Abstract
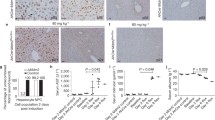
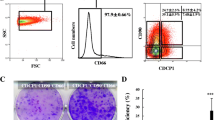
Progenitor cell-derived hepatocytes are critical for hepatocyte replenishment. Therefore, we established a line of human hepatic progenitor (HNK1) cells and determined their biological characteristics for experimental and therapeutic applications. HNK1 cells, isolated from human noncirrhotic liver samples with septal fibrosis, showed high expression of the hepatic progenitor cell (HPC) markers EpCAM, CK7, CK19, alpha-fetoprotein (AFP), CD90 (Thy1), and EFNA1. Expression of CD133 was very low. Ductular reactions at the periphery of cirrhotic nodules were immunohistochemically positive for these HPC markers, including EFNA1. Sodium butyrate, a differentiation inducer, induced hepatocyte-like morphological changes in HNK1 cells. It resulted in down-regulation of the hepatic progenitor cell markers EpCAM, CK7, CK19, AFP, and EFNA1 and up-regulation of mature hepatocyte markers, including albumin, CK8, and CK18. Furthermore, sodium butyrate treatment and a serial passage of HNK1 cells resulted in enhanced albumin secretion, ureagenesis, and CYP enzyme activity, all of which are indicators of differentiation in hepatocytes. However, HNK1 cells at passage 50 did not exhibit anchorage-independent growth capability and caused no tumors in immunodeficient mice, suggesting that they had no spontaneous malignant transformation ability. From this evidence, HNK1 cells were found to be EpCAM+/CD133- hepatic progenitor cells without spontaneous malignant transformation ability. We therefore conclude that HNK1 cells could be useful for experimental and therapeutic applications.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bateman, A.C., and Hubscher, S.G. (2010). Cytokeratin expression as an aid to diagnosis in medical liver biopsies. Histopathology 56, 415–425.
Bioulac-Sage, P., and Balabaud, C. (2010). Human cirrhosis: monoclonal regenerative nodules derived from hepatic progenitor cells abutting ductular reaction. Gastroenterol. Clin. Biol. 34, 267–269.
Clouston, A.D., Powell, E.E., Walsh, M.J., Richardson, M.M., Demetris, A.J., and Jonsson, J.R. (2005). Fibrosis correlates with a ductular reaction in hepatitis C: roles of impaired replication, progenitor cells and steatosis. Hepatology 41, 809–818.
Cui, X.D., Lee, M.J., Yu, G.R., Kim, I.H., Yu, H.C., Yong, S.E., and Kim, D.G. (2010). EFNA1 ligand and its receptor EphA2: potential biomarkers for hepatocellular carcinoma. Int. J. Cancer 126, 940–949.
Desmet, V., Roskams, T., and Van Eyken, P. (1995). Ductular reaction in the liver. Pathol. Res. Pract. 191, 513–524.
Durnez, A., Verslype, C., Nevens, F., Fevery, J., Aerts, R., Pirenne, J., Lesaffre, E., Libbrecht, L., Desmet, V., and Roskams, T. (2006). The clinicopathological and prognostic relevance of cytokeratin 7 and 19 expression in hepatocellular carcinoma. A possible progenitor cell origin. Histopathology 49, 138–151.
Eleazar, J.A., Memeo, L., Jhang, J.S., Mansukhani, M.M., Chin, S., Park, S.M., Lefkowitch, J.H., and Bhagat, G. (2004). Progenitor cell expansion: an important source of hepatocyte regeneration in chronic hepatitis. J. Hepatol. 41, 983–991.
Falkowski, O., An, H.J., Ianus, I.A., Chiriboga, L., Yee, H., West, A.B., and Theise, N.D. (2003). Regeneration of hepatocyte’ buds’ in cirrhosis from intrabiliary stem cells. J. Hepatol. 39, 357–364.
Han, X.J., Lee, M.J., Yu, G.R., Lee, Z.W., Bae, J.Y., Bae, Y.C., Kang, S.H., and Kim, D.G. (2012). Altered dynamics of ubiquitin hybrid proteins during tumor cell apoptosis. Cell Death Dis. 3, e255.
Jung, Y., Brown, K.D., Witek, R.P., Omenetti, A., Yang, L., Vandongen, M., Milton, R.J., Hines, I.N., Rippe, R.A., Spahr, L., et al. (2008). Accumulation of hedgehog-responsive progenitors parallels alcoholic liver disease severity in mice and humans. Gastroenterology 134, 1532–1543.
