Abstract
The objective of our study was to investigate the role of musculoskeletal ultrasound (US) in the assessment of hand and foot small joints in psoriatic arthritis (PsA). Thirteen consecutive patients with PsA of hands or feet underwent B-mode US using a 9- to 13-MHz transducer and simultaneous magnetic resonance imaging (MRI), bone scintigraphy and radiography. US findings were compared with radiography, MRI and scintigraphy in 190, 182 and 109 joints, respectively. To assess the sensitivity and specificity of US, radiography was considered as gold standard for the detection of erosions and osteoproliferations and MRI as gold standard for the detection of joint effusion and synovitis. US, MRI and scintigraphy had a higher sensitivity in the detection of overall joint pathology than radiography in painful and/or swollen joints (71%, 72%, 82% vs 32%) and clinically unaffected joints (17%, 21%, 9% vs 2%). US and radiography detected more erosions and osteoproliferations than MRI, with low agreement between the methods in the detection of erosions. Radiography was superior to US in the visualisation of osteoproliferations. Joint effusions and/or synovitis were more frequently detected by MRI than US. Agreement between both imaging methods was better in carpal joints, carpometacarpal joint I, metacarpophalangeal (MCP)/metatarsophalangeal (MTP) joint I, II and V than in MCP/MTP III, IV, PIP and DIP joints. Compared with MRI, radiography and scintigraphy, the specificity of US ranges between 0.84 and 0.94, depending on the joint pathology. In conclusion, the diagnostic sensitivity of US in the detection of PsA-related synovitis of hands and feet is lower than MRI and depends on the joint region. However, the low cost and the acceptable specificity suggest that US is a useful imaging method in addition to radiography in PsA of hands and feet.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Psoriatic arthritis (PsA) can present as monoarthritis, oligoarthritis, polyarthritis or spondylitis. Involvement of hands or feet is frequent, with transversal arthritis of the distal interphalangeal (DIP) joints or longitudinal involvement of several finger or toe joints as typical presentations.
Plain radiography may reveal the typical coexistence of osteodestructive and osteoproliferative changes of peripheral joints and may thus be helpful in establishing the correct diagnosis. Plain radiographs are useful in the monitoring of disease progression but are insensitive in early arthritis [1]. Bone scintigraphy provides information about the distribution but not the severity of joint involvement and has a low specificity. Data from patients with rheumatoid arthritis indicate that magnetic resonance imaging (MRI) is a more sensitive method than radiography in demonstrating erosions and synovial pathology [2]. Moreover, MRI detects bone marrow oedema and enthesitis that may not be linked to synovitis in PsA [3]. Despite these advantages, the use of MRI is limited by its availability and high cost.
Musculoskeletal ultrasound (US) has become an established adjunctive technique for the diagnosis of rheumatic diseases. By its ability to detect minor synovitic lesions especially in wrist, finger and toe joints, it can differentiate synovitis from tenosynovitis, detect bone erosions [4, 5] and also guide loco-regional procedures [6]. Few studies examined the diagnostic utility of US in finger joints of patients with PsA [7, 8]. More importantly, the role of US in the assessment of small joints has not been compared with other imaging methods (plain radiography, MRI and scintigraphy). For example, the ability of US to detect osteoproliferation as a distinguishing feature of PsA has not been systematically examined. This lack of information prompted us to evaluate the role of US in comparison with other imaging methods.
Materials and methods
Patients
After approval of the ethics committee, consecutive patients were enrolled into the study provided that they had PsA, as diagnosed by the criteria of Moll and Wright [9], involvement of the small joints of the hands and/or feet and were willing to provide written informed consent. Joint involvement was clinically scored by two independent rheumatologists (KW, UAW). Joint involvement was rated as agreement between the two raters. The most affected hand or foot was chosen for further investigations.
Thirteen consecutive patients (male/female 9/4; mean age 45 years; range 32–57 years) were enrolled. The mean disease duration was 5.5 years (range 1–11 years). All subjects had clinically active disease in at least one peripheral joint region, as suggested by joint swelling. Patients were also required to either have elevated levels of C-reactive protein or an elevated blood sedimentation rate.
