Abstract
Kikuchi-Fujimoto’s disease (KFD) or histiocytic necrotising lymphadenitis is a benign and self-limited disease, of unknown aetiology, which affects mainly young women. It presents with localised lymphadenopathy, predominantly in the cervical region, accompanied by fever and leukopenia in up to 50% of the cases. KFD has been rarely described in association with systemic lupus erythematosus (SLE), and its diagnosis can precede, postdate or coincide with the diagnosis of SLE. We present a patient with the diagnosis of SLE characterised by arthritis, leukopenia, malar rash, photosensitivity and positive ANA, besides cervical lymphadenopathy whose biopsy was compatible with KFD, which improved after using prednisone. Although the presence of lymphadenopathy is not uncommon in SLE patients, particularly in the phases of disease activity, the concomitance with KFD has rarely been reported in the literature. Its recognition is necessary because one can avoid laborious investigation for infectious and lymphoproliferative diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Kikuchi-Fujimoto’s disease (KFD), or histiocytic necrotising lymphadenitis, was originally described in Japan in 1972 by Kikuchi [1] and Fujimoto et al. [2] and has been reported in several countries [3] since then, predominantly in Asia [4, 5]. KFD has been reported in association with systemic lupus erythematosus (SLE), although there are few cases reported in the literature [3, 4, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26]. The objective of this paper is to describe a case of KFD associated with SLE, to present a full review of literature on this topic and its possible aetiopathogenic mechanisms.
Case report
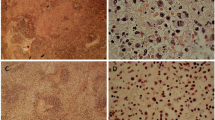
A 20-year-old Brazilian Caucasian woman presented a history of lymphadenomegaly in the cervical region, accompanied by a photosensitive lesion on the face and fever lasting for 30 days. The temperature curve showed an intermittent pattern of fever with a maximum of 38.5°C. In this period the patient experienced asthenia, myalgia in the calves, arthralgia in the interphalangeal joints, alopecia, anorexia, nausea, vomiting and a weight loss of 4 kg. On physical examination she was in general good condition, with a blood pressure level of 130/80 mmHg, heart rate of 72 bpm and respiratory rate of 18 breaths/min. She had a butterfly rash and enlarged lymph nodes in the cervical and in right axillary chain, painless on palpation. Cardiovascular and respiratory examinations were unremarkable. The examination of the extremities revealed oedema in the small joints of the hands. The laboratory investigation showed haemoglobin of 10.7 g/dl, leukocytes of 3400/mm3 with a normal differential count, platelets of 137,000/mm3 and erythrocyte sedimentation rate (ESR) of 55 mm/h. Liver and urine tests were normal. Serologic tests for Toxoplasma, cytomegalovirus (CMV), rubella, Epstein-Barr virus (EBV) and human herpes virus type 1 (HHV-1) were all negative. The antinuclear antibody (ANA) test by IFI in HEp-2 was positive, presenting a speckled pattern with a titre of 1:640. Anti-SSa/Ro antibodies were present in a titre of 1:200 (haemagglutination) as well as anticardiolipin antibodies of IgG isotype (45 GPL). Tests for LE cells and rheumatoid factor as well as the Venereal Disease Research Laboratory (VDRL) slide test were negative. Ultrasonography of the abdomen did not reveal any alterations. Biopsy of the cervical lymph node revealed areas of necrosis with intense karyorrhexis (Fig. 1), surrounded by proliferating histiocytes, without granulomas or aspects of lymphoma. Ziehl-Neelsen and Grocott’s silver stains did not show microorganisms or parasitic elements. The discovery of necrosis with intense karyorrhexis, lymphocytes and histiocytes with crescentic nuclei, and the absence of neutrophils and of haematoxylin bodies were suggestive of KFD (Fig. 2). The skin biopsy showed epidermis with hydropic degeneration of the basal layer and mononuclear infiltration, being compatible with interface dermatitis (Fig. 3). With the diagnosis of KFD associated with SLE, 20 mg/day of prednisone were administered with a gradual improvement of the symptoms. After a follow-up time of 2 years she is taking 400 mg/day of hydroxychloroquine and 5 mg/day of prednisone to control her articular symptoms. The autoantibodies are still present in her serum.
