Abstract
We explored the predictors of foot ulceration in patients with rheumatoid arthritis (RA). The cases were 15 patients with RA reporting foot ulceration in response to a postal survey of patients sampled from a diagnostic register in secondary care (n = 1,130). The controls were 66 patients with RA randomly sampled from the survey respondents (n = 883) after matching for age, sex and disease duration. Patients with co-existent diabetes were excluded. Clinical examination included the assessment of known risk factors for foot ulceration in diabetes including: neuropathy (insensitivity to 10 g monofilament), peripheral vascular disease (ankle brachial pressure index [ABPI]), foot deformity (Platto indices) and raised plantar pressure (PressureStat™ readings). A 44 swollen-joint count, the presence of pre-ulcerative lesions and current steroid therapy were identified through univariate analysis as additional potential predictors in patients with RA. Forward step-wise logistic regression analysis showed that the following variables were significant predictors of ulceration: steroid therapy (OR = 9.70, 95%CI = 2.09–45.11, p = 0.004), abnormal ABPI (OR = 13.45, 95%CI = 1.19–151.43, p = 0.035), the presence of pre-ulcerative lesions (OR = 7.40, 95%CI = 1.51–36.30, p = 0.014) and swollen-joint count (OR = 1.25, 95%CI = 1.02–1.53, p = 0.034). Abnormal sensation, foot deformity and raised plantar pressures were not significant predictors of ulceration. The wide confidence intervals for ABPI were due to sparse data with very few abnormal values, and the results of exact logistic regression (more accurate where data is sparse and case matching employed) found that ABPI was no longer a significant predictor (p = 0.054). The significance of the other predictors did not differ substantially. In this preliminary study, abnormal sensation, foot deformity and raised plantar pressures were not significantly associated with foot ulceration but active disease and current steroid therapy were. The contribution of peripheral vascular disease to risk is unclear and further investigation is needed in a larger cohort.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Whilst the risk factors for foot ulceration in patients with diabetes have been extensively studied, little is known about the epidemiology of foot ulceration in patients with rheumatoid arthritis (RA). Initial investigation has focussed upon establishing the size of the problem, and recent estimates of overall prevalence indicate that 10–13% of patients with RA are affected by past or current ulceration [1, 2]. Furthermore, many patients have reported that they have experienced recurrent episodes of ulceration affecting multiple sites on the foot [1]. This suggests that further work is needed to address risk factors for ulceration in this client group. An understanding of the possible predictors of ulceration in patients with RA is the first step towards evidence-based intervention to reduce both incidence and recurrence rates.
This research was undertaken in the context of a larger study to estimate the validated prevalence of foot ulceration in patients with RA, the findings of which have previously been reported [1]. In this article, we report the findings of a preliminary analysis of the predictors of foot ulceration in patients with RA using a case–control approach. Established risk factors for foot ulceration in patients with diabetes include peripheral neuropathy, peripheral vascular disease, raised plantar pressures, foot deformity and ill-fitting footwear [3]. The evidence is that all these clinical manifestations of disease also occur to a greater or lesser extent in patients with RA, but the contribution of these factors to the risk of foot ulceration in this client group has not been investigated. Additional factors that may also contribute to risk in patients with RA include local disease activity in the joints and soft tissues of the foot, cutaneous vasculitis and nodules. Furthermore, disease-modifying medication such as corticosteroids and methotrexate may cause skin fragility and hinder tissue repair. The individual’s ability to maintain foot health independently may also be compromised by reduced self-care capacity and manual dexterity. The aim of this preliminary study was to identify the predictors of foot ulceration in patients with RA by modelling established risk factors for diabetic foot ulceration combined with RA-specific potential predictors identified through a literature review.
Materials and methods
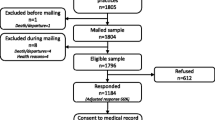
Ethical approval was obtained from the Local Research Ethics Committee, and patients gave informed consent before study participation. Cases and controls were recruited from respondents to the postal survey to establish prevalence data, which achieved a 78% response rate (n = 883) [1]. The sample for the survey were adult patients with a diagnosis of RA identified from the diagnostic database maintained by the hospital’s Rheumatology Department (n = 1,130).
