Abstract
We examined the association between clinical parameters of disease activity and serum hyaluronic acid (HA) level in patients with knee osteoarthritis (OA). Fifty-eight patients with knee OA and 40 healthy individuals were included in this study. Demographic data were noted. The radiological evaluation was done according to Kellgren/Lawrence Grading System. Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) was used in evaluating the clinical disability caused by OA. Erythrocyte sedimentation rate (ESR) and serum C-reactive protein (CRP) level were determined in laboratory evaluations. Serum HA levels (in nanogram per milliliter) of both groups were measured by enzyme-linked immunosorbent assay. The mean ages of patients in OA group and control group were 57.58 (9.55) and 43.3(10.2) years, respectively. There was a significant difference between HA levels of both groups (p = 0.02). Also, there was a significant correlation between HA level and disease duration (p = 0.04), ESR (p = 0.001), and CRP level (p < 0.001). However, there was no significant correlation between HA levels with WOMAC and radiological grade (p > 0.05). There is no satisfactory amount of data regarding HA level in OA. In our study, we demonstrated that HA was in high levels in the serum of patients with OA. We suggest that serum HA levels may be used as a marker to determine the disease activity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoarthritis (OA) is a common cause of pain and disability in adults [1]. Between 2 and 10% of the adult population is affected by OA of the knee [2]. Hyaluronic acid (HA), also known as hyaluronan, is a potential biomarker. HA is a glycosaminoglycan formed from alternating units of glucosamine and glucuronic acid [3]. HA is mainly produced by fibroblasts and other cells of connective tissue. It has a structural role in connective tissue matrix and is involved in intercellular communication. HA is diffused in the body entirely and can be found in synovial fluid as a free molecule. Wells et al. [4] reported that HA is found in synovial fluid in high concentration that is responsible for water retention and joint lubrication. Increased serum HA levels had been reported in patients with liver disease [5], impaired renal function [6], cancer [7, 8], hypothyroidism [9], amyloid-associated arthropathy [10], rheumatoid arthritis (RA) [11], and psoriasis with or without arthropathy [12].
Serum level of HA has been suggested as a possible disease process marker in OA [2]. It has been reported that as a result of the cartilage degradation and synovial inflammation, HA gets in to the circulation, and serum HA levels were increased [13].
Our aim in this study was to investigate the relationship between clinical parameters of disease activity and serum HA level in patients with knee OA.
Materials and methods
Study participants
Fifty-eight patients with knee OA were enrolled in this study. The diagnosis of OA was based on the clinical and radiological criteria of the American College of Rheumatology [14]. Forty healthy individuals who had the radiographic grade 0 according to the Kellgren and Lawrence (K/L) Grading System in both knees were included in this study as the normal healthy controls (NHC). The patients who were suspected to have OA other than knee and rheumatic diseases, chronic liver, and malignant diseases were excluded from the study. Demographic data, disease duration (month), morning stiffness (minute), swollen knee, and comorbid conditions were noted.
Clinical evaluation
All participants underwent bilateral anteroposterior radiography of the knee with weight bearing. The radiological evaluation was done according to the K/L Grading System. K/L grade 0 (normal findings) was defined as the absence of radiographic features of OA. K/L grade 1 (questionable) included a minute radiographic osteophyte of doubtful pathologic significance. Radiographs showing an osteophyte but no joint space narrowing were assigned a K/L grade of 2 (mild); moderate joint space narrowing was assigned a K/L grade of 3 (moderate). K/L grade 4 (severe) was defined by severe joint space narrowing with subchondral bone sclerosis [15].
Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) was used in evaluating the clinical disability of OA [16]. WOMAC is conventionally evaluated in three scales/dimensions, namely, pain (domain A), joint stiffness (domain B), and function (domain C). The WOMAC is a self-rating instrument consisting of 24 items, of which 5 relate to pain, 2 to joint stiffness, and 17 to the function scale. All items are rated on a numerical rating scale (Likert scale) of 0 (no symptoms/disability) to 10 (maximal symptoms/disability) [16].
