Abstract
The objective of this study is to characterize the lipoprotein risk levels in Takayasu arteritis (TA) patients and its possible association with disease activity and glucocorticoid use. Twenty-five female TA patients were consecutively included and compared with 30 age-, gender-, and body mass index-matched healthy controls. Demographic features and the lipid profile were determined and cardiovascular risk levels were evaluated according to NCEP/ATPIII. Total cholesterol (TC), LDL-c, HDL-c, and triglycerides were determined after a 12-h overnight fast. Exclusion criteria were conditions that interfere in the lipid profile. The disease duration was 6.6 ± 7.4 years; 30% had clinical activity and 80% had laboratory activity. Regarding NCEP/ATPIII risk levels, TA patients presented higher frequency of lipid risk compared to controls: high TC (48% vs. 20%, p = 0.04), low HDL-c (20% vs. 0%, p = 0.015), and high triglycerides (36% vs. 10%, p = 0.026). No difference was observed related to LDL-c risk levels between both groups (40% vs. 20%, p = 0.14). Remarkably, 60% of the patients had at least one lipid risk factor for cardiovascular disease. No difference in the lipids was observed between patients with and without clinical activity; however, those with laboratory activity showed lower levels of HDL-c (1.37 ± 0.42 vs. 2.00 ± 0.63 mmol/L, p = 0.012) than patients without this activity. A negative correlation was found between HDL-c and CRP levels (r = −0.42, p = 0.04). Lipids were similar in patients under glucocorticoid compared to those without this therapy. This is the first study to identify that TA, an inflammatory disease, has a proatherogenic lipid profile which is associated to laboratory disease activity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Takayasu arteritis (TA) is a severe, chronic idiopathic inflammatory disease that primarily affects large vessels, thoracic, and abdominal aorta and its main branches [1]. Vessel inflammation and subsequent intimal proliferation of the involved arterial wall may lead to stenosis and occlusion while vessel dilatation or aneurysms may result from inflammatory process involving elastic lamina of arteries [2]. The usual serological finding in Takayasu arteritis is characterized by the presence of persistent high levels of inflammatory markers [3].
Over the last decade, atherosclerosis has also been recognized as an inflammatory vascular disease [4]. Moreover, one of the major risk factors for atherosclerosis is dyslipoproteinemia characterized by high plasma concentrations of cholesterol, in particular low-density lipoprotein (LDL) cholesterol, and low levels of high-density lipoprotein (HDL) cholesterol [5].
In this regard, autopsy studies have revealed atherosclerosis lesions in young patients with Takayasu arteritis [6–9]. Supporting these findings, image evaluation has confirmed early atherosclerosis in Takayasu arteritis [10–15].
We therefore have undertaken the present study to determine the lipid profile in a homogeneous female young TA population in order to determine the cardiovascular lipid risk levels in this disease and its possible association with disease activity and glucocorticoid use.
Materials and methods
Patients
This study was conducted on 25 consecutive pre-menopausal women with TA, according to the American College of Rheumatology criteria [16], who were followed up at the Vasculitis Outpatient Clinic from the Rheumatology Division of São Paulo University Medical School. Thirty age-, sex-, and body mass index (BMI)-matched healthy pre-menopausal females served as controls.
The local ethics committees approved the study and written informed consent was obtained from patients and controls. Patients were clinically evaluated and medical records were extensively reviewed. Serum samples were obtained at the inclusion in the study.
Rigorous exclusion criteria of conditions that interfere in the lipid profile, such as diabetes mellitus, pregnancy, menopause, liver or thyroid disease, ingestion of lipid-raising drugs, or use of statins, were applied.
The following data were collected from all patients: identification, clinical manifestation, disease duration, risk factors for coronary disease, use of glucocorticoids, and clinical and laboratory disease activity. Clinical activity was defined based on the presence of new onset or worsening of fever or musculoskeletal problems, vascular ischaemia, or inflammation such as claudication, diminished or absent pulse, bruit, carotidynia, or asymmetric blood pressure (National Institute of Health (NIH) criteria) [17, 18]. High erythrocyte sedimentation rate (ESR) and/or ultrasensitive C-reactive protein (CRP) levels, in the absence of infection, characterized laboratory activity.