Inclusion criteria were the presence of a foot ulcer, defined by expert consensus as full thickness skin defect occurring in isolation on or below the midline of the malleoli and requiring >14 days to heal. Exclusion criteria were a concomitant diagnosis of diabetes (a known risk factor for foot ulceration) and heel ulceration (deemed more likely to be a pressure ulcer associated with immobility). Ninety-three percent of the participants were white British, and there were no observed differences in ethnic origin between cases and controls.
Clinical examination included the following: insensitivity to 10 g monofilament applied to three sites on the medial and lateral plantar surfaces of the forefoot and the plantar aspect of the great toe (protective sensation absent if two out of three answers are incorrect); ankle brachial pressure index (ABPI abnormal if <1); foot deformity, measured by the Platto index [4], and Manchester grading scale for hallux valgus [5]; plantar pressure measurement, using PressureStat™ footprint mat (Bailey Instruments, Manchester, UK); a 44 swollen-joint count and the presence of pre-ulcerative lesions, which were defined a priori by the research team as a clear area of tissue damage associated either with subcutaneous haemorrhage or discolouration of the skin. History taking included a record of current disease-modifying and steroid therapy. Participants also completed the Foot Impact Scale [6], a two sub-scale, 51-item questionnaire covering the domains of impairments/shoes and activities/participation, and the Stanford Health Assessment Questionnaire, modified for use with British patients [7].
In this preliminary investigation, all respondents reporting open foot ulceration at the time of the survey were invited to take part in the case–control study. Reference to rule of thumb sample size calculations indicated that the numbers of potential participants identified in this nested study would be too small to represent a generalisable population. Hence, this preliminary investigation was undertaken to identify potential predictors of foot ulceration.
The cases
After the exclusion of two patients with co-existent diabetes, 15 open cases were recruited from 21 potential participants identified in the prevalence study. The mean age (SD) of cases was 65 years (13 years) and disease duration (SD) 26 years (10 years). Four of the recruited cases were female and 11 were male. As the cases and controls were matched by age, sex and disease duration to improve the efficiency of the study, any comparison of these variables between the two groups would not be meaningful. Seven of the cases were affected by their first episode of ulceration, which reflects the recruitment of incident cases as they occurred. The remaining eight cases had all experienced multiple sites of involvement ranging from two to five. Between them, the 15 cases had 20 open foot ulcers affecting six sites. The most common site for ulceration was the dorsal aspect of hammer toes (n = 7), followed by the metatarsal heads (n = 5) and the medial aspect of the first metatarsophalangeal joint associated with hallux abductovalgus (n = 4).
The controls
Potential controls were identified from respondents who reported that they were unaffected by past or present foot ulceration. After matching potential controls for age, sex and disease duration to improve the efficiency of the study, four to five randomly sampled controls were recruited per case. A total of 70 controls were recruited from 111 potential participants. The voluntary participation rate of the controls was 70%, which is similar to the participation rate of eligible cases (71%). Sixty-six controls were included in the analyses; four controls were excluded due to incomplete data.
Statistical analysis
The data were entered onto the Statistical Package for the Social Sciences (SPSS version 12.0.1). Initially, univariate analysis was undertaken to identify potential predictors of foot ulceration in RA for inclusion in the modelling. The unadjusted odds ratios and their 95% confidence intervals were computed. Forward step-wise logistic regression was used to assess the relationship between foot ulceration and associated variables. The model tested incorporated variables known to be predictors of foot ulceration in diabetes (insensitivity to 10 g monofilament in the affected or matched limb, abnormal ABPI, foot deformity and raised plantar pressure) and potential predictors in RA identified from the univariate analysis (where p < 0.1). Due to the small sample size, the analysis was re-run using exact logistic regression methods, which are based upon the exact distribution of statistics for the regression parameters and preserve the data in matched form. Exact methods can be more accurate in cases where total numbers are low, where there are few cases relative to controls or where the data are sparse or imbalanced.
Results
Table 1 displays the results of the initial univariate analysis.
In the preliminary analysis, all the factors were associated with an increased likelihood of developing a foot ulcer. Abnormal ABPI and steroid therapy were the factors associated with the greatest increase in the odds ratios.