Serum HA levels and other laboratory tests
Erythrocyte sedimentation rate (ESR) (using the standard Westergren method), liver and kidney function tests, and serum C-reactive protein (CRP) level were determined as the laboratory evaluations of patients. Serum HA levels of patient and control group were used with HA Quantitative Test Kit (Corgenix, USA). Serum HA level was measured by enzyme-linked immunosorbent assay (ELISA) and detected in nanogram per milliliter for each patient.
ELISA
Blood was collected, and sera were separated and immediately stored on ice. The sera were frozen at −20°C within 8 h of collection and then transferred to an −86°C environment for long-term storage. Serum HA level was quantified by the method of uncompetitive sandwich ELISA [17].
Informed consent was obtained from both groups, and the local ethic committee of our hospital approved the study protocol.
Statistical analysis
Statistical Package for Social Sciences software, version 11.0 for Windows, was used for all statistical analysis. Differences between patients and control subjects were evaluated by Student’s t test. For correlation between results, Pearson’s correlation coefficient was performed. The level of significance was set at p value less than 0.05.
Results
The mean ages of patients and NHC were 57.6 (9.5) (range: 33–75) and 43.3 (10.2) (range: 26–64) years respectively. Some of the clinical characteristics of the patient and NHC are given in Table 1. There were no swollen joints in OA patients.
Comorbid disorders were present in 43.1% of OA patients and in 32.5% of NHC patients, and no significant difference was found between the two groups (p = 0.290) (Table 2).
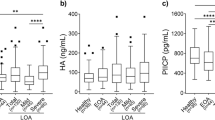
The mean level of serum HA was higher in patients with OA (median 148.2 ng/ml; range: 25.1–1,566.2) than in NHC (median 54.2 ng/ml; range: 1–451.6). There was a significant difference between HA levels of the two groups (p = 0.02). Serum HA levels of both groups are shown in Table 3. The mean scores of WOMAC in OA group were as follows: pain: 12.1 (4.2), stiffness: 3.3 (1.9), and function: 37.8 (12.2). Relationships between serum HA levels and demographic and clinical variables are shown in Table 4. There was a significant correlation between HA level and disease duration (p = 0.04, r = 0.272), ESR (p = 0.001, r = 0.475), and CRP level (p < 0.001, r = 0.556) (Figs. 1 and 2). However, there was no significant correlation between HA level and WOMAC scores (pain p = 0.929, r = 0.012; stiffness p = 0.379, r = 0.119; function p = 0.749, r = 0.043), body mass index (BMI) (p = 0.121, r = 0.210), age (p = 0.947, r = 0.009), and morning stiffness (p = 0.761, r = −0.041).
Patients were assigned into two groups according to radiological grade: group 1 (patients with K/L grade 2) and group 2 (patients with K/L grades 3–4). There was no significant difference in HA levels between both groups (p = 0.494).
Discussion
The results of this study indicate that plasma HA is found in much higher concentrations in OA patients than in NHC. This result suggests that serum HA measurements are useful for assessing patients with OA.
The distribution of HA is not limited to articular tissues but rather encompasses much of the extracellular matrix (ECM). HA is produced locally by cells of the ECM, where it is thought to play a role in cell signaling [18] in addition to its structural properties. Degradation and turnover of the ECM result in the release of HA and HA fragments into the systemic circulation, where they are then cleared by the liver and lymphatics after endocytosis via a HA-specific receptor [17]. Increased production and release of HA from arthritic joints are thought to reflect the localized inflammation occurring in synovial lining [18, 19] and, to a lesser extent, the cartilage degradation [20]. Additionally, it has been suggested that increased HA content in inflamed synovium contributes to the joint stiffness and edema seen in arthritis [4]. Oral administration of corticosteroids has been shown to decrease serum HA production in patients with RA [11].