Joint imaging
US, X-ray, MRI and scintigraphy were performed within 3 days after inclusion into the study. Only hands and feet were included into the study (eight hands and five feet). All joints of the most affected hand or foot were investigated. To evaluate the sensitivities and specificities of US, MRI was chosen as the gold standard imaging method for the detection of synovitis and effusion; radiography was chosen as gold standard for the detection of erosions and osteoproliferations. Finally, joint affection at any quality detected by US was compared with scintigraphy.
Plain radiography
The wrist, finger and toe joints of the patients were examined by conventional radiography. Osteodestructive joint damage and osteoproliferations (periostal proliferations) were scored with the method of Wassenberg et al. [10]. Scoring of the radiographies was performed by the radiologist (MU).
Carpal joints, carpometacarpal I joint, metacarpo-/metatarsophalangeal joints, interphalangeal I joints (IP), proximal interphalangeal joints (PIP) and DIP were scored. Plain radiography was performed in all 13 patients. A total of 190 joints were investigated by radiography (eight hands and five feet); the carpometacarpal I joints were not included in the analysis because of the high frequency of unspecific degenerative joint changes.
Ultrasonography (US)
US was performed in all patients by a third rheumatologist (SMW) experienced in this technique. US images were scored at the moment of acquisition. The sonographer was blinded for the findings in radiography, MRI, scintigraphy, clinical examination and laboratory data. We used a Logiq 200 Pro unit (Kranzbühler, Germany) with a linear array transducer operating at a frequency of 9–13 MHz. Images were acquired according to the guidelines for musculoskeletal ultrasound in rheumatology [5]. The joints were investigated dynamically by moving the joint during examination and by scanning around the joint surface. Longitudinal and transverse scans were carried out dorsal and palmar of the wrist joints, with additional thenar scans. Dorsal and palmar lateral, longitudinal and transverse scans were performed at the proximal and distal interphalangeal and metacarpophalangeal (MCP) I, II and V joints. Dorsal and palmar longitudinal and transverse scans were performed at the MCP joints III and IV. On the feet, the scans were carried out as plantar and dorsal longitudinal and transverse scans, with additional lateral scans on the first and fifth toe. All joints within affected hands or feet were scored.
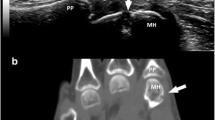
Effusion was defined by abnormal hypoechoic or anechoic intraarticular material, which is displaceable and compressible; synovitis was defined as abnormal hypoechoic intraarticular tissue, which is non-displaceable and poorly compressible [11]. The fact that Doppler sonography was not used in our study impaired a clear distinction of joint effusion and synovitis in some cases. We therefore analysed joint effusion and synovitis in a single group. Intraarticular discontinuities of the bone surface were interpreted as bone erosions. Echoic reflexes proximal of the bone margins were interpreted as osteoproliferations when marked joint space narrowing was absent. All abnormal findings had to be present in two perpendicular planes. Findings were compared with apparently unaffected joints at the same contralateral joint level of the same patient to avoid overinterpretation of ambiguous findings.
Magnetic resonance imaging (MRI)
MRI was obtained on a 1.0-T Siemens Magnetom Impact Unit (Erlangen, Germany). Coronal T1-weighted, axial T2-weighted sequences, T2-weighted coronal inversion recovery sequences and axial sequences after fat suppression were obtained before and after intravenous injection of gadolinium-DTPA (0.1 mmol/kg body weight, Magnevist, Schering, Berlin, Germany). The following pathologic changes were assessed by the radiologist (MU): joint effusion, synovitis, bone marrow oedema, erosions and osteoproliferations (periostal proliferations). Bone marrow oedema was defined as an intraosseous localized (larger than 3 × 3 mm) signal increase in the T2-weighted MR images. Because the study focussed on the imaging of joint involvement, tenosynovitis and enthesitis were excluded from the analysis.
MR images were available in 12 patients (seven hands, five feet). A total of 182 joints (seven carpal joints, seven carpometacarpal joints of the thumb, 60 metacarpo-/metatarsophalangeal joints, 12 interphalangeal joints (IP: seven IP joints of the thumb and five from the first MTP), 48 proximal interphalangeal joints (PIP) and 48 distal interphalangeal joints (DIP) were investigated.