Discussion
KFD is a benign disease, self-limited, with a spontaneous resolution between 1 and 4 months and which predominantly affects young women, in a proportion of 4:1 for women [4, 13, 14, 19, 20, 22], manifesting as localised lymphadenopathy, usually in the cervical region [3, 4, 5, 7, 8, 10, 12, 13, 14, 15, 16, 17, 19, 20, 21, 22, 24, 25, 26, 27, 28, 29, 30, 31, 32], and commonly associated with fever and leukopenia [19, 20, 27, 28].
The aetiology of KFD is as yet unknown. Certain microorganisms (EBV, HTLV-I, herpes human 6 virus,Toxoplasma, parvovirus B19, CMV, Brucella, Yersinia enterocolitica and parainfluenza virus) have been suggested as the causative agents of the disease, initiating a hyper-immune response of the T cells and histiocytes to the infectious agents. However, none of these possibilities have been definitively proven [12, 21, 27]. An autoimmune origin has also been suggested due to a number of cases in which SLE is diagnosed previously, simultaneously or after KFD, demonstrating a strong association between these conditions [4, 19]. Recent studies have demonstrated the occurrence of apoptosis in the affected lymph nodes suggesting that it may play an important role in the histogenesis of KFD [33], although the trigger of apoptosis has not been clearly determined._Hypothetically, apoptotic lymphocytes can deliver nuclear antigens and trigger potentially autoimmune T and B cells for antinuclear antibodies. It is not known if such mechanism participates in the pathogenesis of KFD.
The extra-nodal involvement is uncommon and was only documented in the skin, myocardial and bone marrow [29]. Among these, the skin is the most often reported site in literature [30, 31, 32], characterised by varied and unspecific lesions such as rubella-like maculopapular rash, urticarial papules and cutaneous nodules which are distributed, predominantly, on the face, scalp, trunk and superior members [4, 29, 30, 32]. Likewise, the histological features can also be extremely variable. The skin biopsy of our patient was compatible with interface dermatitis, which has been previously described in cases of KFD [29]. Curiously, a recently described skin manifestation is a lupus-like butterfly rash [31]. In this case it is recommended to perform a biopsy of the lesion for full differentiation.
The diagnosis of KFD is confirmed by the histopathology of the affected lymph node, which reveals a necrotising lymphadenitis, restricted to the cortical and paracortical areas, with partial or complete loss of follicular architecture, associated with marked karyorrhexis. There is also proliferation of hyperplastic histiocytes showing phagocytosis of fragments of karyorrhectic nuclei and of histiocytes with crescentic nuclei [14, 20, 28].
Although SLE and KFD share clinical characteristics such as fever, arthralgia, lymphadenopathy and leukopenia, we believe that our patient presented the two situations simultaneously by the presence of malar rash, arthritis, haematological abnormalities, positive ANA, positive anti-SSa, positive anti-cardiolipin antibodies, chronic evolution, and by the characteristic histological findings of the lymph node. However, sometimes the histological characteristics of KFD have been considered as indistinguishable from those found in lupus, this being the most difficult differential diagnosis [3, 12, 27, 28]. The absence of neutrophils and haematoxylin bodies as have been demonstrated in lupus lymphadenitis may help in the differentiation.
Using the MEDLINE and LILACS databases we identified description of KFD associated with SLE in only 35 cases, distributed all over the world (Table 1). The age of the patients ranged from 10 to 46, with a mean of 29.1±8.98, and the female/male ratio was 3.37:1. SLE was diagnosed previously to KFD in 7 cases [10, 12, 21, 22, 23, 24], simultaneously in 14 cases [10, 11, 12, 15, 18, 19, 20, 22, 25] and afterwards in 14 cases [3, 4, 6, 7, 8, 9, 13, 14, 16, 17, 22, 26]. In the cases in which KFD was diagnosed after or concomitantly with SLE, an obvious tendency was observed to adopt the use of corticosteroids, associated or not with chloroquine, as a standard treatment [12, 21, 23]. On the other hand, when KFD is diagnosed before SLE, it is considered a clinically isolated entity and generally there is no need for treatment due to its benign and self-limited nature, with spontaneous resolution in weeks or months [6, 7, 13, 14, 16].