Serum concentration of HA level can be increased in OA by the same mechanism seen in RA. As a result of synovial inflammation and cartilage degradation, HA gets in to the circulation, and serum levels were increased. Soluble factors of inflammation such as IL-1 and TNF-α can also be responsible for the synovial production of HA [13]. Emlen et al. [21] reported high serum concentration in patients with RA, and also there was a correlation between disease activity and synovial involvement.
Increased serum HA levels have been shown in some patients with severe or active OA, progressive systemic sclerosis, and systemic lupus erythematosus. Authors stated that these findings might be related to growth hormone activity of connective tissue cells in the synovium [2].
It has been shown that serum HA levels were correlated with the degree of joint space narrowing of knee OA [22]. Moreover, Thonar et al. [23] demonstrated that serum HA levels were elevated in canine models of posttraumatic OA. Plasma HA levels in patients with OA have been directly correlated with measurements of functional capacity and estimates of the burden of diseased cartilage [24], as well as indirectly with medial tibiofemoral joint space width [22]. In a prospective study with a 1-year follow-up period in RA patients, the authors concluded that serum HA levels may reflect ongoing joint destruction in RA patients. In our study, we did not show any correlation between serum HA levels and radiological grade. To show the predictive value of serum HA levels in radiologic progression of OA patients, more prospective follow-up studies are needed.
CRP, an acute-phase reactant and marker of systemic inflammation, has been studied as a potential biomarker for OA [25, 26]. In our study, we measured serum CRP levels of the participants and determined a correlation between serum HA levels and this marker of systemic inflammation. In addition, there was a positive correlation between serum HA levels and ESR. In the literature, serum HA has been reported to be correlated with acute-phase proteins and the amino-terminal procollagen type-III peptide [11, 27]. These correlations suggest that serum HA level is a unique and sensitive marker of synovial hyperactivity. Measurement of the level of this molecule in plasma may be useful in conjunction with other blood markers in assessing disease activity and pathogenic changes in joints.
Sharif et al. [2] reported that serum HA level in patients with late stages of OA is more increased than in patients with early stages of OA. Another study demonstrated that there was a positive correlation between the serum HA level and the amount of involved cartilage [24]. We found a correlation between serum HA level and disease duration (Fig. 1). There is a need for further prospective studies to understand the exact relationship between serum HA concentration and disease progression.
In the literature, it has been reported that there was a correlation between the number of swollen joints and the serum HA level [21]. Because of the absence of swollen joints in our OA patients, we could not analyze this relationship.
There is no satisfactory amount of study in the literature regarding the relationship between serum HA level and clinical disability caused by OA. Goldberg et al. [24] showed that the serum HA level in OA patients—but it is interesting to note that not in RA patients—was significantly correlated with the objective functional score. However, it did not show any significant correlation with the subjective functional score in OA patients. Levesque et al. [28] found that serum HA levels did not correlate with clinical or serologic indicators of inflammation. In our study, serum HA levels were significantly correlated with ESR and CRP; however, there was no such correlation between serum HA levels and WOMAC domains. All these contradictory data can be interpreted as: serum HA level is a laboratory indicator of inflammation that does not yet reflect to functional disability related to knee OA.
The main restriction of this study is that there was a significant difference between ages of patients and NHC. OA increases progressively with age at all joint sites. Radiologic changes in OA increase as the individual ages. However, these changes do not always correlate with clinical symptoms or disability [29]. Because of the aim of the study, subjects who had no signs of degenerative changes on knee radiograms were included in the NHC group. As a result of these selection criteria, NHC patients became a younger group when compared to the OA group. We thought that this cannot cause bias. Because as previously reported by Goldberg et al. [24], HA levels and age were not correlated in OA group. Thus, it seems that age has no effect on serum HA levels.
We do not prefer to analyze intraarticular HA level because measuring serum HA levels, which reflects intraarticular HA levels, is much less invasive and easy method when compared to intraarticular HA level measurement.