Scintigraphic assessment
Bone scintigraphy was performed according to standard procedures. Five minutes after intravenous injection of 500–700 MBq of [99mTc]-DPD (TECEOS, Schering, Berlin, Germany), planar images were acquired from anterior and posterior using a double-headed whole body gamma camera (Body scan, Siemens, Erlangen, Germany) with a low-energy all-purpose collimator. Whole body images were acquired at a speed of 0.2 m/min with a 20% energy window around the 140-keV peak. In addition, images of the hands and feet were acquired for 2 min each. The same images were repeated 3 h after injection. Two physicians trained in nuclear medicine read the scans independently. They were aware of the diagnosis but blinded to all other clinical information. Disagreement was solved by consensus reading.
Informed consent for scintigraphy was obtained in seven individuals (four hands, three feet). A total of 109 joints were investigated by scintigraphy.
Results
On clinical examination, 25% of the examined joints were painful and 20% were swollen. In contrast, plain radiography revealed pathologic changes in 33 out of 190 joints (17%), MRI in 68 out of 182 joints (37%), scintigraphy in 38 out of 109 joints (35%) and US in 56 of 182 joints (31%).
Joint erosions
US-detected joint erosions in 26 joints, of which ten were also concordantly detected by conventional radiography and eight by MRI (Tables 1, 2, 3; Fig. 1). Among the joints scored negative for erosions by US, radiography detected erosions in 15 joints and MRI in six joints. US-detected erosions, which were not detected by MRI and radiography in 18 and 14 joints, respectively. When radiography was defined as gold standard, the sensitivity of US in the detection of joint erosions was 10/25 (40%) and the specificity 151/165 (91%).
Erosions detected by US only (e.g. without a correlate within radiography or MRI) were mostly present in PIP and DIP joints (Tables 2 and 3; Fig. 2).
Osteoproliferation
Osteoproliferations were detected by radiography in 21 joints, of whom ten were also detected by US (Table 1; Fig. 3). Thus, osteoproliferations were seen by radiography in 11 joints only. Conversely, osteoproliferative changes without corresponding changes in radiography were suggested by US in six joints. The sensitivity of US in the detection of osteoproliferation was 10/21 (0.48) and the specificity (163/169, 0.96) in comparison to the gold standard radiography. Osteoproliferative changes were detected by MRI in one joint only.
Joint inflammation (effusion and synovitis)
Effusion and synovitis were detected concordantly by US and MRI in 33 joints (Table 1). Thirty four joints were affected by MRI only and 10 joints by US only. The sensitivity of US in the detection of synovitis/joint effusion was 33/68 (0.48) and the specificity (105/115, 0.91) in comparison to MRI. Subgroup analysis showed that the congruence between the two imaging modalities depended on the joint region (Fig. 2). Within carpal joints, the agreement was higher than within MCP/MTP joints; concordance was lowest within the peripheral joints (PIP/DIP). Within MCP/MTP joints, agreement between MRI and US was present only in MCP/MTP I, II and V (15/25 joints, 60%), whereas in MCP/MTP III and IV, the agreement was low (0/13 joints; Fig. 2). The sensitivity of US was higher in carpal joints (5/7, 0.71) and carpometacarpal I joints (4/7, 0.57) compared to MCP/MTP joints (15/33, 0.45) and PIP/DIP joints (9/20, 0.45).
The agreement between US and MRI was higher when at least one of the pathologic findings (effusion/synovitis/erosions/osteoproliferations) were grouped together as an arthritic joint compared to the analysis of single qualities. Compared to joints which were also shown to be affected by MRI, US-detected joint effusion/synovitis and/or bone changes (erosions, osteoproliferations) in six of seven carpal joints (86%), 5/7 carpometacarpal I joints (71%), 15/33 MCP/MTP joints (45%) and 13/21 PIP/DIP joints (62%).
Comparison of US with scintigraphy
One hundred nine joints were examined with both bone scintigraphy and US. Twenty-nine joints were concordantly affected by both methods, nine joints by scintigraphy only and 11 joints by US only. The sensitivity of US was 29/38 (0.76) with a specificity of 60/71 joints (0.84) compared with scintigraphy.