Lipid profiles
Cholesterol (total and fractions) and triglycerides (TG) in the serum were measured enzymatically (Boehringer Mannheim, Argentina, and Merck, Germany, respectively) on a RA 1000 analyser (Technicon Instruments Corp), as well as high-density lipoprotein cholesterol (HDL-c), after a 12-h overnight fast [19–21]. LDL-c was estimated since all samples had triglycerides less than 4.52 mmol/L [22]. Lipid parameters were considered to be altered when total cholesterol >5.18 mmol/L, low-density lipoprotein cholesterol >3.367 mmol/L, high-density lipoprotein cholesterol <1.036 mmol/L, and triglycerides >1.695 mmol/L. The risk levels of the lipid profile were evaluated according to the National Cholesterol Education Program—Adult Treatment Panel III (NCEP/ATPIII) [5].
Inflammatory markers
Ultrasensitive C-reactive protein serum levels were systematically determined in all patients by nephelometry. Erythrocyte sedimentation rate was measured using Westergren method. These parameters were considered to be altered when CRP >5 µg/mL and ESR >13 mm/first hour, according to the normal values from the laboratory of our hospital.
Statistical analysis
Data are reported as mean ± SD, median, and percentage. Demographic and anthropometric variables were compared between patients and controls by the Student's t or Mann–Whitney tests. The frequency values of altered total and fraction cholesterol and triglycerides were compared between patients and controls using chi-square or Fisher’s exact tests. The P values <0.05 were considered to be significant.
Results
Patients and controls had a similar mean age, body mass index, and the frequency of white race. The mean disease duration was 6.6 years. Eighty percent of patients had systemic arterial hypertension. Thirty percent had clinical activity (NIH criteria) and 80% had laboratorial activity (high levels of erythrocyte sedimentation rate and/or C-reactive protein). More than half of the patients were in current use of prednisone (Table 1).
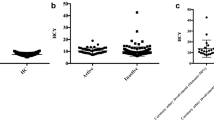
Significant higher levels of total cholesterol (5.18 ± 0.87 vs. 4.65 ± 0.78 mmol/L, p = 0.02) and triglycerides (1.32 ± 0.70 vs. 0.89 ± 0.48 mmol/L, p = 0.01) were observed in patients compared to controls. HDL-c levels (1.49 ± 0.52 vs. 1.54 ± 0.31 mmol/L, p = 0.69) and LDL-c levels (3.08 ± 0.74 vs. 2.71 ± 0.71 mmol/L, p = 0.064) did not differ in both groups.
Regarding NCEP/ATPIII risk levels, TA patients present higher frequency of lipid risk compared to healthy controls: total cholesterol, low HDL-c, and high triglycerides. No difference was observed related to LDL-c risk levels between the two groups (Table 2). Remarkably, 60% of the patients had at least one lipid risk factor for coronary artery disease in spite of young age.
No difference in the lipid profile was observed between patients with and without clinical activity: total cholesterol (5.18 ± 0.81 vs. 5.18 ± 0.92 mmol/L, p = NS), HDL-c (1.32 ± 0.29 vs. 1.57 ± 0.58 mmol/L, p = 0.30), LDL-c (3.19 ± 0.89 vs. 3.04 ± 0.70 mmol/L, p = 0.65), and triglycerides (1.47 ± 0.56 vs. 1.26 ± 0.75 mmol/L, p = 0.52). No significant difference in lipid profile was also found between patients in current use or not of prednisone: total cholesterol (5.25 ± 0.92 vs. 5.09 ± 0.84 mmol/L, p = 0.67), HDL-c (1.49 ± 0.57 vs. 1.50 ± 0.48 mmol/L, p = NS), LDL-c (3.09 ± 0.82 vs. 3.07 ± 0.66 mmol/L, p = NS), and triglycerides (1.46 ± 0.57 vs. 1.14 ± 0.82 mmol/L, p = 0.26).
