data was done by using Statistical Package for Social Sciences (SPSS, Inc., Chicago, IL, USA) software, version 21. The mean and standard deviation of the FA, ADC, CSA, DmL, and DSL of patients and volunteers as well as patients with mild and moderate CTS were calculated. All data revealed results of parametric test with normal distribution. The analysis of data was done to test the statistical significant difference. To compare between two groups, Student’s t test was used. To compare between more than two groups, one-way ANOVA test was used. A receiver-operating characteristic (ROC) curve was done to evaluate the diagnostic capability of FA and ADC for differentiation of CTS from volunteers as well as mild from moderate CTS. The area under the curve (AUC), sensitivity, specificity, and accuracy were calculated. Pearson’s correlation test was done to correlate the FA, ADC with CSA, DML, DSL, and Phalen’s and Tinel’s tests. The correlation coefficient r and P value were calculated. The P value was considered significant if ≤0.05 at a 95% confidence interval.
Results
The mean and SD of FA, ADC, CSA, DSL, and DML of patients and controls are represented in Table 1. The mean and DS of FA, ADC, CSA, DSL, and DML of mild and moderate forms of CTS are represented in Table 2.
The mean FA and ADC of the median nerve were 0.43 ± 0.01 and 1.34 ± 0.02 × 10−3 mm2/s, respectively, in CTS and 0.48 ± 0.01 and 1.26 ± 0.03 × 10−3 mm2/s in volunteers, respectively. There was a statistically significant difference in FA and ADC of the median nerve between patients and volunteers (P = 0.001). The cutoff FA and ADC values of the median nerve used to differentiate CTS from volunteers were 0.45 and 1.31 × 10−3 mm2/s with AUC of 0.98 and 0.95, sensitivity of 84.6 and 84.6%, specificity of 100 and 93%, and accuracy of 88.9 and 87%, respectively (Table 3 and Fig. 2).
Receiver operating characteristic curve of FA and ADC of the median nerve. a The cutoff FA and ADC values of the median nerve used for diagnosis of CTS are 0.45 and 1.31 × 10−3 mm2/s with AUC of 0.985 and 0.954 and accuracy of 88.7 and 87%, respectively. b The cutoff FA and ADC of the median nerve used to differentiate mild from moderate form of CTS are 0.42 and 1.35 × 10−3 mm2/s with AUC of 0.96 and 0.686 and accuracy of 92.3 and 74.4%, respectively
The mean FA and ADC of mild CTS were 0.44 ± 0.012 and 1.33 ± 0.17 × 10−3 mm2/s and those of moderate CTS were 0.41 ± 0.01 and 1.3 ± 0.27 × 10−3 mm2/s, respectively, with significant difference between P = 0.001 and 0.029, respectively. The cutoff FA and ADC values of the median nerve used to differentiate mild from moderate CTS were 0.42 and 1.35 × 10−3 mm2/s with AUC of 0.96 and 0.68, sensitivity of 100 and 90.5%, specificity of 83.3 and 55.6%, and accuracy of 92.3 and 74.4%, respectively (Table 3 and Fig. 2).
The CSA was significantly different (P = 0.001) in CTS (10.0 ± 1.8 mm2) than that of controls (15.7 ± 1.9 mm2). There was a significant difference (P = 0.001) in CSA of median nerve between patients with mild disease (10.9 ± 1.9 mm2) and severe disease (8.9 ± 1.2 mm2). The CSA of CTS well correlated with FA (r = −0.894; P = 0.001) and ADC (r = −0.769, P = 0.001).
The mean DSL and DML in patients with CTS were 6.4 ± 2.87 and 4.9 ± 1.5 m/s and that of control were 2.4 ± 0.26 and 3.2 ± 0.28 m/s with significant difference (P = 0.001). The FA well correlated with DSL (r = −0.953; P = 0.001) and DML (r = −0.935; P = 0.001). The ADC well correlated with DSL (r = 0.781, P = 0.001) and (r = 0.781; P = 0.001), respectively.