Table 2 displays the results of the multivariate forward step-wise logistic regression analysis.
The model was significantly better than the null model (χ 2 = 26.66, df = 4, p < 0.001). The Nagelkerke R-squared statistic indicated that this model accounted for 45.5% of the variance in foot ulcer status, and the percentage of accurate predictions for the ulcer group rose to 53%. The model retained steroid, ABPI, pre-ulcerative lesions and swollen-joint count but did not retain foot deformity and raised plantar pressure. The Hosmer and Lemeshow test showed that the model adequately fitted the data (χ 2 = 7.53, df = 8, p = 0.481).
After adjusting for the effect of all the variables in the step-wise forward logistic regression model, abnormal ABPI remained the predictor of foot ulceration with the greatest odds ratio (OR = 13.45, 95%CI = 1.19–151.44). Current steroid therapy and the presence of pre-ulcerative lesions also remained significant predictors of foot ulceration (OR = 9.70 and 7.40, 95%CI = 2.09–45.11 and 1.51–36.30, respectively), as did swollen-joint count (OR = 1.83, 95%CI = 1.18–3.85).
Table 3 displays the results from the exact logistic regression analysis, accounting for matching.
When the model was re-run using Exact methods and accounting for case matching, the odds ratios were altered for steroid therapy and pre-ulcerative lesions but the overall results did not differ substantially. However, using Exact methods, the p value for the contribution made by ABPI to the model increased to 0.054, meaning that it was no longer a significant predictor of ulceration.
The analysis of the questionnaire data revealed that there was a significant relationship between a higher score on the Foot Impact Scale and foot ulceration (U = 302.500, N 1 = 69, N 2 = 15, p = 0.012, two-tailed). This suggests greater impact of RA on the foot in ulcer patients in the domains of footwear, impairments, activity limitation and participation. Ulcer patients also had greater reduced self-care capacity as measured by the Health Assessment Questionnaire, although this just failed to achieve significance at the conventional 0.05 level (U = 351.50, N 1 = 69, N 2 = 15, p = 0.051, two-tailed).
Discussion
This preliminary case–control study offers an initial insight into the predictors of foot ulceration in patients with RA. Peripheral vascular disease, disease activity and steroid therapy were associated with an increased risk of foot ulceration in this small sample and warrant further investigation in an adequately powered definitive study. Abnormal ABPI was originally the predictor of foot ulceration with the greatest odds ratio in the modelling (OR = 13.45, 95%CI = 1.19–151.44), although it failed to achieve significance when the results were checked using Exact methods. This was due to the unbalanced nature of this variable with relatively few abnormal values, which has implications for future sample size calculations. Peripheral vascular disease is clearly an important area to examine because lack of perfusion to the lower limb decreases tissue resilience and may directly contribute to the progression of tissue damage to ulceration in this client group with important implications for screening in clinical practice. There is growing evidence of a link between RA and peripheral vascular disease, which has been estimated to affect up to 18% of patients over time [8]. In a case–control study investigating the prevalence of sub-clinical atherosclerosis in RA, 25% of cases (n = 40) had an ABPI <1.0 compared with 2.5% of the control group (n = 40) [9].
The other significant predictors of foot ulceration in RA reflected disease activity (swollen-joint count) and potentially related steroid use. Increased disease activity may precipitate ulceration as a result of synovitis, which may cause local deformity of the soft tissues and lower limb oedema, increasing the risks of pressure from footwear. Vasculitis was not included in the multivariate analysis as only one case and one control had a history of vasculitis with no clinical evidence of current disease activity. It was not within the remit of this investigation to look for evidence of sub-clinical small vessel vasculitis. Corticosteroids may be a reflection of disease severity, but the anti-mitotic effect of steroids contributes to delayed epithelialisation and results in a thinned abnormal dermis and epidermis with subsequent increased skin fragility [10]. As corticosteroids interfere with both the inflammatory and proliferative stages of tissue repair, they may also play a role in healing and recurrence rates with increased risks of infections as a result of anti-microbial activity.