Comparison of clinical joint examination and US
The findings of clinical joint examination (joint tenderness and/or joint swelling) were compared with the US findings. US-detected pathological findings in 71% of painful or swollen joints, but 17% of the joints were affected by US and not by clinical examination. MRI and scintigraphy detected pathological changes in 72% and 82% of clinically affected joints and in 21% and 9% of clinically unaffected joints, respectively. In contrast, radiography was significantly less sensitive in detecting joint changes in PsA: 32% and 2% of clinically affected and clinically unaffected joints showed pathologic changes in radiography, respectively.
Discussion
Musculoskeletal US has emerged as a powerful tool in the evaluation of soft tissue involvement in rheumatic diseases. In this pilot study, we evaluated the usefulness of US in the assessment of peripheral joints in PsA. To assess the sensitivity and specificity of US, we compared the US findings with MRI, scintigraphy and plain radiography and investigated all joints at the most affected hand or foot.
In early arthritis, US was slightly superior to MRI with regard to the detection of finger joint effusions or synovitis; the differences were, however, not statistically significant [12]. Other investigators also compared US with MRI in the imaging of MTP synovitis in rheumatoid arthritis and found a 87% sensitivity and 74% specificity of US [13]. In contrast, when we used MRI as the gold standard in the detection of synovitis or joint effusion, US had a relatively low sensitivity (48%) but a high specificity (91%). The low sensitivity of US may be explained by several factors. It is well known that patients with PsA, in contrast to patients with rheumatoid arthritis, may have inflamed joints but only little fluid collections or synovial proliferation [14]. Thus, B-mode US may have limited sensitivity in the detection of hyperperfused early synovitis without effusion, whereas the use of gadolinium in MRI enhances the detection of synovitis. The detection of bone marrow oedema may facilitate the detection of early psoriatic arthritis with MRI [15].
In our study, we used B-mode US. However, the sensitivity of US may be enhanced using colour Doppler or pulse inversion harmonic imaging technology, as shown in several recent studies [16, 17].
US had an acceptable specificity in comparison to MRI with regard to effusions and/or synovitis. However, US-detected effusions and/or synovitis in 10 joints scored negative by MRI. US may visualise mild osteoarthritic changes, which were not scored as synovitis because of the lack of postcontrast enhancement. Moreover, mild hypoechoic rims within joints may also be present in healthy subjects [18]. There was also a site dependency of the sensitivity and specificity of US with better agreement within carpal joints, MCP/MTP I, II and V and poorer results in MCP/MTP III, IV, PIP and DIP joints. These results are consistent with other investigations, which showed a higher sensitivity in the detection of synovitis in wrist (86%) than MCP joints (50%) when compared with MRI [19]. The low agreement between US and MRI with respect to synovitis in MCP III and IV joints may be related to the fact that these joints cannot be scanned from the radial or ulnar side by US. MRI has detected synovitis most frequently at the radial side of the MCP joints in early rheumatoid arthritis, especially in MCP III and MCP IV joints [20].
Studies in patients with rheumatoid arthritis suggest that MRI and US may detect more erosions than radiography [12, 21, 22]. In our study, radiography detected more joint erosions and osteoproliferations than MRI. The reason for this lower sensitivity of MRI in the detection of joint erosions and osteoproliferations may result from the frequent involvement of PIP and DIP joints in PsA. Even if our study was not conducted to evaluate the sensitivity of MRI in PsA, our data suggest that the MRI scanner used in the present study had a low sensitivity in the detection of pathologic changes of bone in the PIP and especially in the DIP joints. The use of new 3-T MRI scanners in these studies may have resulted in a superior sensitivity of MRI compared with radiography.
Compared to radiography and MRI, US had a sensitivity of 40% and 57% for erosions. On the other hand, US and MRI both visualised bone erosions not seen by radiography in our study. In accordance with our data, the study of Backhaus et al. [23] suggested that US and radiography detect more erosions in PIP and DIP joints than MRI. In the same study, however, the concordance of MRI with conventional radiography in the detection of erosions was superior to US, and MRI was the most sensitive diagnostic modality in the detection of early erosions and their follow-up [23]. In rheumatoid arthritis, discrepancies between US, radiography and MRI in the assessment of MTP joints were obviously based on the size of erosions [13].