In conclusion, although KFD is a self-limited condition, it can rarely be associated with SLE and its recognition in this context is important because one can avoid laborious investigation for infectious and lymphoproliferative diseases.
References
Kikuchi M (1972) Lymphadenitis showing focal reticulum cell hyperplasia with nuclear debris and phagocytes. Acta Hematol Jpn 35:379–380
Fujimoto Y, Kojima Y, Yamaguchi K (1972) Cervical subacute necrotizing lymphadenitis. Naika 20:920–927
Dorfman RF, Berry GJ (1988) Kikuchi’s histiocytic necrotizing lymphadenitis: an analysis of 108 cases with emphasis on differential diagnosis. Semin Diagn Pathol 5:329–345
Komócsi A, Tóvari E, Pajor L, Czirják L (2001) Histiocytic necrotizing lymphadenitis preceding systemic lupus erythematosus. J Eur Acad Dermatol Venereol 15:476–480
Graham LE (2002) Kikuchi-Fujimoto disease and peripheral arthritis: a first! Ann Rheum Dis 61:475
Litwin MD, Kirkham B, Henderson DR, Milazzo SC (1992) Histiocytic necrotising lymphadenitis in systemic lupus erythematosus. Ann Rheum Dis 51:805–807
El-Ramahi KM, Karrar A, Ali MA (1994) Kikuchi disease and its association with systemic lupus erythematosus. Lupus 3:409–411
Bousquet E, Tubéry M, Brousset P, Anzieu B, Dubarry B, Massip P, et al. (1996) Kikuchi syndrome, Hashimoto thyroiditis and lupus serology. Apropos of a case. Rev Med Interne 17:836–838
Sire S, Djossou F, Deminière C, Constans J, Ragnaud JM, Aubertin J (1996) Kikuchi-Fujimoto necrotizing histiocytic lymphadenitis: apropos of 2 cases and review of the literature. Rev Med Interne 17:842–845
Biasi D, Caramaschi P, Carletto A, Residori C, Randon M, Friso S, et al. (1996) Three clinical reports of Kikuchi’s lymphadenitis combined with systemic lupus erythematosus. Clin Rheumatol 15:81–83
Adoue D, Rauzy O, Rigal-Huguet F (1997) Kikuchi syndrome, cytomegalovirus infection and lupus. Rev Med Interne 18:338
Martínez-Vázquez C, Hughes G, Bordon J, Alonso-Alonso J, Anibarro-Garcia A, Redondo-Martínez E, et al. (1997) Histiocytic necrotizing lymphadenitis, Kikuchi-Fujimoto’s disease, associated with systemic lupus erythematosus. QJM 90:531–533
Al Salloum AA (1998) Kikuchi’s disease and systemic lupus erythematosus in a Saudi child. Ann Saudi Med 18:51–53
Lopez C, Oliver M, Olavarria R, Sarabia MA, Chopite M (2000) Kikuchi-Fujimoto necrotizing lymphadenitis associated with cutaneous lupus erythematosus: a case report. Am J Dermatopathol 22:328–333
Dalkiliç E, Karakoç Y, Tolunay S, Yurtkuran M (2001) Systemic lupus erythematosus presenting as Kikuchi-Fujimoto disease. Clin Exp Rheumatol 19:226
Vilá LM, Mayor AM, Silvestrini IE (2001) Therapeutic response and long-term follow-up in a systemic lupus erythematosus patient presenting with Kikuchi’s disease. Lupus 10:126–128
Jiménez Sáenz JM, Llorente Arenas EM, Fuentes Solsona F, de Miguel García F, Alvarez Alegret R (2001) Kikuchi-Fujimoto’s disease and the association with systemic lupus erythematosus. An Med Interna 18:429–431
Ben Ghorbel I, Houman MH, Lamloum M, Khanfir M, Miled M, Kchir N, et al. (2002) Concomitant association of Kikuchi disease and systemic lupus erythematosus. Case report. Rev Med Interne 23:797–799