Phalen’s test was positive in 24 patients and Tinel’s test was positive in 12 patients. There was correlation of FA and ADC of the median nerve with Tinel’s test (r = 0.742; P = 0.001; r = 0.684; P = 0.01) and Phalen’s test (r = 0.862; P = 0.001; r = 0.742; P = 0.001).
Discussion
The main findings in this study are that there is significant difference in the FA and ADC values between patients with CTS and volunteers. The FA and ADC can differentiate mild from moderate forms of CTS. The FA and ADC well correlated with clinical tests, nerve conduction parameters, and CSA of the CTS.
In this study, the mean ADC value of the median nerve in patients with CTS was higher than that of volunteers. This may be attributed to free diffusion within the median nerve due to the presence of edema in patients with CTS [14–17]. One study reported that there is decreased FA and increased ADC significantly with age (P = 0.001). There was a significant difference in FA and ADC between healthy volunteers and patients with carpal tunnel syndrome (P = 0.001). An FA threshold of 0.47 and an ADC threshold of 1.054 × 10−3 mm2/s might be used in the diagnosis of carpal tunnel syndrome [22]. Another study added that the carpal tunnel inlet had the largest and most consistent changes in diffusion-tensor imaging parameters. The FA measured at the carpal tunnel inlet had the highest diagnostic accuracy with AUC of 0.82. For an FA threshold of 0.44 or less at the carpal tunnel inlet, sensitivity is 72% and specificity is 82% [15].
In this work, there is significant difference in the FA and ADC values of the median nerve between mild and moderate CTS. One study reported that there is a statistically significant difference in FA and ADC between the normal and mild, mild and moderate, and moderate and severe subgroups. There is significant moderate correlation between diffusion tensor imaging parameters and CTS grades; this likely reflects the degree of nerve compression in various grades of CTS [17].
In this study, the CSA of CTS is well correlated with FA and ADC of the median nerve in patients with CTS. This may be attributed to edema which could potentially increase free water within the endoneural space around axons, or in the extracellular space between nerve fascicles, outside the perineurium [28]. Another study added that there was a significant difference between CSA of CTS and ADC, but not FA, at the distal forearm and proximal carpal tunnel [26].
In this study, the ADC value of the median nerve in CTS well correlated with nerve conduction study results. One study reported that FA and ADC correlated significantly with most nerve conduction study parameters. The highest correlation coefficient was between FA and sensory and motor amplitudes, and only FA had a significant correlation with nerve conduction study parameters [15]. Another study added that diffusion tensor imaging parameters provide helpful information that complements clinical and electrophysiological assessments for evaluating the efficacy of non-surgical treatment of patients with CTS [18]. Another study added that FA and ADC were significantly correlated with the nerve conduction study parameters of CTS [19].
There were few limitations in this study. First, we calculated the FA and ADC of the median nerve manually at one point; further studies measuring FA and ADC at multiple points of the median nerve by advanced post-processing will improve the results [31–33]. Second, we manually calculated the CSA; application of advanced post-processing with automated calculation will improve the results. Third, we applied routine diffusion tensor imaging for calculation of FA; further studies of diffusion tensor imaging of the median nerve using simultaneous multi-slice echo-planar imaging with blipped controlled aliasing in parallel imaging resulting in higher acceleration technique will improve the results [34]. Fourth, diffusion tensor imaging can detect the presence of median nerve neuropathy in patients with carpal tunnel syndrome, but it cannot distinguish compressive versus non-compressive neuropathy. Further studies are recommended to evaluate the value of diffusion tensor imaging in differentiating compressive from non-compressive neuropathy of the median nerve.
We concluded that diffusion tensor imaging can be used for the diagnosis of mild-moderate CTS and well correlated with nerve conduction studies and clinical tests.