Osteoproliferations have been described in up to 25% of patients with PsA and are important findings in the differential diagnosis to other causes of arthritis. An early study by Iurassich et al. [7] described that articular bone heads had a bodkin-like appearance in proximal phalanges in RA, whereas PsA was associated with a hook-like appearance of the distal phalanges. We interpreted this hook-like pattern in US as osteoproliferations. This osteoproliferation may be detectable in US but not in MRI. The sensitivity of US for osteoproliferations, however, was low (48%) when compared to conventional radiography. Nevertheless, six osteoproliferations were identified by US only. However, it is unclear whether the osteoproliferations seen by US represent early degenerative bone changes, such as osteophytes or real inflammatory osteoproliferations. In previous studies, it was suggested that US is able to detect even minimal bone margin changes not visualised by radiography [21], but such small changes may also be overinterpreted. Longitudinal studies will have to show whether erosions seen by US indeed progress to radiologically proven erosions. The current data suggest that radiography should be performed in suspected PsA.
The usefulness of scintigraphy in PsA was addressed in only few reports [24, 25]. In our study, scintigraphy showed a good agreement with US findings when any pathological finding in any of the three categories (synovitis, erosions and osteoproliferations) was judged as joint involvement. It should be kept in mind, however, that scintigraphy has the advantage to simultaneously scan for inflammation in most joints of the body.
US and MRI detected pathologic findings in 71% and 72% of clinically affected joints. The association between US or MRI and clinical examination may, however, be influenced by the restriction of US or MRI analysis to joint involvement, whereas periarticular soft tissue involvement may also occur in the absence of synovitis in some PsA patients. On the other hand, US, MRI and scintigraphy but not radiography detected inflammatory changes in joints not affected by clinical examination at a relatively high frequency (17%, 21% and 9%, respectively). Our observations confirm those of previous studies, suggesting that US may be superior to clinical examination in detecting soft tissue inflammation and effusion [26, 27]. Wakefield et al. [26] found a similar frequency (13%) of subclinical sonographic synovitis in patients with inflammatory oligoarthritis, mainly in MTP (79%) and MCP joints (16%). The higher sensitivity of US and MRI for effusion and synovitis may permit the recognition of a polyarticular pattern in patients who have an oligoarticular pattern clinically. Thus, US or MRI may be included in future studies on the subtyping and classification criteria of PsA. Studies using US and MRI may also be used to investigate whether a proportion of patients with psoriasis and arthralgias without swelling on physical examination has true synovitis. [28].
We conclude that the low sensitivity of radiography (32%) in the detection of overall joint pathology in clinically affected joints calls for additional imaging modalities. US, MRI and scintigraphy detect such joint pathology more frequently. Scintigraphy is suitable in the assessment of the distribution of affected joints but only provides information about overall joint pathology. MRI is superior to US in the detection of inflammation but has the disadvantage of high costs and the inability to detect osteoproliferative bone changes. By focussing on erosions, joint effusion and osteoproliferation and by excluding enthesitis and tenosynovitis from our analysis, we have diminished the diagnostic power of the MRI compared to the other imaging modalities. B-mode US on the other hand is a sensitive and inexpensive imaging method but dependent on the experience of the sonographer.
References
Uhl M (1999) Radiology of the manual skeleton. 1. Inflammatory joint diseases and rheumatology. Radiologe 39:432–449
Klarlund M, Ostergaard M, Jensen KE et al (2000) Magnetic resonance imaging, radiography, and scintigraphy of the finger joints: one year follow up of patients with early arthritis. The TIRA Group. Ann Rheum Dis 59:521–528
McGonagle D (2005) Imaging the joint and enthesis: insights into pathogenesis of psoriatic arthritis. Ann Rheum Dis 64(Suppl 2):ii58–ii60