Kuwahara, R., Kofman, A.V., Landis, C.S., Swenson, E.S., Barendswaard, E., and Theise, N.D. (2008). The hepatic stem cell niche: identification by label-retaining cell assay. Hepatology 47, 1994–2002.
Libbrecht, L., and Roskams, T. (2002). Hepatic progenytor cells in human liver diseases. Semin. Cell Dev. Biol. 13, 389–396.
Lowes, K.N., Brennan, B.A., Yeoh, G.C., and Olynyk, J.K. (1999). Oval cell numbers in human chronic liver diseases are directly related to disease severity. Am. J. Pathol. 154, 537–541.
Ma, S., Chan, K.W., Hu, L., Lee, T.K., Wo, J.Y., Ng, I.O., Zheng, B.J., and Guan, X.Y. (2007). Identification and characterization of tumorigenic liver cancer stem/progenitor cells. Gastroenterology 132, 2542–2556.
Pack, R., Heck, R., Dienes, H.P., Oesch, F., and Steinberg, P. (1993). Isolation, biochemical characterization, long-term culture, and phenotype modulation of oval cells form carcinogen-fed rats. Exp. Cell Res. 204, 198–209.
Piscaglia, A.C., Shupe, T.D., Pani, G., Tesori, V., Gasbarrini, A., and Petersen, B.E. (2009). Establishment of cancer cell lines from rat hepatocholangiocarcinoma and assessment of the role of granulocyte-colony stimulating factor and hepatocyte growth factor in their growth, motility and survival. J. Hepatol. 51, 77–92.
Richardson, M.M., Jonsson, J.R., Powell, E.E., Brunt, E.M., Neuschwander-Tetri, B.A., Bhathal, P.S., Dixon, J.B., Weltman, M.D., Tilg, H., Moschen, A.R., et al. (2007). Progressive fibrosis in nonalcoholic steatohepatitis: association with altered regeneration and a ductular reaction. Gastroenterology 133, 80–90.
Roskams, T., and Desmet, V. (1998). Ductular reaction and its diagnostic significance. Semin. Diagn. Pathol. 15, 259–269.
Roskams, T.A., Theise, N.D., Balabaud, C., Bhagat, G., Bhathal, P.S., Bioulac-Sage, P., Brunt, E.M., Crawford, J.M., Crosby, H. A., Desmet, V., et al. (2004). Nomenclature of the finer branches of the biliary tree: canals, ductules, and ductular reactions in human livers. Hepatology 39, 1739–1745.
Saxena, R., Theise, N.D., and Crawford, J.M. (1999). Microanatomy of the human liver-exploring the hidden interfaces. Hepatology 30, 1339–1346.
Theise, N.D. (2006). Gastrointestinal stem cells. III. Emergent themes of liver stem cell biology: niche, quiescence, self-renewal, and plasticity. Am. J. Physiol. Gastrointest Liver Physiol. 290, G189–G193.
Theise, N.D., Saxena, R., Portmann, B.C., Thung, S.N., Yee, H., Chiriboga, L., and Kumar, A. (1999). The canals of Hering and hepatic stem cells in humans. Hepatology 30, 1425–1433.
Touboul, T., Hannan, N.R., Corbineau, S., Martinez, A., Martinet, C., Branchereau, S., Mainot, S., Strick-Marchand, H., Pedersen, R., Di Santo, J., et al. (2010). Generation of functional hepatocytes from human embryonic stem cells under chemically defined conditions that recapitulate liver development. Hepatology 51, 1754–1765.
Wang, P., Cong, M., Liu, T.H., Yang, A.T., Cong, R., Wu, P., Tang, S.Z., Xu, Y., Wang, H., Wang, B.E., et al. (2010). Primary isolated hepatic oval cells maintain progenitor cell phenotypes after two-year prolonged cultivation. J. Hepatol. 53, 863–871.
Yamashita, T., Ji, J., Budhu, A., Forgues, M., Yang, W., Wang, H.Y., Jia, H., Ye, Q., Qin, L.X., Wauthier, E., et al. (2009). EpCAMpositive hepatocellular carcinoma cells are tumor-initiating cells with stem/progenitor cell features. Gastroenterology 136, 1012–1024.
Yoon, S.M., Gerasimidou, D., Kuwahara, R., Hytiroglou, P., Yoo, J.E., Park, Y.N., and Theise, N.D. (2011). Epithelial cell adhesion molecule (EpCAM) marks hepatocytes newly derived from stem/progenitor cells in humans. Hepatology 53, 964–967.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hao, PP., Lee, MJ., Yu, GR. et al. Isolation of EpCAM+/CD133− hepatic progenitor cells. Mol Cells 36, 424–431 (2013). https://doi.org/10.1007/s10059-013-0190-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-013-0190-y