Abbreviations
- ADC:
-
Apparent diffusion coefficient
- APB:
-
Abductor pollicis brevis
- CSA:
-
Cross-sectional area
- CTS:
-
Carpal tunnel syndrome
- DML:
-
Distal motor latency
- DSL:
-
Distal sensory latency
- FA:
-
Fractional anisotropy
References
Gillig JD, White SD, Rachel JN (2016) Acute carpal tunnel syndrome: a review of current literature. Orthop Clin North Am 47:599–607
Goldfarb CA (2016) The clinical practice guideline on carpal tunnel syndrome and workers’ compensation. J Hand Surg Am 41:723–725
Duckworth AD, Jenkins PJ, McEachan JE (2014) Diagnosing carpal tunnel syndrome. J Hand Surg Am 39:1403–1407
Lalonde DH (2014) Evidence-based medicine: carpal tunnel syndrome. Plast Reconstr Surg 133:1234–1240
Amirfeyz R, Gozzard C, Leslie IJ (2005) Hand elevation test for assessment of carpal tunnel syndrome. J Hand Surg Br 30:361–364
Goldberg G, Zeckser JM, Mummaneni R, Tucker JD (2016) Electrosonodiagnosis in carpal tunnel syndrome: a proposed diagnostic algorithm based on an analytic literature review. PM R 8:463–474
Razek AA, El-Basyouni SR (2016) Ultrasound of knee osteoarthritis: interobserver agreement and correlation with Western Ontario and McMaster Universities Osteoarthritis. Clin Rheumatol 35:997–1001
Razek AA, Fouda NS, Elmetwaley N, Elbogdady E (2009) Sonography of the knee joint. J Ultrasound 12:53–60
McDonagh C, Alexander M, Kane D (2015) The role of ultrasound in the diagnosis and management of carpal tunnel syndrome: a new paradigm. Rheumatology 54:9–19
Miyamoto H, Halpern EJ, Kastlunger M, Gabl M, Arora R, Bellmann-Weiler R et al (2014) Carpal tunnel syndrome: diagnosis by means of median nerve elasticity—improved diagnostic accuracy of US with sonoelastography. Radiology 270:481–486
Martins RS, Siqueira MG, Simplicio H, Agapito D, Medeiros M (2008) Magnetic resonance imaging of idiopathic carpal tunnel syndrome: correlation with clinical findings and electrophysiological investigation. Clin Neurol Neurosurg 110:38–45
Abdel Razek AA, Elkammary S, Elmorsy AS, Elshafey M, Elhadedy T (2011) Characterization of mediastinal lymphadenopathy with diffusion-weighted imaging. Magn Reson Imaging 29:167–172
Abdel Razek AA, Gaballa G, Denewer A, Tawakol I (2010) Diffusion weighted MR imaging of the breast. Acad Radiol 17:382–386
Wang H, Ma J, Zhao L, Wang Y, Jia X (2016) Utility of MRI diffusion tensor imaging in carpal tunnel syndrome: a meta-analysis. Med Sci Monit 22:736–742
Kwon BC, Koh SH, Hwang SY (2015) Optimal parameters and location for diffusion-tensor imaging in the diagnosis of carpal tunnel syndrome: a prospective matched case-control study. AJR Am J Roentgenol 204:1248–1254
Koh SH, Kwon BC, Park C, Hwang SY, Lee JW, Kim SS (2014) A comparison of the performance of anatomical MRI and DTI in diagnosing carpal tunnel syndrome. Eur J Radiol 83:2065–2073
Bulut HT, Yildirim A, Ekmekci B, Gunbey HP (2014) The diagnostic and grading value of diffusion tensor imaging in patients with carpal tunnel syndrome. Acad Radiol 21:767–773
Yildirim A, Bulut HT, Ekmekci B, Surucu GD, Karabiber M (2014) Use of diffusion tensor imaging for nonsurgical treatments of carpal tunnel syndrome. Muscle Nerve 50:950–955
Brienza M, Pujia F, Colaiacomo MC, Anastasio MG, Pierelli F, Di Biasi C et al (2014) 3T diffusion tensor imaging and electroneurography of peripheral nerve: a morphofunctional analysis in carpal tunnel syndrome. J Neuroradiol 41:124–130