On the other hand, patients with laboratory activity showed lower levels of HDL-c than patients without this activity. No differences related to total cholesterol, LDL-c, and triglycerides were found between both groups (Table 3). In addition, a negative Spearman correlation was found between HDL-c and CRP levels (r = −0.42, p = 0.04).
With regard to inflammatory markers, patients with Takayasu arteritis exhibited an increased mean level of CRP (8.7 ± 9.3 mcg/mL) and ESR (21.7 ± 12.2 mm/h). Importantly, 56% (n = 14) of Takayasu patients had a high level of CRP and 68% (n = 17) had high ESR levels.
Discussion
This is the first study to identify that Takayasu arteritis, an inflammatory disease, has a proatherogenic lipid profile, mainly characterized by low HDL levels during laboratory disease activity.
The accuracy of our data is supported by the study design with strict inclusion criteria. The selection of only pre-menopausal female patients was defined as an essential aspect to evaluate lipid profile in this disease since gender is an important parameter that must be accounted for in dyslipidemia studies [23]. Concentrations of total cholesterol, LDL-c, and plasma triglycerides are lower, and HDL-c concentration is higher in pre-menopausal women compared with men, partially due to the effects of estrogen [24]. In addition, women normally have greater activity in lipoprotein transport and removal of VLDL-c from the plasma than men do [24]. Moreover, as patients are living longer, lipid abnormalities due to increasing age become relevant [25].
Importantly, all our patients were matched by BMI and also were within normal range for this parameter. Actually, being overweight and obesity are well known factors that modify the lipid profile [24].
Furthermore, the rigorous exclusion criteria used to select Takayasu patients without other conditions that could interfere with lipid metabolism such as diabetes, thyroid disease, renal and hepatic involvements, and use of lipid-lowering drugs provided a unique opportunity to define more accurately the lipid profile in this population [26–29].
Another important factor that could play a role in vascular atherogenesis, mainly in inflammatory disease, is the glucocorticoid treatment. This drug is the gold standard for TA treatment [30] and long-term treatment with these drugs is known to cause hyperlipidemia due to induced LDL hepatic synthesis and decreased LDL catabolism [31, 32]. In this study, LDL-c NCEP/ATPIII risk levels were comparable in TA patients and controls. Furthermore, our data did not show any association between use of glucocorticoids and lipid profile alterations; however, more studies in large cohorts are necessary to substantiate the role of corticosteroid in TA atherogenesis.
The main finding of the present study was to demonstrate that during laboratory activity TA patients display low levels of HDL-c, and this particle was negatively correlated with CRP. Low HDL-c is a well-established independent risk factor for atherosclerosis [33]. This lipid alteration is also observed during flares of systemic lupus erythematosus (SLE) and rheumatoid arthritis. In fact, the lipid profile of patients with active and untreated RA is primarily characterized by a decrease in serum levels of high-density lipoprotein cholesterol [34]. Similarly, our group studying female inactive SLE patients without corticosteroids has demonstrated that these patients have low levels of HDL-c and high triglyceride levels compared to healthy controls, and these abnormalities are enhanced by lupus disease activity. Moreover, these lipid alterations were particularly associated with vasculitis [35, 36]. Recently, Quéméneur et al. have demonstrated, in 33 patients with systemic vasculitis (including two patients with Takayasu arteritis), a negative association between CRP and HDL-c as observed in the present study [37]. These data suggest that the inflammatory process leading to vasculitis seems to be extremely relevant to induce an atherogenic lipid profile.
The remarkable association of low HDL-c with elevated CRP levels, demonstrated herein, reinforces the role of inflammation and disease activity in the atherosclerosis process in Takayasu patients.
This is the first study to identify that patients with Takayasu’s arteritis have a proatherogenic lipid profile. Longitudinal studies are needed to evaluate if lipid preventive intervention could be therefore recommended for these patients.
