Abstract
We report a 29-year-old Japanese woman with disseminated intravascular coagulation (DIC) and adult onset Still’s disease (AOSD). Her disease was refractory to high-dose glucocorticoids, two courses of steroid pulse therapy, and addition of cyclosporine (3.5 mg/kg/day). The serum interleukin-6 level was markedly elevated. Therefore, we administered an anti-interleukin-6 receptor antibody (tocilizumab, 8 mg/kg fortnightly), which dramatically improved her symptoms and the levels of acute-phase proteins. In addition, rapid tapering of the glucocorticoid dose was possible. Four months later, she was maintained on tocilizumab infusion once a month with low-dose steroid therapy. Cyclosporine is one of the first-line immunosuppressants for AOSD, especially when associated with DIC, hepatic failure, or hemophagocytic syndrome. In patients with cyclosporine-resistant AOSD, tocilizumab may be another useful option.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Adult onset Still’s disease (AOSD) is an uncommon disorder that typically features an evanescent rash, sore throat, intermittent spiking fever, and polyarthritis. Glucocorticoids are the mainstay of treatment, but some patients require additional immunosuppressants, including methotrexate, cyclosporine, anti-TNF agents, or the IL-1 receptor antagonist anakinra [1]. Here, we report a patient with treatment-resistant AOSD complicated by disseminated intravascular coagulation (DIC), successfully managed with tocilizumab, a humanized monoclonal anti-IL-6 receptor antibody.
Case report
A 29-year-old Japanese woman was admitted to her local hospital with a high fever, sore throat, and arthritis. Bacterial infection was suspected due to elevation of the leukocyte count and C-reactive protein (CRP). Administration of imipenem (1 g/day) was immediately started, but symptoms and laboratory findings did not improve. Spiking fever (maximum: 40°C) continued for 2 weeks. No evidence of infection or connective tissue disease was obtained. AOSD was suggested by an elevated serum ferritin level, so she was referred to our hospital.
On admission, she had an evanescent rash on her anterior chest, upper arms, and thighs. Cervical lymph nodes were palpable, and she complained of arthralgia in the shoulders, knees, and ankles. Laboratory tests showed a leukocyte count of 14.7 × 109/l (93% neutrophils), hemoglobin of 10.6 g/dl, platelet count of 87 × 109/l, and ESR of 35 mm/h (normal <15). Aspartate aminotransferase was 213 U/l (normal range 11–30), alanine aminotransferase was 65 U/l (4–30), lactate dehydrogenase was 1,690 U/l (109-216), CRP was 26.1 mg/dl, and serum ferritin was 34,891.5 ng/ml (3.0–59.4). Serum IL-6 was markedly elevated to 52 pg/ml (normal <4). Fibrinogen was 270 mg/dl, fibrin degradation products (FDP) were 119.2 μg/ml (normal <5.0), and D-dimer was 99.2 μg/ml (0.5–1.5). Her chest X-ray film and abdominal computed tomography (CT) demonstrated bilateral pleural effusions and mild splenomegaly, respectively. From these findings, a diagnosis of AOSD with DIC was made.
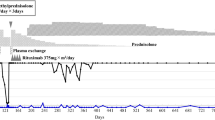
Intravenous methylprednisolone (1 g/day) was given for 3 days, followed by oral prednisolone at 60 mg/day (1 mg/kg). Continuous intravenous infusion of heparin was also done (Fig. 1). On the next day, fibrinogen decreased to 77 mg/dl, and purpura appeared on her chest and abdomen. Fresh frozen plasma was administered, and heparin was changed to nafamostat mesilate. Oral cyclosporine (2.5 mg/kg/day) was added for worsening of liver dysfunction. Two days after finishing methylprednisolone, spiking fever recurred and CRP rebounded. Prednisolone was changed to oral dexamethasone at 6 mg/day (0.12 mg/kg), cyclosporine was increased to 3.5 mg/kg/day, and intravenous dexamethasone (100 mg/day) was added for 3 days. DIC and liver function tests improved with resolution of her fever, but arthralgia and a high CRP persisted.
Summary of the clinical course. Hep heparin, NAF nafamostat mesilate, mPSL methylprednisolone, PSL prednisolone, CyA cyclosporine, Dex dexamethasone, TCZ tocilizumab. Reference values are as follows: IL-18 (normal <259.4), IL-1β (normal <10), TNF-α (normal <2.8), IL-6 (normal <4), and IFN-γ (normal <1.2)
Since we had previously achieved dramatic improvement of intractable AOSD by using tocilizumab, we decided to try it. After oral informed consent was obtained, we administered 400 mg (8 mg/kg) of tocilizumab intravenously. On the next day, fatigue and arthralgia were markedly improved, and CRP was almost normalized within 1 week. Fortnightly administration of tocilizumab was continued, and dexamethasone was tapered to 2 mg/day at discharge. Three months later, cyclosporine has been discontinued, and 4 months later, she was maintained on tocilizumab every 4 weeks with prednisolone 10 mg/day.
Discussion
Various cytokines, including TNF-α, IFN-γ, IL-1β, IL-6, and IL-18, are elevated in AOSD [1]. Although IL-6 is thought to be important in this disease, its elevation is not observed consistently [2–6]. High circulating levels of a cytokine do not necessarily reflect its importance for AOSD [2]. Anti-TNF agents are less effective in patients with refractory AOSD [7]. A causative role of IL-1 in AOSD has been demonstrated through the response to its direct inhibition by anakinra [2, 3, 8]. We previously encountered a patient with refractory AOSD who responded to treatment with an anti-IL-6 receptor monoclonal antibody [9]. A role of IL-6 in AOSD could also be confirmed by direct blockade of its receptor.