The results from this preliminary study suggest that loss of protective sensation, foot deformity and raised plantar pressures are not significant predictors of foot ulceration in patients with RA. Whilst peripheral neuropathy rarely occurs with the same severity in RA as in diabetes, patients with RA are still at increased risk. Entrapment or distal sensory neuropathies are thought to be more common than mononeuritis multiplex and sensorimotor neuropathy, which is associated with vasculitis. Ultimately, however, the risk of ulceration arises from a reduction in protective sensation which means that the individual is not alerted to tissue damage occurring at an early stage from, for example, ill-fitting footwear. A recent study estimated the prevalence of reduced protective sensation in the feet of patients with RA at 59% compared with just 12.5% of the controls [11]. It is possible that the threshold for risk relates to the level of sensory loss, but in this study, no significant difference was detected between the groups after neurological assessment using the 10-g Semmes–Weinstein monofilament.
It was more surprising that foot deformity did not emerge as a significant predictor of ulceration as the location data and history taking suggested otherwise. It is possible that the measurement tool, the Platto foot deformity score, lacked sensitivity in relation to the degree of deformity present, as foot deformity was a very common finding in both groups with a mean Platto score of 8.93 (SD = 1.534) in cases and 7.10 (SD = 3.273) in controls. The more likely explanation though is that by matching for disease duration, any differences between cases and controls were reduced. Participants of longstanding disease duration were more likely to have greater foot deformity and raised plantar pressures, and the fact that they were matched on this variable decreases the ability of the study to detect a significant change in the odds ratio.
All the sites of open ulceration in this study, with the exception of one case of inter-digital ulceration caused by pressure from a neighbouring toe, occurred at the interface between bony prominences and footwear. Footwear fit was not evaluated due to the lack of an objective measurement tool, although ill-fitting footwear appeared to be a contributory factor to many cases of ulceration. Indeed, the modification of footwear, such as the temporary wearing of sandals instead of shoes, appeared to be a common means of self-management to enable healing to take place. The focus of this study was on intrinsic risk factors for foot ulceration, but future work will also need to address extrinsic factors, including footwear fit and the role of compressive and shearing forces.
The case mix of participants in this study reflects the findings of the prevalence study in which the dorsal aspect of hammer toes was the most common site of both open and healed ulceration followed by metatarsal heads and the medial aspect of the first metatarsophalangeal joint associated hallux abductovalgus [1]. Research in diabetes has focussed on plantar ulceration and demonstrates a strong association between risks of high foot pressures and ulceration, particularly in association with neuropathy and callus formation [12–14]. Plantar pressure measurements in patients with RA have consistently recorded raised pressures over the metatarsal heads as a result of forefoot deformity and subsequent pressure distribution, limited joint mobility and changes in gait patterns [15–18]. In this small study, there were only five cases of open ulceration over the metatarsal heads, all with callus at the site. The PressureStat system could only measure interface pressures on the plantar aspect of the foot, whereas the majority of ulcers transpired to occur at other sites. In diabetes, site of ulceration and aetiology are related and future sample size calculations need to allow for the stratification of patients to facilitate predictive modelling for plantar and dorsal ulceration.
This study is the first of its kind to explore the risk factors for foot ulceration in patients with RA. However, the reliability of the findings is weakened by the low numbers of open ulcer cases at the time of the study. Clinical epidemiology is not a clearly defined field and many researchers apply methods without due regard for understanding the temporal relationship between risk and the occurrence of disease. Whilst focussing solely on the examination of incident cases is rarely feasible, the inclusion of healed cases was specifically avoided in this study as risk factors may be modified after the progression of tissue damage to ulceration. A larger study will not only generate more valid and reliable findings, but will also facilitate an exploration of risk factors that may vary according to the location of ulceration. Whilst matching was employed to improve the efficiency of the study, this may reduce the potential to detect differences in exposure between cases and controls and in the future should only be used to account for known risk factors that are likely to be important confounders [19].
This preliminary investigation raises the awareness of the factors which may contribute to risk in this client group that warrant further investigation, including disease activity, steroid use and peripheral vascular disease. The findings from this study suggest that the case mix and aetiology of foot ulceration in patients with RA may have both similarities and differences with foot ulceration in diabetes, but further work is needed in this area. A larger multi-centre case–control study is planned, and an unmatched analysis will be undertaken in conjunction with latent class modelling. This will enable the inclusion of continuous latent variables and multi-level modelling to explore the relationship between the predictors and identify any clusters of risk factors that can be used for prediction.