Lindberg PG, Feydy A, Le Viet D, Maier MA, Drapé JL (2013) Diffusion tensor imaging of the median nerve in recurrent carpal tunnel syndrome—initial experience. Eur Radiol 23:3115–3123
Naraghi A, da Gama LL, Menezes R, Khanna M, Sussman M, Anastakis D et al (2013) Diffusion tensor imaging of the median nerve before and after carpal tunnel release in patients with carpal tunnel syndrome: feasibility study. Skelet Radiol 42:1403–1412
Guggenberger R, Markovic D, Eppenberger P, Chhabra A, Schiller A, Nanz D et al (2012) Assessment of median nerve with MR neurography by using diffusion-tensor imaging: normative and pathologic diffusion values. Radiology 265:194–203
Tasdelen N, Gurses B, Kilickesmez O, Firat Z, Karlikaya G, Tercan M et al (2012) Diffusion tensor imaging in carpal tunnel syndrome. Diagn Interv Radiol 18:60–66
Wang C-K, Jou I-M, Huang H-W, Chen P-Y, Tsai H-M, Liu Y-S et al (2012) Carpal tunnel syndrome assessed with diffusion tensor imaging: comparison with electrophysiological studies of patients and healthy volunteers. Eur J Radiol 81:3378–3383
Stein D, Neufeld A, Pasternak O, Graif M, Patish H, Schwimmer E et al (2009) Diffusion tensor imaging of the median nerve in healthy and carpal tunnel syndrome subjects. J Magn Reson Imaging 29:657–662
Yao L, Gai N (2009) Median nerve cross-sectional area and MRI diffusion characteristics: normative values at the carpal tunnel. Skelet Radiol 38:355–361
Khalil C, Hancart C, Le Thuc V, Chantelot C, Chechin D, Cotten A (2008) Diffusion tensor imaging and tractography of the median nerve in carpal tunnel syndrome: preliminary results. Eur Radiol 18:2283–2291
Kabakci N, Gurses B, Firat Z, Bayram A, Ulug AM, Kovanlikaya A et al (2007) Diffusion tensor imaging and tractography of median nerve: normative diffusion values. AJR Am J Roentgenol 189:923–927
Altrocchi P, Daube J, Frishberg B et al (1993) Practice parameter for carpal-tunnel syndrome. Neurology 43:2406–2409
Bland JDP (2000) A neuro physiological grading scale for carpal tunnel syndrome. Muscle Nerve 23:1280–1283
Razek A, Nada N, Ghaniem M, Elkhamary S (2012) Assessment of soft tissue tumours of the extremities with diffusion echoplanar MR imaging. Radiol Med 117:96–101
El-Serougy L, Abdel Razek AA, Ezzat A, Eldawoody H, El-Morsy A (2016) Assessment of diffusion tensor imaging metrics in differentiating low-grade from high-grade gliomas. Neuroradiol J 29:400–407
Razek AA, Sieza S, Maha B (2009) Assessment of nasal and paranasal sinus masses by diffusion-weighted MR imaging. J Neuroradiol 36:206–211
Filli L, Piccirelli M, Kenkel D, Boss A, Manoliu A, Andreisek G et al (2016) Accelerated magnetic resonance diffusion tensor imaging of the median nerve using simultaneous multi-slice echo planar imaging with blipped CAIPIRINHA. Eur Radiol 26:1921–1928
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Institutional review board approval was obtained, and informed consents were obtained from the patients and controls.
Disclosure
None.
Rights and permissions
About this article
Cite this article
Razek, A.A.K.A., Shabana, A.A.E., El Saied, T.O. et al. Diffusion tensor imaging of mild-moderate carpal tunnel syndrome: correlation with nerve conduction study and clinical tests. Clin Rheumatol 36, 2319–2324 (2017). https://doi.org/10.1007/s10067-016-3463-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-016-3463-y