References
Yamato M, Lecky JW, Hiramatsu K, Kohda E (1986) Takayasu arteritis: radiographic and angiographic findings in 59 patients. Radiology 161:329–334
Numano F (2002) Takayasu's arteritis: clinical aspects. In: Hoffman GS, Weyand CM (eds) Inflammatory diseases of blood vessels. Marcel Dekker, New York, pp 455–466
Jennette JC, Falk RJ, Andrassy K, Bacon PA, Churg J, Gross WL et al (1994) Nomenclature of systemic vasculitides: proposal of an international consensus conference. Arthritis Rheum 37:187–192
Ross R (1999) Atherosclerosis—an inflammatory disease. N Engl J Med 340:115–126
Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (2001) Executive summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 285:2486–2496
Numano F (2000) Vasa vasoritis, vasculitis and atherosclerosis. Int J Cardiol 75(Suppl 1):S1–S8
Numano F, Kishi Y, Tanaka A, Ohkawara M, Kakuta T, Kobayashi Y (2000) Inflammation and atherosclerosis. Atherosclerotic lesions in Takayasu arteritis. Ann NY Acad Sci 902:65–76
Numano F, Okawara M, Inomata H, Kobayashi Y (2000) Takayasu's arteritis. Lancet 356:1023–1025
Filer A, Nicholls D, Corston R, Carey P, Bacon P (2001) Takayasu arteritis and atherosclerosis: illustrating the consequences of endothelial damage. J Rheumatol 28:2752–2753
Sun Y, Yip PK, Jeng JS, Hwang BS, Lin WH (1996) Ultrasonographic study and long-term follow-up of Takayasu's arteritis. Stroke 27:2178–2182
Sharma S, Sharma S, Taneja K, Gupta AK, Rajani M (1996) Morphologic mural changes in the aorta revealed by CT in patients with nonspecific aortoarteritis (Takayasu's arteritis). Am J Roentgenol 167:1321–1325
Taniguchi N, Itoh K, Honda M, Obayashi T, Nakamura M, Kawai F et al (1997) Comparative ultrasonographic and angiographic study of carotid arterial lesions in Takayasu's arteritis. Angiology 48:9–20
Lefebvre C, Rance A, Paul JF, Beguin C, Bletry O, Amoura Z et al (2000) The role of B-mode ultrasonography and electron beam computed tomography in evaluation of Takayasu's arteritis: a study of 43 patients. Semin Arthritis Rheum 30:25–32
Park SH, Chung JW, Lee JW, Han MH, Park JH (2001) Carotid artery involvement in Takayasu's arteritis: evaluation of the activity by ultrasonography. J Ultrasound Med 20:371–378
Seyahi E, Ugurlu S, Cumali R, Balci H, Seyahi N, Yurdakul S, Yazici H (2006) Atherosclerosis in Takayasu arteritis. Ann Rheum Dis 65:1202–1207
Arend WP, Michel BA, Bloch DA, Hunder GG, Calabrese LH, Edworthy SM et al (1990) The American College of Rheumatology 1990 criteria for the classification of Takayasu arteritis. Arthritis Rheum 33:1129–1134
Kerr GS, Hallahan CW, Giordano J, Leavitt RY, Fauci AS, Rottem M, Hoffman GS (1994) Takayasu arteritis. Ann Intern Med 120:919–929
Bezerra MC, Calomeni GD, Caparbo VF, Gebrim ES, Rocha MS, Pereira RM (2005) Low bone density and low serum levels of soluble RANK ligand are associated with severe arterial calcification in patients with Takayasu arteritis. Rheumatol (Oxford) 44:1503–1506
Siedel J, Hägele EO, Zielgenhorn J, Wahlefeld AW (1983) Reagent for the enzymatic determination of serum total cholesterol with improved lipolytic efficiency. Clin Chem 29:1075–1080
Fossati P, Prencipe L (1982) Serum triglycerides determined colorimetrically with an enzyme that produces hydrogen peroxide. Clin Chem 28:2077–2080