Grassi W, Cervini C (1998) Ultrasonography in rheumatology: an evolving technique. Ann Rheum Dis 57:268–271
Backhaus M, Burmester GR, Gerber T et al (2001) Guidelines for musculoskeletal ultrasound in rheumatology. Ann Rheum Dis 60:641–649
Grassi W, Lamanna G, Farina A et al (1999) Synovitis of small joints: sonographic guided diagnostic and therapeutic approach. Ann Rheum Dis 58:595–597
Iurassich S, Rossi E, Carbone M et al (1999) Ultrasound patterns of rheumatoid and psoriatic arthritis in finger joints. Radiol Med (Torino) 98:15–18
Kane D, Greaney T, Bresnihan B et al (1999) Ultrasonography in the diagnosis and management of psoriatic dactylitis. J Rheumatol 26:1746–1751
Moll JM, Wright V (1973) Psoriatic arthritis. Semin Arthritis Rheum 3:55–78
Wassenberg S, Fischer-Kahle V, Herborn G et al (2001) A method to score radiographic change in psoriatic arthritis. Z Rheumatol 60:156–166
Wakefield RJ, Balint PV, Szkudlarek M et al (2005) Musculoskeletal ultrasound including definitions for ultrasonographic pathology. J Rheumatol 32:2485–2487
Backhaus M, Kamradt T, Sandrock D et al (1999) Arthritis of the finger joints: a comprehensive approach comparing conventional radiography, scintigraphy, ultrasound, and contrast-enhanced magnetic resonance imaging. Arthritis Rheum 42:1232–1245
Szkudlarek M, Narvestad E, Klarlund M et al (2004) Ultrasonography of the metatarsophalangeal joints in rheumatoid arthritis: comparison with magnetic resonance imaging, conventional radiography, and clinical examination. Arthritis Rheum 50:2103–2112
Schmidt WA (2005) Clinical efficacy of power and color Doppler. Ann Rheum Dis 64:8–9
Totterman SM (2004) Magnetic resonance imaging of psoriatic arthritis: insight from traditional and three-dimensional analysis. Curr Rheumatol Rep 6:317–321
Szkudlarek M, Court-Payen M, Strandberg C et al (2001) Power Doppler ultrasonography for assessment of synovitis in the metacarpophalangeal joints of patients with rheumatoid arthritis: a comparison with dynamic magnetic resonance imaging. Arthritis Rheum 44:2018–2023
Schueller-Weidekamm C, Krestan C, Schueller G et al (2007) Power Doppler sonography and pulse-inversion harmonic imaging in evaluation of rheumatoid arthritis synovitis. AJR Am J Roentgenol 188:504–508
Schmidt WA, Schmidt H, Schicke B et al (2004) Standard reference values for musculoskeletal ultrasonography. Ann Rheum Dis 63:988–994
Scheel AK, Schmidt WA, Hermann KG et al (2005) Interobserver reliability of rheumatologists performing musculoskeletal ultrasonography: results from a EULAR “Train the trainers” course. Ann Rheum Dis 64:1043–1049
Tan AL, Tanner SF, Conaghan PG et al (2003) Role of metacarpophalangeal joint anatomic factors in the distribution of synovitis and bone erosion in early rheumatoid arthritis. Arthritis Rheum 48:1214–1222
Grassi W, Filippucci E, Farina A et al (2001) Ultrasonography in the evaluation of bone erosions. Ann Rheum Dis 60:98–103
Wakefield RJ, Gibbon WW, Conaghan PG et al (2000) The value of sonography in the detection of bone erosions in patients with rheumatoid arthritis: a comparison with conventional radiography. Arthritis Rheum 43:2762–2770
Backhaus M, Burmester GR, Sandrock D et al (2002) Prospective two year follow up study comparing novel and conventional imaging procedures in patients with arthritic finger joints. Ann Rheum Dis 61:895–904
Holzmann H, Krause BJ, Kaltwasser JP et al (1996) Psoriatic osteoarthropathy and bone scintigraphy. Hautarzt 47:427–431
Stoeger A, Mur E, Penz-Schneeweiss D et al (1994) Technetium-99m human immunoglobulin scintigraphy in psoriatic arthropathy: first results. Eur J Nucl Med 21:342–4
Wakefield RJ, Green MJ, Marzo-Ortega H et al (2004) Should oligoarthritis be reclassified? Ultrasound reveals a high prevalence of subclinical disease. Ann Rheum Dis 63:382–385
Hauzeur JP, Mathy L, De Maertelaer V (1999) Comparison between clinical evaluation and ultrasonography in detecting hydrarthrosis of the knee. J Rheumatol 26:2681–2683
Zanolli MD, Wikle JS (1992) Joint complaints in psoriasis patients. Int J Dermatol 31:488–491
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Weiner, S.M., Jurenz, S., Uhl, M. et al. Ultrasonography in the assessment of peripheral joint involvement in psoriatic arthritis. Clin Rheumatol 27, 983–989 (2008). https://doi.org/10.1007/s10067-008-0835-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-008-0835-y