There is no satisfactory amount of data related to HA level in OA. In our study, we demonstrated that HA was in high levels in the serum of patients with OA. This may be due to degeneration of cartilage, which results in high serum HA levels. We suggest that serum HA levels may be used as laboratory test to determine disease activity. Further studies must be conducted to search the effect of disease progression and its application in new treatment options for OA.
References
Elliott AL, Kraus VB, Luta G, Stabler T, Renner JB, Woodard J, Dragomir AD, Helmick CG, Hochberg MC, Jordan JM (2005) Serum hyaluronan levels and radiographic knee and hip osteoarthritis in African Americans and Caucasians in the Johnston County Osteoarthritis Project. Arthritis Rheum 52:105–111
Sharif M, George E, Shepstone L, Knudson W, Thonar EJ, Cushnaghan J, Dieppe P (1995) Serum hyaluronic acid level as a predictor of disease progression in osteoarthritis of the knee. Arthritis Rheum 38:760–767
Garnero P, Rousseau JC, Delmas PD (2000) Molecular basis and clinical use of biochemical markers of bone, cartilage, and synovium in joint diseases. Arthritis Rheum 43:953–968
Wells AF, Klareskog L, Lindblad S, Laurent TC (1992) Correlation between increased hyaluronan localized in arthritic synovium and the presence of proliferating cells: a role for macrophage-derived factors. Arthritis Rheum 35:391–396
Laurent TC, Dahl IM, Dahl LB, Engstrom-Laurent A, Eriksson S, Fraser JR, Granath KA, Laurent C, Laurent UB, Lilja K et al (1986) The catabolic fate of hyaluronic acid. Connect Tissue Res 15:33–41
Hallgren R, Engstrom-Laurent A, Nisbeth U (1987) Circulating hyaluronate: a potential marker of altered metabolism of the connective tissue in uremia. Nephron 46:150–154
Wilkinson CR, Bower LM, Warren C (1996) The relationship between hyaluronidase activity and hyaluronic acid concentration in sera from normal controls and from patients with disseminated neoplasm. Clin Chim Acta 256:165–173
Manley G, Warren C (1987) Serum hyaluronic acid in patients with disseminated neoplasm. J Clin Pathol 1987 40:626–630
Faber J, Horslev-Petersen K, Perrild H, Lorenzen I (1990) Different effects of thyroid disease on serum levels of procollagen III N-peptide and hyaluronic acid. J Clin Endocrinol Metab 71:1016–1021
Ozasa H, Chichibu K, Tanaka Y, Kondo T, Kitajima K, Ota K (1992) Relationship between plasma levels of hyaluronic acid and amyloid-associated osteoarthropathy in chronic hemodialysis patients. Nephron 61:187–191
Engstrom-Laurent A, Hallgren R (1985) Circulating hyaluronate in rheumatoid arthritis: relationship to inflammatory activity and the effect of corticosteroid therapy. Ann Rheum Dis 44:83–88
Lundin A, Engstrom-Laurent A, Hallgren R, Michaelsson G (1985) Circulating hyaluronate in psoriasis. Br J Dermatol 112:663–671
Nishida Y, D’Souza AL, Thonar EJM, Knudson W (2000) Stimulation of hyaluronan metabolism by interleukin 1α in human articular cartilage. Arthritis Rheum 43:1315–1326
Altman RD (1991) Criteria for classification of clinical osteoarthritis. J Rheumatol 18(Suppl 27):19–23
Kellgren JH, Lawrence JS (eds) (1963) The epidemiology of chronic rheumatism, atlas of standard radiographs. Blackwell, Oxford, UK
Bellamy N, Buchanan WW, Goldsmith CH, Campbell J, Stitt L (1988) Validation study of WOMAC: a health status instrument for measuring clinically-important patient relevant outcomes following total hip or knee arthroplasty in osteoarthritis. J Orthop Rheumatol 1:95–108