Warnick GR, Cheung NC, Albers JJ (1979) Comparison of current methods for high density lipoprotein cholesterol quantification. Clin Chem 25:596–604
Friedewald WT, Levy RI, Fredrickson DS (1972) Estimation of the concentration of low density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 18:499–502
Williams CM (2004) Lipid metabolism in women. Proc Nutr Soc 63:153–160
Regitz-Zagrosek V, Lehmkuhl E, Mahmoodzadeh S (2007) Gender aspects of the role of the metabolic syndrome as a risk factor for cardiovascular disease. Gend Med 4(Suppl B):S162–S177
Heiss G, Tamir I, Davis CE, Tyroler HA, Rifkand BM, Schonfeld G et al (1980) Lipoprotein-cholesterol distribution in selected North American populations: the lipid research clinics program prevalence study. Circulation 61:302–315
Kannel WB, Hjortland MC, McNamara PM, Gordon T (1976) Menopause and risk of cardiovascular disease. The Framingham Study. Ann Intern Med 85:447–452
Brown WV (1994) Lipoprotein disorders in diabetes mellitus. Med Clin North Am 78:143–161
Thompson GR, Soutar AK, Spengel FA, Jadhav A, Gavigan SJ, Myant NB (1981) Defects of receptor-mediated low density lipoprotein catabolism in homozygous familial hypercholesterolemia and hypothyroidism in vivo. Proc Natl Acad Sci USA 78:2591–2595
Attman PO, Alaupovic P (1991) Lipid and apolipoprotein profiles of uremic dyslipoproteinemia—relation to renal function and dialysis. Nephron 57:401–410
Seko Y (2007) Giant cell and Takayasu arteritis. Curr Opin Rheumatol 19:39–43
Nashel DJ (1986) Is atherosclerosis a complication of long-term corticosteroid treatment. Am J Med 80:925–929
Al Rayyes O, Wallmark A, Florén CH (1997) Additive inhibitory effect of hydrocortisone and cyclosporine on low-density lipoprotein receptor activity in cultured HepG2 cells. Hepatology 26:967–971
Downs JR, Clearfield M, Weis S, Whitney E, Shapiro DR, Beere PA, Langendorfer A, Stein EA, Kruyer W, Gotto AM Jr (1998) Primary prevention of acute coronary events with lovastatin in men and women with average cholesterol levels: results of AFCAPS/TexCAPS. Air Force/Texas Coronary Atherosclerosis Prevention Study. JAMA 279:1615–1622
Park YB, Lee SK, Lee WK, Suh CH, Lee CW, Lee CH (1999) Lipid profiles in untreated patients with rheumatoid arthritis. J Rheumatol 26:1701–1704
Borba EF, Bonfá E (1997) Dyslipoproteinemias in systemic lupus erythematosus: influence of disease, activity, and anticardiolipin antibodies. Lupus 6:533–539
de Carvalho JF, Borba EF, Viana VS, Bueno C, Leon EP, Bonfá E (2004) Anti-lipoprotein lipase antibodies: a new player in the complex atherosclerotic process in systemic lupus erythematosus? Arthritis Rheum 50:3610–3615
Quéméneur T, Martin-Nizard F, Kandoussi A et al (2007) PON1, a new biomarker of cardiovascular disease, is low in patients with systemic vasculitis. Semin Arthritis Rheum 37:149–155
Acknowledgments
This work was supported by grants of Conselho Nacional de Desenvolvimento Científico e Tecnológico [305691/2006-6 to R.M.R.P., 305468/2006-5 to E.B.] and Federico Foundation to E.B.
Disclosures
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Carvalho, J.F., Bonfá, E., Bezerra, M.C. et al. High frequency of lipoprotein risk levels for cardiovascular disease in Takayasu arteritis. Clin Rheumatol 28, 801–805 (2009). https://doi.org/10.1007/s10067-009-1153-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-009-1153-8