Quintás-Cardama A, Fraga M, Cozzi SN, Caparrini A, Maceiras F, Forteza J (2003) Fatal Kikuchi-Fujimoto disease: the lupus connection. Ann Hematol 82:186–188
Martins EP, Tanure LA, Sasaki M, Viana RS, Carvalho GT, Nicodemo EL, et al. (2003) Doença de Kikuchi-Fujimoto. Relato de três casos e revisão de literatura. Rev Bras Reumatol 43:62–68
Danowski A, Bica B, Baptista R, Nentzinsky W, Azavedo MN (2003) Linfadenite de Kikuchi associada ao lúpus eritematoso sistêmico. Rev Bras Reumatol 43:58–61
Hu S, Kuo TT, Hong HS (2003) Lupus lymphadenitis simulating Kikuchi’s lymphadenitis in patients with systemic lupus erythematosus: a clinicopathological analysis of six cases and review of the literature. Pathol Int 53:221–226
Wano Y, Ebata K, Masaki Y, Takeshita S, Ogawa N, Kim CG, et al. (2000) Histiocytic necrotizing lymphadenitis (Kikuchi-Fujimoto’s disease) accompanied by hemophagocytosis and salivary gland swelling in a patient with systemic lupus erythematosus. Rinsho Ketsueki 41:54–60
Tumiati B, Bellelli A, Portioli I, Prandi S (1991) Kikuchi’s disease in systemic lupus erythematosus: an independent or dependent event? Clin Rheumatol 10:90–93
Meyer O, Ribard P, Belmatoug N, Kahn MF, Grossin M, Fournet JC, et al. (1991) 3 cases of Kituchi’s lymphadenitis in systemic lupus erythematosus. Role of the parvovirus B19. Ann Med Interne (Paris) 142:259–264
Raison-Peyron N, Meunier L, Diebold J, Meynadier J (1996) Kikuchi’s disease and systemic lupus erythematosus. Eur J Dermatol 6:447–449
Menasce LP, Banerjee SS, Edmondson D, Harris M (1998) Histiocytic necrotizing lymphadenitis (Kikuchi-Fujimoto disease): continuing diagnostic difficulties. Histopathology 33:248–254
Norris AH, Krasinskas AM, Salhany KE, Gluckman SJ (1996) Kikuchi-Fujimoto disease: a benign cause of fever and lymphadenopathy. Am J Med 101:401–405
Spies J, Foucar K, Thompson CT, LeBoit PE (1999) The histopathology of cutaneous lesions of Kikuchi’s disease (necrotizing lymphadenitis): a report of five cases. Am J Surg Pathol 23:1040–1047
Kaur S, Thami GP, Kanwar AJ (2002) Kikuchi’s disease, skin and systemic lupus erythematosus. Br J Dermatol 146:167–168
Okuzawa C, Kuroiwa T, Kaneko Y, Ueki K, Tsukada Y, Nojima Y (2003) Kikuchi’s disease accompanied by lupus-like butterfly rash. J Rheumatol 30:857–859
Yasukawa K, Matsumura T, Sato-Matsumura KC, Takahashi T, Fujioka Y, Kobayashi H, et al. (2001) Kikuchi’s disease and the skin: case report and review of the literature. Br J Dermatol 144:885–889
Iguchi H, Sunami K, Yamane H, Konishi K, Takayama M, Nakai Y, et al. (1998) Apoptotic cell death in Kikuchi’s disease: a TEM study. Acta Otolaryngol 53 [Suppl 8]:250–253
Acknowledgement
We thank Dr. Almir Ramos Junior for helping with the histopathological analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Santana, A., Lessa, B., Galrão, L. et al. Kikuchi-Fujimoto’s disease associated with systemic lupus erythematosus: case report and review of the literature. Clin Rheumatol 24, 60–63 (2005). https://doi.org/10.1007/s10067-004-0923-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-004-0923-6