The role of a cytokine cascade in AOSD was proposed by Fitzgerald et al. [2]. It is considered that TNF-α induces IL-1, which stimulates the production of IL-6. Accordingly, it may be better to directly inhibit IL-6, the downstream cytokine, by using tocilizumab than to block IL-1 with anakinra. A prospective study could properly define the most effective agents for treating AOSD that is refractory to current immunosuppressants, but its low prevalence precludes such an investigation. Tocilizumab may be more convenient than anakinra for clinical use because of less frequent administration, since daily subcutaneous injection of anakinra is needed, and interruption of therapy can trigger recurrence [2].
Efficacy of cyclosporine has been reported in patients with treatment-resistant AOSD [10, 11]. It is also effective for AOSD associated with DIC or hemophagocytic syndrome [12, 13], and we have previously obtained dramatic improvement of AOSD associated with hepatic failure by administration of cyclosporine [14]. In the present patient, we added cyclosporine due to worsening of liver function tests. Although her fever, DIC, and liver function tests improved to some extent, cyclosporine did not improve arthralgia or the levels of acute-phase proteins. Thus, cyclosporine is not always effective for steroid-resistant AOSD.
Long-term tocilizumab therapy can induce remission of AOSD. Although spontaneous disease remission also occurs, our first patient has been disease-free with no medications for 7 years after 18 months of tocilizumab therapy (unpublished observation). Remission has also been achieved in patients with systemic-onset juvenile idiopathic arthritis receiving tocilizumab [15], but it remains unclear how long anti-cytokine therapy should be continued for AOSD.
References
Efthimiou P, Georgy S (2006) Pathogenesis and management of adult-onset Still’s disease. Semin Arthritis Rheum 36:144–152
Fitzgerald AA, Leclercq SA, Yan A et al (2005) Rapid responses to anakinra in patients with refractory adult-onset Still’s disease. Arthritis Rheum 52:1794–1803
Kötter I, Wacker A, Koch S et al (2007) Anakinra in patients with treatment-resistant adult-onset Still’s disease: four case reports with serial cytokine measurements and a review of the literature. Semin Arthritis Rheum 37:189–197
Fujii T, Nojima T, Yasuoka H et al (2001) Cytokine and immunogenetic profiles in Japanese patients with adult Still’s disease. Association with chronic articular disease. Rheumatology (Oxford) 40:1398–1404
Scheinberg MA, Chapira E, Fernandes ML et al (1996) Interleukin 6: a possible marker of disease activity in adult onset Still’s disease. Clin Exp Rheumatol 14:653–655
Hoshino T, Ohta A, Yang D et al (1998) Elevated serum interleukin 6, interferon-gamma, and tumor necrosis factor-alpha levels in patients with adult Still’s disease. J Rheumatol 25:396–398
Fautrel B, Sibilia J, Mariette X et al (2005) Tumour necrosis factor alpha blocking agents in refractory adult Still’s disease: an observational study of 20 cases. Ann Rheum Dis 64:262–266
Kalliolias GD, Georgiou PE, Antonopoulos IA et al (2007) Anakinra treatment in patients with adult-onset Still’s disease is fast, effective, safe and steroid sparing: experience from an uncontrolled trial. Ann Rheum Dis 66:842–843
Iwamoto M, Nara H, Hirata D et al (2002) Humanized monoclonal anti-interleukin-6 receptor antibody for treatment of intractable adult-onset Still’s disease. Arthritis Rheum 46:3388–3389
Marchesoni A, Ceravolo GP, Battafarano N et al (1997) Cyclosporin A in the treatment of adult onset Still’s disease. J Rheumatol 24:1582–1587
Shojania K, Chalmers A, Rangno K (1995) Cyclosporin A in the treatment of adult Still’s disease. J Rheumatol 22:1391–1392
Park JH, Bae JH, Choi YS et al (2004) Adult-onset Still’s disease with disseminated intravascular coagulation and multiple organ dysfunctions dramatically treated with cyclosporine A. J Korean Med Sci 19:137–141
Nishida T, Suzuki K, Kuwada N et al (2001) Hemophagocytic syndrome and adult Still’s disease associated with meningoencephalitis and unconsciousness. Intern Med 40:1037–1040
Nagashima T, Aoki Y, Onishi S et al (2008) Steroid-refractory severe hepatic failure in adult onset Still’s disease responding to cyclosporine. Clin Rheumatol 27:1451–1453
Yokota S, Imagawa T, Miyamae T et al (2004) Long-term therapeutic experience of humanized anti-IL-6 receptor monoclonal antibody (aIL-6R Mab) in systemic-onset juvenile idiopathic arthritis (So-JIA). Arthritis Rheum 50:S438
Disclosures
Dr. Iwamoto and Minota have received royalties from Chugai Pharmaceutical Company, Ltd., Tokyo, Japan (less than $10,000). The other authors have disclosed no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Matsumoto, K., Nagashima, T., Takatori, S. et al. Glucocorticoid and cyclosporine refractory adult onset Still’s disease successfully treated with tocilizumab. Clin Rheumatol 28, 485–487 (2009). https://doi.org/10.1007/s10067-009-1097-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-009-1097-z