References
Firth J, Hale CA, Helliwell PS, Hill J, Nelson EA (2008) The prevalence of foot ulceration in patients with rheumatoid arthritis. Arthritis Rheum 59(2):200–205
Matricali GA, Boonen A, Verduyckt J, Taelman V, Verschueren P, Sileghem A, Corluy L, Westhovens R (2006) The presence of forefoot problems and the role of surgery in patients with rheumatoid arthritis. Ann Rheum Dis 65(9):1254–1255
Boulton AJ (2000) The pathway to ulceration. In: Boulton AJM, Connor H, Cavanagh PR (eds) The foot in diabetes. Wiley, London
Platto MJ, O’Connell PG, Hicks JE, Gerber LH (1991) The relationship of pain and deformity of the rheumatoid foot to gait and an index of functional ambulation. J Rheumatol 18(1):38–43
Garrow AP, Papageorgiou A, Silman AJ, Thomas E, Jayson MIV, Macfarlane GJ (2001) The grading of Hallux Valgus: the manchester scale. J Am Podiatr Med Assoc 91(2):74–78
Helliwell PS, Reay N, Gilworth G, Redmond A, Slade A, Tennant A, Woodburn J (2005) Development of a foot impact scale for rheumatoid arthritis. Arthritis Rheum 53(3):418–422
Kirwan JR, Reeback JS (1986) Stanford health assessment questionnaire modified to assess disability in British patients with rheumatoid arthritis. Br J Rheumatol 25:206–209
Maradit-Kremers H, Nicola PJ, Crowson CS, Ballman KV, Gabriel SE (2005) Cardiovascular death in rheumatoid arthritis. Arthritis Rheum 52(3):722–732
Alkaabi JK, Ho M, Levison R, Pullar T, Belch JJ (2003) Rheumatoid arthritis and macrovascular disease. Rheumatology 42(2):292–297
Anstead GM (1998) Steroids, retinoids, and wound healing. Adv Wound Care 11:277–285
Wilson O, Kirwan JR (2006) Measuring sensation in the feet of patients with rheumatoid arthritis. Musculoskeletal Care 4(1):12–23
Lavery LA, Armstrong DG, Vela SA, Quebedeaux TL, Fleischli JG (1998) Practical criteria for screening patients at high risk for diabetic foot ulceration. Arch Intern Med 158(2):157–162
Murray HJ, Young MJ, Hollis S, Boulton AJ (1996) The association between callus formation, high pressures and neuropathy in diabetic foot ulceration. Diabet Med 13(11):979–982
Veves A, Murray HJ, Young MJ, Boulton AJM (1992) The risk of foot ulceration in diabetic patients with high foot pressure: a prospective study. Diabetologia 35:660–663
Collis WJMF, Jayson MIV (1972) Measurement of pedal pressures: An illustration of a method. Ann Rheum Dis 31:215–217
Minns RJ, Craxford AD (1984) Pressure under the forefoot in rheumatoid arthritis. A comparison of static and dynamic methods of assessment. Clin Orthop Relat Res 187:235–242
Tuna H, Birtane M, Tastekin N, Kokino S (2005) Pedobarography and its relation to radiologic erosion scores in rheumatoid arthritis. Rheumatol Int 26:42–47
Woodburn J, Helliwell PS (1996) Relation between heel position and the distribution of forefoot plantar pressures and skin callosities in rheumatoid arthritis. Ann Rheum Dis 55(11):806–810
Elwood M (1998) Critical appraisal of epidemiological studies and clinical trials, 2nd edn. Oxford University Press, Oxford
Funding
This work was funded by the award of a Doctoral Nursing Studentship to Jill Firth from the Smith and Nephew Foundation. Dr. Firth is currently the recipient of the Smith and Nephew Post Doctoral Research Fellowship.
Disclosures
No disclosures.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Firth, J., Helliwell, P., Hale, C. et al. The predictors of foot ulceration in patients with rheumatoid arthritis: a preliminary investigation. Clin Rheumatol 27, 1423–1428 (2008). https://doi.org/10.1007/s10067-008-0940-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-008-0940-y