Weigel JA, Raymond RC, McGary C, Singh A, Weigel PH (2003) A blocking antibody to the hyaluronan receptor for endocytosis (HARE) inhibits hyaluronan clearance by perfused liver. J Biol Chem 278:9808–9812
Poole AR, Witter J, Roberts N, Piccolo F, Brandt R, Paquin J, Baron M (1990) Inflammation and cartilage metabolism in rheumatoid arthritis: studies of the blood markers hyaluronic acid, orosomucoid, and keratan sulfate. Arthritis Rheum 33:790–799
Manicourt DH, Triki R, Fukuda K, Devogelaer JP, de Deuxchasnes CN, Thonar EJ (1993) Levels of circulating tumor necrosis factor and interleukin-6 in patients with rheumatoid arthritis: relationship to serum levels of hyaluronan and antigenic keratan sulfate. Arthritis Rheum 36:490–499
Hedin PJ, Weitoft T, Hedin H, Engstrom-Laurent A, Saxne T (1991) Serum concentrations of hyaluronan and proteoglycan in joint disease: lack of association. J Rheumatol 18:1601–1605
Emlen W, Niebur J, Flanders G, Rutledge J (1996) Measurement of serum hyaluronic acid in patients with rheumatoid arthritis: correlation with disease activity. J Rheumatol 23:974–978
Sharma L, Hurwitz DE, Thonar EJ, Sum JA, Lenz ME, Dunlop DD, Schnitzer TJ, Kirwan-Mellis G, Andriacchi TP (1998) Knee adduction moment, serum hyaluronan level, and disease severity in medial tibiofemoral osteoarthritis. Arthritis Rheum 41(7):1233–1240
Thonar EJ, Masuda K, Lenz ME, Hauselmann HJ, Kuettner KE, Manicourt DH (1995) Serum markers of systemic disease processes in osteoarthritis. J Rheumatol Suppl 43:68–70
Goldberg RL, Huff JP, Lenz ME, Glickman P, Katz R, Thonar EJ (1991) Elevated plasma levels of hyaluronate in patients with osteoarthritis and rheumatoid arthritis. Arthritis Rheum 34:799–807
Sowers M, Jannausch M, Stein E, Jamadar D, Hochberg M, Lachance L (2002) C-reactive protein as a biomarker of emergent osteoarthritis. Osteoarthr Cartil 10:595–601
Spector TD, Hart DJ, Nandra D, Doyle DV, Mackillop N, Gallimore JR, Pepys MB (1997) Low-level increases in serum C-reactive protein are present in early osteoarthritis of the knee and predict progressive disease. Arthritis Rheum 40:723–727
Horslev-Petersen K, Bentsen KD, Engström-Laurent A, Junker P, Halberg P, Lorenzen I (1988) Serum amino terminal type III procollagen peptide and serum hyaluronan in rheumatoid arthritis: relation to clinical and serological parameters of inflammation during 8 and 24 months treatment with levamisole, penicillamine, or azothioprine. Ann Rheum Dis 47:116–126
Levesque H, Delpech B, Le Loet X, Deshayes P (1988) Serum hyaluronate in rheumatoid arthritis: study by affino-immunoenzymatic assay. Br J Rheumatol 27:445–449
Di Cesare PE, Abramson SB (2005) Pathogenesis of osteoarthritis. In: Harris ED (ed) Kelley’s textbook of rheumatology, 7th edn. Elsevier, Philadelphia, PA, pp 1493–1513
Author information
Authors and Affiliations
Corresponding author
Additional information
This manuscript has been accepted as poster presentation in the Annual European Congress of Rheumatology, 8–11 June 2005, in Vienna, Austria.
Rights and permissions
About this article
Cite this article
Turan, Y., Bal, S., Gurgan, A. et al. Serum hyaluronan levels in patients with knee osteoarthritis. Clin Rheumatol 26, 1293–1298 (2007). https://doi.org/10.1007/s10067-006-0499-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-006-0499-4